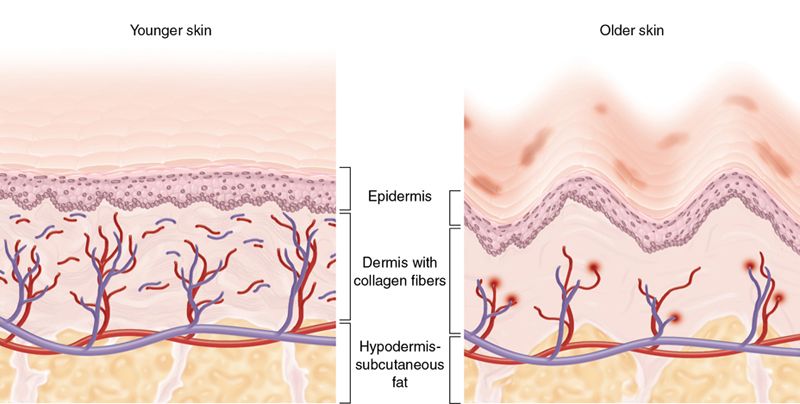
There is an ongoing loss of collagen and elasticity in the skin, resulting in the skin becoming lax.3 Collagen loss causes tissue atrophy and thinning of the skin, with increased rhytid (wrinkle) formation (Fig. 24-2). Loss of the underlying fat causes descent of the overlying structures Fig. 24-3 A).4,5
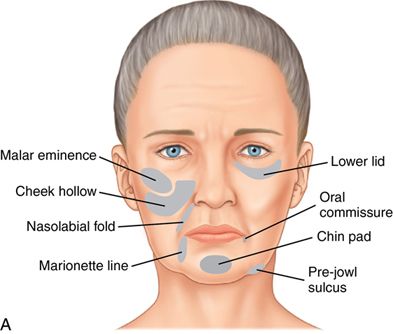
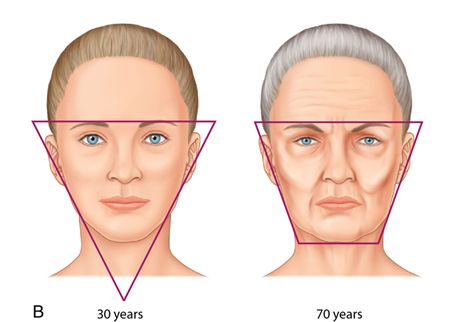
These changes lead to loss of the volume and curves of the cheeks, resulting in bony contours. Tissue descent also causes increased nasolabial and labiomandibular folds and increased “jowling” (Fig. 24-3 B).
There are two types of wrinkles: dynamic wrinkles and static wrinkles. The dynamic wrinkle is caused by animation or muscle function (Fig. 24-4 A).
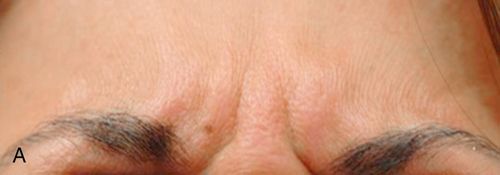
Use of botulinum toxin is appropriate for treating dynamic wrinkles because it weakens the underlying muscle and causes a “chemical denervation.” This prevents wrinkling of the overlying skin.
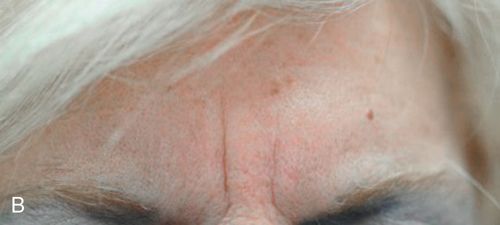
A static wrinkle occurs in the absence of facial movement when the musculature is completely relaxed (Fig. 24-4 B).
Botulinum toxin cannot be used alone to treat static wrinkles because it has little effect on wrinkles that appear when the facial muscles are completely at rest. In these cases, filler or combination therapy (with the Botulinum toxin correcting against dynamic wrinkles that can occur in conjunction with static wrinkles) would be preferable.
Botulinum toxin
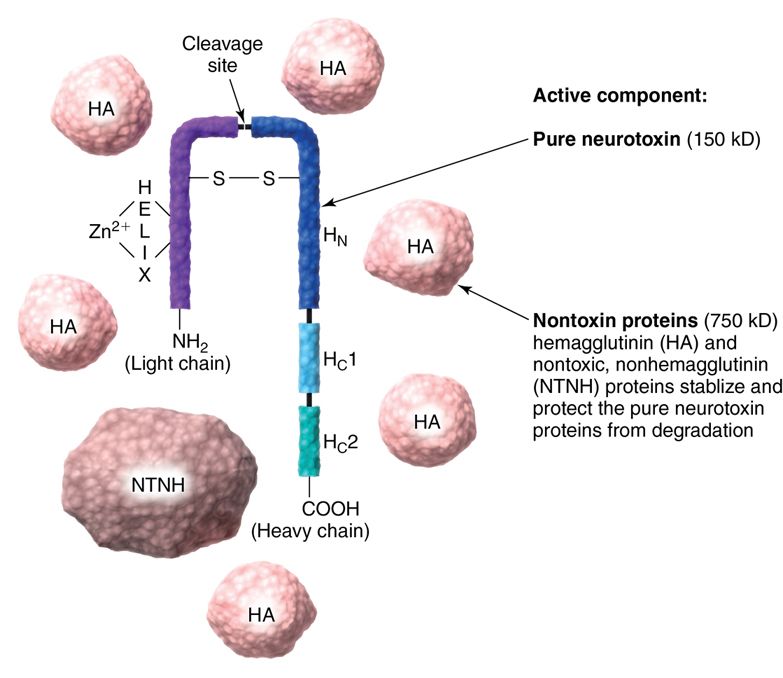
Botulinum toxin is a clear fluid medication in a lyophilized (freeze-dried) form. It contains a pure neurotoxin that is surrounded by non-toxin proteins, which are used to stabilize and protect the neurotoxin (Fig. 24-5).
The practitioner mixes the toxin with saline and injects the medication subcutaneously or intramuscularly, with the intention of weakening the target muscle. Contrary to popular belief, it does not “fill” lines, nor does it “smooth” wrinkles.
History
Botulinum toxin is derived from Clostridium botulinum, a gram-positive, spore-producing bacteria first identified by Emile Van Ermengen in 18976 when he was investigating fatal cases of food poisoning associated with sausage meat. ( Botulus is Latin for sausage.) Serotypes A, B, and E cause the classic food-borne disease, which can cause flaccid paralysis of motor and autonomic nerves.
During World War II, scientists investigated botulinum toxin as a means of chemical warfare and isolated serotype A. This research was going on simultaneously in the United States, the United Kingdom, and Russia.7 Research products first created in the United States and the United Kingdom eventually became the products Botox and Dysport, respectively.
In 1978, botulinum toxin was tested in clinical trials for strabismus.8–10 Throughout the 1980s it was used extensively to treat blepharospasm,11, 12 hemifacial spasm,13, 14 and cervical dystonia.15–17
In 1987, Jean Carruthers, an ophthalmologist, was treating her blepharospasm patients with botulinum toxin, while her husband, Alastair Carruthers, a dermatologist, noticed that the patient’s wrinkles were “disappearing” in the treated areas.18 In 2002, the FDA approved botulinum toxin type A (Botox) for cosmetic use.
Currently, three types of commercial botulinum toxin Type A are available for cosmetic use in the United States: Botox (Allergan, Inc.), Dysport (Medicis Aesthetic), and Xeomin (Merz Aesthetics, Inc.). One type of commercially available botulinum toxin Type B, Myobloc (Elan Pharmaceuticals), is licensed for the treatment of cervical dystonia. Type B toxin differs from Type A toxin by serotype and is not approved for cosmetic use.
Mechanism of action
Botulinum toxin’s effects occur at the motor end plate. Muscle contraction occurs when motor nerve terminal impulse reaches a nerve ending, which in turn releases acetylcholine across the neuromuscular gap to the muscle signaling muscle contraction. Botulinum toxin blocks acetylcholine release. This blocking of acetylcholine occurs as a four-step process (Fig. 24-6).
1. Binding. The free end of the neurotoxin molecule binds to the cholinergic receptors located on the presynaptic neuron of the neuromuscular junction.
2. Internalization. The botulinum toxin passes through the cell membrane and cytoplasm via endocytosis.
3. Release. Botulinum toxin is released into the cytoplasm.
4. Block acetylcholine release. Botulinum toxin blocks acetylcholine release by cleaving SNAP25. SNAP25 is an integral protein required for successful docking and release of acetylcholine from vesicles situated in the nerve endings.
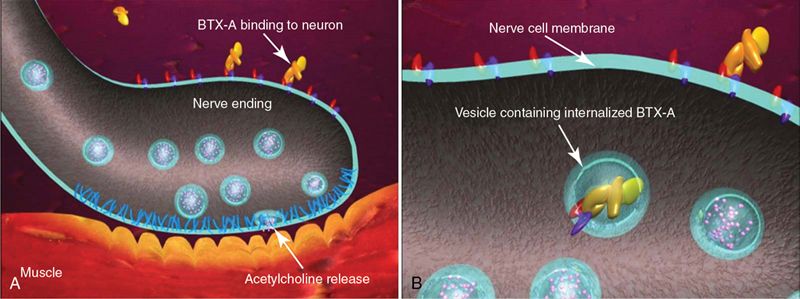
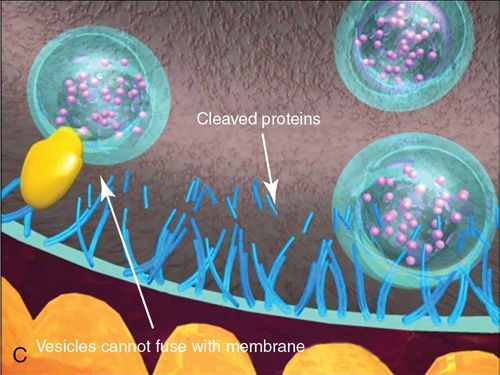
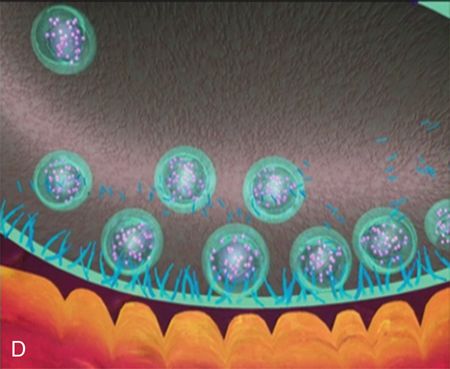
The toxin therefore causes a “chemical denervation” of the muscle. If acetylcholine cannot be released from the nerve ending, the muscle cannot contract. If the muscle cannot contract, then the overlying skin cannot wrinkle.
Wrinkle formation of the overlying skin can be compared to “Roman shades” (Fig. 24-7).

When the shade is raised, it folds over upon itself. Similarly, muscle contraction causes the overlying skin to shorten or fold over upon itself. This wrinkle is perpendicular to the direction of the muscle fibers. For example, horizontal wrinkles are caused by vertical fibers of the frontalis muscle, whereas radial wrinkles run perpendicular to the fibers of the orbicularis oculi.
Onset of action and duration
The onset of action of botulinum toxin varies, depending on the type of formulation, ranging from 24 hours to 5 days. Final results may take 2 weeks. Results should last for 3 to 4 months. Toward the end of this period, most patients will feel a gradual return of muscle function, and repeat therapy can be initiated. The repetitive use of botulinum toxin has not resulted in muscle degeneration or atrophy.19
Safety and contraindications
Examination of nearly 20 years of research reveals a high degree of safety and efficacy.20 A review of 36 randomized trials with 1425 patients revealed no serious adverse events.21
Most adverse events are reported to be transient in nature. They include lack of intended cosmetic effect, injection site reaction, ptosis (drooping of the eyelid), muscle weakness, and headache.22
There are relatively few contraindications for botulinum toxin therapy. These include muscle movement disorders (e.g., amyotrophic sclerosis [ALS], myasthenia gravis and Lambert-Eaton syndrome) and known allergy to botulinum toxin, which is extremely rare.23 Botulinum toxin is a pregnancy category C drug and therefore is not recommended during pregnancy or nursing. Caution is advised in the presence of pre-existing skin infections or rashes at the intended injection site.
Storage, preparation, and injection technique
Most types of botulinum toxin require refrigeration. The toxin is packaged in lyophilized (freeze-dried) form, in a multidose vial (Fig. 24-8 A).
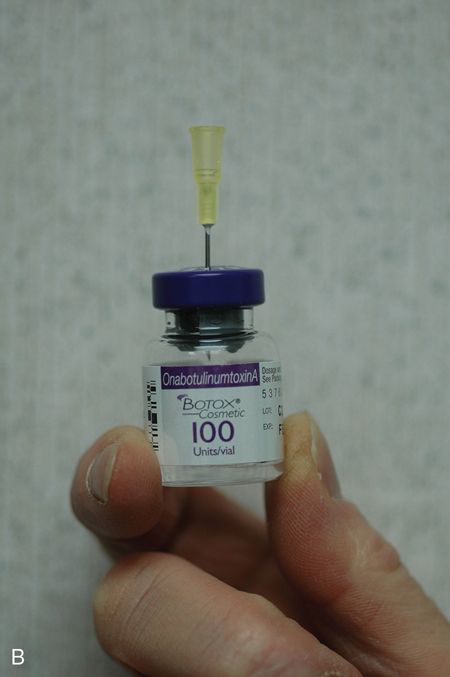
The toxin is constituted with saline before use by drawing the desired amount of saline from another vial and transferring it into the vial containing the toxin. According to the manufacturers, the toxin should be drawn up through the rubber stopper on top of the vial, and then a needle tip should be left behind to draw up future doses.
Many practitioners choose to open the multidose vial by removing the rubber stopper, aseptically drawing up the desired toxin into an insulin syringe (Fig. 24-8 C), and then recapping the vial for future use.
The insulin syringe has a very fine needle, which can make receiving the injection much more comfortable for the patient than if a larger-diameter needle were to be used. Once constituted, all neurotoxin should be stored in the refrigerator. Sterile techniques must be used as with any multidose vial and the constituted, unused neurotoxin must be used within a given time frame.
The desired units are drawn up in a syringe using a fine-gauge needle (30 or 31 gauge), and injected into the pre-marked areas. For most areas, either a subcutaneous or intramuscular injection is acceptable. However, some areas require deeper injections, to ensure that the neurotoxin will infiltrate the target muscle. No local anesthetic is required. However, for added comfort, some patients prefer to place an ice-pack on the area before injection for about 30 seconds.
Areas of treatment
Classically, botulinum toxin was used in the muscles of the upper face. The FDA approval for Botox Cosmetic reads, “indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity in adult patients ≤65 years of age.”24 Since FDA approval for cosmetic use, it has been used in all areas of the face and neck. For example, an injection can be given intraorally for a gummy smile or into the temporalis tendon muscle for TMJ pain; however, these are off- label uses.
Upper face.
In the upper face, botulinum toxin is generally used in three major areas, the forehead, glabella, and peri-orbital areas.
Forehead.
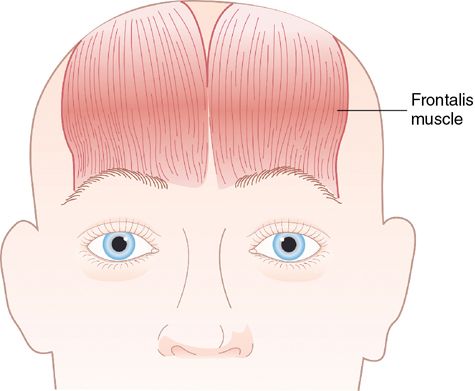
Horizontal forehead wrinkles are caused by contraction of the frontalis muscles.
The frontalis muscles are two large fanlike muscles that extend from the eyebrow region to the top of the forehead (Fig. 24-9). Using multiple low dose injections, the practitioner can relax the frontalis muscles. The key to achieving an aesthetically pleasing result is to maintain or enhance eyebrow position and movement. By relaxing the forehead wrinkles without paralyzing brow movement, the patient receives a very natural result.
Glabellar complex.
The glabellar complex is made up of two muscles, the corrugator and procerus (Fig. 24-10).
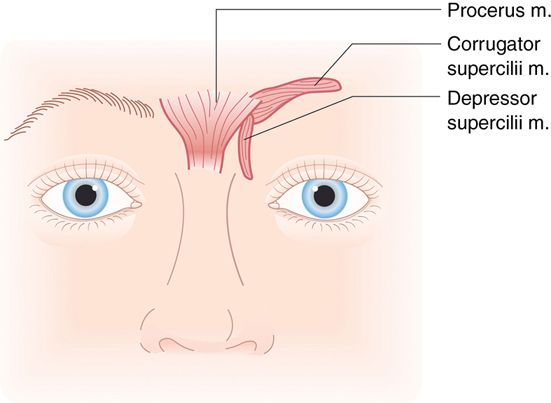
The corrugator muscle is a longitudinal muscle that goes from the medial of the orbits to the mid eyebrow, running approximately 30 degrees above horizontal. The procerus in a flat vertical muscle extending from the bridge of the nose and interdigitates with the corrugator, frontalis, and orbicularis oculi. These two muscles, working in concert, move the eyebrows downward and medial. This causes a perpendicular wrinkling of the overlying skin, that can appear as vertical fan lines (sometimes referred to as “frown lines” or “eleven lines”) over the medial eyebrows. Injecting botulinum toxin in a V pattern in this area usually relieves these wrinkles.
In many patients, the deepest of the vertical lines in the glabella complex become static lines. (After treatment with botulinum toxin, these lines remain despite the musculature being at rest.) Therefore combination therapy of botulinum toxin and filler material is required. The filler should be threaded directly into the deep wrinkles to correct them.
Periorbital area.
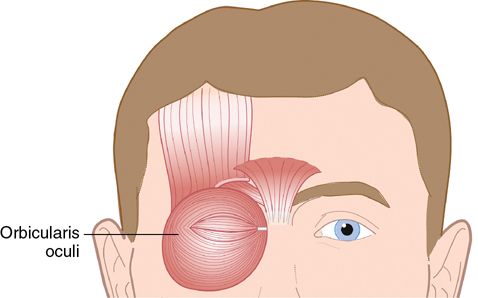
The lateral canthus area is a common area for wrinkles that form lateral to the eye with aging. These wrinkles form in a radial fashion (“crow’s feet”) because the underlying orbicularis oculi muscle is round (Fig. 24-11). Following the curve of the lateral orbital rim, three to five low-dose injections of botulinum toxin can greatly reduce or eliminate these wrinkles.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses