Fig. 8.1
Causes of intrinsic stains
8.2.1 Systemic Causes of Posteruptive Intrinsic Stains
The developing dentition can be affected by a number of systemic factors, including metabolic and genetic diseases that cause intrinsic tooth discoloration (Plotino et al. 2008). Systemic factors also include drug-induced discolorations, including those from the intake of antibiotics and excess fluoride. In the case of fluoride its source can be either iatrogenic or from natural sources, such as drinking water.
Although most publications refer to aging as a local cause of intrinsic discoloration, the physiological continuous deposition of secondary dentin throughout the life of an individual may be considered both a systemic and a local cause of intrinsic discoloration, as reactionary tertiary dentin forms in response to external stimuli, such as tooth attrition and bacteria from caries lesions. The deposition of both secondary dentin and tertiary dentin results is a narrower pulp space with time. Thicker dentin affects the light-transmitting properties of teeth, resulting in a gradual darkening of the tooth and increased opacity. In addition, the chemical structure and physical properties of dental hard tissues change over time.
8.2.2 Local Causes of Posteruptive Intrinsic Stains
Local causes of posteruptive intrinsic stains include intrapulpal hemorrhage, residual pulpal tissue after endodontic therapy, calcific metamorphosis, endodontic materials, restorative materials such as amalgam, and root resorption. Other less frequent causes are trauma to developing teeth and periapical infection of primary teeth (Hattab et al. 1999). Besides being responsible for posteruptive pigmentation of vital teeth (Chap. 6), the antibiotic minocycline also causes discoloration of nonvital teeth. In one case, minocycline was utilized in the root canal as a component of antibiotic paste resulting in blue discoloration of immature necrotic permanent teeth in children (Dabbagh et al. 2002). In another case, minocycline was applied as a component of a triple antibiotic mixture inside a root canal of a tooth with a necrotic pulp, as an attempt to disinfect the root canal system for revascularization. Minocycline resulted in tooth discoloration 6 weeks after the triple antibiotic paste had been applied (Kim et al. 2010).
Necrotic pulps may cause tooth discoloration as a result of the decomposition of the pulpal tissues producing colored byproducts that infiltrate the dentinal tubules. Discoloration related to improper endodontic treatment may be caused by trauma inflicted during pulp extirpation or obturation materials left in the pulp chamber. Another cause of discoloration in endodontically treated teeth is the residual pulp tissue left in the pulp horns when the access to the pulp chamber is under-prepared (Brown 1965; Faunce 1983). Evidence suggests that stains in endodontically treated teeth are not just confined to the pulp chamber, but they penetrate into the dentin substrate showing through dentin and enamel (van der Burgt and Plasschaert 1986).
Fitch (1861) stated, “the disintegration of the red corpuscles of the blood is another source of discoloration. The hematine, or iron, which is supposed to constitute the pigment of these globules, passes readily into the tubules of the dentine from the pulp chamber of the tooth, whenever the red disks are disintegrated, and the discoloration becomes more or less permanent. These red globules may be dissolved or broken up.” Spasser (1961) reported that “in many cases, the hemolysis of red blood cells with the release of hemoglobin, especially when associated with hemorrhage following pulp extirpation or trauma” was responsible for the discoloration of endodontically treated teeth. More recently other authors have corroborated this theory. Diffusion of blood components into the dentinal tubules caused by pulp extirpation or traumatically induced internal pulp bleeding is one of the causes for discoloration of nonvital teeth (Arens 1989). Products of blood (erythrocytes) decomposition, such as hemosiderin, release iron. The iron can be transformed to ferric sulfide with hydrogen sulfide produced by bacteria, which causes a dark brownish discoloration of the tooth (Brown 1965; Glockner et al. 1999; Attin et al. 2003). Other end products of hemoglobin decomposition, biliverdin and bilirubin, are themselves known causes of color alteration in the skin and mucosae, being responsible for jaundice in some systemic diseases (Shibahara et al. 2002; Leuschner 2003).
Coronal discoloration of endodontically treated teeth may also be caused by endodontic sealers (Fig. 8.2), such as Grossman’s cement and AH26 silverfree (Dentsply Caulk) (van der Burgt and Plasschaert 1985; van der Burgt et al. 1986). Temporary restorative materials, such as classical ZOE, Cavit (3M ESPE), and I.R.M. (Dentsply Caulk), also result in discoloration of endodontically treated teeth (van der Burgt et al. 1986). Two mineral trioxide aggregate (MTA)-based endodontic materials, ProRoot MTA (Dentsply Tulsa Dental Specialties) and MTA Angelus (Angelus Indústria de Produtos Odontológicos), have also been reported to cause tooth discoloration when used over a period of 12 weeks (Jang et al. 2013). The discoloration was observed at the MTA-dentin interface and intracoronal dentin surface. Removal of the discolored MTA resolved the discoloration.
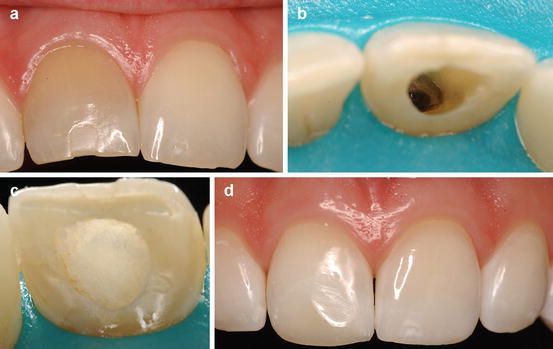

Fig. 8.2
Clinical case of a recent discoloration (a) caused by endodontic sealer (b) in the pulp chamber. This tooth lighten with the walk bleach technique using sodium perborate mixed with distilled water after only one session (c, d)
8.3 Treatment Plan Considerations
When referring to whitening of nonvital teeth, Salvas (1938) wrote, “bleaching teeth is, at best, more or less of an unsatisfactory operation. Although we may succeed in restoring the color of a tooth, it is seldom permanent.” The unpredictability of intracoronal whitening has not changed considerably since Salvas wrote these statements in 1938.
Taking into consideration that the treatment outcome as well as the durability of the treatment varies for each clinical situation, the patient must be informed of these inherent limitations of intracoronal whitening. Another detail that the patient must be aware of is that root dentin does not respond well to bleaching, either external or internal (Kwon 2011). This is especially significant in case there is gingival recession that has resulted in exposed root surface.
The indications and contraindications for intracoronal whitening are displayed in Table 8.1. Prior to starting an internal whitening procedure to lighten single or multiple teeth, it is crucial to understand all treatment options available for each specific clinical case. The prognosis of internal whitening depends on several factors, including the etiology and the duration of the discoloration. Additionally, a history of traumatic injury to the tooth or teeth may be associated with subsequent external cervical resorption. It is also important to understand that the intrinsic discoloration in endodontically treated teeth affects dentin but not enamel (van der Burgt et al. 1986). A precise diagnosis relies on accurate clinical and radiographic exams. The presence of craze lines on the enamel structure and marginal microgaps in existing restorations may result in seepage of the intracoronal bleaching agents to the surrounding tissues. For this reason, the use of transillumination to diagnose microgaps and craze lines is extremely important. The treatment plan must be based on existing evidence to improve the chance of clinical success.
Table 8.1
Indications and contraindications for internal whitening of endodontically treated teeth
|
Indications
|
Contraindications
|
|---|---|
|
Discolorations from pulpal trauma or pulp remains
|
Inadequate root canal treatment
|
|
Discolorations that do not respond to external bleaching techniques
|
Untreated caries lesions and abfraction lesions
|
|
Dentin discolorations of various origins, including endodontic sealers
|
Loss of coronal tooth structure that prevents sealing of the bleaching material inside the pulp chamber
|
|
Defective restorations or enamel craze lines that may result in seepage of the bleaching material to the periodontal tissues
|
|
|
Patient’s high expectations
|
|
|
Pregnancy
|
|
|
Discoloration caused by oxidation of metals (silver, amalgam)
|
|
|
Discolorations restricted to enamel
|
Intraoral photographs are essential to document the baseline color. Although the color of darkened endodontically treated teeth is not usually within the range of universal shade guides, the clinician may still take a photograph with a tooth tab from the Vita Classical A1-D4 shade guide (VITA Zahnfabrik H. Rauter GmbH & Co. KG) next to the darkened tooth. This will provide an excellent comparative reference for the postoperative versus the preoperative color.
8.4 Whitening Techniques for Endodontically Treated Teeth
Superoxol used to be the most commonly used intracoronal bleaching material. More recently, water has been used (instead of hydrogen peroxide) mixed with sodium perborate (Ari and Ungör 2002; Plotino et al. 2008). Sodium perborate (NaBO3) is a white crystalline powder that contains about 95 % of the perborate corresponding to 9.9 % available oxygen (Rotstein and Friedman 1991). In contact with water, sodium perborate decomposes with the liberation of hydrogen peroxide and later of oxygen. Sodium perborate is, therefore, a hydrogen peroxide precursor (Ari and Ungör 2002). Warm air and acidic solutions also initiate the decomposition of sodium perborate. A thick creamy paste of sodium perborate with water is currently recommended, left in the pulp chamber for varying periods of time.
Two techniques for bleaching endodontically treated teeth have been used through the years, namely, the thermocatalytic technique and the walking bleach technique. Other bleaching techniques include combined thermocatalytic and walking bleach techniques (Freccia et al. 1982).
8.4.1 Thermocatalytic Technique
In the thermocatalytic technique, heat is used to activate the release of nascent oxygen from the oxidizing agent or agents, often hydrogen peroxide and sodium perborate (Ari and Ungör 2002). Several sources of heat have been utilized as adjunct activation methods, including ultraviolet (UV) lights, infrared lights, flamed instruments, and electrical sources of light and heat. Fischer (1911) reported heating hydrogen peroxide with a special mercury arc light with a quartz lens, or Kromeyer’s lamp, to irradiate the teeth with ultraviolet (UV) rays to mimic the sunlight. Prinz (1924) recommended using heated solutions consisting of sodium perborate and Superoxol for cleaning the pulp cavity. Dietz (1957) recommended a 20-in. infrared light for the thermocatalytic technique in case only one appointment was feasible. In 1963, the use of 30 % hydrogen peroxide with a source of light and heat from a distance of 5 cm was reported (Weisman 1963).
In light of the risk of side effects from heating hydrogen peroxide in the pulp chamber space (Madison and Walton 1990), including external cervical resorption, the thermocatalytic technique is no longer advocated.
8.4.2 Walking Bleach Technique
For the walking bleach technique, hydrogen peroxide or water is mixed with sodium perborate, but heat is not used to trigger the release of nascent oxygen. The use of an intracoronal mixture of sodium perborate mixed with Superoxol or water has been described as a successful technique (Kirk 1893; Nutting and Poe 1967; Rotstein et al. 1993a). Saline and anesthetic have also been used instead of water (Madison and Walton 1990). Propylene glycol, which is largely used as in the pharmaceutical and food processing industry as a humectant and preservative, has also been suggested in some countries as a vehicle for the intracoronal bleaching paste in lieu of water. There is, however, no evidence that propylene glycol is more effective than water to mix with sodium perborate for internal whitening.
The first description of the walking bleach technique using a mixture of sodium perborate and distilled water was published in 1938 (Salvas 1938). The mixture was left in the pulp space for a few days, while the access cavity was sealed with provisional cement. This same technique was revived in 1961 (Spasser 1961) and modified in 1967 (Nutting and Poe 1967), when 30 % hydrogen peroxide was used instead of water to improve the bleaching effectiveness of the mixture. Although 30 % hydrogen peroxide may boost the efficacy of internal whitening, the procedure can be successfully accomplished without hydrogen peroxide (Salvas 1938; Spasser 1961).
Clinically, the walking bleach technique of sodium perborate mixed with water has been reported to be effective. In 57 out of 95 teeth (60 %), a good or acceptable result was obtained after one or two visits (Holmstrup et al. 1988). The remaining 38 teeth (40 %) were treated over 3–9 visits. The method used sodium perborate moistened with water and the access cavity was temporarily restored with Cavit (3M ESPE) between visits. While 55/69 (79.7 %) of the teeth examined at 3 years still had a good or acceptable result, 14 teeth (20.3 %) showed recurrence of discoloration that was considered unacceptable. The total number of teeth with an unacceptable result either initially or after 3 years was 25 % of the treated teeth. Despite recurrence, all examined teeth showed less discoloration than before bleaching. Interestingly, it has been reported that patients treated with the walking bleach technique brush more often than other patients, developing a superior dental hygiene regimen (Abou-Rass 1988).
In vitro and clinical studies have shown that three applications of sodium perborate mixed with water are equally effective as applying sodium perborate and the 30 % hydrogen peroxide solution (Rotstein et al. 1991c; Rotstein et al. 1993a; Holmstrup et al. 1988).
With the advent of the at-home vital whitening technique (Chap. 6), carbamide peroxide (also known as urea peroxide) has been advocated for the walking bleach technique alone or in combination with sodium perborate (de Souza-Zaroni et al. 2009). Aldecoa and Mayordomo (1992) suggested a 4-week treatment with 10 % carbamide peroxide mixed with sodium perborate as a “second phase” of internal whitening once the first phase of bleaching with hydrogen peroxide mixed with sodium perborate was completed. The association of carbamide peroxide with sodium perborate is more effective than that of water with sodium perborate (Yui et al. 2008). Another study reported contradictory findings, as the sodium perborate mixed with 37 % carbamide peroxide association proved to be as effective as sodium perborate mixed with distilled water for intracoronal bleaching (de Souza-Zaroni et al. 2009).
As several concentrations of carbamide peroxide gels are available for the at-home whitening technique, the use of 10, 17, or 37 % carbamide peroxide has been studied regarding the ensuing infiltration of peroxide into radicular dentin. All three concentrations resulted in significantly lower penetration of peroxide into radicular dentin than a combination of Superoxol with sodium perborate. However, 37 % carbamide peroxide resulted in significantly more penetration of peroxide that 10 or 17 % carbamide peroxide (Gokay et al. 2008).
Although there are different materials currently available for the walking bleach technique, the combination of water with sodium perborate is still preferred, as it may reduce the risks of side effects.
8.4.3 Combined Techniques
Freccia et al. (1982) stained extracted teeth with blood and then compared the results of three nonvital bleaching techniques: thermocatalytic technique, walking bleach, and a combined technique. Although the techniques were equally effective in bleaching blood stained teeth, the walking bleach technique consumed the least operator time.
8.5 Factors That Influence the Prognosis of Intracoronal Whitening
8.5.1 Duration of Discoloration
The prognosis may depend on the duration of the discoloration (Brown 1965; van der Burgt and Plasschaert 1986). Brown (1965) reported that the shorter the time the tooth has been discolored, the more successful bleaching is. Teeth that had been discolored for less than 1 year had an 87.5 % success rate, while for teeth that had been discolored over 5 years, the success rate dropped to 66 %.
8.5.2 Intensity of Discoloration
Severely discolored teeth have less chance of successful bleaching (75 %) than teeth with moderate or slight discolorations (90–100 %) (Brown 1965). Gradual discolorations tend to bleach more efficiently than rapid discolorations.
8.5.3 Potential for Color Regression
Some color relapse may occur in about 50 % of bleached teeth after 1 year, and even more after a longer period (Brown 1965; Howell 1981). Teeth that are more difficult to bleach are more likely to discolor again (Howell 1981). Specific endodontic sealers result is higher risk of color relapse than others (van der Burgt and Plasschaert 1986). The probability of color reversal is much higher when the discoloration is due to metallic stains or silver-containing medicaments (Freccia et al. 1982).
Brown (1965) performed a survey on 80 teeth that had been bleached using 30 % hydrogen peroxide, and compared standardized photographic records of the teeth taken before and after bleaching, and at intervals of l–5 years. The author found that 20 (25 %) teeth failed, of which 14 teeth failed to respond to treatment at all, while in 6 teeth the color relapsed after initially successful bleaching. Of the 60 (75 %) successfully bleached teeth, 23 showed no postoperative change, but in 37 teeth there was some color regression.
Stability of nonvital discolored teeth subjected to combined thermocatalytic and walking bleach intracoronal techniques was evaluated at 16 years (1989–2005) (Amato et al. 2006). The series comprised 50 patients (age range 7–30). After 16 years, 35 cases were evaluated. In 22 of these cases (62.9 %) the color had remained stable and was similar to that of adjacent teeth, indicating a successful outcome of the combined bleaching technique. There were 13 cases (37.1 %) classified as failures because of marked color relapse. Radiographically none of the cases re-examined underwent internal or external root resorption. However, there is insufficient evidence in terms of efficacy and safety to substantiate the use of the combined technique over the walking bleach technique.
8.5.4 Patient’s Age
The success rate of the internal whitening technique is 50–90% without a direct relationship between success and patient’s age (Brown 1965; Howell 1981). However, other authors have stated that young teeth bleach faster than old teeth because the dentinal tubules are wider in younger teeth (van der Burgt and Plasschaert 1986).
Dietz (1957) reported a direct relation between the age of a tooth and its resistance to bleaching, suggesting that more permanent results were obtained with teeth of the older age groups. Camps et al. (2007) evaluated the diffusion of hydrogen peroxide through human dentin in patients under 20 years old and in patients between 40 and 60 years old. The teeth were endodontically treated, and a defect was created at the CEJ. The access cavities were filled with 20 % hydrogen peroxide gel. The amount of diffusing hydrogen peroxide was assessed at 1, 24, 48, and 120 h. Diffusive flux and maximal diffusion were higher through young teeth than through old teeth.
8.5.5 Type of Discoloration
The prognosis for success of any bleaching technique depends on the cause of the discoloration (Freccia et al. 1982). When the discoloration is a result of products of pulpal decomposition within the dentinal tubules, the prognosis is usually very good. Recent discolorations from endodontic sealers are also easy to whiten (van der Burgt and Plasschaert 1986) (Fig. 8.2). When the discoloration is due to metallic stains or silver-containing medicaments, bleaching is more difficult and it is sometimes not possible to achieve satisfactory results. In fact, internal discoloration caused by oxidation of metals (silver, amalgam) cannot be removed by whitening treatments (Attin et al. 2003).
8.6 Adverse Effects
Hydrogen peroxide is the main toxic substance that has been used for internal bleaching (Chaps. 4 and 5). Hydrogen peroxide is a reactive oxygen species (ROS) (Bax et al. 1992) with oxidative ability (Kashima-Tanaka et al. 2003), as seen in Chap. 3. It is well known that free radicals and ROS exert biological actions such as inflammation, carcinogenesis, aging, and mutation (Valko et al. 2007). ROS also play an important role in tissue injury at sites of inflammation in various diseases (Valko et al. 2007).
8.6.1 External Cervical Resorption (ECR)
Table 8.2 displays a summary of the findings in clinical studies of internal whitening and the reported number of external cervical resorption (ECR) cases. ECR is the loss of tooth hard tissue as a result of odontoclastic activity (Patel et al. 2009). Figure 8.3 shows cone-beam computed tomography images of ECR in endodontically treated teeth that had been unsuccessfully whitened internally prior to the restorative procedures.
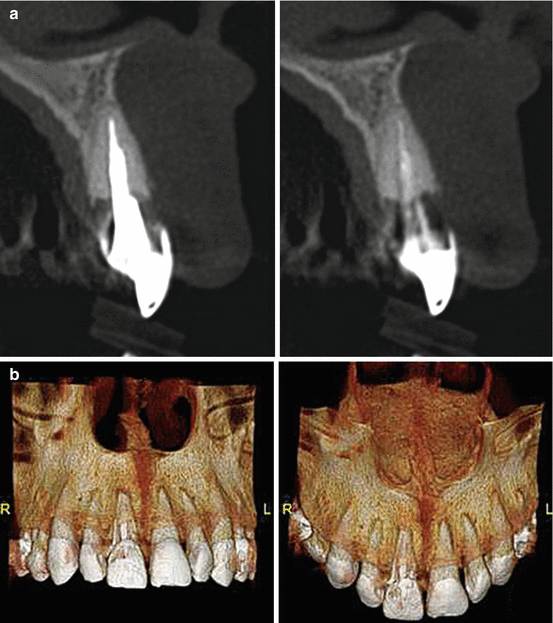
Table 8.2
Clinical studies of internal whitening
|
Number of teeth
|
ECR
|
History of trauma
|
Outcome
|
|
|---|---|---|---|---|
|
Harrington and Natkin (1979)
|
4 central incisors were root canal treated in 4 patients whose age ranged from 11 to 15 years of age. In 3 of the 4 cases, the thermocatalytic bleaching technique was carried out from 6 to 15 years after the trauma and completion of root canal therapy
Same authors reported 3 extra cases that developed ECR
|
All with ECR; all resorptive lesions occurred in the cervical third of the root
|
All 4 teeth
|
No reported outcome
|
|
Cvek and Lindvall (1985)
|
11 teeth with ECR after bleaching with 30 % H2O2
|
2 teeth – superficial ECR that did not progress;
5 teeth – ECR followed by ankylosis. 4 teeth – ECR was progressive and associated with radiolucency in the adjacent alveolar bone
|
10/11, when patients were 11–16 years old
|
|
|
Abou-Rass (1988)
|
112 severely tetracycline-stained teeth in 20 patients were root canal treated and internally bleached with a thick paste of sodium perborate in 30 % H2O2. Procedure was repeated after 1 week if needed.
|
No report of ECR after 3–15 years
|
No history of trauma
|
7 % failure – 8/112 teeth were noticeably dark at the cervical zone
Intracoronal restorative failures were relatively high (7 %), endodontic failure was only 2 % and there was no evidence of external root resorption
|
|
Friedman et al. (1988)
|
58 bleached teeth were re-examined after 1–8 years
|
4/58 (6.9 %); resorption started apically; 2 teeth had advanced ECR; 2 teeth had arrested ECR, one of them had been bleached with the walking bleach technique
|
No trauma for the 4 teeth with ECR
|
43 teeth (74 %) were bleached once, 15 teeth (26 %) were more than once; all teeth bleached with 30 % H2O2; 29/58 teeth (50 %) were found esthetically satisfactory; 17/58 teeth (29 %) were clinically acceptable; 12/58 teeth (21 %) were unacceptable, of which 4 had received full-coverage restorations
|
|
Holmstrup et al. (1988)
|
95 teeth, walking bleach technique with sodium perborate moistened with water
|
No report of ECR at 3 years
|
91/95 teeth had history of trauma
|
In 57 teeth (60 %), a good or acceptable result after 1 or 2 visits. The remaining 38 teeth were treated over 3–9 visits. Satisfactory initial result in 90 % of the cases After 3 years, recurrence of discoloration was observed in 20 % of the teeth
|
|
258 intact tetracycline-stained teeth underwent elective root-canal treatment; GIC cervical barrier 1 mm below CEJ, walking bleach technique with 30 % H2O2 and sodium perborate; repeated 2–3 times every 4 weeks. A mix of 10 % carbamide peroxide + sodium perborate was then applied for 4–6 weeks
|
No report of ECR at 4 and 6 years
|
No history of trauma
|
Color relapse for 6/258 teeth (2.3 %) at the 4-year recall; 10 % of teeth after 6 years
|
|
|
Heithersay et al. (1994)
|
204 teeth were re-examined after 1–19 years; all teeth had been treated with a combination of thermocatalytic and walking bleach procedures using 30 % H2O2
|
4 teeth (1.96 %) developed invasive ECR. All of these teeth had a history of traumatic injury and the level of gutta-percha was at the CEJ without a barrier
|
151/204 (77.9 %) had history of traumatic injury
|
Not reported, as this study was based on radiographic evaluations
|
|
Glockner et al. (1999)
|
5-year clinical follow-up of teeth bleached with 30 % H2O2 with sodium perborate, walking bleach technique for 1 week; procedure repeated until satisfactory results obtained
|
Not reported
|
Not reported
|
Treatment was successful in 68 patients (79 %) after 5 years
|
|
Amato et al. (2006)
|
Thermocatalytic technique used with 35 % H2O2 and sodium perborate heated with light source; 50 teeth initially selected, 35 were evaluated at 16 years
|
None of the 13 failures had radiographic signs of ECR. However, authors stated that for the 9 teeth for which the root canal that had been re-treated, 2 of them showed fistula, pain and a peri–radicular and/or latero–radicular bone lysis area that had failed to disappear or had reappeared
|
42 of the initial 50 teeth has a history of trauma
|
22 teeth (62.9 %) the color had remained stable and was similar to that of adjacent teeth; 13 cases (37.1 %) classified as failures because of marked color relapse
|

Fig. 8.3
Cone-beam computed tomography (a) with reconstructed three-dimensional (3D) image (b) showing an ECR lesion on tooth #8 (FDI 1.1). This tooth had been treated with intracoronal whitening in two different occasions, but did not respond to the treatment. The ECR lesion was diagnosed after the tooth was restored with a cast post-and-core and a full-coverage restoration (Images courtesy of Prof. Eduardo Vilain de Melo, Florianópolis, Brazil)
It has been reported that hydrogen peroxide is the cause of dentin and cementum alterations leading to complications such as ECR (Harrington and Natkin 1979; Lado et al. 1983; Montgomery 1984). Hydrogen peroxide seeps through the dentinal tubules into the surrounding tissues causing destruction of cells, which triggers an inflammatory process that may lead to ECR. A study suggested that the combination of heat and hydrogen peroxide is responsible for ECR (Madison and Walton 1990). However, other studies reported clinical cases of ECR in which heat was not used (Goon et al. 1986; Friedman et al. 1988). Even though the walking bleach technique with sodium perborate and hydrogen peroxide is less harmful than the thermocatalytic bleaching technique, ECR may also occur with the walking bleach technique (Goon et al. 1986; Latcham 1986; Friedman et al. 1988). As hydrogen peroxide has been used in most studies that reported ECR, several authors have opposed the use of hydrogen peroxide for internal whitening to prevent ECR (Montgomery 1984; Cvek and Lindvall 1985).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


