2
Nonsurgical Periodontal Therapy
Case 1: Hand and Automated Instrumentation
Helen Livson, DMD, MMSc
Case 2: Local Drug Delivery
Emilio I. Arguello, DDS, MSc
Case 3: Systemic Antibiotics
Ricardo Teles, DDS, DMSc
Case 1
Hand and Automated Instrumentation
CASE STORY
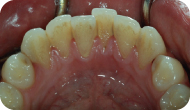
A 25-year-old Caucasian male presented with a chief compliant of: “I need a cleaning.” The patient had noticed significant staining as well as plaque and calculus buildup, particularly in the mandibular anterior region (Figures 1, 2, and 3).
Figure 1: Frontal view.
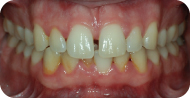
Figure 2: Buccal view, mandibular anterior.
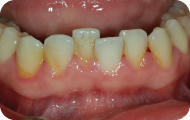
Figure 3: Lingual view, mandibular anterior.
LEARNING GOALS AND OBJECTIVES
- To identify appropriate instruments that may be used for scaling and/or scaling and root planing
- To learn the importance of Initial phase, reevaluation, and maintenance therapy
- To learn the advantages and disadvantages of hand versus power instrumentation
- To understand the limitations of these instruments in the nonsurgical management of periodontal disease
Medical History
The patient reported a history of asthma and used an albuterol inhaler once per day. Otherwise, there were no significant medical problems, and the patient had no known allergies.
Review of Systems
- Vital signs
- Blood pressure: 120/65 mm Hg
- Pulse rate: 72 beats/minute (regular)
- Respiration: 15 breaths/minute
Social History
The patient did not drink alcohol. He reported smoking one time per week.
Extraoral Examination
No significant findings. The patient had no masses or swelling and the temporomandibular joint was within normal limits.
Intraoral Examination
- The soft tissues of the mouth, including the tongue, appeared normal.
- A gingival examination revealed a localized marginal erythema, with rolled margins and some inflamed papillae (Figures 1, 2, and 3).
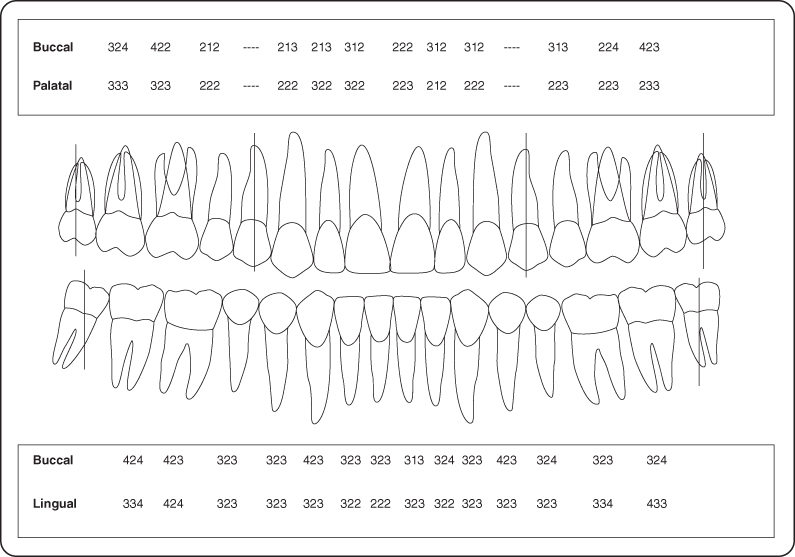
- Periodontal examination and charting were completed (Figure 4).
Figure 4: Probing pocket depth measurements
Occlusion
There were no occlusal discrepancies or interferences.
Radiographic Examination
Bite-wing radiographs were ordered given that the patient was young, and there was no history of caries (Figure 5).
Figure 5: Bite-wing radiographs
Diagnosis
After reviewing the patient’s history as well as the clinical and radiographic examination, a differential diagnosis of generalized moderate gingivitis was made.
Treatment Plan
The treatment plan for this patient includes oral hygiene instructions, followed by scaling and polishing using hand instruments as well as powered mechanical instruments.
Treatment
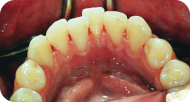
After thorough oral hygiene instructions were reviewed, the patient received a scaling and polishing. The procedure was started with an ultrasonic-powered mechanical instrument to loosen any tenacious supragingival deposits and initial stain removal. Hand scalers were then used, including a sickle scaler for removal of supragingival calculus and stain in the mandibular anterior areas, Gracey curettes were used in the interproximal areas posteriorly, and universal curettes were used for the buccal and lingual aspects of the teeth. The ultrasonic was then used again for a final lavage, and the teeth were finally polished for any additional stain elimination using prophylaxis paste and a rubber cup (Figures 6, 7, and 8).
Figure 6: Frontal view.
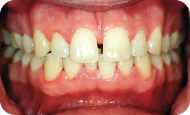
Figure 7: Buccal view, mandibular anterior.
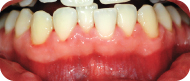
Figure 8: Lingual view, mandibular anterior.
Discussion
The patient presented with heavy supragingival plaque and calculus, gingival inflammation and generalized bleeding on probing with no concurrent bone loss around the teeth. After a detailed medical history was completed, a diagnosis of plaque-induced gingivitis was given. To restore the patient to periodontal health, comprehensive oral hygiene instructions were provided, and a professional scaling was arranged.
A combination of hand instruments and power-driven instruments were used for removal of plaque, calculus, and stain from the teeth. Hand instruments are available in various lengths, shapes, and designs to effectively reach and treat all areas of the teeth. An ultrasonic instrument (Cavitron) was used as an adjunct for stain removal, mechanical debridement, lavage, and cavitation. Regularly scheduled reevaluations and prophylaxis in addition to routine home care are recommended for maintaining optimal oral hygiene and periodontal health in this patient.
Self-Study Questions
A. What are the chief tools available in our armamentarium for scaling and root planing?
B. What are some examples of hand instruments?
C. What is the mechanism of action of powered mechanical instruments?
D. What are the limitations to prophylaxis and scaling and root planing?
E. Are there any contraindications to using power mechanical instruments?
F. Are there differences in efficacy of treatment between hand scalers and automated scalers?
G. What are some advantages and disadvantages of hand instruments versus power mechanical instruments?
Answers located at the end of the chapter.
References
1. Ewen SJ, Glickstein BA. Ultrasonic Therapy in Periodontics. Springfield, IL: Charles C. Thomas, 1968. pp.12–34.
2. Clarke PR, Hill CR. Physical and chemical aspects of ultrasonic disruption of cells. J Acoust Soc Am 1970;47:649–653.
3. Waerhaug J. Healing of the dento-epithelial junction following subgingival plaque control II: as observed on extracted teeth. J Periodontol 1978;49:119–134.
4. Caffesse RG, Sweeney PL, et al. Scaling and root planing with and without periodontal flap surgery. J Clin Periodontol 1986;13:205–210.
5. Lindhe J, Socransky S, et al. Critical probing depths in periodontal therapy. J Clin Periodontol 1982;9:323–336.
6. Philstrom BL, McHugh RB, et al. Comparison of surgical and nonsurgical treatment of periodontal disease. A review of current studies and additional results after 6½ years. J Clin Periodontol 1983;10:524–541.
7. Isidor F, Karring T. Long-term effect of surgical and non-surgical periodontal treatment. A 5-year clinical study. J Periodontal Res 1986;21:462–472.
8. Gantes BG, Nilveus R. The effects of different hygiene instruments on titanium surfaces: SEM observations. Int J Periodontics Restorative Dent 1991;11:225–239.
9. Miller CS, Leonelli FM, et al. Selective interference with pacemaker activity by electrical dental devices. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1998;85:33–36.
10. Breininger D, O’Leary T, et al. Comparative effectiveness of ultrasonic and hand scaling on removal of subgingival plaque and calculus. J Periodontol 1987;58:9–18.
11. Thornton S, Garnick J. Comparison of ultrasonic to hand instruments in the removal of subgingival plaque. J Periodontol 1982;53:35–37.
12. Baeni P, Thilo B, et al. Effects of ultrasonic and sonic scalers on dental plaque microflora in vitro and in vivo. J Clin Periodontol 1992;19:455–459.
13. Nishimine D, O’Leary TJ. Hand instrumentation versus ultrasonics in the removal of endotoxins from root surface. J Periodontol 1979;50:345–349.
14. Hunter R, P’Leary T, et al. The effectiveness of hand versus instrumentation in open-flap root planing. J Periodontol 1984;55:697–703.
15. Matia J, Bissada N, et al. Efficiency of scaling the molar furcation area with and without surgical access. Int J Periodontics Restorative Dent 1986;6:24–35.
16. Gellin R. The effectiveness of the Titan-S sonic scaler versus curettes in the removal of subgingival calculus. A human evaluation. J Periodontol 1986;57:672–680.
17. Bower RC. Furcation morphology relative to periodontal treatment – furcation entrance architecture. J Periodontol 1979;50:23–27.
18. Dragoo M. A clinical evaluation of hand and ultrasonic instruments on subgingival debridement. Part I. With unmodified and modified ultrasonic inserts. Int J Periodontics Restorative Dent 1992;12:311–313.
19. Leon L, Vogel R. A comparison of the effectiveness of hand scaling and ultrasonic debridement in furcations as evaluated with dark-field microscopy. J Periodontol 1987;58:86–94.
20. Ritz L, Hefti A, et al. An in vitro investigation on the loss of root substance in scaling with various instruments. J Clin Periodontol 1991;18:643–647.
21. Larato DC, Ruskin PF, et al. Effect of an ultrasonic scaler on bacterial counts in air. J Periodontol 1967;38:550–554.
TAKE-HOME POINTS
The most fundamental objective of primary phase nonsurgical therapy is to reestablish periodontal health in a patient by reduction of putative periodontal pathogens from the periodontium. Literature has shown that instrumentation can effectively decrease these periodontal pathogens and restore a healthier bacterial flora in the mouth.
A. In our armamentarium the chief tools available for scaling and root planing include:
a. Manual mechanical instrumentation: scalers and curettes
b. Powered mechanical instrumentation: ultrasonics (magnetostrictive, piezoelectric)
c. Air abrasives/rubber cups (for stain removal)
B. Manual instrumentation is an essential skill that dental practitioners need to obtain good results from initial nonsurgical therapy. This involves a thorough understanding of the available tools:
- Scalers are designed for removal of supragingival calculus. They include two straight cutting edges and a pointed toe (e.g., sickle scalers; Figures 9A and B).
- Curettes are designed for subgingival instrumentation but can be used for supragingival scaling as well.
Figure 9: (A,B) Sickle scalers; (C) universal curettes; (D,E) Gracey curettes; (F) magnetorestrictive ultrasonic scalers; (G) piezoelectric ultrasonic scalers.
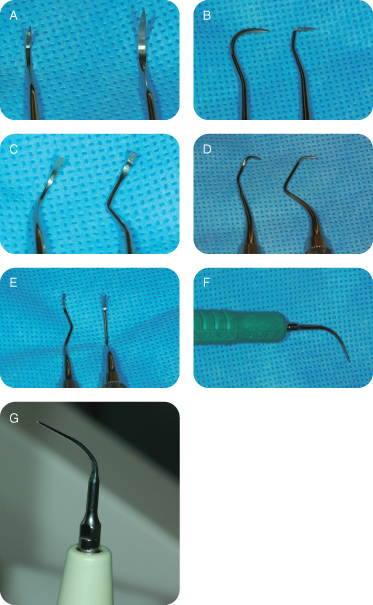
C. Powered mechanical instruments such as sonic and ultrasonic scalers have been used by practitioners to remove plaque, calculus, and stain from tooth and root surfaces. These automated scalers work by way of vibration; as the tip oscillates, it helps break apart adherent deposits that may be present on the teeth. The tip also sprays a jet of water that flushes away any debris and helps dissipate heat produced by the instrument. Additionally, when the water passes over the vibrations of the oscillating tip of an ultrasonic instrument, cavitation is observed. Cavitation is a process by which formation and destruction of small air cavities releases energy that may disrupt bacterial cell membranes [1,2].
Powered mechanical instruments are classified based on their frequencies, and are very effective for nonsurgical debridement of teeth:
- Sonic scalers operate at a frequency of 3000–8000 cycles per second and are driven by compressed air from the dental unit. A rotor system is then activated and the sonic tip oscillates in an elliptical to circular pattern.
- Ultrasonic scalers can be further classified into:
i. Magnetorestrictive ultrasonic scalers: Operate inaudibly, and the tip oscillates at 18,000–42,000 kHz (cycles per second). The motion of the tip is elliptical to circular in motion, and all surfaces of the tip are useful in debridement (Figure 9F).
ii. Piezoelectric ultrasonic scalers: Operate inaudibly, and the tip oscillates at 24,000–45,000 kHz (cycles per second). The motion of the tip is linear, so unlike the magnetorestrictive ultrasonics, only its lateral surfaces are useful in debridement (Figure 9G)
D. Although hand and power instruments are effective in treating periodontal and gingival disease as a part of the initial phase of therapy, there are some limitations. The literature has shown that even with all currently available instrumentation, there is a certain pocket depth up to which we can effectively eliminate subgingival plaque and calculus [3]. Caffesse and associates found that the extent of residual calculus is directly related to pocket depth; as pocket depth increased, the percentage of the tooth surfaces completely free of calculus gradually decreased [4]. This probing depth, referred to as the “critical probing depth,” is 2.9 mm for scaling and root planing. Any probing depth beyond 4.2 mm benefits from surgical periodontal therapy rather than nonsurgical treatment due to the limits of the effectiveness of hand or powered instruments [5]. Nonsurgical treatments are usually attempted first; if appropriate results are not achieved at reevaluation, surgical treatment should be pursued. Whether nonsurgical or surgical periodontal treatment modalities are used, longitudinal studies show that if treated appropriately and followed closely, patients with periodontal disease can be maintained in the long term [6,7].
E. Concerns that the practitioner should be aware of if using powered mechanical instruments are both dental and medical in nature.
Dental
Sonic and ultrasonic instruments should not be used on dental implants because the metal tips can scratch the softer titanium. Ideally, hand periodontal scalers made of plastic, teflon, wood, titanium, or gold-plated scalers should be used for cleaning dental implants [8]. If used too vigorously, these instruments may damage certain restorative margins. As with manual instrumentation, these mechanical instruments may remove excessive tooth structure causing subsequent hypersensitivity.
Medical
Cardiac pacemakers, particularly those placed before the mid-1980s, may get electromagnetic interference from magnetorestrictive ultrasonics (piezoelectric scalers are a safe alternative) [9]. Patients with significant respiratory problems may be intolerant to the aerosols produced during power instrumentation. Powered mechanical instruments should be avoided in patients with infectious diseases that may be transmitted through aerosol production.
F. Several studies have looked at the efficacy of plaque and calculus removal using manual versus automated instrumentation. Studies have demonstrated that both hand instrumentation and ultrasonic instrumentation are very effective in plaque removal [10]. There are no significant differences found between the use of manual instruments and ultrasonic instruments [11] or between sonic and ultrasonic instrumentation in terms of the ability to remove plaque [12].
Some studies have shown that manual instrumentation is more effective than ultrasonic instrumentation in calculus removal [13,14]. However, power instruments have been shown to access concavities and furcations of roots better than manual instruments [15]. According to Gellin, using these methods concurrently is more effective than using either method alone [16].
G. See Table 1.
Table 1: Advantages and Disadvantages of Using Hand Instruments versus Power Mechanical Instruments
| Advantages | Disadvantages | |
| Hand instruments |
* Not as damaging to porcelain and composite restorations * More effective at removing endotoxin from periodontally involved root surfaces [13] |
* Larger tip than furcation width [17] * Fatigue [18] * Longer scaling times [18] |
| Power mechanical instruments |
* Lavage effect of irrigation * Cavitation [1,2] * Slightly shorter scaling times [18] * Less skill required to be competent [18] * More effective in class II and III furcations [19] * Less removal of tooth structure [20] * Require no sharpening |
* May be contraindicated for patients with older cardiac pacemakers [9] * Production of aerosols (30-fold increase of airborne microorganisms in the treatment room) [21] |
Case 2
Local Drug Delivery
CASE STORY
A 35-year-old Caucasian male presented with a chief compliant of: “My dentist told me I have gum disease.” The patient had not experienced pain, discomfort, or swelling associated with his gums; however, he had noticed slight bleeding whenever he brushes and flosses. The patient claimed to brush his teeth at least once daily and he flossed once a week (Figures 1, 2, and 3).
Figure 1: Preoperative view of maxillary anterior teeth and a15-mm periodontal probe demonstrating a 5-mm probing on mesial of tooth #8 with bleeding on probing.
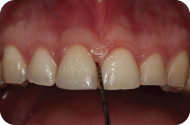
Figure 2: Preoperative periapical radiograph of tooth #8 demonstrating localized mild vertical bone loss.
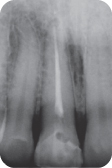
Figure 3: Preoperative view of maxillary posterior teeth and a15-mm periodontal probe demonstrating a 4-mm probing on the distal of tooth #3. with bleeding on probing.
LEARNING GOALS AND OBJECTIVES
- To be able to diagnose periodontitis
- To identify the possible etiological and contributing factors and to address them
- To be able to elaborate an initial treatment plan with a comprehensive phase 1 therapy to achieve initial periodontal stability
- To be able to identify the different local chemotherapeutic agents and how, when, and why to use them
Medical History
There were no significant medical problems and the patient had no known allergies. He had no known medical illnesses. On questioning the patient stated he was not taking any medications and he had no allergies.
Review of Systems
- Vital signs
- Blood pressure: 128/83 mm Hg
- Pulse rate: 69 beats/minute (regular)
- Respiration: 16 breaths/minute
Social History
The patient did not smoke and drank alcohol twice a month in social gatherings.
Extraoral Examination
There were no significant findings. The patient had no masses or swelling, and the temporomandibular joint was within normal limits.
Intraoral Examination
- The soft tissues of the mouth (except gingiva) including the tongue appeared normal.
- A gingival examination revealed a mild marginal erythema and swollen papillae (Figures 1 and 3).
- The patient exhibited localized areas with bleeding on probing as well as localized areas with probing depths of 4 and 5 mm (Figures 1 and 3).
- A periodontal chart was completed. Note that the clinical attachment levels (CALs) are not displayed because all gingival margins are at the level of the cementoenamel junction, thus the CAL equals probing depth (Figure 4).
Figure 4: Probing pocket depth measurements.
Occlusion
There were no occlusal discrepancies or interferences.
Radiographic Examination
A full-mouth set of radiographs was ordered; there were localized areas of mild vertical bone loss on the mesial and distal surfaces of tooth #8 (Figure 2).
Diagnosis
After reviewing the history and the clinical and radiographic examinations, the periodontal diagnosis was localized chronic mild periodontitis. The American Dental Association diagnosis is type II.
Treatment Plan
The treatment plan consisted of addressing the primary etiology that causes the periodontal disease for this patient (phase 1) in the following order: oral hygiene instruction, subgingival scaling and root planing of all sites with initial probing depths >3 mm, and supragingival scaling and polishing of all other surfaces; concomitant administration of a local antibiotic immediately after scaling (Figures 5 and 6) and only for sites with probing depths >3 mm followed by a minimum of 4- to 6-week reevaluation to assess the need for further periodontal therapy (phase 2).
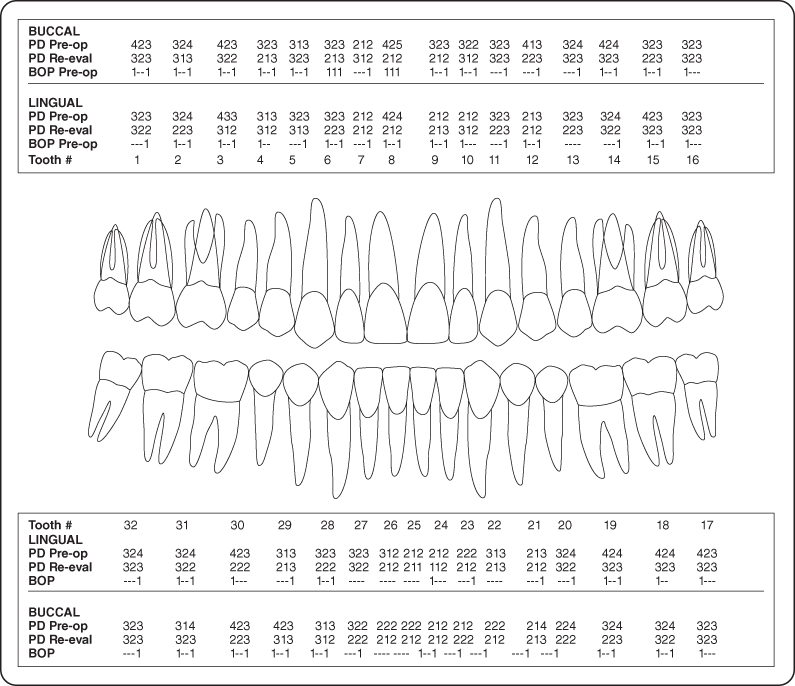
Figure 5: Application of local delivery Atridox on a 5-mm pocket mesial of tooth #8 immediately after scaling and root planing.
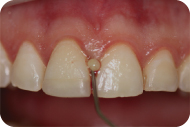
Figure 6: Application of local delivery Atridox on a 4-mm pocket distal of tooth #3 immediately after scaling and root planing.
Reevaluation of Post-Initial Treatment
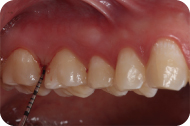
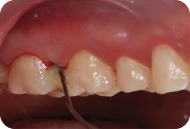
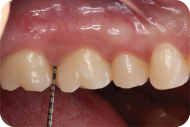
At the 6-week reevaluation, the clinical parameters had improved, and certain probing depths that were initially >3 mm were reduced (Figures 7 and 8). Thus no further periodontal treatment is needed; only periodontal maintenance therapy every 3 months is required.
Figure 7: A 6-week postoperative image of maxillary anterior teeth demonstrating a 2-mm probing on mesial of tooth #8.
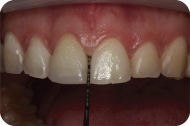
Figure 8: A 6-week postoperative image of maxillary posterior teeth demonstrating a 3-mm probing on distal of tooth #3.
Discussion
Elements that will lead to a successful outcome include an accurate diagnosis based on a thorough history and a periodontal examination, as well as the knowledge of the etiologic factors that lead to disease. Next, a variety of treatment choices must be selected with the understanding that the primary goal will be to provide initial stability of the disease or condition – in this particular case, periodontitis. Once the initial stability has been achieved, either one can proceed to further treatment such as restorative procedures or have the patient on a 3-month periodontal maintenance protocol. However, initial therapy or phase 1 with or without the concomitant use of local chemotherapeutics may not achieve the optimal periodontal stability desired, and thus, it is the responsibility of the treating clinician to identify what areas will need to proceed to the second phase of therapy that could involve periodontal surgery.
ACKNOWLEDGMENTS
Thanks to Dr. Campo Elias Perez for his contribution to this chapter.
Self-Study Questions
A. What is the rationale for using local antibiotics and other agents for the treatment of periodontal disease?
B. Which local drug delivery agents are available?
C. What type of patients will benefit from the use of a local antibiotic?
D. What is the existing research that justifies the use of local drug delivery?
E. What are the techniques and devices used to deliver local antibiotics?
F. What are the considerations for patients to observe after receiving the local antibiotic?
G. What are the determinants of success in local drug delivery therapy?
H. What does rescue therapy mean, and how can local antibiotics help patients with periodontal disease that recurs over time?
Answers located at the end of the chapter.
References
1. Haffajee AD, Arguello EI, et al. Controlling the plaque biofilm. Int Dent J 2003;53(Suppl 3):191–199.
2. Haffajee AD, Socransky SS. Introduction to microbial aspects of periodontal biofilm communities, development and treatment Periodontol 2000 2006;42:7–12.
3. Hanes P, Purvis J. Local anti-infective therapy: pharmacological agents. A systematic review. Ann Periodontol 2003;8:79–98.
4. Golub LM, Goodson JM, et al. Tetracyclines inhibit tissue collagenases. J Periodontol 1985;56(Suppl):93–97.
5. Slot DE, Kranendonk AA, et al. The effect of a pulsed Nd:YAG laser in non-surgical periodontal therapy. J Periodontol 2009;80:1041–1056.
6. Sanz M, Teughels W; Group A of European Workshop on Periodontology. Innovations in non-surgical periodontal therapy: Consensus Report of the Sixth European Workshop on Periodontology. J Clin Periodontol 2008;35(8 Suppl):3–7.
7. Armitage GC. Development of a classification system for periodontal diseases and conditions. Ann Periodontol 1999;4:1–6.
8. Caffesse RG, Sweeney PL, et al. Scaling and root planing with and without periodontal flap surgery. J Clin Periodontol 1986;13:205–210.
9. Cobb CM. Non-surgical pocket therapy: mechanical. Ann Periodontol 1996;1:443–490.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


