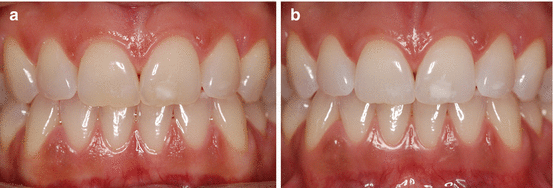
Fig. 6.1
Retracted view of a 28-year-old patient who bleached his maxillary teeth with 10 % carbamide peroxide gel (Opalescence 10 %, Ultradent Products, Inc.) in a custom-fitted tray overnight for 3 weeks. In the first appointment patient had stated that he did not want to whiten the mandibular teeth. He later decided to have the lower teeth bleached with 10 % carbamide peroxide in a custom-fitted tray
- 1.
Dentist-administered in-office whitening followed by at-home whitening with a peroxide-based gel in a custom-fitted tray, also known as jump-start technique. In-office bleaching is performed first by a dental professional to provide an initial jump-start bleaching effect. Then, the patient is prescribed at-home whitening (Kugel et al. 1997), usually 10–20 % carbamide peroxide gel for daily application, which is to be used until the desired shade is obtained (Deliperi et al. 2004).
- 2.
Dentist-administered, custom-fitted bleaching tray containing a higher concentration of carbamide peroxide (usually ≈ 35 %), also known as waiting-room whitening. Patient wears the tray filled with carbamide peroxide gel for periods of 30 min to 1 h while waiting in the dental office.
- 3.
Over-the-counter (OTC) tooth-whitening products for home use without professional supervision, such as gels, rinses, paint-on films, strips (Fig. 6.2), and kits with prefabricated “thermoforming” or “thermofitting” trays that can be molded in hot water by the user at home (please see Chaps. 1 and 4 for more information on OTC whitening products). Dual-arch trays prefilled with a silicone material, from which the patient can make a custom-fitted prosthesis, are available online from auction sites.
 Fig. 6.2Frontal view of a 38-year-old patient 5 min after she applied a bleaching strip to her maxillary teeth
Fig. 6.2Frontal view of a 38-year-old patient 5 min after she applied a bleaching strip to her maxillary teeth
OTC tooth-whitening products were not included in the list of accepted whitening agents by the now extinct American Dental Association Seal of Acceptance Program. Some toothpastes are also marketed as having a whitening effect. They typically contain an abrasive to remove and/or prevent surface stains, such as hydrated silica, calcium carbonate, dicalcium phosphate, dihydrate, calcium pyrophosphate, alumina, perlite, and sodium bicarbonate (Joiner 2010). The inclusion of peroxides in toothpaste is much more challenging in terms of formulation and the short exposure time. A toothpaste with 0.5 % calcium peroxide has been shown to reduce natural extrinsic stain after 6 weeks (Ayad et al. 1999). In the European Union, the maximum concentration of peroxide allowed in toothpastes and mouth rinses is 0.1 % (European Commission Scientific Committee on Consumer Products 2007). According to the same document, most clinical studies with peroxide-containing toothpastes are sponsored by the respective manufacturers and rarely published.
Indications for at-home whitening with carbamide peroxide gel in a custom-fitted tray:
-
Tetracycline staining, especially degrees I and II (Jordan and Boksman 1984) (Fig. 6.3), which is discussed in Sect. 6.4.4.2.
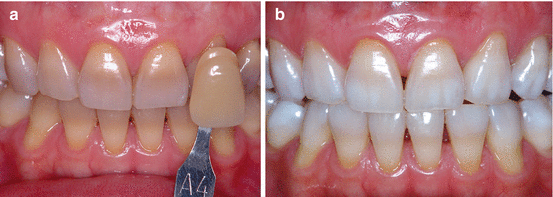 Fig. 6.3(a) A 23-year-old patient with a history of antibiotic intake when she was a child. Although patient did not recall which type of antibiotic she had been prescribed, the clinical exam suggested that the discoloration was compatible with tetracycline staining. (b) Aspect after 4 months of at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) overnight in a custom-fitted tray. Patient returned to clinic for monthly recalls. She did not experience any sensitivity or any other side effects
Fig. 6.3(a) A 23-year-old patient with a history of antibiotic intake when she was a child. Although patient did not recall which type of antibiotic she had been prescribed, the clinical exam suggested that the discoloration was compatible with tetracycline staining. (b) Aspect after 4 months of at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) overnight in a custom-fitted tray. Patient returned to clinic for monthly recalls. She did not experience any sensitivity or any other side effects -
Yellow/brown stains from enamel fluorosis (Fig. 6.4) or from idiopathic causes, which are discussed in more detail in Sect. 6.4.4.3. Clinical solutions related to this topic can be found in Chaps. 12, 13, and 15.
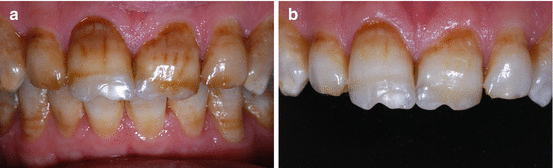 Fig. 6.4(a) This 38-year-old patient was born and raised overseas in an “area where everybody had brown teeth”. He used to drink water from a water well when he was a child in his home country. According to the patient’s account, all his siblings who lived in the same area had “brown teeth.” The medical history revealed no significant findings. After an intra-oral exam, patient was informed that at-home whitening might improve the appearance of his teeth as long as he understood that the treatment could span over a few months. Patient agreed to have his teeth whitened with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) at-home in a custom-fitted tray. Patient was scheduled for monthly recalls. (b) After 3 months of treatment, the appearance of the teeth improved considerably. Patient was very happy, in spite of confessing that he did not wear the tray on a daily basis. Patient chose to stop the treatment for a few months and then restart (Reprinted with Permission from Perdigao J (2010) Dental whitening – revisiting the myths. Northwest Dent 89:19–21, 23–6. (Northwest Dentistry, The Journal of the Minnesota Dental Association))
Fig. 6.4(a) This 38-year-old patient was born and raised overseas in an “area where everybody had brown teeth”. He used to drink water from a water well when he was a child in his home country. According to the patient’s account, all his siblings who lived in the same area had “brown teeth.” The medical history revealed no significant findings. After an intra-oral exam, patient was informed that at-home whitening might improve the appearance of his teeth as long as he understood that the treatment could span over a few months. Patient agreed to have his teeth whitened with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) at-home in a custom-fitted tray. Patient was scheduled for monthly recalls. (b) After 3 months of treatment, the appearance of the teeth improved considerably. Patient was very happy, in spite of confessing that he did not wear the tray on a daily basis. Patient chose to stop the treatment for a few months and then restart (Reprinted with Permission from Perdigao J (2010) Dental whitening – revisiting the myths. Northwest Dent 89:19–21, 23–6. (Northwest Dentistry, The Journal of the Minnesota Dental Association)) -
Discolored tooth caused by calcific metamorphosis (Fig. 6.5), which is discussed in more detail in Sect. 6.4.4.4.
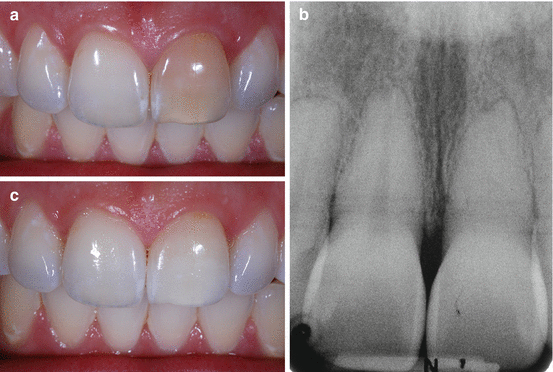 Fig. 6.5(a) Frontal view of maxillary incisors in a 26-year-old patient whose chief complaint was her discolored tooth #9 (FDI 2.1). Patient had had a traumatic injury to this tooth when she was 12 years old. No other signs or symptoms were associated with this tooth. Response to percussion was identical in all her maxillary anterior teeth. Although the response to cold was negative, the pulp responded to the electric pulp tester. (b) Periapical radiograph showing a calcified pulp space in tooth #9 (FDI 2.1). (c) Clinical aspect after 2 weeks of at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray. Patient decided to bleach for another period of 2 weeks, but she did not return for the recall appointment
Fig. 6.5(a) Frontal view of maxillary incisors in a 26-year-old patient whose chief complaint was her discolored tooth #9 (FDI 2.1). Patient had had a traumatic injury to this tooth when she was 12 years old. No other signs or symptoms were associated with this tooth. Response to percussion was identical in all her maxillary anterior teeth. Although the response to cold was negative, the pulp responded to the electric pulp tester. (b) Periapical radiograph showing a calcified pulp space in tooth #9 (FDI 2.1). (c) Clinical aspect after 2 weeks of at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray. Patient decided to bleach for another period of 2 weeks, but she did not return for the recall appointment -
Whitening of anterior teeth prior to esthetic rehabilitation with veneers (Fig. 6.6) or direct resin-based composite restorations (Chap. 14).
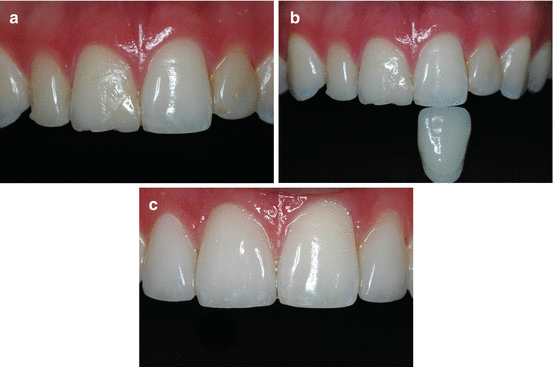 Fig. 6.6(a) A 22-year-old patient with several resin-based composite restorations in his discolored maxillary incisors. Tooth #9 (FDI 2.1) was not restored. (b) Clinical aspect after 2 weeks of overnight at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray. (c) Porcelain veneers were bonded with a etch-and-rinse adhesive and a light-cure resin-based luting cement on teeth #7 (FDI 1.2), #8 (FDI 1.1), and #10 (FDI 2.2) 5 weeks after patient completed the bleaching treatment
Fig. 6.6(a) A 22-year-old patient with several resin-based composite restorations in his discolored maxillary incisors. Tooth #9 (FDI 2.1) was not restored. (b) Clinical aspect after 2 weeks of overnight at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray. (c) Porcelain veneers were bonded with a etch-and-rinse adhesive and a light-cure resin-based luting cement on teeth #7 (FDI 1.2), #8 (FDI 1.1), and #10 (FDI 2.2) 5 weeks after patient completed the bleaching treatment -
Dietary stains
Contraindications for at-home whitening with carbamide peroxide gel in a custom-fitted tray:
-
Patient’s unrealistic expectations.
-
Patient unsure if he/she is willing to carry out the treatment on a daily basis.
-
Smoking. As a precautionary measure, patients are informed to refrain from smoking between 2 h before inserting the tray and 2 h after removing the tray. This relative contraindication derives from the findings of the “hamster study” (Weitzman et al. 1986), as discussed in detail in Chap. 3. More recently, it has been reported that at-home bleaching does not induce DNA damage to the gingival tissue of smokers during a 3-week treatment (de Geus et al. 2015c). However, smokers had slightly darker teeth after 1 month of at-home bleaching. This may be an important detail of information to disclose to patients prior to starting the bleaching treatment (de Geus et al. 2015a). A clinical study also concluded that bleaching with 10 % carbamide peroxide during 3 h daily for 3 weeks is effective in smokers even after 1 year, but dental prophylaxis may be necessary to remove extrinsic stains caused by diet and smoking (de Geus et al. 2015b).
-
Pregnancy and breastfeeding. There is not enough available evidence concerning either the teratogenicity or the safety for breastfeeding infants of the ingredients of whitening agents.
-
Root (dentin) hypersensitivity. Preexisting tooth sensitivity must be treated prior to starting the whitening treatment to block any patent dentinal tubules.
-
Gingival recession with discolored roots, especially in elderly patients. Radicular dentin does not respond to whitening as well as coronal dentin does (Haywood 2003).
-
Possible allergy to inactive components of the bleaching gel.
-
Acatalasemia. A hereditary disorder in which the blood catalase activity level is below normal (European Commission Scientific Committee on Consumer Products 2007).
-
G6PD deficiency. A genetic disorder of erythrocytes that lack the enzyme G6PD, causing them to destroy prematurely and preventing patient with this disorder to break down hydrogen peroxide (European Commission Scientific Committee on Consumer Products 2007).
-
Some patients with xerostomia, as dry mouth may affect the degradation of hydrogen peroxide (European Commission Scientific Committee on Consumer Products 2007).
-
Discolored endodontically treated teeth do not typically respond to external tray whitening as well as vital teeth do. However, some cases of light staining in endodontically treated teeth that have darkened recently can be successfully bleached externally using ≈ 20 % carbamide peroxide gel in a specially designed bleaching tray (Fig. 6.7). The prognosis of the at-home whitening treatment in these cases depends on the nature and duration of the discoloration.
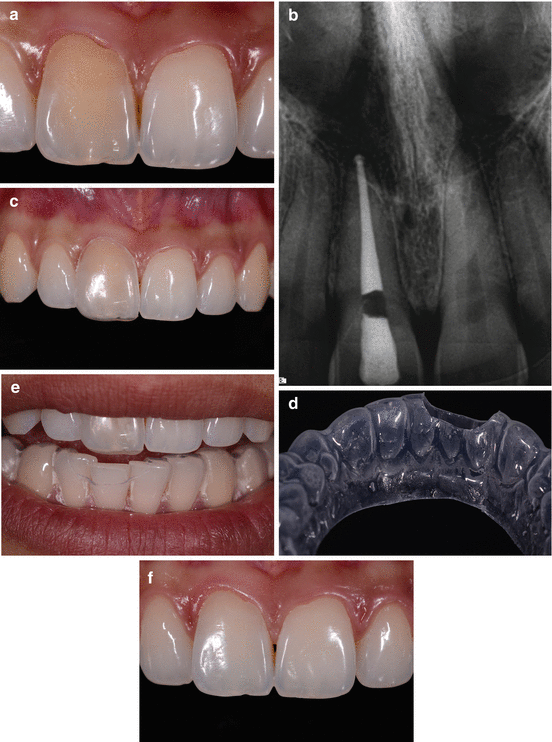 Fig. 6.7(a) A 24-year-old patient suffered a traumatic injury to her tooth #8 (FDI 2.1). After 4 years, her tooth became darker without any symptoms. The patient immediately visited her dentist who diagnosed pulpal necrosis. (b) A root canal treatment was performed and the lingual access preparation restored with a resin-based composite material. (c) A special bleaching tray was fabricated to bleach the discolored tooth following an at-home regimen. (d) A lower tray was fabricated and customized to serve as stabilizer for the upper one-tooth tray. The lower tray was not used as a bleaching tray. (e) Patient wearing the upper bleaching tray and the lower stabilizing tray. (f) After 5 days of at-home whitening with 22 % carbamide peroxide gel (Whiteness Perfect 22 %, FGM) for 2 h twice daily
Fig. 6.7(a) A 24-year-old patient suffered a traumatic injury to her tooth #8 (FDI 2.1). After 4 years, her tooth became darker without any symptoms. The patient immediately visited her dentist who diagnosed pulpal necrosis. (b) A root canal treatment was performed and the lingual access preparation restored with a resin-based composite material. (c) A special bleaching tray was fabricated to bleach the discolored tooth following an at-home regimen. (d) A lower tray was fabricated and customized to serve as stabilizer for the upper one-tooth tray. The lower tray was not used as a bleaching tray. (e) Patient wearing the upper bleaching tray and the lower stabilizing tray. (f) After 5 days of at-home whitening with 22 % carbamide peroxide gel (Whiteness Perfect 22 %, FGM) for 2 h twice daily -
Deep white spot lesions. White spots deeper than 0.5 mm may be better camouflaged with other treatment modalities, including microabrasion, enamel etching followed by resin infiltration (Chaps. 9, 10, 12, and 13), or removal of the discolored area and restoration with a dentin adhesive and a resin-based composite material (Chap. 15, Sect. 15.3).
6.2 Advantages and Disadvantages of At-Home Whitening
Two major advantages of at-home whitening are its efficacy (Fig. 6.1) and stability of posttreatment color (Table 6.1) (Swift et al. 1999; Ritter et al. 2002). After 10 years, 43 % of patients that whitened their teeth with 10 % carbamide peroxide for 6 weeks deemed the color to be stable (Ritter et al. 2002).
Table 6.1
Advantages and disadvantages of at-home whitening with a custom-fitted tray supervised by a dental professional
|
Advantages
|
Disadvantages
|
|---|---|
|
Very effective, durable whitening
|
Tooth sensitivity
|
|
Backed with clinical and laboratory research
|
Patient compliance
|
|
Safe
|
Relatively long treatment time
|
|
Low cost compared to in-office procedures
|
Over-the-counter whitening methods are less expensive
|
A major disadvantage of at-home whitening is patient’s compliance (Table 6.1), as the dental professional is unable to monitor the daily treatment. Meireles et al. (2008a) asked subjects to return all used and unused bleaching gel syringes to ensure compliance based on the amount of gel used. This method, however, may be difficult to implement on a regular basis.
Another disadvantage of at-home whitening compared to in-office whitening is the longer treatment time for the former. However, one session of in-office whitening is not usually sufficient to achieve optimal results (Al Shethri et al. 2003), resulting in a similar overall treatment time for the two techniques.
6.3 Efficacy and Durability
There are several variables that influence the treatment outcome, including the technique, the type of bleaching agent, the concentration, and the application time (Joiner 2006; Buchalla and Attin 2007; Meireles et al. 2008b; Matis et al. 2009a).
6.3.1 At-Home Whitening in a Custom-Fitted Tray
Dentist-prescribed overnight bleaching with carbamide peroxide in a custom-fitted tray has been shown to be the safest, most effective method of tooth whitening (Haywood 2003; Matis 2004; Matis et al. 2009a).
Two clinical studies evaluated the 2-year effectiveness of tray whitening with carbamide peroxide (Swift et al. 1999; Meireles et al. 2010). In the first study, 29 patients had their maxillary teeth treated with a 10 % carbamide peroxide gel nightly for 2 weeks. Teeth became eight shades lighter after 2 weeks of treatment when color was measured with the Vita Classical A1-D4 shade guide (VITA Zahnfabrik H. Rauter GmbH & Co. KG) organized by value (lighter to darker). Twenty-four patients were recalled after 2 years. Teeth in 20 patients (83.3 %) had darkened an average of two shades, which occurred during the first 6 weeks posttreatment. The lightening effect remained statistically significant at 2 years. Overall, patients were satisfied with the shade. In the second study (Meireles et al. 2010), 92 patients whitened their maxillary anterior teeth with 10 % carbamide peroxide or with 16 % carbamide peroxide in a custom-fitted tray for 2 h/day during 3 weeks. Shade evaluations were carried out at baseline, 1 month, 6 months (Meireles et al. 2008b), 1 year (Meireles et al. 2009), and 2 year post-bleaching (Meireles et al. 2010). Although the 16 % carbamide peroxide group showed some reversal of the whitening effect at 1 year (Meireles et al. 2009), both treatment groups had the same median tooth shade 1 year after bleaching, which was lighter than at baseline. At 2 years, the median tooth shade remained lighter than at baseline for both carbamide peroxide concentrations tested.
Boushell et al. (2012) evaluated patients’ satisfaction and reported side effects of at-home whitening with 10 % carbamide peroxide in a custom-fitted tray up to 17 years posttreatment. Thirty-one participants who had completed clinical studies using 10 % carbamide peroxide were contacted at least 10 years posttreatment. Patient satisfaction with tray whitening was determined to last an average of 12.3 years posttreatment.
In case the patient perceives that there has been a color regression and is willing to touch-up the teeth color, the original bleaching tray may be applied for 2 or 3 nights using the same 10 % carbamide peroxide gel. In case the tray no longer fits the patient’s teeth as a result of recent restorations or extracted teeth, disposable trays prefilled with 6 %, 10 %, or 15 % hydrogen peroxide gel (Opalescence Go, Ultradent Products, Inc.) may be used 2 or 3 days. The respective manufacturer recommends decreasing contact times with increasing hydrogen peroxide concentration. For the lowest concentration, the manufacturer suggests that patients wear the disposable tray for 60–90 min daily, whereas for the highest hydrogen peroxide concentration the recommended contact time is 15–20 min daily.
Patients often inquire if they need to refrain from a potentially staining diet during and after at-home whitening. Current evidence from controlled clinical trials suggests that coffee does not interfere with the outcome of whitening nor does it affect tooth sensitivity (Rezende et al. 2013). Peroxide-based whitening agents are effective in preventing any staining from coffee or red wine during the treatment (Cortes et al. 2013). As a result, the need for a white diet during the bleaching treatment has been challenged. A study determined whether a white diet is necessary by evaluating the effects of coffee, tea, wine, and dark fruits on tooth whitening during the bleaching process (Matis et al. 2015). From five published studies, the authors concluded that a nonwhite diet was not significantly associated with less tooth whitening, and there was only a weak positive association between tooth whitening and diet for subjects who consumed large amounts of coffee/tea.
After the whitening regimen is completed, both coffee and red wine cause enamel color change. Red wine, however, stains enamel more intensely than coffee (Cortes et al. 2013).
6.3.2 Jump-Start Whitening
The objective of the jump-start technique is to boost the bleaching effect with the in-office treatment, then improve color stability with the at-home component to reach a more esthetic result compared to in-office bleaching alone (Deliperi et al. 2004; Matis et al. 2009b). Clinical evidence, however, does not support this assumption. Two recent clinical studies reported that the results of the combined in-office/at-home technique were similar to those obtained only with the at-home technique (Bernardon et al. 2010; Dawson et al. 2011). Therefore, the in-office component of the combined jump-start technique does not improve the treatment outcome and may be considered redundant. Nevertheless, this technique may motivate some patients, as the whitening effect is visible immediately.
A more recent version of the jump-start technique is known as deep whitening technique (Kör Whitening, Evolve Dental Technologies, Inc) (Sulieman 2008). This technique currently includes three different modalities, according to the severity of the discoloration: (A) 2 weeks of at-home overnight whitening with 16 % carbamide peroxide, followed by one in-office whitening session with 34 % Tri-Barrel Hydremide™ peroxide2; (B) in-office “conditioning” visit with 13 % Tri-Barrel Hydremide™ peroxide in whitening trays followed by 3–4 weeks of at-home overnight whitening, and a final in-office whitening session with 34 % Tri-Barrel Hydremide™ peroxide; (C) in-office “conditioning” visit with 13 % Tri-Barrel Hydremide™ peroxide in whitening trays, followed by 6–8 weeks of at-home overnight whitening, and a final in-office whitening session with 34 % Tri-Barrel Hydremide™ peroxide. All three methods require periodic at-home maintenance after the treatment.
Currently, there is no independent scientific evidence to back the use of the so-called deep whitening technique.
The trays used in the deep bleaching technique are specially made trays (Kurthy 2001). As per the respective manufacturer, these trays provide better sealing than conventional bleaching trays, enabling the whitening agent to be active all night as opposed to other tray whitening methods (Kör Whitening 2015). The manufacturer’s website also states: “Clinical Research associates as well as other researchers have found that whitening gel in conventional whitening trays is only strongly active for 25–35 minutes. This is due to rapid contamination of the whitening gel by saliva.” However, this statement is not supported by independent research. It has been shown that hydrogen peroxide releases all of its peroxide in 30–60 min, with a quick decline, while carbamide peroxide releases about 50 % of its peroxide in 4 h, then experiences a slow decline (Haywood 2005).
More than 50 % of the carbamide peroxide active agent is available after 2 h. The percentage of carbamide peroxide recovered from tray and teeth is 10 % at 10 h (Matis et al. 1999).
The use of light sources to allegedly activate the peroxide during the in-office component of the jump-start technique has been used in many dental offices. According to Christensen (2003), “all whitening methods are successful to some degree” but “the use of lights with bleaching has been mainly a marketing tool.”
6.3.3 Over-the-Counter (OTC) Whitening
Sales of OTC bleaching products have increased dramatically in recent years (Chap. 1), driven not only by their lower cost compared to professional tooth-whitening techniques but also by strong consumer demand for esthetic dental care and easy access through online auctions and e-commerce sites. Additionally, OTC bleaching products are easy to use and convenient for the patient (Kugel 2003). Concentrations as high as 44 % carbamide peroxide are available from online auctions sites and retailers. Non-dental options are the latest trend, including mall kiosks, salons, and spas. More recently, whitening has been performed in passenger ship cruises (ADA Council on Scientific Affairs 2010).
How does the efficacy of OTC whitening products compare to that of the dentist-prescribed at-home whitening? While there are many studies comparing OTC whitening with dentist-prescribed whitening, there are only a few independent clinical studies (Serraglio et al. 2016). A study (Bizhang et al. 2009) measured tooth shade with spectrophotometry and concluded that 6 % hydrogen peroxide whitening strips applied twice a day for 30 min each for 2 weeks are not as effective as at-home whitening with 10 % carbamide peroxide overnight for 2 weeks. Kishta-Derani et al. (2007) evaluated four paint-on films self-adhering solutions that are brushed on the tooth surface. These paint-on films contained hydrogen peroxide, sodium percarbonate or carbamide peroxide. Two of the paint-on films did not result in any significant whitening effect after 2 weeks of daily application. The results obtained with OTC whitening are not as pleasant and the procedure is not as safe as those methods prescribed by a dental professional (Haywood 2003). For similar concentrations of hydrogen peroxide, OTC bleaching strips cause more gingival irritation and tooth sensitivity than at-home tray whitening, as discussed in Chap. 4.
Given that OTC whitening products are not custom-fitted to the patient’s mouth, they are not the ideal vehicle for the application of peroxide-based gels. Ill-fitting trays may result in soft tissue injury, poor patient compliance, and malocclusion problems (Kugel 2003).
6.4 At-Home Whitening with a Custom-Fitted Tray Supervised by a Dental Professional
6.4.1 Treatment Plan
The correct diagnosis of the origin of the discoloration is critical, as different treatment options lead to different clinical outcomes. It is, therefore, imperative that the dental professional understands the etiology of each specific tooth discoloration case to be able to diagnose and prescribe the proper treatment for each patient. Please refer to Chap. 1 for further details.
A full-mouth exam and recent periapical radiographs of the anterior teeth are essential during the diagnostic appointment. Intra-oral photographs are extremely valuable to document the pretreatment tooth color for future comparisons and to include in the patient’s record. Pulp testing is always necessary for single-tooth discolorations. Patient must be informed that existing anterior esthetic restorations, including porcelain and resin-based composites, will not lighten with bleaching agents, except for superficial extrinsic stains (Fig. 6.8). These restorations must be replaced after the whitening treatment is completed to ensure an acceptable esthetic outcome. Additionally, patient must also be informed that amalgam restorations that come in contact with the bleaching gel may generate a “greening effect” of the tooth structure in areas immediately adjacent to the amalgam material (Haywood 2002).
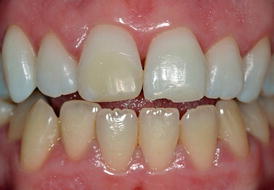

Fig. 6.8
Existing resin-based composite restorations on teeth #8 (FDI 1.1) and #9 (FDI 2.1) after at-home whitening of the maxillary arch with 10 % carbamide peroxide with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray for 3 weeks
Haywood (2003) defined nightguard vital bleaching as a three-step technique:
- 1.
Whitening material, which is usually a thick peroxide-based gel
- 2.
Application prosthesis, currently known as the bleaching tray
- 3.
Treatment regimen
Haywood (2003) suggested that wearing a tray on only one arch might improve patient’s compliance, as patient can directly observe the color change in one arch compared to the arch that is not undergoing treatment. Additionally, the interocclusal thickness of both maxillary and mandibular trays may exacerbate TMJ disorder symptoms (Robinson and Haywood 2000).
6.4.2 Whitening Material
Carbamide peroxide in concentrations between 10 and 22 %,3 and hydrogen peroxide in concentrations from 4 to 8 % have been used for at-home bleaching for different periods of time (Joiner 2006; Meireles et al. 2008b; Matis et al. 2009a). A recent systematic review and meta-analysis of at-home whitening concluded that carbamide peroxide results in a slightly better whitening efficacy than hydrogen peroxide when applied in a custom-fitted tray (Luque-Martinez et al. 2016).
The bleaching agent for at-home application that has been more frequently scrutinized in the dental literature is 10 % carbamide peroxide (Matis 2004). Chemically, carbamide peroxide is a crystalline material containing a molecule of urea complexed with a single molecule of hydrogen peroxide – 10 % carbamide peroxide contains approximately 3.3–3.5 % hydrogen peroxide (Cooper et al. 1992; Sulieman 2008; ADA Council on Scientific Affairs 2010). Carbamide peroxide is preferred over hydrogen peroxide because it is more stable than hydrogen peroxide, providing a nonaqueous formula of available hydrogen peroxide (Fischer 1995).
Current carbamide peroxide bleaching gels contain glycerin as a humectant and flavor enhancer; and a thickener, usually a polymer (Carbopol,4 The Lubrizol Corporation). Carbopol polymers are cross-linked high molecular weight homo- and copolymers of acrylic acid, therefore containing active carboxyl groups. These polymers are slightly acidic, which lowers the pH of the bleaching gel. Accordingly, bases such as sodium hydroxide may be used to make the gel less acidic. Similar thickeners and bases are also used in the composition of hydrogen-peroxide-containing OTC bleaching strips.
6.4.3 Bleaching Tray Design
Several brands of thermoplastic materials are available to fabricate bleaching trays. We currently use a 0.035″-thick ethylene vinyl acetate material that is heated prior to forming the tray around the stone model in a vacuum or pressure device, as shown in the video Fabrication of a Whitening Tray. After cooling, the tray is then trimmed in a horseshoe shape (Fig. 6.9). The fine trimming must follow the scalloped contour of the free gingival margin (Haywood 1997b). The design has evolved to a scalloped tray slightly short of the free gingival margin (0.5–1.0 mm) to prevent possible irritation caused by the contact of the gel with the soft tissues (Chap. 4). The scalloped design is contraindicated with low-viscosity bleaching gels, as the gel is more likely to leak to the mouth and irritate the tongue and lips (Haywood 2003). In specific situations, including clinical cases of one-tooth whitening, the tray may be slightly extended gingivally. In case of inadvertent fabrication of shortened trays, successful whitening still occurs beyond the borders of the short tray without demarcation lines on the teeth (Oliver and Haywood 1999), as peroxides diffuse easily through enamel.
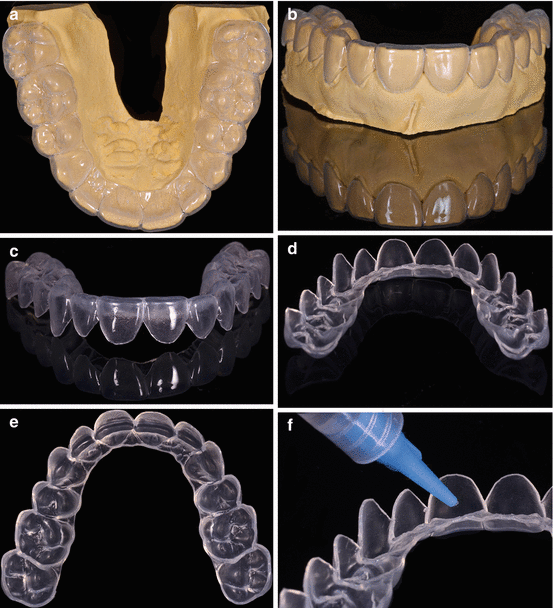

Fig. 6.9
Custom-made bleaching tray for at-home whitening. (a) Occlusal view of the tray inserted onto the stone model. The model has been trimmed to remove the palatal area to enhance the vacuum over the teeth and obtain a tighter adaptation of the heated tray material to the teeth. (b) Frontal view of the model after the tray was scalloped around the gingival margins. (c) Frontal view of the scalloped tray. (d) Lingual view of the scalloped tray. (e) Incisal view of the scalloped tray. (f) Demonstrating to patient how to load the bleaching gel into the tray
The use of tray reservoirs to make space to retain the bleaching gel has been patented (Fischer 1992). It remains, nevertheless, a controversial issue. Light-cured block-out resin spacers are recommended by some manufacturers, but the use of spacers to create reservoirs for the bleaching gel does not seem to increase the success of home bleaching (Javaheri and Janis 2000; Matis et al. 2002). The bleaching gel remains active for longer periods when reservoirs are used (Matis et al. 2002), which may be the reason why tray reservoirs result in higher rates and higher intensity of gingival inflammation during at-home bleaching (Kirsten et al. 2009).
The tray is then tried-in after fine trimming to check for a tight fit, making sure that the patient does not feel any sharp edges. The dental professional must examine the soft tissues very carefully at this stage to identify areas of compression that may cause discomfort to the patient. It is crucial to demonstrate how to dispense the right amount of gel into the tray, usually one drop (Fig. 6.9f). To verify the gel covers the buccal aspect of the tooth the patient is instructed to ensure that a very slight amount of gel has extruded from the tray at its gingival border. Then, the excess gel is wiped out with a toothbrush or a cotton swab to prevent the contact of the gel with the mucosa. The bleaching gel may also be applied from the lingual in case the buccal enamel is covered with restorative material (Fig. 6.10). Haywood and Parker (1999) described a case of porcelain veneers bonded to tetracycline-stained teeth that resulted in a graying of the veneers. A custom-fitted tray with no reservoirs and no gingival scalloping was used to bleach the teeth with 10 % carbamide peroxide applied nightly for 9 months.
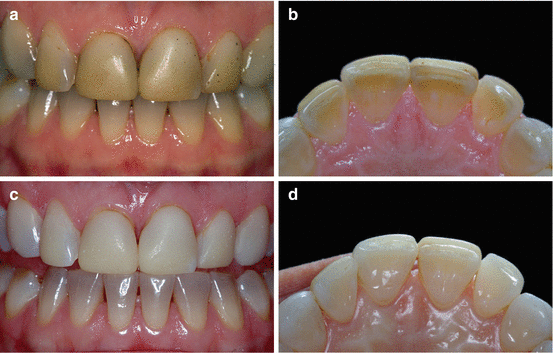

Fig. 6.10
(a) A 43-year-old patient visited the University of Minnesota School of Dentistry Comprehensive Care Clinic to ask for a second opinion about her front maxillary teeth. She had direct resin-based composite veneers placed approximately 20 years back, but the restorative material had become discolored with “black spots all over.” The patient was not sure if porcelain veneers were indicated for her clinical situation. (b) The lingual view of the maxillary incisors depicted a slight grayish dentin discoloration. Although the medical history was negative for antibiotic ingestion, the patient vaguely recalled having some fever episodes and possibly taking antibiotics in her childhood. We then informed the patient that we might be able to whiten her teeth if she agreed to wear a tray with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) for 2–6 months at night. After the patient agreed and signed the respective consent form, a custom-fitted tray was fabricated and the patient instructed to apply the whitening gel into the lingual aspect of the tray to whiten the teeth from the lingual surface. Patient was also instructed to return to the clinic every month. (c) Retracted frontal view after three months of at-home whitening. Note that the composite stains were removed by the peroxide oxidative action. Patient did not experience any sensitivity or any alterations of the soft tissues at each periodical recall. (d) Lingual view after 3 months. Compare the shade with that of b. Old resin-based composite restorations were removed at a subsequent appointment and enamel polished with diamond pastes. After observing the final result, patient was unsure whether or not she wanted veneers. She decided that she did not want any other treatment (Reprinted with Permission from Perdigao J (2010) Dental whitening – revisiting the myths. Northwest Dent 89:19–21, 23–6. (Northwest Dentistry, The Journal of the Minnesota Dental Association)
-
The use of spacers for the bleaching gel does not improve the success of home bleaching.
-
The bleaching gel remains active for longer periods of time when spacers are used.
-
The use of a reservoir in the tray may result in higher intensity of gingival inflammation.
6.4.4 Treatment Regimen
6.4.4.1 Physiological Discoloration
The recommended duration of the treatment for the original nightguard vital whitening technique with 10 % carbamide peroxide was 2–6 weeks (Haywood and Heymann 1989). Currently, the typical treatment time for teeth that are inherently discolored by aging or discolored by diet and chromogenic diet is from 2 to 4 weeks, especially if the treatment is carried out overnight.
Although higher concentrations of peroxides result in a faster rate of whitening than 10 % carbamide peroxide, they reach a similar final result (Matis et al. 2000; Meireles et al. 2009; Basting et al. 2012). Higher concentrations, however, increase the incidence of tooth sensitivity (Matis et al. 2000). We have only prescribed 10 % carbamide peroxide for at-home whitening of physiological discoloration in the last 10 years. This concentration is the only one that has been approved by the American Dental Association (ADA Seal Product Category 2015).
A clinical study tested four different application times of 10 % carbamide peroxide – 15 min, 30 min, 1 h or 8 h. After 16 days, 15 out of 15 (100 %) subjects that had bleached for 8 h/day were satisfied with the results, while only 5/15 subjects that had bleached 1 h per day were satisfied with the results (Cardoso et al. 2010). Matis et al. (2009a) pooled data from nine clinical studies from the same research center, which included in-office and tray whitening. These authors concluded (1) whitening is most effective when bleaching gel is placed in trays and the trays are used overnight; and (2) tray whitening during the daytime for shorter periods of time was the second most effective whitening method.
Current clinical evidence suggests that 10 % carbamide peroxide is as effective as higher concentrations but results in lower incidence of sensitivity than higher concentrations (Matis et al. 2000). Overnight tray whitening with 10 % carbamide peroxide results in whiter teeth and more durable results than whitening for a few hours during the daytime (Matis et al. 2009a; Cardoso et al. 2010).
Should the recommended treatment be 2–4 weeks overnight for all patients? This treatment regimen is usually adequate for shades A and B (reddish-brownish and reddish-yellowish, respectively) in the Vita Classical A1-D4 shade guide (VITA Zahnfabrik H. Rauter GmbH & Co. KG). When the tooth color has a gray component (C and D shades, Vita Classical A1-D4 shade guide) or when teeth are discolored by the accumulation of tetracycline stains in dentin, teeth do not respond to whitening as well, especially when the stain accumulates in the cervical third.
The prescription of at-home bleaching treatments to child and teenage patients has become a pertinent issue, as parents often ask their family dentists about the possibility of whitening young patients’ teeth. Croll (1994) described a protocol for “at-home” tooth bleaching in young patients. According to Croll and Donly (2014), tray whitening of the permanent dentition in children and teenagers is safe and can be performed in a similar manner as for adults. The American Academy of Pediatric Dentistry has published a policy since 2009 on the use of dental bleaching for child and adolescent patients (American Academy of Pediatric Dentistry Council on Clinical Affairs 2015). However, this policy does not address the recommended contact time of the gel with the dentition of young patients. While there is an abundant amount of information on the safety of at-home bleaching gels for adults, studies focused on the tolerable carbamide peroxide concentration and respective contact time with the tooth surface for young patients in terms of pulpal health are lacking. With this in mind, stronger evidence may be needed to start recommending tray whitening in child and teenage patients on a regular basis.
6.4.4.2 Tetracycline-Stained Teeth
Tetracyclines and their derivatives are broad-spectrum antibiotics active against both Gram-positive and Gram-negative bacteria as well as infections caused by Mycoplasma, Rickettsia, and Chlamydia. They are also used in rheumatoid arthritis, chronic respiratory diseases, and in the management of periodontal disease (Seymour and Heasman 1995; Tilley et al. 1995; Sánchez et al. 2004; Tredwin et al. 2005). Tetracyclines are contraindicated during pregnancy because they cross the placenta and are toxic to the developing fetus (Sánchez et al. 2004), causing tooth discoloration and enamel hypoplasia if administered during the period of tooth development.
The affinity of tetracycline for dental tissues was first described by Shwachman et al. (1958–1959) in pediatric patients with cystic fibrosis of the pancreas treated with long-term antibiotic therapy. Soon thereafter, Zegarelli et al. (1960) reported similar findings in 38 of 52 children with cystic fibrosis of the pancreas treated with tetracycline. In 1962, Davies and coworkers suggested that tetracycline is deposited on the organic matrix of bones and teeth prior to calcification (Davies et al. 1962). The fluorescence of the pigment and the histological findings confirmed the clinical observation that the pigmentation was due to tetracycline (Wallman and Hilton 1962). When 50 out of 64 newborns that had been given tetracycline in the neonatal period were followed up, 46 of them were found to have yellow or brown discoloration of the teeth, with or without enamel hypoplasia. The greater the total dose of tetracycline per birth weight, the greater was the change. The severity of the stain and its pattern depended on the tetracycline type, dosage, and duration of therapy (Wallman and Hilton 1962).
The affinity of tetracycline for mineralizing tissue is the result of binding to calcium to form a tetracycline-calcium orthophosphate insoluble complex (Gassner and Sayegh 1968; Eisenberg 1975). Chelation with iron has also been reported for tetracycline-induced tooth discoloration (Salman et al. 1985; Bowles and Bokmeyer 1997). Teeth with tetracycline deposits emit yellow fluorescence when observed under ultraviolet light in a darkened room as opposed to the bluish fluorescence characteristic of nonpigmented teeth. The tetracycline stain undergoes degradation by exposure to light, which results in darker staining with age (Abou-Rass 1988).
In 1978, it was reported that minocycline was a viable alternative to treat cases of acne that did not respond to treatment with other tetracyclines (Cullen 1978). Minocycline is a semisynthetic tetracycline derivative used for the treatment of acne for those suffering from rheumatoid arthritis, and chronic respiratory infections (Tilley et al. 1995; Tredwin et al. 2005). In 1980, in a letter to the editor of the Journal of the American Academy of Dermatology, a dermatologist described a 42-year-old patient who had been on minocycline, 100 mg two to three times a day, for approximately 4–5 years (Caro 1980). The patient’s dental hygienist had noted the development of a gray discoloration of the patient’s teeth. Additionally, the patient also stated that she retained a tan for longer than normal and that the skin and fingernails had a gray appearance. The patient’s dental crowns had to be restained to match the gray color of the natural dentition. In 1985, a retrospective cohort study found that 4 of 72 patients who had minocycline therapy during adolescence had minocycline-associated tooth discoloration, which occurred after only 4 weeks of treatment in one case (Poliak et al. 1985). Other cases of post-eruptive tooth staining with minocycline have been described (Salman et al. 1985; Bowles and Bokmeyer 1997; Cheek and Heymann 1999). Discoloration caused by minocycline is usually green-gray/blue-gray (Tredwin et al. 2005). Besides discoloration of teeth, minocycline may also cause a blue staining of the sclera, ears, and oral mucosa, which may be irreversible (Dodd et al. 1998; LaPorta et al. 2005; Johnston 2013). Minocycline also causes discoloration of nonvital teeth (Dabbagh et al. 2002; Kim et al. 2010) as discussed in Chap. 8.
The esthetic management of patients with tetracycline-stained teeth is a challenge since the degree of staining varies from mild to severe (Jordan and Boksman 1984).
- 1.
Mild tetracycline staining (Fig. 6.11) is usually very receptive to whitening. This staining is yellow to gray with minimal or no banding and is uniformly spread throughout the tooth, but more confined to the incisal three-quarters of the crown.
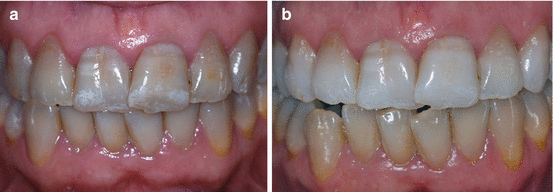 Fig. 6.11(a) 38 year old patient with history of tetracycline ingestion. She was diagnosed with mild tetracycline staining. Additionally, the maxillary central incisors had white spot areas in the incisal third. (b) After 3 months of at-home whitening with 10 % carbamide peroxide with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray with monthly recalls. Both the tetracycline stains and the white spot areas were successfully camouflaged, in spite of a residual gray band in the cervical third
Fig. 6.11(a) 38 year old patient with history of tetracycline ingestion. She was diagnosed with mild tetracycline staining. Additionally, the maxillary central incisors had white spot areas in the incisal third. (b) After 3 months of at-home whitening with 10 % carbamide peroxide with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray with monthly recalls. Both the tetracycline stains and the white spot areas were successfully camouflaged, in spite of a residual gray band in the cervical third - 2.
Moderate tetracycline staining (Fig. 6.12) may vary from a uniform deep yellow discoloration, which is responsive to bleaching, to a dark-gray discoloration band located between the cervical fifth of the crown and the tooth surface located incisally to the band.
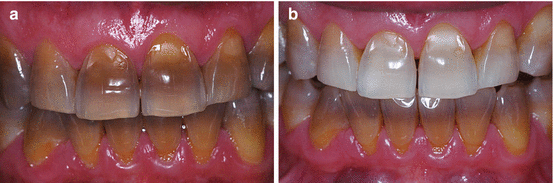 Fig. 6.12(a) A 39-year-old patient with a history of tetracycline ingestion during infancy. He was informed that long-term whitening (2–6 months) might lighten his teeth. However, there was no assurance given of the final whitening result. Patient agreed to carry out the treatment by wearing a custom-fitted tray with 10 % carbamide peroxide gel (Opalescence 10 %, Ultradent Products, Inc.) every night. Instructions were carefully given to the patient, and a new appointment set up for within 1 month (and every month thereafter). (b) Final result after 6 months. No sensitivity was reported at any recall period; no alterations of soft tissues were observed. Patient started whitening the lower arch immediately after the completion of the treatment in the upper arch (Reprinted with Permission from Perdigao J (2010) Dental whitening – revisiting the myths. Northwest Dent 89:19–21, 23–6. (Northwest Dentistry, The Journal of the Minnesota Dental Association))
Fig. 6.12(a) A 39-year-old patient with a history of tetracycline ingestion during infancy. He was informed that long-term whitening (2–6 months) might lighten his teeth. However, there was no assurance given of the final whitening result. Patient agreed to carry out the treatment by wearing a custom-fitted tray with 10 % carbamide peroxide gel (Opalescence 10 %, Ultradent Products, Inc.) every night. Instructions were carefully given to the patient, and a new appointment set up for within 1 month (and every month thereafter). (b) Final result after 6 months. No sensitivity was reported at any recall period; no alterations of soft tissues were observed. Patient started whitening the lower arch immediately after the completion of the treatment in the upper arch (Reprinted with Permission from Perdigao J (2010) Dental whitening – revisiting the myths. Northwest Dent 89:19–21, 23–6. (Northwest Dentistry, The Journal of the Minnesota Dental Association)) - 3.
Severe tetracycline staining (Fig. 6.13) appearing blue-gray or dark gray, accompanied by significant banding across the tooth. Although whitening will somehow lighten these teeth, they may not become esthetically acceptable without bonded restorations.
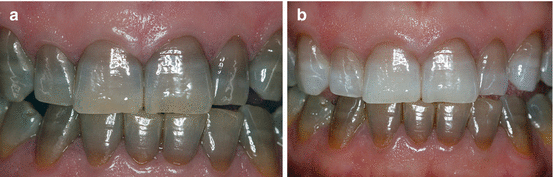 Fig. 6.13(a) Severe tetracycline staining in a 42-year-old patient. (b) Final aspect after 6 months of at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray with monthly recalls. As expected, and as the patient had been informed, the cervical third was the most resistant area to whitening
Fig. 6.13(a) Severe tetracycline staining in a 42-year-old patient. (b) Final aspect after 6 months of at-home whitening with 10 % carbamide peroxide gel with potassium nitrate and sodium fluoride (Opalescence 10 % PF, Ultradent Products, Inc.) in a custom-fitted tray with monthly recalls. As expected, and as the patient had been informed, the cervical third was the most resistant area to whitening
Clinical studies have demonstrated that mild-to-moderate tetracycline stains can be removed relatively well (Figs. 6.3, 6.11, and 6.12) using the at-home whitening technique with carbamide peroxide in a custom-fitted tray, even though an extended treatment time may be required to achieve satisfactory results (Leonard et al. 2003). For mild-to-moderate tetracycline-stained teeth, the recommended treatment is 2–6 months with monthly recalls to evaluate the tooth color and potential side effects (irritation of soft issues, exacerbation of symptoms from TMJ disorders, and tooth sensitivity). The stains that are most difficult to remove are those located at the cervical third. If no improvement in tooth color is observed within the first 3 months, it is unlikely that any improvement will occur (Deliperi et al. 2006). In fact, the maximum lightening effect occurs during the first month (Matis et al. 2006). Therefore, patients with tetracycline-stained teeth must be informed that a residual gray stain may still be perceptible at the end of the treatment at the cervical third. These clinical cases may need a longer bleaching regimen (Matis et al. 2006).
Patients with tetracycline-stained teeth participated in a clinical trial of tray whitening with 10 % carbamide peroxide for 6 months. The 90-month follow-up determined the stability, posttreatment side effects, and patient satisfaction (Leonard et al. 2003). Shade was stable at least 90 months after treatment. Patients in this study were overwhelmingly positive about the procedure in terms of shade retention and lack of posttreatment side effects, as 60 % of the subjects reported no obvious shade change or only a slight darkening not noticed by others.
A total of 44 subjects bleached their tetracycline-stained teeth overnight for 6 months using trays with reservoirs, and then followed for 5 years. This was a split-mouth design study that used two of three different concentrations of carbamide peroxide – 10 %, 15 %, or 20 %. More than 65 % of the maximum tooth whitening remained for all carbamide peroxide concentrations. However, 15 and 20 % carbamide peroxide caused significantly more sensitivity than 10 % carbamide peroxide (Matis et al. 2006). In this study, there was a reversal of color change in tetracycline-stained teeth at 5 years.
Although at-home whitening with 10 % carbamide peroxide for up to 6 months remains the first choice for whitening tetracycline-stained teeth, these patients may need to rebleach or touch-up the tooth color approximately 5 years after the original treatment.
6.4.4.3 Fluororis and Fluorosis-Like Enamel Hypocalcifications
Excessive fluoride intake may result is dental fluorosis, which is a hypomineralization of enamel characterized by opaque white areas or discolorations ranging from yellow to dark brown (Horowitz et al. 1984). The severity of fluorosis is correlated with the amount and duration of fluoride ingestion during tooth development (Robinson and Kirkham 1990). In more severe cases the enamel surface becomes pitted, displaying porosities on the surface (Chap. 15). The degree of enamel hypomineralization may vary on different parts of the tooth surface due to the variation in enamel thickness (Fejerskov et al. 1990). Not all white or brown demineralized enamel areas are caused by fluorosis; therefore, they may be considered idiopathic (Cutress and Suckling 1990; Croll 2009) (Fig. 6.14). The term enamel “dysmineralization” has been used when referring to fluorosis-like enamel discolorations (Croll 1990).