Fig. 7.1
Metabolic pathways in infection-driven carcinogenesis (Picture prepared by Bascones-Martinez)
7.4 Acetaldehyde Production by Oral Microorganisms
Alcohol is not carcinogenic but the first metabolite of ethanol, acetaldehyde, is highly carcinogenic. Although the liver is the organism responsible for 75–90 % of ethanol metabolism, extrahepatic pathways also exist. Ethanol is oxidized also by mucosal and microbial cells yielding acetaldehyde, formed by alcohol dehydrogenase enzyme. Acetaldehyde is further metabolized by aldehyde dehydrogenase yielding acetone which is less toxic and less harmful compound. This, in turn, is oxidized to carbon dioxide and then eliminated from the body.
Homann et al. (1997) were the first to show high levels of acetaldehyde in saliva after intake of alcohol. This group further showed that drinking alcohol and smoking concomitantly increased salivary acetaldehyde concentrations and that poor oral hygiene associated with this risk (Homann et al. 2000, 2001) (see Fig. 7.2). Later, also oral Candida species were shown to produce acetaldehyde from ethanol partly explaining why Candida infections as such have been linked to oral cancer (Nieminen et al. 2009; Uittamo et al. 2011). Moritani et al. (2015) reported that indeed considerable numbers of oral bacteria have the capability to produce acetaldehyde from ethanol. Today it seems clear that the ethanol metabolism here discussed is an evident pathologic mechanism in the development of oral and upper gastrointestinal tract cancer.
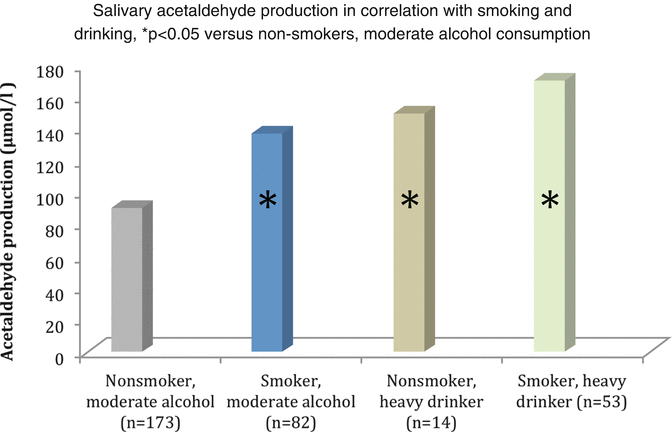

Fig. 7.2
Relationship between drinking alcohol and smoking and salivary acetaldehyde concentration (Modified from Homann et al. 2000)
7.5 Caries, Periodontitis, and Cancer
Chronic periodontitis in particular associates with oral cancer risk. In a large study from the USA comprising more than 13000 subjects, clinical attachment loss, a proxy for periodontitis, associated with the presence of tumor (OR 4.57, 95 % CI 2.25–9.30) and premalignant lesions (OR 1.55, 95 % CI 1.06–2.27), respectively (Tezal et al. 2005). Periodontitis was found to also associate with tongue cancer risk (OR 5.23, 95 % CI 2.64–10.35) (Tezal et al. 2007). Concomitant HPV infection seems to play a role in this regard too (Tezal et al. 2009).
As regards dental caries, there might be an inverse relationship between this dental disease and cancer. Tezal et al. (2013) investigated in a case–control study how cardiological status parameters link to head and neck cancer. It appeared that caries lesions showed an OR 0.55 (95 % CI 0.30–101) regarding cancer. The authors suggest that the lactobacilli prevalent in caries lesions might exert beneficial effect and enhance the immune system against cancer.
Virtanen et al. (2014), on the other hand, showed in their observational study of 1390 subjects with 24 years of duration that dental infections in periodontally healthy subjects associated with the incidence of any cancer (OR 2.62, 95 % CI 1.18–5.78). Gingivitis was also shown to link to cancer in this same Swedish cohort study (Söder et al. 2015). Furthermore, after 26 years of observation in the same study, high gingival index score associated with the incidence of any cancer with OR 1.29 (95 % CI 1.00–1.65). The statistical association between oral infections and cancer has also been observed specifically with certain types of cancer. Söder et al. (2011) found in their cohort that missing any molar tooth from the mandible associated with incidence of breast cancer with OR 2.36 (95 % CI 1.07–5.21). Missing molars were the proxy for history of dental infections. It is evident, however, that more studies are needed for final conclusion regarding the associations between caries, periodontal disease, and cancer.
7.6 Role of Saliva in the Oral Infection-Linked Carcinogenesis
Little is known about the role of saliva in oral infection-related carcinogenesis. It has been observed that salivary characteristics differ in patients with and without head and neck tumors so that saliva form those with malignancy showed more cytotoxic effect on fibroblasts than that from healthy controls (Bloching et al. 2007). Oral microorganisms may also metabolize dietary components into carcinogenic substances. Saliva may contain nitrosamines, for example (Bahar et al. 2007). However, more studies are called for before any conclusions can be drawn.
Conclusion
The highly prevalent oral infections and dental diseases in particular pose threat to systemic health if not diagnosed in time and properly treated. Studies have shown that statistical associations exist between chronic dental infections and development of cancer. Head and neck and upper gastrointestinal tract cancer may be affected by direct oral microbial metabolism leading to carcinogenic substances thus triggering malignant development in the tissue. But malignancy in any organ may in fact be affected by infection.
References
Bloching MB, Barnes J, Aust W, Knipping S, Neumann K, Grummt T, Naim R. Saliva as a biomarker for head and neck squamous cell carcinoma: in vitro detection of cytotoxic effects by using the plating efficiency index. Oncol Rep. 2007;18:1551–6.PubMed
Chang AH, Parsonnet J. Role of bacteria in oncogenesis. Clin Microbiol Rev. 2010;23:837–57.PubMedCentralCrossRefPubMed
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


