and cation 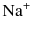 and then forms an equilibrium with hypochlorous acid (HOCl) that is pH dependent (Fig. 5.1). Pure sodium hypochlorite has a pH of around 12 at which
and then forms an equilibrium with hypochlorous acid (HOCl) that is pH dependent (Fig. 5.1). Pure sodium hypochlorite has a pH of around 12 at which 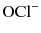 predominates [21]. Both
predominates [21]. Both 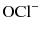 and HOCl are powerful oxidizing agents and are the active agents which exert the antimicrobial activity of sodium hypochlorite. Given it is typically used in its pure form, the high pH of sodium hypochlorite also provides additional tissue dissolving effects [22].
and HOCl are powerful oxidizing agents and are the active agents which exert the antimicrobial activity of sodium hypochlorite. Given it is typically used in its pure form, the high pH of sodium hypochlorite also provides additional tissue dissolving effects [22].

Fig. 5.1
Mechanism of action of sodium hypochlorite (NaOCl)
Sodium hypochlorite is a strong base (pH >11). The hypochlorite anion OCl− can exert its action through non-specific degradation of proteins on cell wall, membranes and within cells. This causes the irreversible inhibition of normal cell metabolism that ultimately results in cell death and cell dissolution [22]. As this action is non-specific, sodium hypochlorite is an effective agent against bacteria, viruses and fungi but it is also active against normal healthy human tissue. This is advantageous when wanting to dissolve and remove pulp tissue but not when this irrigant is extruded beyond the confines of the canal affecting the periapical tissue.
Sodium hypochlorite is commonly used in concentrations between 0.5 and 6 %. Brystöm and Sundqvist studied the effect of irrigation concentration in necrotic root canals. These investigators showed that using 0.5 % or 5 % NaOCl, with or without EDTA for irrigation, resulted in similar reductions in bacterial counts in the canals when compared to normal saline. Nevertheless, it was very difficult to render the canals completely free of bacteria, even after repeated sessions [6, 23]. In contrast Clegg and colleagues using an ex vivo biofilm study, comparing the effectiveness of 3 % and 6 % NaOCl, showed the higher concentration was more effective [24].
Higher concentrations of NaOCl have a better tissue-dissolving ability [25], although lower concentrations when used in high volumes with more frequent intervals to replenish the solution can be equally as effective [26]. The risks of toxicity increase as the concentration increases with any inadvertent extrusion beyond the confines of the root canal system risking serious irritation [27]. Furthermore, a 5.25 % solution significantly decreases the elastic modulus and flexural strength of human dentine compared to physiological saline, whilst a 0.5 % solution does not [28].
An alternative approach to increasing the effectiveness of NaOCl is to increase the temperature of low-concentration NaOCl solutions. The ability of 1 % NaOCl at 45 °C to dissolve human dental pulps was found to be equal to that of 5.25 % solution at 20 °C [29]. Even fast-acting biocides such as NaOCl require sufficient working time to reach their potential and the optimal time it needs to remain in the canal system is an issue yet to be resolved [14]. In summary the concentration of NaOCl is not critical, and the use of 1 % NaOCl can be recommended on the basis of less risk of tissue toxicity provided fresh hypochlorite solution is used and replenished frequently within the canal system.
As with any product used in the medical or dental field, potential toxicity is always an important consideration. Sodium hypochlorite is not genotoxic but has a non-specific cytotoxicity and can cause substantial injury if not used carefully [27, 30]. The same tissue-dissolving property that is desirable within the root canal will be equally applied if inadvertently extruded into the periapical tissues. And even though higher concentrations are more cytotoxic, lower concentrations can still cause tissue irritation and damage [31, 32].
Numerous case reports have been published in the literature of sodium hypochlorite accidents following its use as an endodontic irrigant. Symptoms include immediate severe pain, oedema of neighbouring soft tissue including possible extension to the lip and infra-orbital region, profuse bleeding within the canal, ecchymosis within the skin an mucosa of in the affected area, possible secondary infection and reversible or irreversible damage to vital structures. Inadvertent extrusion of sodium hypochlorite may be the result of open apical foramens due to incomplete root development or apical resorption, iatrogenic or pathologic perforations, lack of control of the irrigation needle depth, needle binding in the canal and use of extreme pressure when irrigating [27].
Extrusion of sodium hypochlorite can result in tissue necrosis to varying degrees depending on the volume and concentration of the irrigant extruded. It is considered to be a self-limiting process but can be extremely traumatic for patients and practitioners alike. Current treatment protocols for NaOCl accidents have been largely determined from numerous case reports. Early recognition is crucial and treatment is typically prescribed on a case-by-case basis including simple palliative care in the form of patient reassurance, pain relief and local anti-inflammatory measures. Antibiotics and steroids may be beneficial as adjunctive therapy and referral for specialist oral surgical management may be needed in severe cases [27] (Fig. 5.11 and Case 5.1).
Shelf life, storage and handling are also important factors that can affect NaOCl and its efficacy. Higher concentrations have been shown to lose their effectiveness faster than lower concentration [29, 33]. Even so they still have a very acceptable shelf life with concentrations over 3 % still easily lasting at least in the range of 3–6 months [21]. In contrast, lower concentrations of 1 % or under can remain stable at room temperature for up to 23 months [34]. This is provided they are stored are stored in appropriate conditions under 30 ° C and in their original high pH form. There is a school of thought that sodium hypochlorite buffered to a lower concentration has a superior antibacterial effectiveness, but this drastically reduces the shelf life of sodium hypochlorite [35].
The primary limitation of sodium hypochlorite is its inability to dissolve inorganic tissue. In the context of endodontic treatment, this translates to an inability to remove the smear layer that is caused during the instrumentation phase of treatment [15–19]. Ethylenediamine tetraacetic acid (EDTA) is a chelating agent used in endodontics for its ability to remove the inorganic component of smear layer (Fig. 5.2). EDTA acts by reacting with the calcium ions of hydroxyapatite to form soluble chelates that can then be rinsed from the root canal system [19]. Scanning electron microscopy has shown that with the use of EDTA, much less smear layer and thus much cleaner canals walls can be achieved than with using sodium hypochlorite alone [36].
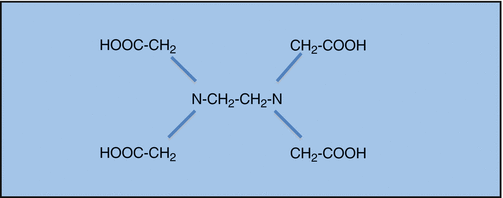

Fig. 5.2
Structural formula of EDTA (ethylenediaminetetraacetic acid)
However, EDTA alone has too many limitations to be used as a sole endodontic irrigant. It has very poor antibacterial properties. When Dunavant and colleagues tested EDTA against an E. faecalis biofilm it only had a 26 % kill rate compared to sodium hypochlorite’s 99 % [37]. Furthermore, while EDTA is very effective at helping dissolve inorganic material it not effective at dissolving organic matter. Thus, it cannot be relied upon for tissue dissolution. For these reasons, EDTA is best considered an adjunctive irrigant to be used in combination with other irrigants to supplement their antimicrobial and tissue dissolution properties.
EDTA is therefore most commonly used in combination with sodium hypochlorite. Indeed, smear layer removal has been shown to be most effective when both EDTA and sodium hypochlorite are used in conjunction [36, 38]. This is not surprising when considering the fact that EDTA will remove the inorganic component of the smear layer followed by NaOCl, which can then remove the exposed residual organic component. However, care must be taken when using EDTA and NaOCl together because it has been shown that NaOCl is deactivated in the presence of EDTA affecting its tissue dissolution ability and probably its antibacterial efficacy as well [39–41]. In contrast EDTA retains it chelating ability in the presence of NaOCl [42].
While it has been established that using sodium hypochlorite and EDTA in combination provides the cleanest canals, the ideal irrigation regime or sequence is a topic of some debate. A range of different combinations has been recommended. However, Goldman and colleagues found that the cleanest canals were achieved by using NaOCl throughout instrumentation and then finishing with a final rinse of EDTA followed by further NaOCl. It was surmised that the NaOCl dealt better with the larger amount of organic tissue that needed dissolving early in the instrumentation phase of treatment and thus reducing the amount of overall smear layer produced [40, 41].
Chlorhexidine digluconate (CHX) is a widely used disinfectant in dentistry because of its good antimicrobial activity. It consists of two symmetric 4-chlorophenyl rings and two biguanide groups connected by a central hexamethylene chain [42]. It is a wide-spectrum antimicrobial agent that is effective against bacteria and yeasts (Candida albicans) [43]. It is able to permeate the cell wall or outer membrane causing disruption of the bacterial cytoplasm, inner membrane or the yeast plasma membrane. Its low toxicity and inherent substantivity, due to its ability to bind to hard tissue [44], has led to its application in endodontics (especially re-treatment cases) as both an intra-canal medicament and irrigant (Fig. 5.3).
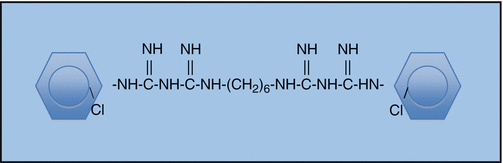

Fig. 5.3
Structural formula of chlorhexidine (CHX)
Numerous “in vitro” studies have evaluated the use of CHX comparing its antibacterial properties to NaOCl. Vianna and colleagues investigated the antimicrobial activity of two forms of CHX (gel and liquid) using three concentrations (0.2, 1 and 2 %) against common endodontic pathogens. The results were compared to those achieved using NaOCl at five concentrations (0.5, 1, 2.5, 4 and 5.25 %) [45]. Contrasting results were found when using a biofilm model. Spratt and colleagues evaluated the effectiveness of 2.25 % NaOCl, 0.2 % CHX, 10 % povidone iodine, 5 parts per million colloidal silver and phosphate buffered solution (PBS) as a control against common monoculture biofilm isolates. They found the most effective irrigants were NaOCl, followed by iodine solution [46].
Unlike NaOCl, CHX has no tissue-dissolving capability so its sole use as an endodontic irrigant cannot be justified. Furthermore its effects on biofilm disruption have been shown to be less effective when compared to NaOCl solution [46].
The mixing of CHX with sodium hypochlorite solution has been shown to produce an orange-brown colour change and the formation of a precipitate. Basrani and colleagues used x-ray photoelectron spectroscopy (XPS) and time of flight secondary ion mass spectrometry (TOF-SIMS) to identify this precipitate. They showed the precipitate contained a significant amount of para-chloroaniline (PCA); a hydrolysis product of CHX [47]. PCA has industrial uses in pesticides and dyes and has been demonstrated to be carcinogenic. This has led to two main concerns, namely, the risk of precipitate leaching into the surrounding periapical tissues and also the occluding of dentinal tubules that could lead to reduced disinfection and/or resin penetration [48, 49]. Use of intermediate flushes of saline or distilled water and drying the canal prior to CHX irrigation may prevent the formation of PCA.
The combination of CHX and EDTA is known to produce a white precipitate. An investigation using reverse-phase high-performance chromatography analysed the precipitate formed when mixing 17 % EDTA and 2 % or 20 % CHX. Based on the results, CHX forms a salt with EDTA rather than undergoing a chemical reaction [50].
Iodine compounds exhibit significant antimicrobial properties against both Gram-positive and Gram-negative bacteria and yeasts (Candida albicans). Iodine potassium iodide (IKI) is the most commonly used iodine compound as an endodontic irrigant as a solution of 2 % iodine in 4 % potassium iodide [51]. Two percent IKI has shown less toxicity and tissue irritation compared to formocresol, camphorated monoparachlorophenol, CHX and sodium hypochlorite [52]. There is a body of opinion that has suggested the use of alternative intra-canal irrigation protocols when undergoing root canal retreatments. The school of thought has been that the causative persistent microbe responsible for failing or failed cases may be more resistant to the common endodontic irrigants used [53, 54]. Safavi and colleagues demonstrated in vitro using human teeth that 2 % IKI treatment for a period of 1–2 h was sufficient to disinfect dentine. In contrast bacteria remained viable in the dentine even after relatively extended periods of calcium hydroxide treatment (24 h) [55]. IKI has no tissue-dissolving capacity, and so its use like CHX may be recommended following completion of chemomechanical preparation as an adjunctive intra-canal medicament or final antiseptic rinse. Use of iodine should be avoided in patients with known allergies.
From a historical perspective, hydrogen peroxide has been used in concentrations ranging from 3 to 5 % alternating with sodium hypochlorite as part of the irrigation protocol. The release of nascent oxygen, which produces effervescence, was thought to help dislodge debris, which could drain out of the canal with the irrigant [56]. Accidental extrusion of hydrogen peroxide beyond the confines of the canal has resulted in sudden, severe pain, swelling, emphysema and crepitus, as reported in the literature [57]. Its use as an endodontic irrigant can therefore no longer be recommended.
Recently several new irrigants have been introduced to the profession in an attempt to further enhance effective debridement of the root canal system and eradicate intra-radicular infections. MTAD (Biopure, Dentsply, Tulsa Dental, Tulsa, OK) has been recommended as an alternative solution to EDTA for removing the smear layer. It is a mixture of tetracycline (3 % doxycycline hyclate), acid (4.25 % citric acid) and detergent (0.5 % polysorbate (Tween) 80) [58]. Tetraclean (Ogna Laboratori Farmaceutici, Muggio, Italy) is another combination product containing doxycycline (50 mg/mL compared to 150 mg/mL in MTAD), an acid and a detergent (polypropylene glycol) [59]. The combination of smear layer removal and the potential ability to exert an antibacterial effect are the main advantages when considering use of this irrigants as a final rinse following disinfection protocols. Disadvantages include tetracycline resistance amongst common root canal isolates, intrinsic staining and potential sensitivity.
The two most commonly used chelators EDTA and citric acid have been shown to react with sodium hypochlorite solution resulting in diminished antimicrobial and tissue-dissolving potential. HEBP (1-hydroxyethylidene-1,1-bisphosphonate) (etidronic acid) is a chelator that has been recommended as an alternative to EDTA that can be used in combination with sodium hypochlorite solution without affecting its proteolytic or antimicrobial properties [40].
QMiX was introduced in 2011 as a novel endodontic irrigant containing EDTA, CHX and a detergent used for smear layer removal and added disinfection [60]. This combination single solution product is available as a clear ready to use solution with no chair-side mixing involved. It has been recommended for use at the end of instrumentation as a final rinse. If NaOCl has been used then saline is used to rinse out any remaining NaOCl prior to its use to prevent potential PCA formation [47–49].
A laser is an acronym for light amplification by stimulated emission of radiation. The radiation involved in generating laser light is non-ionizing and the energy produced can be harnessed for disinfection purposes either thermally or chemically [61]. The neodymium: yttrium-aluminium-garnet (Nd: YAG), erbium: chromium: yttrium-scandium-gallium-garnet (Er: Cr: YSGG) and the erbium: yttrium-aluminium-garnet (Er: YAG) lasers have shown the most promise in endodontics. The Nd: YAG laser emits a wavelength of 1064nm close to the infrared range allowing flexible conductors to be used in narrow and curved canals yielding bactericidal effects both within the root canal walls and deeper dentine layers [62]. The Er: Cr: YSGG laser (developed by Waterlase MD, Biolase Technology, Irvine, CA, USA) is equipped with a 200 μm radially emitting laser tip equivalent to a number 20 file. It emits a wavelength of 2780 nm, which correlates close to the absorption maximum of hydroxyapatite acting through photo ablation. It can be used to remove smear layer and debris from the root canal walls thereby reducing bacterial loads [63]. The Er: YAG (developed by Powerlase, Lares research, Chico, CA, USA) has photon-induced photoacoutstic streaming (PIPS) capability to create short microsecond pulse rates (50μs at a wavelength of 2940 nm). Irrigating solutions can be activated by the transfer of pulsed energy, enhancing the removal of organic tissue and microbes, resulting in improved tubular dentine disinfection [64, 65].
Photo activated disinfection (PAD) also known as light-activated disinfection, photodynamic antimicrobial chemotherapy and photodynamic therapy has been used to target microorganisms in root canals as an adjunct to current endodontic disinfection techniques. Methylene blue and toluidine blue are well-established photosensitizers that have been used in PAD for targeting both gram negative and positive microorganisms. The photosensitizer binds to the surface of the microorganism and following light activation of appropriate wavelength results in the generation of singlet oxygen and free radicals that are cytotoxic to the microbial cell wall [66, 67].
Efficient irrigation that can reach the canal terminus requires not only a suitable irrigant delivery system but one that can also work in a safe manner without causing harm to the patient. Root canal delivery irrigation systems can be broadly classified into two categories: manual and machine-assisted agitation techniques. The most common method of manual passive irrigation involves the use of a 27–30 gauge side-venting needle without binding it on the canal walls. The irrigation solution is dispensed approximately 1mm deeper than the tip of the needle. Depth of needle placement is determined by the size of the canal, canal curvature and corresponding needle size used. One must bear in mind the closer the needle tip is positioned to the apical tissues, the greater the chance of apical extrusion of the irrigant and potential for catastrophic accident (hypochlorite accident) [68, 69].
A 30-gauge irrigation needle covered with a brush (NaviTip FX; Ultradent Products Inc., South Jordan, UT) is available commercially as an adjunct for canal debridement [70]. Inherent friction created when using the brush may risk dislodgement of the radiolucent bristles in the canals.
The method of manual dynamic irrigation is well recognized as a simple method to ensure direct contact of irrigant with canal wall to enhance effectiveness of irrigant action. Gently moving a well-fitting master gutta-percha cone up and down in short 2–3 mm amplitude strokes (manual dynamic irrigation) within an instrumented canal produces a hydrodynamic effect improving irrigant displacement and exchange [71]. A recent study using a collagen “bio-molecular” ex vivo tooth model demonstrated that manual-dynamic irrigation was significantly more effective than an automated-dynamic irrigation system (RinseEndo) [72]. Despite ease of use and no costs involved the routine use of this hand-activated method has been shunned in clinical practice due to the perception of it being too labour intensive by some.
Machine assisted agitation systems have been developed to facilitate debris and smear layer removal. Rotary brushes such as CanalBrush (Coltene Whaledent, Langenau, Germany) has introduced a rotary handpiece-attached micro brush constructed from polypropylene. The micro brush is intended to rotate at 600rpm allowing and has been shown to effectively remove debris from simulated canal extensions and irregularities [73].
Sonic activation operating at low frequencies (1–6 KHz) has been shown to be an effective method for disinfecting root canals. A recently introduced sonically driven canal irrigation system known as the Endoactivator (Dentsply Tulsa Dental Specialities, Tulsa, OK) is commercially available. It consists of a plastic disposable polymer tip available in three different sizes (ISO tip 20, 25 and 30) that are easily attached to a sonic hand piece that can vibrate up to 10,000 cycles per minute [74]. A study has reported that the use of Endoactivator facilitates irrigant penetration and mechanical cleansing compared to conventional irrigation syringes and needles, with no increase in the risk of irrigant extrusion beyond the apex [75].
The use of ultrasonic energy for cleaning root canals and facilitating canal disinfection is well recognized. Ultrasonic files are oscillated at ultrasonic frequencies (25–30 kHz) resulting in characteristic patterns of antinodes and nodes along the length of the file. Passive ultrasonic irrigation whereby the ultrasonic file is vibrated within the canal following canal preparation has been advocated as a means to enhance irrigation without the adverse effect of dentine cutting and iatrogenic damage. Energy transferred from the oscillating file to the irrigant in the root canal system induces acoustic streaming and cavitation of the irrigant [76–78]. Acoustic streaming is defined as the movement of fluid, which occurs as a result of the ultrasound energy creating mechanical pressure changes within the tissues. Cavitation is defined as the formation and collapse of gas and vapour-filled bubbles or cavities in a fluid. Studies have demonstrated the effectiveness of bacterial elimination [79, 80], removal of pulpal tissue and dentine debris removal [81] and removal of smear layer when using passive ultrasonic irrigation [81, 82].
Machine assisted pressure alternation devices have been introduced in a further attempt to overcome the disadvantages of conventional syringes and needle systems. For any irrigation solution to be effective at mechanically removing root canal debris it must reach the apex, create a current, and carry the debris away [68, 69]. Furthermore air entrapment within the confines of the root canal space (apical vapour lock) can theoretically prevent both irrigation exchange and adequate flow.
EndoVac (Discus Dental, Culver City, CA, USA) was introduced as a means of concomitant irrigant delivery and aspiration using a negative pressure-approach lowering the risks of hypochlorite accident and the phenomenon of apical vapour lock [83]. It consists of a disposable syringe, a macro-cannula and a micro-cannula allowing irrigant to be placed in the pulp chamber and the ability for the irrigant to be sucked down the root canal and back up again with minimal risk of apical extrusion. A study by Brito and colleagues comparing NaviTip needles (Ultradent, South Jordon, UT, USA), EndoActivator and the EndoVac system concluded that there was no evidence of antibacterial superiority with any of the techniques used [84].
The RinseEndo system (Dürr Dental Co) is another root canal irrigant device based on a pressure-suction mechanism with approximately 100 cycles per minute [85]. 65 μL of the rinsing solution is oscillated at a frequency of 1.6 Hz and drawn from the attached syringe allowing irrigant transfer to the canal via an adapted cannula. During the suction phase, the used solution and air are extracted from the root canal and automatically merged with fresh rinsing solution thereby ensuring constant irrigant exchange and replenishment. The effectiveness of this system in cleaning canal walls has been challenged using the previously mentioned solubilized collagen-staining model that attempts to stimulate a bacterial biofilm within the canal walls. When comparing RinseEndo to manual-dynamic irrigation (using the pumping action of a master gutta-percha cone) the former proved less effective at removing the stained collagen [72]. Not enough clinical data is available to draw any firm conclusions as to the benefits of using such a system.
In summary irrigation is a key part of successful root canal treatment. It has several important functions including: reducing friction between the instrument and dentine, improving the cutting effectiveness of the files, dissolving organic tissue remnants, cooling both the file and tooth, and furthermore, it has a washing effect and an antimicrobial effect. Irrigation is also the only way to have a positive impact on areas of the root canal wall not touched by mechanical instrumentation. NaOCl is the main irrigating solution used to dissolve organic matter and kill microbes effectively. EDTA is needed as a final rinse to remove the smear layer (see Fig. 5.4). Sterile water or saline may be used between these two main irrigants, however, they must not be the only solutions used. Different means of delivery are used for root canal irrigation, from traditional syringe-needle delivery to various machine-driven systems, including automatic pumps and sonic or ultrasonic energy. In selecting any irrigant and delivery system, consideration must be given to both safety and efficacy [86].
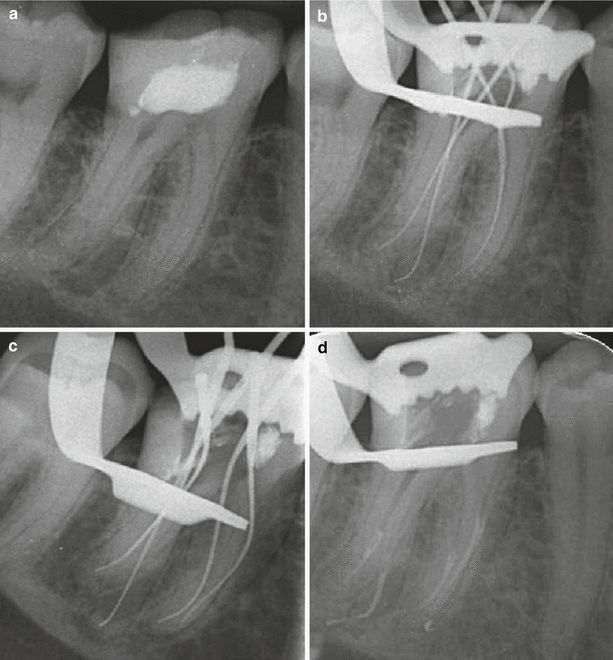
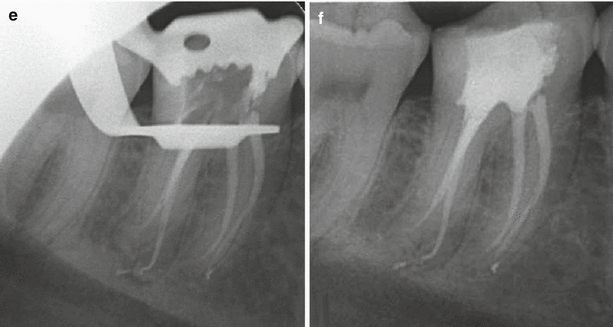


Fig. 5.4
Clinical radiographs demonstrating non-surgical root canal treatment of tooth 46 using 1 % sodium hypochlorite solution and 17 % EDTA solution. Note (a) preoperative radiograph (b) initial apical file radiograph (c) master apical file radiograph (d) mid-fill (e) backfill and (f) post-operative view. Note sealer puffs in the mesial and distal root apices confirming apical patency. Sodium hypochlorite solution functions as the main bactericidal irrigant and allows dissolution of organic tissue and lubrication during preparation. EDTA is an organic acid, which is used to eliminate the inorganic mineral known as the “smear layer”. The inability to effectively remove this layer can allow further thickening and condensing that can potentially close entrances of dentinal tubules, lateral and accessory canals and the main canal. Blockages or both organic or inorganic tissue remnants can be difficult to remove often resulting in incomplete canal preparation that can lead to failure
5.2 Common Endodontic Irrigants
5.2.1 Sodium Hypochlorite
Of all the currently available choices of irrigants, NaOCl appears to be the most ideal, covering most of the requirements for endodontic irrigation. Its ability to dissolve necrotic tissue and the organic components of the smear layer as well as antimicrobial action is of paramount importance when carrying out cleaning and shaping procedures (Fig. 5.5).


Fig. 5.5
Clinical photographs demonstrating sodium hypochlorite tissue-dissolving ability. Note experiment using organic pulp tissue immersed in 1 % NaOCI solution for (a) 1 min, (b) 10 min, (c) 20 min and (d) 30 min, respectively. This crude experiment shows that time is needed for tissue-dissolving capability. Solution replenishment and use of heat will both decrease time taken to dissolve tissue
The antibacterial efficacy and tissue-dissolving capability is a function of its concentration, but so too its toxicity. Typical concentrations used for irrigation purposes range from 0.5 to 6 % depending on preference. It is useful to remember that severe tissue irritations usually occur when using higher concentrations of solution, which is inadvertently forced into the periapical tissues. There is no added antimicrobial advantage when using 0.5 % NaOCl solution or 5 % solution. Replenishment of irrigant is key during treatment procedures with fresh hypochlorite solution continuously replaced.
Increasing the temperature of NaOCl solution also improves the effectiveness of irrigant solution within the canal system with respect to tissue-dissolving capacity. The ability of a 1 % NaOCl at 45 °C to dissolve human pulp tissue is equal to that of 5.25 % at 20 °C.
The most important factor to ensure optimal desired effects when using NaOCl is the factor of time. This is particularly important in regard to the concept of modern preparation techniques whereby the time taken to instrument and shape a canal has reduced significantly. The “chemo” aspect of preparation, nevertheless, requires adequate working time for the irrigant to make contact with the non-instrumented surfaces and effectively dissolve necrotic pulp tissue remnants, organic aspects of the smear layer and exerts optimal antimicrobial effects.
5.2.2 Chlorhexidine
CHX is a potent antiseptic and is commonly used in concentrations ranging from 0.1 to 2 % (the latter is the concentration of root canal irrigation commonly used in the endodontic literature). Its use has been advocated as a final irrigant owing to its substantivity, rather than the main irrigant to be used throughout the endodontic procedure. This is due to its inability to dissolve necrotic tissue remnants. One must also bear in mind that primary endodontic cases are typically a polymicrobial infection predominated by Gram-negative species. CHX has a greater effectiveness against Gram-positive bacteria.
5.2.3 Iodine Potassium Iodide
The endodontic literature has shown that 2 % IKI has been proven to be beneficial in some resistant failing cases. The use of iodine must be cautioned particularly in patients with known allergy. Like CHX it has no tissue-dissolving capability and so its use may be beneficial as part of a final irrigation protocol following chemomechanical preparation with NaOCl solution.
5.3 Removal of Smear Layer
Instrumentation procedures that make contact with the root canal walls (using hand or rotary instruments, ultrasonic tips and burs) can produce a 0.5–2 μ thick smear layer consisting of organic (pulp tissue remnants, bacteria and biofilm) and inorganic (mineralized dentine and predentine particles) components. The adjunctive use of chelating agents such as EDTA has therefore been recommended. Removal of smear layer may allow for disruption of biofilm plaques that are adherent to canal walls and opening of dentinal tubules that further optimizes either antibacterial irrigation penetration or intra-canal medicaments which may diffuse further without constraints.
The recommended protocol for smear layer removal is the use of NaOCl followed by EDTA or citric acid. One to two minutes should be sufficient working time for EDTA action prior to inactivation with saline.
EDTA should be used in liquid form and typical concentrations for endodontic use range from 15 to 17 %. The clinician must be aware of the fact that EDTA in the presence of NaOCl solution reduces the available chlorine in solution, rendering NaOCl less effective with regards to both tissue-dissolving capability and antimicrobial activity. This is the very reason why the use of chelating agents in paste form which were once recommended during the canal shaping procedure is no longer recommended.
5.4 Alcohol
The use of 95 % ethyl alcohol has been recommended as a pre-irrigation step or final irrigation step prior to intra-canal medicament placement or obturation of the canal. This is based on anecdotal practice with the assumption that the alcohol reduces the surface tension of either the irrigant fluid to be used thereafter or sealer during obturation. The evaporation of the alcohol is thought to aid either irrigant or sealer penetration. A final rinse of approximately 3–5 ml of 95 % Ethyl alcohol can be recommended in order to improve the sealing ability of the root canal filling.
5.5 Irrigant Interactions
5.5.1 Sodium Hypochlorite and EDTA
The combination of NaOCl and EDTA results in EDTA retaining its calcium-complexing ability with a reduced amount of chlorine available in solution. NaOCl on the other hand has reduced tissue-dissolving capability and antimicrobial effectiveness. When using NaOCl and EDTA, an alternating regime should be used with copious amounts of the former to ensure that any remnants of EDTA have been removed.
5.5.2 Sodium Hypochlorite and Chlorhexidine
The combination of NaOCl and CHX results in the formation of an orange-brown precipitate consisting possibly of para-chloroaniline (PCA). The amount of PCA formed is directly linked to the increasing concentration of NaOCl. Due to the potential toxicity of PCA, the ability of the precipitate to occlude the dentinal tubules and potential for the insoluble precipitate to interfere with the final seal of the root filling caution should be excised when irrigating with both of these solutions.
Clinical recommendations to avoid precipitate formation include the use of absolute alcohol, saline or distilled water following use of NaOCl and prior to the introduction of CHX within the root canal. An alternative is to use QMiX that does not appear to form any precipitate in the presence of NaOCl solution. Nevertheless manufacturers recommend use of a saline irrigation prior to QMiX use.
5.5.3 EDTA and Chlorhexidine
The combination of EDTA and CHX results in the formation of a white precipitate due to the chlorhexidine forming a salt with EDTA rather than undergoing a chemical reaction. Clinical recommendation will be to flush out the EDTA with saline after recommended working time (usually 1–3 min) prior to introduction of CHX as a final rinse (Fig. 5.6).


Fig. 5.6




Clinical photographs demonstrating potential irrigant interactions. Note (a) sodium hypochlorite solution (NaOCI) 1 %. (b) 1 % NaOCI solution and chlorhexidine 2 % (CHX) mix. (c) NaOCI and CHX after 5 min. (d) NaOCI and CHX after 10 min. (e) NaOCI and CHX after 20 min. Orange-brown precipitate possibly consisting of para-chloroaniline (PCA). (f) The combination of ethylenediaminetetraacetic acid (EDTA) and CHX results in the formation of a white precipitate due to the CHX forming a salt with EDTA rather than undergoing a chemical reaction
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


