Neurologic Evaluation and Management
A good neurologic examination, in combination with a thoughtful battery of tests, will invariably achieve the correct diagnosis. It is vital for the surgeon to become equally comfortable with the rapid assessment of the trauma patient and the potential for neurologic involvement. Once initial stabilization of the patient has been achieved, management of the neurologic injury is determined by specific causative conditions. Rapid recognition of potentially reversible causes (e.g., basilar artery occlusion [BAO], nonconvulsive status epilepticus, herniation), followed by the appropriate emergency consultations, can often prevent or reduce morbidity and mortality.1–3
Initial Assessment
Resuscitation: Initial Trauma Management
Surgeons’ recognition of these patterns of injury have led to the development of the Advanced Trauma Life Support (ATLS) approach by the American College of Surgeons. ATLS is the standard of care for trauma patients; it is built around a standardized protocol for patient evaluation. This protocol ensures that the most immediate life-threatening conditions are actively identified and addressed in the order of their risk potential. The objectives of the initial evaluation of the trauma patient are as follows: (1) to stabilize the trauma patient; (2) to identify life-threatening injuries and to initiate adequate supportive therapy; and (3) to organize definitive therapy or transfer to a facility that provides definitive therapy efficiently and rapidly.4–7
A, airway maintenance with cervical spine protection
C, circulation with hemorrhage control
On completion of the primary survey, resuscitation efforts are well established, the vital signs are normalizing, and the secondary survey is begun. This may occur during transport or on arrival at the hospital. The secondary survey consists of a head to toe systematic assessment of the abdominal, pelvic and thoracic areas, complete inspection of the body surface to find all injuries, and a neurologic examination (Table 8-1). It also includes a complete history and physical examination, including the reassessment of all vital signs. Each region of the body must be fully examined. The purpose of the secondary survey is to identify all injuries so that they may be treated. A missed injury is one that is not found during the initial assessment (e.g., as a patient is brought into a hospital’s ED), but rather manifests at a later point in time. Radiographs and further imaging indicated by examination are obtained. If at any time during the secondary survey the patient deteriorates, another primary survey is carried out because a potential life threat may be present. The person should be removed from the hard spine board and placed on a firm mattress as soon as reasonably feasible because the spine board can rapidly cause skin breakdown and pain whereas a firm mattress provides equivalent stability for potential spinal fractures.6
History
More than 3 million facial injuries occur in the United States each year. Maxillofacial injuries are commonly encountered in emergency and trauma settings, with most of them being secondary to assaults and motor vehicle accidents (MVAs). Information about the causes of facial fractures depends on the country and location of the trauma center; therefore, reported statistics vary widely. More than 50% of patients with these injuries have multisystem trauma requiring coordinated management among ED physicians, trauma surgeons, maxillofacial trauma surgeons, and other medical and surgical specialists as required for the best outcome for each patient.8,9
Trauma to the maxillofacial anatomy mandates special attention. The maxillofacial region is a complex area that controls specialized functions, including seeing, hearing, smelling, breathing, eating, and talking. Also, the vital structures in the head and neck region are intimately associated. It is also of great importance to remember that the psychological impact of disfigurement can be devastating. The associated head injures and damage to the neurologic system are important to identify early in a patient who has sustained facial trauma. It is imperative that the maxillofacial surgeon be scrupulous during the initial history taking and physical examination of the patient to avoid any ill-intended oversight of an injury. Once the initial stabilization has been performed, a detailed history and complete physical examination are essential.10,11 If the patient is unable to give an accurate history, witnesses to the injury must be questioned. Essentials of the history that must be ascertained through questioning include the following:
1. Was there a witnessed loss of consciousness following the injury? How much time was involved?
2. Was there a lucid interval immediately after the injury? And, did this precede a loss of consciousness?
3. Has there been a change or deterioration in the patient’s mental status?
4. Did the patient have any seizures or syncopal events following the injury? Was there tonic deviation of the eyes toward one particular side?
5. Was there associated spinal column trauma? Did the patient complain of tenderness along the spine?
Evaluation
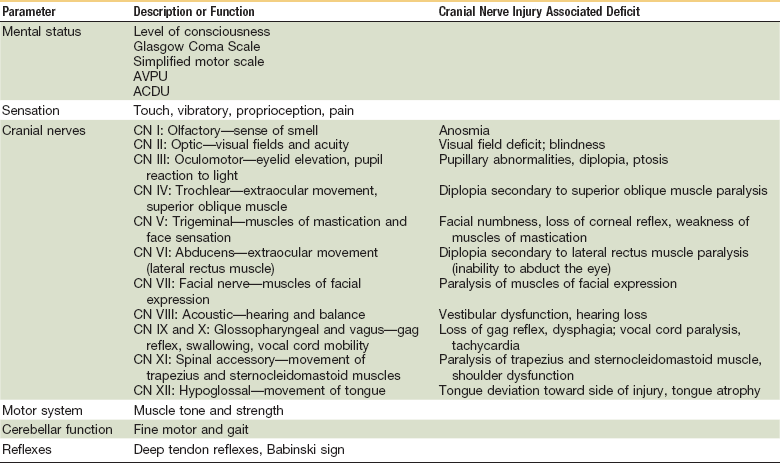
Mental Status
Mental status alterations involve changes in alertness, cognition, and behavior. When referring to an individual’s alertness, one is indicating a person’s ability to interact with his or her environment. Cognition is how one processes and integrates information and behavior refers to how we react to the environment. An altered mental status could be caused by a traumatic injury, a manifestation of a primary neurologic condition, an acute systemic process, or a metabolic imbalance. During the clinical examination, it is important to distinguish among memory problems, disorders of thought, and an altered arousal or level of consciousness. The presence of memory problems or disorientation is indicative of a medical or neurologic illness, whereas thought disorders suggest a psychiatric illness.12 A depressed level of consciousness represents failure of global brain function (Table 8-2).13
TABLE 8-2
Terms Describing Altered Mental Status
| Term | Definition |
| Coma | State of total unresponsiveness |
| Stupor | State of severely impaired arousal with some unresponsiveness to vigorous stimuli |
| Obtundation | Decreased arousal state with some responsiveness to touch or voice |
| Lethargy (somnolence) | Decreased arousal state maintained spontaneously or with light stimulation |
| Confusion | State of impaired attention but adequate arousal |
| Delirium | Acute state of confusion with periods of agitation and sometimes hypervigilance, active irritability, and hallucinations; typically, alternating periods during which the level of arousal is depressed |
| Dementia | Insidious onset of progressive and irreversible decline in intellectual function; patients may experience also superimposed delirium, normal arousability, alertness |
An immediate assessment of the patient’s level of consciousness should be made in the trauma patient, because a decrease could represent an emergent condition. A patient with a normal level of consciousness is one who is awake, alert, and aware.14 Quick assessment tools such as the Glasgow Coma Scale (GCS), AVPU, ACDU, and Simplified Motor Score (SMS; Table 8-3) can be used to rule out conditions that warrant immediate neurosurgical intervention but are not a substitute for a formal neuropsychiatric evaluation when it has been determined that it is indicated. It is also important to know the patient’s baseline functional status, any history of altered behavior, current medications, medical comorbidities, prior psychiatric history, and any relevant circumstances that preceded the trauma event. The assistance of family members to obtain this pertinent history might be necessary in the trauma patient secondary to the presence of an artificial airway, sedation, or altered level of consciousness. An assessment of orientation, attention, memory, and executive function forms the foundation of the comprehensive cognitive mental status examination.15
TABLE 8-3
Neurologic Scales for Assessment of Level of Consciousness
| Scale | Score |
| SIMPLIFIED MOTOR SCALE | |
| Obeys commands | 2 |
| Localizes pain | 1 |
| Withdrawal to pain or less response | 0 |
| AVPU | |
| Alert | A |
| Responds to verbal stimuli | V |
| Responds to painful stimuli | P |
| Unresponsiveness to all stimuli | U |
| ACDU | |
| Alert | A |
| Confused | C |
| Drowsy | D |
| Unresponsive | U |
| GLASGOW COMA SCALE | |
| Eye Opening | |
| Spontaneous | 4 |
| To voice | 3 |
| To pain | 2 |
| None | 1 |
| Verbal Response | |
| Oriented, normal conversation | 5 |
| Confused conversation | 4 |
| Words but not coherent | 3 |
| Incomprehensible sounds | 2 |
| None | 1 |
| Motor Response | |
| Obeys commands | 6 |
| Localizes to pain | 5 |
| Withdraws to pain | 4 |
| Abnormal flexion (decorticate posture) | 3 |
| Abnormal extension (decerebrate posture) | 2 |
| None | 1 |
The GCS score is the most commonly used tool for the assessment of altered levels of consciousness. It also guides therapeutic and diagnostic decision making and allows estimation of prognosis in TBIs.16 It was developed in 1974 to improve communication among health care providers caring for patients with brain injuries. The current 15-point scale was published in 1976,17 with the modification of the best motor response as having six levels and the division of flexion into two categories. Its three components assess the function of the cerebral cortex and the upper brainstem but do not assess cognition or differentiate delirium from dementia. Sedation and endotracheal intubation can affect its predictive value of the severity of the brain injury, need for neurosurgical intervention, and long-term outcome. The GCS was originally established for in-hospital use; however, it has been demonstrated that there is a correlation between field GCS scores and patient outcome and the need for neurosurgical intervention.18 Reporting each GCS component in conjunction with the total score provides a better estimate of prognosis, because up to 38 combinations with 13 different sum scores are possible.19–21 There is variation in scoring according to providers and institutions, which can lead to unnecessary referrals and neurosurgical interventions.22 The neurologic assessment should be repeated multiple times, with each interval documented to obtain reliable clinical information about the patient’s neurologic status. The best eye opening, best verbal response, and best motor response are reported using one of two schemes, EVM or EMV. The former is recommended, because it follows a logical clinical evaluation; the patient is asked to open the eyes and a verbal response is prompted, followed by a motor response.22
AVPU is an acronym used in ATLS. AVPU scores roughly correlate to a GCS score of 15, 13, 8, and 6 respectively.23 As with the GCS, the AVPU system does not differentiate causes of altered mental status. The ACDU mnemonic was devised in Sweden. In a study comparing it to AVPU and the GCS scale in neurosurgical patients, ACDU scores roughly correlated to a GCS score of 15, 13, 10, and 6.24 This study also found that the AVPU scale was not as effective for detecting early changes in level of consciousness and that the ACDU was easier to use, in particular in patients with small decreased level of consciousness. As such, it could be considered for use as a screening tool for ward patients.
The SMS was developed from the motor component of the GCS because it was thought that the individual parts of the GCS score were as predictive of outcome as the GCS score as a whole. The motor component alone of the GCS appears to be as sensitive as the GCS total score when predicting the severity of head injury in trauma patients and their risk of dying.25,26 The SMS scale is as follows: obeys commands (2), localizes pain (1), and withdrawal to pain or less response (0). The three-point SMS demonstrates almost identical discriminatory value when compared with the six-point motor component of the GCS, emphasizing the importance of the 4 and 6 range in the GCS in predicting the need for intubation, neurosurgical intervention, and outcomes in TBI.27,28 Furthermore, it has been found to have better interrater reliability than GCS, AVPU, and ACDU in patients presenting to the ED with an altered level of consciousness of traumatic or nontraumatic cause.29
Cranial Nerve Examination
Cranial nerve injury is an important component of neurotrauma, which may not be readily apparent in the emergency room to which the patient is brought after having sustained a maxillofacial injury. The incidence of cranial nerve injury in craniocerebral trauma varies between 5% and 23%.30 The initial evaluation includes a detailed history and physical and neurologic evaluation, in which sensory and cortical function and hemodynamic status are assessed. It is also important to evaluate the ocular movements, pupils, facial symmetry, and laryngeal function. This will help determine the functional integrity of the cranial nerves. A detailed evaluation of each individual cranial nerve is a time-consuming and elaborate exercise, even in a conscious patient, and is not feasible in a comatose patient. Injury to the cranial nerves can occur by shearing forces, rapid acceleration or deceleration, injury to the skull base, and penetrating craniocerebral injuries.
Olfactory Nerve
Head injury is the most common cause of injury to the olfactory nerve31,32 and the olfactory nerve is the most common cranial nerve damaged in head injury.33 Diagnosis of post-traumatic anosmia may not be of importance in immediate management. However, it may assume medicolegal importance for the patient in the future. Olfaction is impaired because of injury to the olfactory nerve filaments at the cribriform plate or because of injury to the olfactory bulb or olfactory tracts. The olfactory nerve filaments can get torn by fractures involving the cribriform plate of the ethmoid bone. Frontal or occipital blows can shear off the olfactory filaments through a shearing or stretching mechanism of the nerve fibers and any fractures in the area can lacerate the filaments. Olfaction can also be impaired by edema, hematoma, or ischemia.
Closed head injury can produce impairment of olfactory recognition despite preserved olfactory detection, secondary to injury to the orbitofrontal and temporal lobes.34 Olfactory nerves and tracts can also be injured iatrogenically during surgical interventions involving the anterior skull base. Olfactory nerve damage is suspected in the presence of nasal bleed, periorbital ecchymosis, proptosis, and cerebrospinal fluid (CSF) rhinorrhea in the setting of craniocerebral injury. Clinically, patients often complain of an altered taste sensation.35 In TBI with olfactory nerve dysfunction, almost 40% recover their olfaction. Most of these patients recover it within 3 months, and may continue up to 2 years.36 In injuries in which there is severe disruption, spontaneous reconnection of the nerve fibers is not possible, making anosmia permanent.37
Optic Nerve and Chiasm
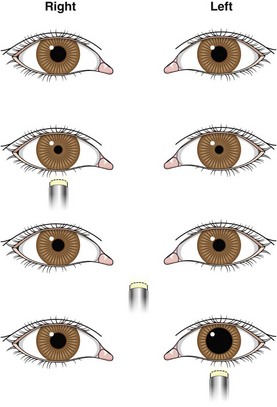
Optic nerve injury secondary to trauma has been described as a rare injury,38 with only a few series reported in the literature.39–41 Loss of vision is the chief complaint of a conscious patient with an optic nerve injury. A history of loss of consciousness is elicited in almost 90% of cases of optic nerve damage40 and the presence of an associated epistaxis (Maurer’s triad—head injury, epistaxis caused by traumatic pseudoaneurysm of the internal carotid artery, uniocular blindness) should alert the clinician to the possibility of an accompanying optic nerve injury.42,43 Diagnosis of optic nerve injury is difficult in patients with severe head injury with altered sensorium. An afferent pupillary defect (Marcus-Gunn pupil) is seen in an isolated optic nerve injury; bright light shone on the normal side elicits a consensual reflex on the affected side, whereas light shone on the injured side elicits relative pupillary dilatation (Fig. 8-1). Optic nerve atrophy sets in approximately 4 to 6 weeks after an injury has taken place.
Operative versus nonoperative management of an optic nerve injury is controversial. Mahapatra and Bhatia were the first to carry out a protocol based on a prospective study and established the role of visual evoked potentials in the management of optic nerve injury.44 They performed studies following different protocols, and showed spontaneous visual recovery in 51% to 57% cases with conservative management.
The treatment used is as follows:
1. All patients are given large doses of dexamethasone (initial dose, 0.75 mg/kg, followed by 0.33 mg/kg every 6 hours for the next 24 hours)45 Steroids are beneficial by reducing cellular edema, a membrane-stabilizing action, and reducing circulatory spasm. Studies have shown various recovery rates, from 30% to 100%, after nonoperative treatment.46,47
2. No patient is operated within 3 weeks. The visual evoked potential (VEP) test is given every week.
3. In cases of visual recovery or in patients with no perception of light, no surgery is carried out.
4. In patients with partial recovery that becomes static, optic nerve decompression can be carried out.
5. In patients in whom identifiable bony fragments and hematomas are present, optic nerve decompression is carried out immediately.48
Chiasmal injury is rare, seen in only 0.3% of all cases of head injury.49 It is often associated with a severe acceleration-deceleration strain; therefore, it is likely that the low incidence of chiasmal injury is related to the low overall survival from this severe type of head injury. Severe frontal impact can cause fracture in the sella and clinoid regions. There may be multiple facial fractures, causing separation of the optic foramina and splitting of the chiasm.50 Skull base fracture can also extend to the sella and clinoids, causing direct injury to the chiasm. During clinical examination, bitemporal field defects are noted and, in the case of associated optic nerve injury, there will be a visual loss in the involved eye. Funduscopic examination also reveals pallor of the optic disc on the nasal side. Other cranial nerves may be involved in a chiasmal injury and the management of this type of injury is restricted to relieving the compressive bony fragment or a hematoma.
Oculomotor Nerve
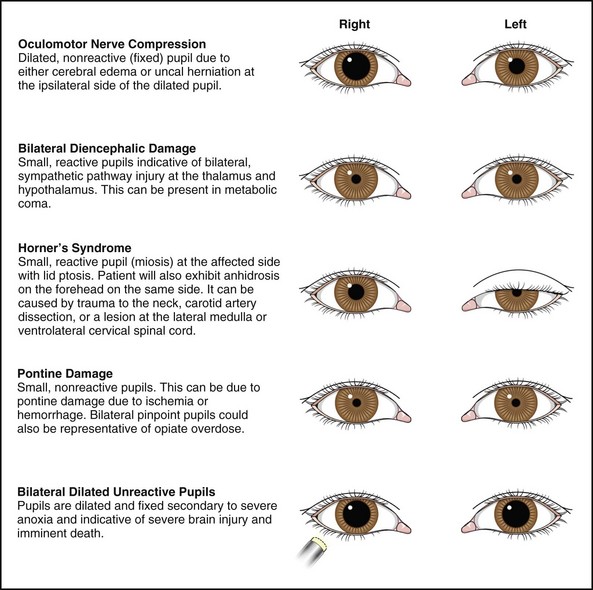
Head trauma accounts for 8% to 16% of all oculomotor palsies,51,52 which is seen in 2.9% of all head injuries, including patients with multiple cranial nerve involvement.53 Of all the cranial nerves, oculomotor palsy in a patient with head injury imparts a sense of urgency in imaging and management because of the possibility of an expanding intracranial hematoma (Fig. 8-2). Midbrain hematoma in the tectal region can present with oculomotor palsy, which often results from compression of the nerve at the tentorial hiatus by the uncus during transtentorial herniation. Avulsion or stretching of the nerve at the mesencephalopontine junction can also result in an isolated oculomotor palsy.54 Frequently, oculomotor palsy occurs in conjunction with other ocular motor nerves contained in the cavernous sinus in the case of skull base fracture. Injury at the superior orbital fissure, orbit, or maxillofacial injury can result in injury to the superior or inferior divisions of the nerve. The oculomotor nerve can also be injured in its course in the brainstem by a shearing injury.
In the setting of a head injury, unilateral mydriasis (Hutchinson’s pupil) assumes ominous importance as being representative of an ipsilateral, supratentorial, expanding hematoma. Invariably, this is accompanied by concurrent alteration in sensorium. Mydriasis may also be accompanied by ptosis and extraocular muscle weakness. An avulsion or stretch oculomotor nerve injury is suggested by the rapid recovery of consciousness in an otherwise complete motor palsy present in the clinical examination.55 This injury may be relatively mild.56 In a patient with proptosis, time should be allowed for the swelling to regress before ocular movements can be assessed appropriately. Detection of ocular pulsations and bruit over the eyeball makes the diagnosis of a caroticocavernous fistula obvious.
Ocular motility should be assessed after improvement in the level of consciousness and resolution of periorbital swelling. Palsy caused by a compressive lesion is likely to resolve on removal of the lesion, whereas injury to the fascicles is likely to give rise to an aberrant regeneration, in which axons terminate in inappropriate structures. Such a regeneration can result in elevation of the optotic eyelid on adduction and a pupil that is poorly reactive to light but constricts with adduction.57 Management of unresolved oculomotor palsy is difficult; extraocular muscle surgery can achieve binocular vision in the primary position and reading positions.
Trochlear Nerve
The incidence of trochlear nerve injury is 2.14% in head injuries.53 It is often accompanied by injury to other ocular motor nerves. When the trochlear nerve is injured in isolation, this tends to occur at its subarachnoid course. A sudden deceleration impact or blow to the head may cause the brain to move back and the brainstem to impact against the tentorium, resulting in a trochlear nerve injury. The presence of bilateral trochlear nerve injury is always caused by trauma.58,59
Diagnosis of trochlear nerve injury is generally made in a conscious and cooperative patient. The patient complains of diplopia with a perceived slant in the environment, which is compensated by adopting a characteristic head tilt (Bielschowsky’s head tilt), with the head being tilted away from the affected eye. Clinical examination reveals hypertropia (visual axis higher in the affected eye) that worsens on lateral gaze. Bilateral trochlear palsy is diagnosed by the presence of alternating hyperdeviation (upward deviation of one eye) in various positions of upward gaze. In the presence of concomitant oculomotor nerve palsy, trochlear nerve palsy can be suspected in the absence of intorsion of the eye. Spontaneous recovery occurs in 65% of patients with a unilateral trochlear nerve palsy.60 The use of an eye patch or prisms pasted onto spectacles can be useful for achieving binocular single vision. In case of incomplete recovery after 12 months, corrective ocular muscle surgery can be carried out.
Abducens Nerve
Head injury accounts for almost 3% to 15% of abducens palsies.52 Its long intradural course, with passage over the petrous ridge, its relative fixed course under the petroclinoid ligament, and its location in the cavernous sinus makes it vulnerable to stretch or tear.61 Hyperextension trauma to the cervical spine can also lead to an abducens nerve palsy, which is typically accompanied by lower cranial nerve palsies.62
Trigeminal Nerve
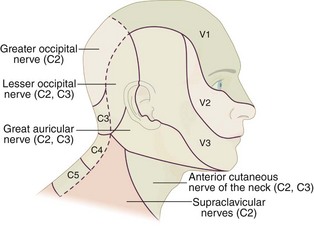
Branches of the trigeminal nerve are often injured during severe maxillofacial and skull base injury, because these nerves exit the various foramina from the skull. The supraorbital and infraorbital nerves are injured in trauma to the forehead, orbit, or maxilla. The inferior dental branch and the mandibular division (V3) of the trigeminal nerve can be injured in fractures of the mandible. Skull base injury with injury to the cavernous sinus region can result in ocular motor dysfunction, with sensory impairment over the ophthalmic division (V1). Closed or penetrating injuries can cause injury to the trigeminal ganglion.63 Skull fractures involving the middle fossa can extend into the foramen ovale and foramen rotundum and damage the exiting nerves.
Sensation along the cutaneous distribution of the involved nerve is affected (Fig. 8-3). Hyperpathia can occur along the distribution of the affected nerve and patients should be evaluated for corneal anesthesia, because the eye requires sensation to be protected against exposure keratitis and corneal ulceration. Patients who develop a hyperalgesia can be treated with carbamazepine or gabapentin. Alternatively, in intractable cases, the involved root can be sectioned or radiofrequency ablation of the ganglion can be done to relieve pain.
Petrous Fractures
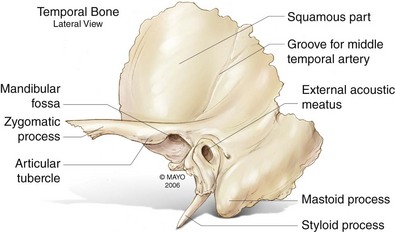
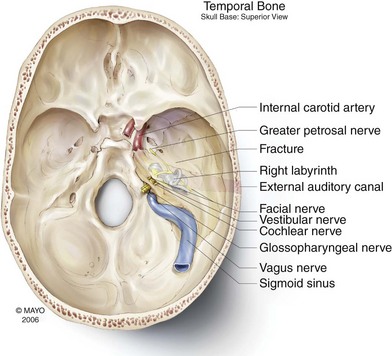
The temporal bone itself is composed of five parts—the squamous, petrous, mastoid, and tympanic portions, as well as the styloid process (Fig. 8-4). The squamous portion is smooth and convex; the temporalis muscle attaches to this region. The zygomatic arch projects forward from the inferior part of the squamous portion, giving rise to the articular tubercle just anterior to the glenoid fossa. The fossa is bounded posteriorly by the tympanic portion of the temporal bone and the bony external auditory canal. The pyramid-shaped petrous portion of the temporal bone is wedged between the sphenoid and occipital bones at the base of the skull. There are many important structures that pass through it, including the carotid canal, internal jugular vein, and facial nerve (Fig. 8-5). An injury to the facial and vestibulocochlear nerves is common after head injury and a temporal bone fracture is a frequent accompaniment to this injury. As such, it would be pertinent to review briefly this type of fracture here.64
Longitudinal fractures are most frequently caused by a lateral blow to the skull in the parietal region. Transverse fractures are more commonly caused by an intense blow to the occipital region or by a direct frontal injury. The latter are more frequently serious or fatal. A fracture with significant comminution or fracture lines running in both the longitudinal and transverse directions is termed a mixed fracture. Because studies have suggested that the traditional classification system may not predict the presence of sequelae, such as facial nerve injury or CSF leak, alternatives to the traditional classification scheme have been developed. One alternative scheme that has demonstrated better correlation classifies fractures based on whether they are otic capsule–sparing or otic capsule–violating. Others have suggested that fracture classifications should include more specifics regarding fracture location because of the difficulty in describing many fractures as longitudinal or transverse, or should describe fractures in a manner that correlates with the optimal surgical approach to repair the facial nerve.64
Facial Nerve
Trauma is the second most common cause of facial paralysis after Bell’s palsy.65 During a deceleration head injury, the facial nerve is injured at the geniculate ganglion, where it is tethered by the greater superficial petrosal nerve. The shearing force results in an intraneural contusion, edema, and hemorrhage. Transection can also occur in severe head injury cases. The presence of a fracture of the otic capsule indicates severe trauma, and the facial nerve damage is generally complete in such cases.66 Facial nerve palsy can be seen in 45% to 50% of patients with a gunshot wound to the face.67 A missile fragment enters from the lateral or inferior aspect of the temporal bone and, as it lodges in the bone, it dissipates its energy, damaging the facial nerve. The facial nerve is commonly injured at its vertical segment as it exits the cranium at the stylomastoid foramen.68 Immediate paralysis carries a worse prognosis and is caused by transection of the nerve or another form of severe neural trauma. Delayed paralysis, longer than 24 hours, is caused by nerve edema and swelling of the nerve within its sheath or epineurium; this carries a better prognosis. Delayed paralysis can also be caused by external compression by an expanding hematoma or edema of the loose fibrous tissue and periosteum between the nerve and bony canal.69
In patients with a facial nerve paralysis, care is taken to avoid exposure keratitis and dryness of the cornea at the affected site. Earlier recommendations of waiting for 3 weeks have now become obsolete for patients requiring surgical intervention, with findings of experimental studies advocating early surgery when appropriate. Decompression of the facial nerve improves recovery if performed within 48 hours; slitting of the epineurium does not seem to confer any benefit.70 Presence or absence of anacusis determines the approach to be taken for decompression of the facial nerve. In anacusis, the translabyrinthine approach is used because it allows excellent access to the entire intratemporal segment of the nerve. In the presence of hearing, a combined transmastoid middle fossa approach is used, which permits access to the perigeniculate and vertical segments of the nerve.70 Results of facial nerve grafting appear to favor early repair over delayed grafting. Anatomically intact facial nerve has a higher potential for recovery. However, there may be unpleasant sequelae of recovery in the form of dysacusis, ageusia, epiphora, and gustatory lacrimation. Late sequelae can be in the form of facial contractures, tics, spasm, and synkinesis. Recovery from facial palsy is graded according to the House-Brackmann grading system.
Vestibulocochlear Nerve
Following head injury, hearing loss can occur because of damage to the vestibulocochlear nerve. Transverse fractures of the petrous bone can damage the anterior portion of the vestibule and cochlea. These organs can also suffer a concussion. An injury can occur with damage to the central auditory pathways in the brainstem. Pressure waves generated by a blow to the head can damage the hair cells in the cochlea and cause high-frequency hearing loss and tinnitus.71 In addition to fracture of the bony otic capsule, the nerve can also be damaged at the internal acoustic meatus.
Glossopharyngeal, Vagus, and Accessory Nerves
The three lower cranial nerves are injured together because of their close proximity with one another in the jugular foramen. These nerves, after exiting the brainstem, enter the jugular foramen and a fracture of this region can lead to injury of the three nerves. This type of injury is uncommon because of the infrequent injury pattern of the posterior skull base in comparison to that of the anterior and middle skull base. More often, the nerves are injured in their extracranial course because of stab wounds, gunshot injuries, or stretch from high falls. Occipital condyle fracture can also lead to injury to the ninth and tenth nerves.72
Hypoglossal Nerve
The hypoglossal nerve is one of the least injured nerves in traumatic head injuries. Fractures of the occipital condyle can damage the hypoglossal nerve as it passes medial to the condyle. Hypoglossal palsy can occur late, after a minor head trauma and condylar injury.73,74 More often, the hypoglossal nerve is injured in the course of surgery on the neck while operating on the submandibular gland and on C2-C3 cervical disc procedures.
Motor System
The power of the individual muscle groups is measured on a scale from 0 to 5:
• Grade 1—palpable muscle contraction; no joint motion
• Grade 2—complete range of motion with gravity eliminated
• Grade 3—complete range of motion against gravity; no resistance
• Grade 4—complete range of motion against gravity with some resistance
• Grade 5—complete range of motion against gravity with full resistance
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses