4
Regenerative Therapy
Case 1: Treatment of Furcations
Soo-Woo Kim, DMD, MS, and Myron Nevins, DDS
Case 2: Treatment of Intrabony Defects Using Allografts
Kevin Guze, DMD, DMSc
Case 3: Treatment of Intrabony Defects Using Growth Factors
Marc L. Nevins, DMD, MMSc, and Vinicius Souza Rodrigues, DDS
Case 4: Treatment of Intrabony Defects Using Alloplastic Materials
N. Joseph Laborde III, DDS, and Giuseppe Intini, DDS, MS, PhD
Case 5: Guided Bone Regeneration
Kevin Guze, DMD, DMSc
Case 1
Treatment of Furcations
CASE STORY
A 47-year-old Caucasian female was referred for periodontal evaluation of the maxillary posterior teeth by a dental hygienist at the office of a general dentist. She was aware of bleeding when brushing but had no other clinical symptoms. However, the hygienist was concerned by gingival distension and the deep probing depth. The patient reported that she brushed her teeth two to three times a day and flossed regularly.
LEARNING GOALS AND OBJECTIVES
- To be able to diagnose/classify furcation involvement
- To identify possible etiologic factors
- To be aware of appropriate treatments for furcation involvement
Medical History
There was no significant medical history. She was not currently taking any medication except multivitamins, and the patient had no known drug or food allergies.
Review of Systems
- Vital signs on the day of initial visit
- Blood pressure: 128/83 mm Hg
- Pulse: 74 beats/minutes (regular)
- Respiratory rate: 16 breaths/minute
- No significant problems were reported.
Social History
The patient was a well-spoken certified public accountant. She denied any use of tobacco, alcohol, or recreational drugs. There were no parafunctional habits reported such as clenching, bruxism, tongue thrusting, and mouth breathing.
Extraoral Examination
This articulate patient appeared to be under no apparent distress. The extraoral examination revealed no masses, swelling, or lymphadenopathy. Her temporomandibular joints were in full range of motion and within normal limits.
Intraoral Examination
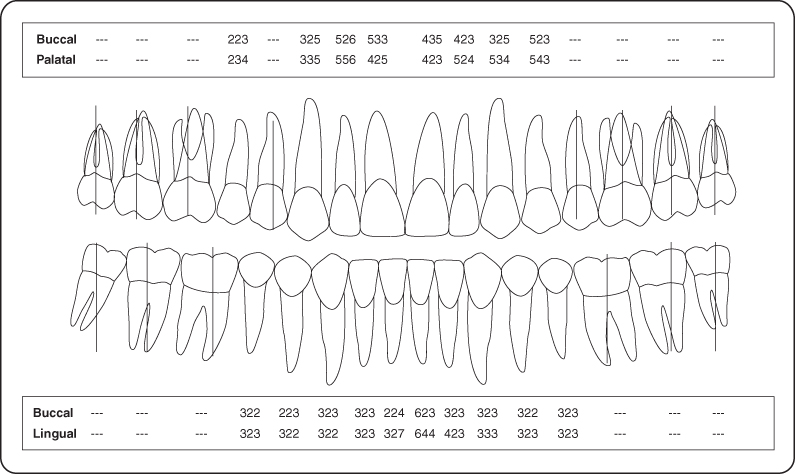
The oral mucosa including lips, tongue, and palate demonstrated no aberrations. The periodontal examination revealed generalized pink gingiva with localized marginal erythema. There was a deviated papilla, and exudate was present at tooth #14. Periodontal charting was completed (Figure 1).
Occlusion
There was a minimal discrepancy between centric relation and centric occlusion but no eccentric interferences.
Figure 1: Periodontal charting.
Diagnosis
Review of the full-mouth radiographic survey demonstrated minimal osseous loss excluding tooth #14, giving a diagnosis of generalized mild chronic periodontitis. A vertical probing depth of 6 mm and horizontal probing depth of 5 mm were present at the site of the buccal furcation on tooth#14. The palatal root prevented further horizontal probing. The nonsurgical diagnosis of this furcation was a class II furcation involvement in the buccal of #14 according to Glickman’s classification.
Treatment Plan
The treatment plan was to address the generalized mild chronic periodontitis with four quadrants of root planning under local anesthesia complemented by oral hygiene instruction. Surgical intervention would be necessary to treat #14 with the hope that periodontal regeneration could be performed. It was impossible to determine mesial and distal extension of furcation involvement until the time of surgery.
Treatment
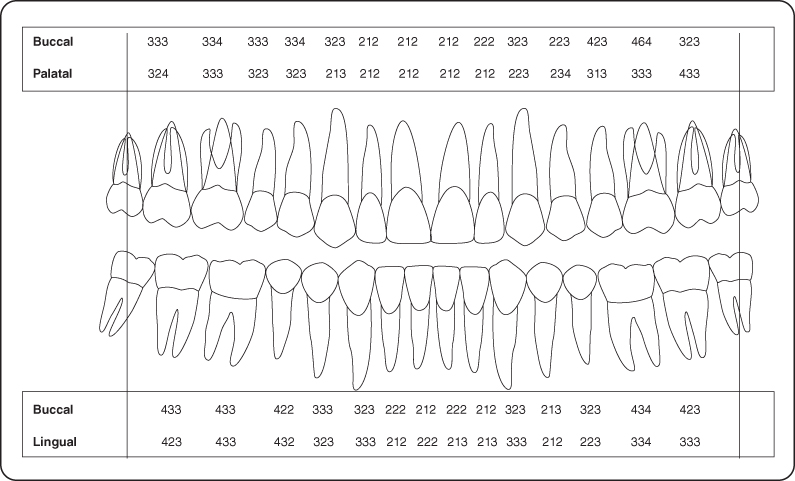
The left maxillary quadrant was anesthetized buccally and palatally around #14 with 2% Xylocaine with 1:50,000 of epinephrine. A full-thickness buccal flap was reflected using mesial and distal vertical incisions to obtain the maximum visibility. All granulation tissue was removed from the affected area, and root planning removed all accretions from the tooth. This was the appropriate time to evaluate any loss of proximal structure between the palatal and two buccal roots. The internal proximal areas were evaluated with a #23 explorer and Kramer Nevins #4 curette, but there was no proximal extension. We therefore concluded that this problem was limited to the buccal furcation and we could treat the area to achieve periodontal regeneration. A tetracycline slurry was introduced to decontaminate the surface. The area was thoroughly washed with sterile saline. Autogenous bone was harvested with a trephine from an edentulous mandibular posterior site. The cores were ground with a bone mill and adapted to the defect. Ti-reinforced membrane (Gore, Newark, DE, USA) was fitted to cover the defect, apically tacked with a fixation screw, and sutured coronally with 4.0 Gore-Tex suture. Then flap was repositioned coronally to protect the surgical site. The area was reopened to remove the membrane at 6 months. There was clinical evidence of periodontal regeneration interradicularly and to a lesser extent on the radicular surface of the buccal roots. The patient was under observation at the periodontal office for a year and returned to the family practice (Figure 2A–D).
Figure 2: (A) Full-thickness flap raised showing furcation involvement; (B) autogenous bone in place; (C) membrane with fixation screw; (D) 6-month postoperative view.
Discussion
The interradicular loss of periodontium of a multirooted tooth is referred to as furcation involvement. The compromised furcation is most successfully treated when its loss of structure is minimal or incipient and is categorized as class I.
The opportunities for successful periodontal regeneration are limited for maxillary molars. The best case scenario is when the buccal furcation between the two buccal roots is involved. If the loss of attachment has continued between the palatal root and the two buccal roots, the most difficult item for the regeneration effort is to eliminate the infected granulation tissues and the accretions on the root surface. Standard curettes are frequently larger than the interradicular space, and many clinicians use ultrasonic instruments as a supplement.
If the procedure only requires the treatment of a buccal furcation, the next decision relates to the selection of the osteogenic materials. This tooth was treated with autogenous bone and a nonresorbable membrane, but there are many products available. They include allografts, xenografts, and alloplasts used with or without a barrier membrane. The only treatment regimen that has satisfied the definition of periodontal regeneration for a furcation defect in the form of human histological evidence is the use of recombinant platelet-derived growth factor (rhPDGF-bb) in combination with an allograft [1,2].
Self-Study Questions
A. How would you classify/define furcation involvement?
B. What information would you need to make a correct diagnosis of furcation involvement?
C. What etiologic and anatomic factors influence furcation involvement?
D. How would you treat different furcation involvements?
E. What are the factors that affect treatment outcome?
F. What is the long-term prognosis of treatment of furcation involvement?
G. What are possible complications from the treatment of furcation involvement?
H. What would be the maintenance protocol after the treatment of furcation involvement?
Answers located at the end of the chapter.
References
1. Camelo M, Nevins M, et al. Periodontal regeneration in human class II furcations using purified recombinant human platelet-derived growth factor-BB (rhPDGF-BB) with bone allograft. Int J Periodontics Restorative Dent 2003;23:213–225.
2. Nevins M, Camelo M, et al. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J Periodontol 2003;74:1282–1292.
3. Glickman I. The treatment of bifurcation and trifurcation involvement. In: Clinical Periodontology. Philadelphia, PA: Saunders, 1958. pp693–704.
4. Hamp SE, Nyman S, et al. Periodontal treatment of multirooted teeth. Results after 5 years. J Clin Periodontol 1975;2:126–135.
5. Eskow RN, Kapin SH. Furcation invasions: correlating a classification system with therapeutic considerations. Part I. Examination, diagnosis, and classification. Compend Contin Educ Dent 1984;5:479–483, 487.
6. Tarnow D, Fletcher P. Classification of the vertical component of furcation involvement. J Periodontol 1984;55:283–284.
7. Ricchetti PA A furcation classification based on pulp chamber-furcation relationships and vertical radiographic bone loss. Int J Periodontics Restorative Dent 1982;2:50–59.
8. Al-Shammari KF, Kazor CE, et al. Molar root anatomy and management of furcation defects. J Clin Periodontol 2001;28:730–740.
9. Zappa U, Grosso L, et al. Clinical furcation diagnoses and interradicular bone defects. J Periodontol 1993;64:219–227.
10. Mealey BL, Neubauer MF, et al. Use of furcal bone sounding to improve accuracy of furcation diagnosis. J Periodontol 1994;65:649–657.
11. Abdallah F, Kon S, et al. The furcation problem: etiology, diagnosis, therapy, and prognosis. J West Soc Periodontol Periodontal Abstr 1987;35:129–141.
12. Larato DC. Furcation involvements: incidence and distribution. J Periodontol 1970;41:499–501.
13. Tal H. Relationship between the depths of furcal defects and alveolar bone loss. J Periodontol 1982;53:631–634.
14. Hermann DW, Gher ME Jr, et al. The potential attachment area of the maxillary first molar. J Periodontol 1983;54:431–434.
15. Bower RC Furcation morphology relative to periodontal treatment. Furcation entrance architecture. J Periodontol 1979;50:23–27.
16. Everett FG, Jump EB, et al. The intermediate bifurcational ridge: a study of the morphology of the bifurcation of the lower first molar. J Dent Res 1958;37:162–169.
17. Gutmann JL Prevalence, location, and patency of accessory canals in the furcation region of permanent molars. J Periodontol 1978;49:21–26.
18. Moskow BS, Canut PM Studies on root enamel (2). Enamel pearls. A review of their morphology, localization, nomenclature, occurrence, classification, histogenesis and incidence. J Clin Periodontol 1990;17:275–281.
19. Masters D, Hoskins SW. Projection of cerival enamel into molar furcations. J Periodontol 1964;35:49–53.
20. Pontoriero R, Lindhe J. Guided tissue regeneration in the treatment of degree II furcations in maxillary molars. J Clin Periodontol 1995;22:756–763.
21. Filipowicz F, Umstott P, et al. Vital root resection in maxillary molar teeth: a longitudinal study. J Endod 1984;10:264–268.
22. Langer B, Stein SD, et al. An evaluation of root resections. A ten-year study. J Periodontol 1981;52:719–722.
23. Newell DH. The role of the prosthodontist in restoring root-resected molars: a study of 70 molar root resections. J Prosthet Dent 1991;65:7–15.
24. Nevins M, Langer B. The successful application of osseointegrated implants to the posterior jaw: a long-term retrospective study. Int J Oral Maxillofac Implants 1993;8:428–432.
25. Bahat O, Handelsman M. Use of wide implants and double implants in the posterior jaw: a clinical report. Int J Oral Maxillofac Implants 1996;11:379–386.
26. Bowers GM, Schallhorn RG, et al. Factors influencing the outcome of regenerative therapy in mandibular Class II furcations: Part I. J Periodontol 2003;74:1255–1268.
27. Mellonig JT, Valderrama Mdel P. Histological and clinical evaluation of recombinant human platelet-derived growth factor combined with beta tricalcium phosphate for the treatment of human class III furcation defects. Int J Periodontics Restorative Dent 2009;29:169–177.
28. Karring T, Cortellini P. Regenerative therapy: furcation defects. Periodontol 2000 1999;19:115–137.
29. Pontoriero R, Nyman S, et al. The angular bony defect in the maintenance of the periodontal patient. J Clin Periodontol 1988;15:200–204.
TAKE-HOME POINTS
A.
a. Glickman’s classification of furcation involvement [3]
i. Grade I: Early furcation involvement just into the flute of the furcation is present, but the interradicular bone is intact. There is no significant destruction of bone or connective tissue in the furcation proper.
ii. Grade II: Distinct horizontal destruction of the furcation area including interradicular bone is present. Destruction should not be extended to the other side of furcation. Vertical bone loss may or may not be present.
iii. Grade III: Destruction of interradicular bone and connective tissue all the way through the furcation so that an instrument can be passed through the furcation. The furcation defect is not yet visible clinically.
iv. Grade IV: Severe destruction of interradicular bone and connective tissue all the way through the furcation so that the tunnel is completely visible through the furcation defect when viewed clinically.
b. Horizontal furcation classification [4]
i. Degree I: Horizontal loss of periodontal tissue support <3 mm.
ii. Degree II: Horizontal loss of support >3 mm but not encompassing the total width of the furcation area.
iii. Degree III: Horizontal “through-and-through” destruction of the periodontal tissue in the furcation.
c. Vertical furcation classification [5,6]
In addition to the horizontal classification:
i. Subclass A: Probeable vertical depth of 1–3 mm from the roof of the furcation apically.
ii. Subclass B: Probeable vertical depth of 4–6 mm from the roof of the furcation apically.
iii. Subclass C: Probeable vertical depth >7 mm from the roof of the furcation apically.
d. Horizontal and vertical furcation classification [7]
Ricchetti’s furcation classification system divides the molar into buccal, middle, and lingual/palatal thirds. The classifications are as follows:
i. Class I: Incipient involvement; horizontal involvement just into the interradicular area.
ii. Class Ia: Involvement into approximately the first half of the buccal or lingual third.
iii. Class II: Horizontal involvement beyond class Ia but not into the middle third of the molar.
iv. Class IIa: Horizontal involvement into the middle third of the molar but not beyond halfway.
v. Class III: Horizontal involvement beyond half of the tooth width.
B. Al-Shammari et al [8]
a. Radiographs: may assist in the diagnosis of furcation defects but have a limited value if used as the sole diagnostic tool, especially in early and moderate defects.
b. Clinical examination: Clinical measurements alone also have limited value [9]. However, the combination of radiographic and clinical examinations improves detection to 65% for maxillary molars but only to 23% for mandibular molars. Probing the deepest interradicular site does not measure the true pocket depth or the attachment level of the furcation area. This indicates that the probing measurement recorded the depth of probe penetrating into the inflamed connective tissue rather than the true pocket depth.
c. Bone sounding: Bone sounding with local anesthesia may assist in the diagnosis of furcation defects by more accurately determining the underlying bony contours [10].
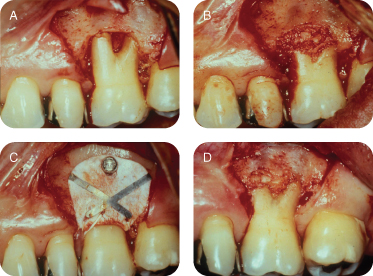
d. Diagnosing furcation invasion is best accomplished using a combination of radiographs, periodontal probing with a curved explorer or Nabers probe, and bone sounding (Figure 3).
Figure 3: Mandibular furcation involvement, showing a probe entering the furcation.
C.
a. Primary factor
i. Bacterial plaque
1. The extension of inflammatory periodontal disease into the furcation area leads to interradicular bone resorption and formation of furcation defects [11].
ii. Age
1. The average number of furcation involvements increases with age [12].
b. Occlusal trauma
i. Glickman showed that the hyperfunction of rat molar makes the furcation area susceptible to attachment loss. The heavy occlusal load on molar teeth may render them susceptible to increased bone loss in the furcation areas if inflammation is present.
c. Predisposing anatomic factors:
i. Root trunk length
1. The distance from the cementoenamel junction (CEJ) to the entrance of the furcation varies by individual. The shorter the trunk length, the less attachment loss needs before the furcation involvement [13].
ii. Root length
1. Teeth with long root trunks and short roots may have lost most of their support by the time the furcation becomes affected [14].
iii. Root form
1. Furcation entrance diameter
a. 81% of entrances on molars are <1 mm in width, and 58% are <0.75 mm, which is less than the diameter of the blade of standard curettes [15]. A small furcation entrance further complicates the proper maintenance of good hygiene.
2. Root concavity [15]
a. Mandibular first molars have concavities in 100% of mesial roots and 99% of distal roots. Deeper concavity is found in the mesial root than the distal root.
b. Maxillary first molars have concavities in 94% of mesiobuccal roots, 31% of distobuccal roots, and 17% of palatal roots. Deepest concavity is found in the mesiobuccal root.
iv. Interradicular dimension
1. Root proximity can be a precipitating factor. Closely proximated or fused roots can impede complete periodontal treatment or maintenance once they get involved.
v. Anatomy of furcation
1. The presence of bifurcational ridges, a concavity in the dome [16], and possible accessory canals [17] can complicate appropriate periodontal treatment and maintenance therapy.
vi. Developmental anomaly
1. Developmental anomalies are enamel pearls (EPs) and cervical enamel projection (CEPs). EPs are larger round deposits of enamel, and CEPs are flat ectopic extensions of enamel beyond the CEJ junction. EPs are seen in 1–5% of permanent molars [18]. CEPs are seen in 29% of mandibular molars and 17% of maxillary molars [19]. These anatomic structures interfere with the attachment apparatus, making furcation more vulnerable to the disease process.
d. Other factors
i. Orthodontics
1. Poorly planned orthodontic procedures may make teeth super-erupt, exposing furcations.
ii. Pulpal pathology
1. When present, especially with accessory canals, it is very likely that furcation would be involved.
iii. Vertical root fractures
1. Vertical root fractures are associated with rapid and localized alveolar bone loss.
iv. Iatrogenic factors
1. Overhanging margin of restorations near furcations can lead to furcation involvement.
D.
a. Class I
i. Limited to the incipient lesions
1. Scaling and root planning in conjunction with debridement
a. Good root preparation is the key to successful therapy. Efficacy decreases with more roots involved. Presence of precipitating anatomic factors also hinders the success of therapy.
2. Apically positioned flap in conjunction with osteoplasty/osteoectomy
a. This provides an environment for improved oral hygiene application with the reduced pocket depth postoperatively.
b. Class II
i. Buccal and lingual mandibular/buccal maxillary furcations
1. Apically positioned flap in conjunction with osteoplasty/osteoectomy
a. A shallow class II mandibular lesion with divergent roots will benefit from this procedure.
2. Guided tissue regeneration (GTR)
a. Combination treatment (debridement + bone graft + membrane) at buccal class II furcations results in decreased probing depth and bone gain and reduces the amount of soft tissue recession above what was accomplished by flap debridement alone. [20].
b. Introduction of growth factor. Use of growth factors has permitted the treatment of many mandibular class III defects and maxillary class II defects with a regenerative approach. Nevins et al [2] used rhPDGF-BB with DFDBA to successfully treat human class II furcation defects.
3. Root resection (see other chapters)
a. Factors that influence the outcome of root resections include (1) the patency of the root canal system, (2) occlusal forces, (3) the length of the edentulous span, and (4) the length, width, and shape of the root.
b. Root resection in conjunction with endodontic therapy is necessary because long-term survival of vital root resection is poor [21].
c. In the short term of 3 or 4 years, this treatment has been highly successful, but after 10 years approximately a third fail. A substantial number of failures were attributed to recurrent periodontal disease, recurrent caries, root fractures, or endodontic failures [22,23].
4. Extraction and implant (see other chapters)
a. This treatment completely removes etiology and successfully restores the function.
b. It is highly predictable with success rate of 99% [24]. Wide implant (diameter >5 mm) also favors successful treatments of furcation involvements [25].
ii. Other proximal furcations
1. Access to proximal maxillary furcation must proceed with individual defect analysis based on surgical access.
iii. It is critical not to allow class II to become class III.
c. Class III furcation
i. Debridement treatment
a. Maintenance and no invasive therapy
ii. Root resection (see other chapters)
iii. Tunneling
a. This treatment will convert grade III and deep grade II furcations into grade IV furcation to improve access for oral hygiene.
b. Success is challenged with interradicular caries with limited access to repair; size of pulp chamber is 2 mm.
iv. Extract and implant (see other chapters)
v. Regeneration with growth factors
a. Studies report favorable results in mandibular class II furcation; less favorable results were found in mandibular class III defects and maxillary class II defects [20,26]. In general, class III has a less favorable outcome than class II.
b. Histologic evidence of regeneration in class III furcation is not available yet. Mellonig et al used rhPDGF-BB with BCP for improving human class III furcation defect [27]. However, predictability remains questionable.
E. Karring and Cortellini [28]
a. Patient factors
i. Oral hygiene: Patient compliance
ii. Smoking
iii. Systemic conditions (diabetics, metabolic disorders, etc.)
b. Defect factors
i. Mandibular versus maxillary teeth
ii. Location of the defect (buccal class II defects have better prognosis [29])
iii. Vertical height of the defect (<3 mm of vertical defect: better prognosis)
c. Technique factors
i. Operators skill (technique sensitive)
ii. Incomplete removal of etiologic factors
iii. Postoperative infection control
iv. Adjunctive systemic antibiotics (minimal benefits?)
F.
a. Depending on diagnostic skills, all of these methods have been clinically demonstrated as efficacious; none are exclusive
G.
a. The patient should be seen with periodicity of 3 months because they already have demonstrated their susceptibility to periodontal disease.
b. Successful treatment resulting in minimal probing depth will benefit both the oral hygienist and the patient.
c. Proximal brush for interproximal furcation.
Case 2
Treatment of Intrabony Defects Using Allografts
CASE STORY
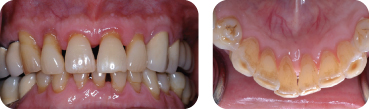
The patient was a 63-year-old male who presented to the Department of Periodontology for consultation regarding several “loose teeth” in both the maxilla and mandible. His chief complaint was his missing teeth and his desire to have fixed restorations to replace them (Figure 1).
Figure 1: Intraoral photo of both maxillary and mandibular teeth in occlusion and occlusal view of maxillary arch.
LEARNING GOALS AND OBJECTIVES
- To identify and appropriately diagnose patients requiring guided tissue regeneration (GTR)
- To understand preoperative and postoperative issues that lead to successful GTR therapy
- To be introduced to surgical techniques and biomaterials used to perform GTR
Medical History
The patient had been diagnosed with type 1 hypertension several years ago and had been under treatment with his physician ever since. His physician had prescribed atenolol 50 mg once daily and hydrochlorothiazide 25 mg daily. He regularly monitored his condition and had been stable since prescription treatment began. Otherwise this patient was in good health, did not have any other significant medical problems, and did not report any allergies or history of diabetes.
Review of Systems
- Vital signs
- Blood pressure: 126/75 mm Hg
- Pulse rate: 58 beats/minute (regular)
Social History
The patient was married with three children. He did not consume alcohol and did not smoke.
Extraoral Examination
No significant findings were noted. The patient had no masses or swelling, and the temporomandibular joints were within normal limits. Figure 2 shows pocket depth measurements after phase 1 therapy.
Figure 2: Probing pocket depth measurements (following phase 1 therapy).
Intraoral Examination
- The soft tissues of the mouth including tongue appeared normal. Oral cancer screen was negative.
- The gingival examination revealed a generalized moderate marginal erythema. Several areas of boggy tissue and mobile teeth were noted.
- A hard tissue examination was completed.
- The edentulous ridge in the maxillary right quadrant had deficiencies bucco-lingually and apico-coronally (Seibert class 3).
- Minimal salivary flow of mucus consistency.
Occlusion
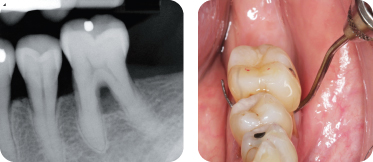
There was a lack of posterior support in the right quadrant and unprotected occlusion. First occlusal contacts were on anterior teeth (Figure 3).
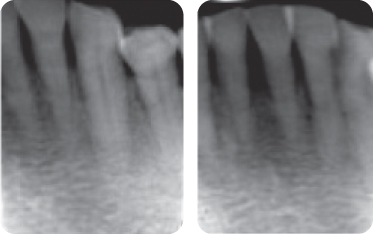
Figure 3: Radiographic evaluation (peri-apicals of the mandibular anterior teeth).
Treatment Plan
The primary care physician was contacted to obtain comprehensive information regarding the patient’s cardiovascular status and to suggest alternative medication to increase salivary flow. Prosthodontic and endodontic consultations were required to establish a comprehensive treatment plan. A cone beam computed tomography scan was ordered for the maxillary and mandibular arch for potential implant placement in edentulous areas.
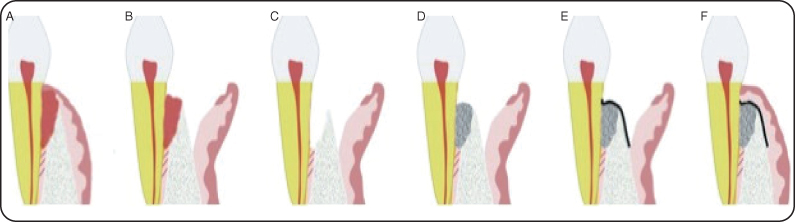
The final treatment plan included extensive periodontic, prosthetic, and occlusal rehabilitation with implant-supported crowns. Guided tissue regeneration (GTR) procedures involved several teeth including tooth #24. Figure 4 illustrates the steps involved in GTR surgery.
Figure 4: (A) Vertical defect evident with granulation tissue fill. (B) Sulcular incision is made and tissue is reflected to expose osseous defect. (C) Complete debridement of defect and root planning of root surface. (D) Defect filled with allograft bone. (E) Membrane placed over grafts and endogenous bone. (F) Primary soft tissue closure is obtained and sutured.
Treatment
The patient received oral hygiene instructions and three complete scaling and root planing sessions prior to surgical therapy. The patient was placed on a permanent saliva substitute program. Pocket depths shown (Figure 2) were recorded following phase 1 therapy.
Preoperative Consultation
The medical history was reviewed. His blood pressure was again monitored. The consent form addressing benefits and risks associated with the procedure was reviewed with the patient. Accessibility to the surgical site was clinically assessed. Significant (Class II) mobility of tooth #24 was noted. The following prescriptions were delivered to the patient: amoxicillin 500 mg (tid for 10 days starting 24 hours before the procedure), ibuprofen 600 mg (every 4–6 hours as needed for pain), and Peridex 0.12% (bid).
Guided Tissue Regeneration Procedure
Bilateral mental nerve blocks and local lingual infiltration were achieved with lidocaine 2% (epinephrine 1/100,000). A full-thickness flap was elevated buccally and lingually from distal of teeth #23 to mesial #27 to achieve access (Figure 5A–D). The teeth in this region were thoroughly scaled and root planed, and the osseous defect was completely degranulated (Figure 5E and F). A rotary finishing bur was used to remove additional debris and conservatively recontour surrounding bone. Following a thorough rinse of the teeth, rehydrated freeze-dried mineralized bone allograft (FDBA) was placed in the defect to the crest of the surrounding bone (Figure 5G). A resorbable collagen membrane was placed over the augmented site (Figure 5H) and the flap was reapproximated to achieve primary closure and then sutured (Figure 5I). The six mandibular anterior teeth were splinted together with orthodontic wire and composite resin to stabilize the mobile tooth #24 (Figure 5J). Healing was uneventful thereafter (Figure 6).
Figure 5: (A–J) Intraoral surgical photos.
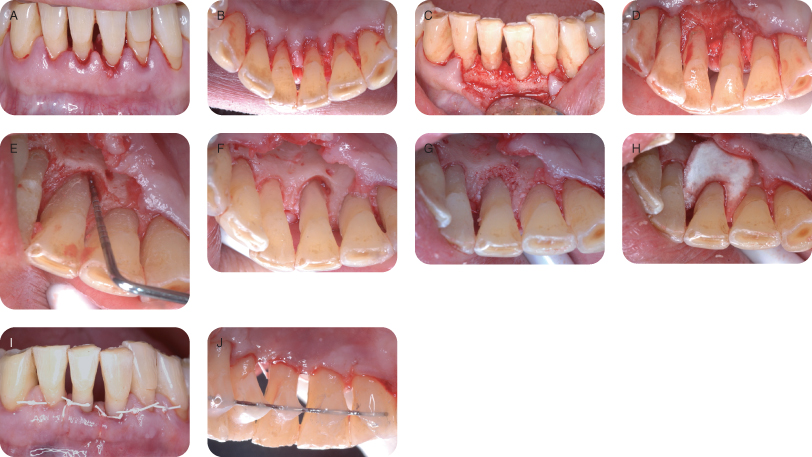
Figure 6: Periapical radiographs (A) initial; (B) following 6 months of healing.
Discussion
GTR is a predictable procedure for regenerating bone adjacent to teeth that was lost due to inflammatory periodontal disease. It is important to understand the limitations of this procedure in order to achieve these results. Successful oral hygiene management as well as appropriate maintenance intervals is an essential part of the treatment. Good communication with the referring general dentist and/or hygienist is necessary in order to provide patients with optimal care.
Although we were unable to change this patient’s blood pressure medication, we were able to provide saliva substitutes that will provide an additional defense against future periodontal breakdown.
The surgery was successful and uneventful. The patient was seen for postoperative visits at 2 weeks to remove the sutures and at 3 and 6 weeks for evaluation. After 6 months the fixed splint was removed from this region and periapical radiographs were taken to assess the amount of bone regenerated (Figure 5G). Tooth #24 was now only slightly mobile compared with the +2 mobility originally recorded. Final occlusal rehabilitation by the prosthodontist following additional periodontic therapy provided the patient with a stable atraumatic occlusion.
Self-Study Questions
A. What is the rationale for performing GTR as opposed to other available surgical techniques?
B. What are the techniques used in GTR therapy?
C. Which biomaterials can be used in GTR? Is there histologic evidence of bone formation?
D. What are the determinants of success in GTR?
E. Do traumatic occlusal events affect the success of treatment with GTR?
F. What are the complications associated with GTR? How do you manage these complications?
Answers located at the end of the chapter.
References
1. Melcher AH. On the repair potential of periodontal tissues. J Periodontol 1976;47:256–260.
2. Becker W, Becker BE, et al. New attachment after treatment with root isolation procedures: Report for treated class III and class II furcations and vertical osseous defects. Int J Periodontics Restorative Dent 1988;8:8–23.
3. Dahlin C, Linde A, et al. Healing of bone defects by guided tissue regeneration. Plast Reconstr Surg 1988;81:672–676.
4. Garrett JS, Crigger M, et al. Effects of citric acid on diseased root surfaces. J Periodontal Res 1978;13:155–163.
5. Stahl SS, Froum SJ, et al. Healing responses of human intraosseous lesions following the use of debridement, grafting and citric acid root treatment. II. Clinical and histologic observations: One year postsurgery. J Periodontol 1983;54:325–338.
6. Albair WB, Cobb CM, et al. Connective tissue attachment to periodontally diseased roots after citric acid demineralization. J Periodontol 1982;53:515–526.
7. Terranova VP, Franzetti LC, Hic S, et al. A biochemical approach to periodontal regeneration: tetracycline treatment of dentin promotes fibroblast adhesion and growth. J Periodontal Res 1986;21:330–337.
8. Blomlof J, Blomlof L, et al. Effect of different concentrations of EDTA on smear removal and collagen exposure in periodontitis-affected root surfaces. J Clin Periodontol 1997;24:534–537.
9. Blomlof J, Jansson L, et al. Root surface etching at neutral pH promotes periodontal healing. J Clin Periodontol 1996;23:50–55.
10. Caffesse RG, Kerry GJ, et al. Clinical evaluation of the use of citric acid and autologous fibronectin in periodontal surgery. J Periodontol 1988;59:565–569.
11. Mariotti A. Efficacy of clinical root surface modifiers in the treatment of periodontal disease. A systematic review. Ann Periodontol 2003;8:205–226.
12. Newman MG, Takei H, et al. Carranza’s Clinical Periodontology. 3rd ed. Maryland Heights, MO: Elsevier/Saunders, 1995.
13. Carranza FA Sr. A technic for reattachment, J Periodontol 1954;25:272.
14. Moskow BS, Tannenbaum P. Enhanced repair and regeneration of periodontal lesions in tetracycline-treated patients: case reports. J Periodontol 1991;62:341.
15. Lindhe J, Karring T, et al. Clinical Periodontology and Implant Dentistry. 4th ed. Hoboken, NJ: Wiley-Blackwell, 2003.
16. Zellin G, Gritli-Linde A, et al. Healing of mandibular defects with different biodegradable and non-biodegradable membranes: an experimental study in rats. Biomaterials 1995;16:601.
17. Friedmann A, Strietzel FP, et al. Histological assessment of augmented jaw bone utilizing a new collagen barrier membrane compared to a standard barrier membrane to protect a granular bone substitute material. Clin Oral Implant Res 2002;13:587.
18. Sandberg E, Dahlin C, et al. Bone regeneration by the osteopromotive technique using bioabsorbable membranes: an experimental study in rats. Int J Oral Maxillofac Surg 1993;51:1106.
19. Hollinger JO, Brekke J, et al. Role of bone substitutes. Clin Orthop 1996;324:55–65.
20. Tolman DE. Reconstructive procedures with endosseous implants in grafted bone: a review of the literature. Int J Oral Maxillofac Implants 1995;10:275–294.
21. Mowlem R. Cancellous chip bone grafts: Report on 75 cases. Lancet 1944;2:746–748.
22. Mellonig JT. Autogenous and allogeneic bone grafts in periodontal therapy. Crit Rev Oral Biol Med 1992;3:333–352.
23. Mulliken JB, Glowacki J. Induced osteogenesis for repair and construction in the craniofacial region. Plast Reconstr Surg 1980;65:553–560.
24. Cammack GV II, Nevins M, Clem DS III, et al. Histologic evaluation of mineralized and demineralized freeze-dried bone allograft for ridge and sinus augmentations. Int J Periodontics Restorative Dent 2005;25:231–237.
25. Simion M, Trisi P, et al. Vertical ridge augmentation using a membrane technique associated with osseointegrated implants. Int J Periodontics Restorative Dent 1994;14:496–511.
26. Cochran DL, Douglas HB. Augmentation of osseous tissue around nonsubmerged />
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


