Fig. 5.1
Patient subjected to a professional in-office tooth bleaching therapy, which can be esthetically regarded as a success. However, the patient reported postbleaching sensitivity in the anterior teeth (Images provided by Dr. Heraldo Riehl) (a) Clinical condition before bleaching therapy, (b) In-office bleaching therapy with a 35% H2O2 gel, (c) Post-bleaching clinical condition.
Recent clinical studies have demonstrated that the prevalence and intensity of tooth sensitivity is restricted to anterior teeth and proportional to the tooth size, with incisors being the most susceptible teeth (de Almeida et al. 2012; Bonafé et al. 2013). Therefore, the toxic potential of traditional in-office therapy on pulp cells has been the focus of a number of preliminary studies carried out by our research group. When a 35 % H2O2 gel was applied for three consecutive periods of 15 min onto 3.5-mm thick enamel/dentin discs (simulating upper central incisors) adapted to artificial pulp chambers (APC), gel components that diffused through dental structure (extract) caused toxic effects to pulp cells. In such laboratorial studies the extracts were applied to the cells for 1 or 24 h. After a 1-h contact time, the odontoblast-like cells, an immortalized cell lineage from rat dental papillae, featured 50–60 % of cell viability reduction (Soares et al. 2013a, b; Duque et al. 2014; de Almeida et al. 2015; Soares et al. 2015a). Almost 100 % of cell viability reduction was observed after 24-h exposure of pulp cells to these extracts (Coldebella et al. 2009; Trindade et al. 2009). Cell morphology was completely disturbed, with cells showing apoptotic body-like structures, as shown on Fig. 5.2. Human dental pulp cells exposed to the bleaching gel extracts were even more vulnerable, corroborating data previously reported in the literature. Around 97 % of cell viability reduction was observed in human pulp cells after only 1-h contact time. The intensity of the negative effects caused by the 35 % H2O2 bleaching gel on pulp cells was proportional to their contact time with dental structure, which was directly related to the amount of H2O2 capable of diffusing through enamel/dentin discs (Soares et al. 2013a, b, 2015a; Duque et al. 2014). Also, association of this in-office bleaching technique with light sources increased significantly the in vitro cytotoxicity (Dias Ribeiro et al. 2009), since this procedure leads to more intense H2O2 diffusion through tooth structure, as previously demonstrated (Camargo et al. 2009).
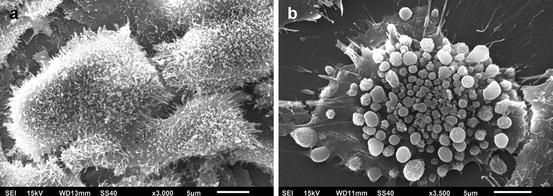

Fig. 5.2
(a) Scanning electron micrograph showing the morphology of normal odontoblast-like MDPC-23 cells. Original magnification = ×3,000. (b) Scanning electron micrograph showing a MDPC-23 cell exposed for 1 h to the extract obtained after simulated traditional in-office bleaching therapy. Original magnification = ×3,500
According to our data, pulp cell viability reduction after tooth bleaching seems to be associated with cell membrane rupture and oxidative stress generation, in a time/concentration dependent fashion. In fact, these effects are proportional to the H2O2 dosage capable of reaching the cells, with high concentrations leading to cell death by necrosis following two pathways: (1) direct contact with free radicals from extracellular H2O2 decomposition, leading to cell membrane rupture; (2) the onset of a pathologic oxidative stress condition after H2O2 diffusion through cell membrane and further decomposition into free radicals on cytoplasm, culminating with lipid peroxidation (Soares et al. 2014a, 2015c).
Since the amount of H2O2 capable of reaching cells after trans-enamel and trans-dentinal diffusion is the main pathway for bleaching-induced cell toxicity, reducing this phenomenon is highly required for minimizing the oxidative damage on pulp cells, turning bleaching into a safe procedure compatible with pulp health. More recent data collected by our research group determined that decreasing the H2O2 concentration in the bleaching gel by 50 %, and applying the gel from 5 to 45 min on enamel, may decrease 11.3- to 4.5-fold, respectively, the toxicity of in-office tooth bleaching to pulp cells compared with simulated traditional therapy (35 %-H2O2 3 × 15 min) (Soares et al. 2014a). These alternative protocols using a 17.5 % H2O2 gel allowed such cells to overcome the low initial oxidative damage and feature a regenerative phenotype through time (Soares et al. 2014a, 2015b).
According to our data, the degree of disturbance on the expression of odontoblastic markers (ALP, DSPP, DMP-1, and mineralized nodule deposition) after tooth bleaching, was related to the oxidative stress intensity and to the overexpression of IL-1β, TNF-α, IL-6, and COX-2 on remaining cells (Soares et al. 2015b, 2016a). It has been demonstrated that both oxidative stress intensity and pro-inflammatory mediator dosage have direct relationship on pulp tissue regeneration capability. Exposure of human dental pulp cells to high doses of pro-inflammatory mediators, such as TNF-α and IL1-β, may negatively interfere with their odontoblastic markers expression and mineralization rate, whereas low dosages seem to have an opposite effect (Min et al. 2006; Paula-Silva et al. 2009; Alongi et al. 2010). Nevertheless, it was demonstrated that human dental pulp cells under intense oxidative stress triggered by long-term exposure to H2O2 had the expression of DSPP and DMP-1 mRNA downregulated associated with no mineralized matrix deposition. Conversely, the treatment of these cells with nontoxic concentrations of H2O2 enhanced ALP activity and calcified nodule deposition as well as increased DSPP, OPN, and OCN expression (Lee et al. 2006, 2013; Min et al. 2008; Matsui et al. 2009).
Therefore, the lower the concentration of H2O2 in contact with pulp cells after tooth bleaching, the higher the regenerative capability of human pulp tissue to overcome the oxidative damage.
With this in mind, future studies should focus on strategies for reducing the amount of residual H2O2 and other free radicals capable of diffusing through the dental structure to reach the pulp chamber, since these toxic molecules play a central role on pulp tissue damage.
Recent data obtained in our lab demonstrated that bleaching gels containing 8–10 % H2O2 minimize significantly the initial toxicity on human dental pulp cells (Soares et al. 2015a). Also, we observed that these bleaching gels are capable of promoting the same color alteration on thin enamel/dentin discs as high concentrated gels applied on thick discs (unpublished data). Therefore, tailoring the bleaching regimen to the tooth size appears to be an interesting alternative to achieve an effective, safe, and biocompatible in-office bleaching technique.
Therefore, tailoring the bleaching regimen to the tooth size appears to be an interesting alternative to achieve an effective, safe, and biocompatible in-office bleaching technique.
5.3 Effects of At-Home Bleaching on Pulp Cells In Vitro
The at-home bleaching therapy with 10 % carbamide peroxide (CP) agents has been considered the safest method for tooth bleaching since minimal clinical adverse effects have been reported in literature (Boushell et al. 2012). The scientific data obtained in recent years by our research group has demonstrated that the application of 10 % CP gel from 1.5 to 8 h onto dental structure does not result in significant toxic effects on both odontoblast-like cells and human dental pulp cells (Soares et al. 2011; Lima et al. 2013; Duque et al. 2014; Almeida et al. 2015). According to our data, application of 10 % CP gel for 4 h onto 3.5 mm enamel/dentin discs results in 16 times less intense H2O2 diffusion in comparison to the traditional in-office bleaching protocol, i.e., 35 % H2O2 gel 3 × 15 min (Duque et al. 2014). Nevertheless, higher CP concentrations (15–22 %) are available for at-home tooth bleaching, with the appeal of promoting a faster bleaching outcome.
Data collected from our group showed that increasing CP concentration from 10 to 16 % increases the toxicity of this treatment to pulp cells (Soares et al. 2011). Taking into consideration that clinical studies have demonstrated that higher concentrations of CP gels cause the same bleaching outcome as that achieved by using 10 % CP gel (Matis 2003; Meireles et al. 2010, Basting et al. 2012), the use of higher concentration products has no advantage and may be more toxic to pulp tissue.
H2O2-based at-home bleaching products are also available. We have observed that the application of 6 % H2O2 gels from 45 min to 1.5 h have no toxic effect on odontoblast-like cells, instead the amount of H2O2 capable of diffusing through dental structure and interact with the cells was twice as high as that of the 10 % CP gel (Almeida et al. 2015). Also, a 10 % H2O2 whitening strip (WS) applied for 1 h on tooth structure did not cause trans-enamel and trans-dentinal cytotoxicity to this same pulp cell lineage. The WS resulted in about 13 times lower H2O2 diffusion than that observed for traditional in-office bleaching therapy with the 35 % H2O2 gel (Soares et al. 2013b). Despite the interesting results obtained with WS, this treatment is currently carried out without direct professional supervision. Therefore, there is a risk of overuse associated with this over-the-counter at-home bleaching technique, which, in turn, may cause higher toxic effects to pulp cells. According to our data, the toxic effect of the WS and CP was time dependent; the toxicity of these products increases significantly after 1-h daily application during 5 consecutive days (Lima et al. 2013; Soares et al. 2013b).
The main concern regarding at-home bleaching relies on the fact that it is a patient-applied therapy; therefore, there is a risk of gel application on exposed dentin areas, craze lines, and restoration interfaces that may increase H2O2 diffusion through hard tooth structures. Also, the inappropriate use of the tray may result in gel overflow, with extended soft-tissue exposure and likely material ingestion. Therefore, it is imperative that this treatment is performed under professional supervision. Also, the professional should perform a detailed clinical evaluation before prescribing this therapy.
5.4 Histopathological Analysis of Human Dental Pulp After In-Office Bleaching
Based on data collected in in vitro studies, it is believed that the presence of high concentrations of H2O2 within the pulp chamber is responsible for the intense oxidative stress condition that causes cell membrane disruption associated with pulp cell death by necrosis (Soares et al. 2014a, 2015b, c, 2016a). This kind of cytotoxic effect also promotes extensive damage on the neighboring tissue, since lysosome enzymes and other toxic substances are leached from dying cells, causing a ripple effect. Consequently, acute inflammatory reaction is expected with expression of a plethora of pro-inflammatory cytokines and chemokines, followed by expression of hyperalgesia mediators, such as prostaglandins (Cooper et al. 2010; Markowitz 2010; Pashley 2013; Cooper et al. 2014). These cellular events have been correlated with the clinical symptoms of tooth sensitivity. In order to detect these events on human pulp tissue, our group associated with other research groups around the world has conducted histopathological analysis on human teeth subjected to professional in-office bleaching therapies. In many of these studies, sound premolars and mandibular incisors scheduled to be extracted for orthodontic reasons were selected, after respective approval by Ethical Committees. Therefore, the volunteers (or legal guardians for patients below 18 years of age) received all necessary explanations including the experiment rationale, the clinical procedures to be performed and possible risks, and signed a consent form explaining the research protocol.
When a bleaching gel with high concentration of H2O2 (38 %) was applied for three times of 10 min each in sound mandibular incisors from young patients (age mean 16.2 years), intense pulpal damage occurred in about 75 % of samples. Pulp necrosis was observed in a wide area of coronal pulp tissue. Deposition of reactionary dentin in part of the coronal and root pulp tissue associated to mild local inflammatory response was also detected in bleached incisors (Fig. 5.3).
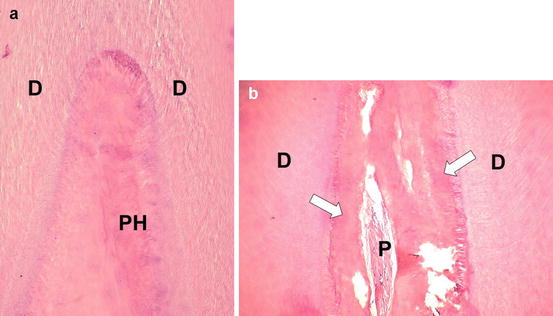

Fig. 5.3
(a) Pulp horn (PH) of a human lower incisor subjected to a professional in-office bleaching with 38 % H2O2. Note the large area of necrosis. H/E, ×64. (b ) Intense deposition of tertiary dentin is observed (arrows) in the radicular pulp chamber, in which a small area of residual pulp (P) tissue with inflammation can be seen. H/E, ×64. (D dentin)
On the other hand, pulps of premolars subjected to the same bleaching protocol with the 38 % H2O2 gel showed the following histopathological features: tubular dentin and predentin, intact odontoblastic layer, and cell-free zone and cell-rich zone, such as observed in control groups (nonbleached incisors and premolars) (control teeth – Fig. 5.4).
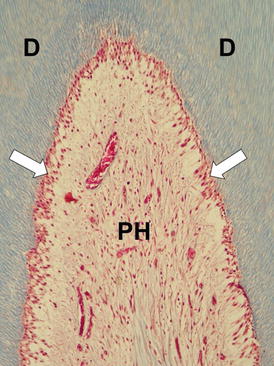

Fig. 5.4
Pulp horn (PH) of a human tooth exhibiting normal histological features. Note the odontoblasts (arrows) underlying the tubular dentin (D) and the subjacent cell-free zone. A number of small blood vessels among collagen fibers and fibroblasts can be observed. Masson’s Trichrome, ×125
In this experiment, the mean of dentin thickness was 1.82 ± 0.08 mm for bleached incisors and 3.10 ± 0.11 mm for bleached premolars. Only patients who had their incisors bleached reported tooth sensitivity (de Souza Costa et al. 2010). Similar results were found when sound mandibular incisors (mean age 18.2 years) were bleached with a 35 %-H2O2 gel applied on enamel for 3 consecutive periods of 15 min each or 1 period of 45 min. Similar pulp tissue responses were found for both tested protocols with the 35 %-H2O2 gel. The majority of bleached incisors (80 %) exhibited large zone of coagulation necrosis in the coronal pulp tissue associated to mild/moderate inflammatory response on surrounding tissue. Tertiary dentin adjacent to the necrotic tissue was observed in 25 % of those teeth, associated with reactionary dentin in the lateral walls of the coronal and root pulp chambers (Fig. 5.5). Moderate deposition of reactionary dentin with no coronal pulp necrosis occurred in only 20 % of samples, which exhibited mild inflammatory pulp reaction. All patients subjected to bleaching protocol reported tooth sensitivity (Roderjan et al. 2015).
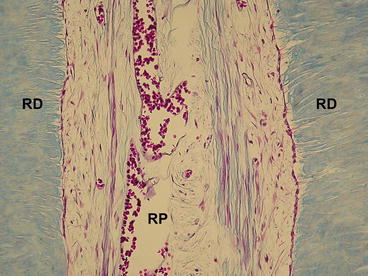

Fig. 5.5
Radicular pulp (RP) tissue of a bleached human lower incisor. Note the lateral deposition of reactionary dentin (RD), which exhibits a reduced number of tubules with cytoplasm processes within them. Masson’s Trichrome, ×180
These data corroborate those obtained from clinical investigations, in which the authors demonstrated that tooth sensitivity is restricted to anterior teeth subjected to in-office bleaching (de Almeida et al. 2012; Bonafé et al. 2013). According to these authors, teeth bleached with 35 %-H2O2 gel applied on enamel for 3 consecutive periods of 15 min each had tooth sensitivity in 76.6 % of lateral incisors, 53.3 % of central incisors, and 30 % of canines, with no discomfort reports for premolars. Thus, one can conclude that enamel/dentin thickness plays an important role in H2O2 diffusion across hard tooth structures to reach pulp chamber, causing tissue damage and postbleaching sensitivity, which are more intense in anterior than in posterior teeth.
Another investigation showed that partial pulp necrosis occurred in 60 % of mandibular incisors of elderly patients (54–62 years old; mean 58.2 ± 4.3) in comparison with 100 % of young patients (18–30 years old; mean 20.1 ± 4.3) that were bleached using the same in-office bleaching therapy. Additionally, areas of pulp necrosis were larger in young teeth. The main dentin thickness on young and elderly patients was 1.77 ± 0.08 mm and 1.99 ± 0.10 mm, respectively. As observed in the previous study, the histological sections evidenced a number of differentiated odontoblast-like cells that deposited a layer of reparative dentin below the necrotic area. These pulp responses against bleaching products are similar to those observed after calcium hydroxide application on pulps of sound teeth mechanically exposed (Roderjan et al. 2014). Therefore, it seems that even in human pulps strongly damaged by in-office tooth bleaching procedures, the pulp cells subjacent to the necrotic tissue are capable of maintaining their phenotype, confirming the data collected in previous in vitro studies carried out by our research group.
Based on these data, tooth sensitivity experienced by patients subjected to in-office bleaching with 35–38 % H2O2 may be associated with inflammatory reaction caused by oxidizing toxic compounds from bleaching gels capable of reaching the pulp chamber, producing an intense chemical irritation of pulp cells. Because of the fact that in-office tooth bleaching causes pulp damage, the release of cell-derived factors, such as prostaglandins, would excite or sensitize pulpal nociceptors. Additionally, we believe that the fluid shifts that occur in dentinal tubules due to vasodilation and increased pulp pressure during local tissue inflammation may trigger impulses in the intradentinal pulpal nerve fiber endings, causing the intense tooth sensitivity that has been claimed by patients subjected to this professional in-office therapy. However, further studies are needed to assess in detail this topic.
5.5 Tooth/Restoration Interface as a Pathway for Bleaching Inducing Toxicity
Recent clinical trials showed that the intensity of tooth sensitivity is increased when traditional in-office tooth bleaching is performed in anterior teeth that have adhesive restorations with no clinical sign of margins degradation (Bonafé et al. 2013). It seems that the presence of restorations in tooth to be bleached may enhance H2O2 diffusion into the pulp chamber. In addition, dental materials used to restore dental cavities interfere significantly with this H2O2 diffusion through enamel and dentin (Gökay et al. 2000, 2004; Benetti et al. 2004; Camargo et al. 2007). Therefore, as several restorative materials and bleaching protocols are available in clinical practice, the question related to the safety of tooth bleaching performed in restored teeth remains a concern (Fig. 5.6).
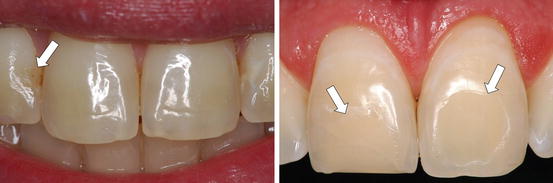

Fig. 5.6
Anterior teeth with resin composite restorations (arrows) that were selected by a clinician for professional bleaching (Images provided by Dr. Heraldo Riehl)
Previous studies demonstrated that different adhesive systems have variable degrees of susceptibility to H2O2, as follows: one-step self-etch > two-step self-etch > etch-and-rinse systems (Van Landuyt et al. 2009; Didier et al. 2013; Dudek et al. 2013; Roubickova et al. 2013). According to the results collected in our laboratory, self-etch adhesive interfaces act as a pathway for H2O2 diffusion from tooth surface into pulp chamber, increasing significantly the toxicity of a 35 %-H2O2 gel on pulp cells (Soares et al. 2015d). Nevertheless, no significant difference concerning cytotoxicity and trans-enamel and trans-dentinal diffusion of H2O2 was observed when etch-and-rinse adhesive restorations where bleached with a 20 or 35 % H2O2 gels for 45 min (Soares et al. 2014c). One can conclude that the compromised bond performance of some self-etch adhesives to enamel and dentin creates a more permeable tooth/restoration interface, facilitating H2O2 diffusion through dental structure. Resin-modified glass-ionomer cements (RMGIC) interface seem to present the same susceptibility to H2O2 as self-etch adhesive systems. The shear bond strength of RMGIC to tooth structure is significantly lower than those observed for etch-and-rinse adhesive systems, due to the low cohesive strength of GIC (Marquezan et al. 2011; Bonifácio et al. 2012; Nujella et al. 2012). According to our results, applying a 35 %-H2O2 gel onto enamel/dentin discs containing RMGIC interfaces subjected to hydrolytic degradation allowed for a more intense H2O2 diffusion through hard dental structures, as well as increased the in vitro cytotoxicity to odontoblast-like cells, leading to increased oxidative stress and IL-1β overexpression as well as disturbing the odontoblastic markers expression (Soares et al. 2016b).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


