Management of Nonpenetrating Chest Trauma
History of Chest Trauma
Trauma is the leading cause of morbidity and mortality in Americans younger than 45 years and the third highest overall cause of death in the United States.1 Thoracic trauma can cause significant mortality, because 25% of traumatic deaths are secondary to thoracic injuries.2 The written history of chest trauma can be traced back to 1600 BC in Egypt; 48 cases of trauma were reported, and four of these involving the chest were described. One detailed flail chest, which at that time was deemed hopeless. In 400 BC, Hippocrates described wrapping of chest with linen to stabilize fractures for 20-40 days. This practice continued until the twentieth century. In 1773, William Bromfield performed the first thoracentesis for traumatic pneumothorax and described tension pneumothorax.
Over the last 100 years, most of what we have learned about chest trauma was taken from wartime. During WWI, 37% of deaths were related to chest injuries.2 In WWII, 34% of deaths were related to chest injuries. However, the overall mortality from WWI to WWII decreased from 24% to 8%. Advances in antimicrobials, anatomy, and physiology have helped improved morbidity and mortality associated with chest trauma.
Motor Vehicle Accidents and Thoracic Injuries
The number of motor vehicles on the roads worldwide has increased steadily since World War II and, with the increase in high-speed motor vehicle travel, there has been a correlated increase in the number of motor vehicle accidents (MVAs). MVAs are leading cause of death for people age 5 to 34 years in the United States.3 Each year, more than 6 million people are injured in MVAs. Of those, 45% to 50% have thoracic injuries and approximately 150,000 of these victims die. A total of 25% of these 150,000 deaths are caused entirely by thoracic injuries and in another 50% of victims, injuries of the thorax are a major contributing factor to mortality4–6 The male-to-female ratio is 6.5 : 1. Also, nonfatal chest injuries, such as rib fractures and pneumothorax or hemothorax, are frequently seen in victims of MVAs. MVAs are the most common cause of blunt chest trauma, accounting for 70% to 80% of blunt thoracic injuries. The costs of crash-related deaths and injuries among drivers and passengers in 2003 totaled $70 billion dollars.
The mortality rate from isolated blunt chest trauma 25 years ago approached 50%. Fortunately, improvements in prehospital care and the development of rapid transport systems have lowered the mortality rate to less than 10%. Less than 10% of the patients suffering blunt chest trauma require a thoracotomy. Prevention of closed chest injuries associated with MVAs has been the focus of improved highway construction and design, as well as vehicular safety features and innovations such as air bag systems, safety belts with shoulder harnesses, antilock brakes, use of energy-absorbing materials, and improved design. Enforcing the use of safety belts and impaired driver legislation have also decreased the severity of MVAs and associated injuries.7 It is hoped that once the patient reaches the hospital, the prompt recognition of chest injuries, coupled with an improved understanding of their pathophysiology and management, will further reduce the number of deaths caused by nonpenetrating thoracic trauma.8
General Management of Blunt Chest Trauma
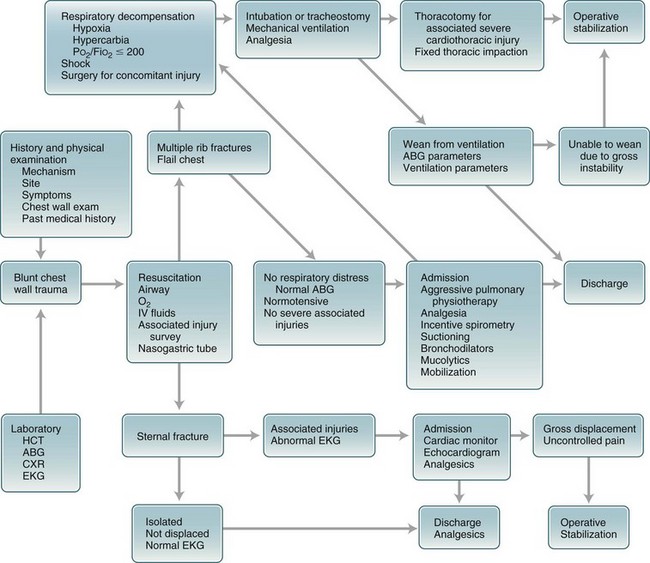
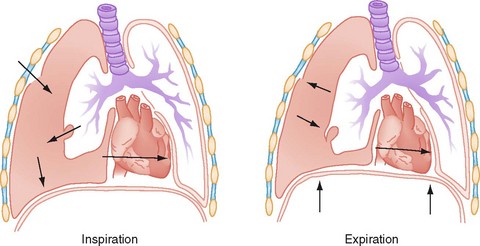
The most common injury associated with blunt chest trauma is rib fracture, which occurs in 56% of patients reported in a study of chest trauma victims, followed by pneumothorax in 40% of patients evaluated, pulmonary contusion (35%), flail chest (13%), and injury to the heart and major vessels in 5% of cases.9 Initial management of patients with blunt chest trauma should focus on performance of the ABCs of trauma care,10 including establishment of a patent airway, initiation or support of adequate ventilation, and treatment of shock (Fig. 6-1).
Treatment of severe chest wall injury must focus on the identification and care of specific injuries after establishing adequate ventilation and oxygenation. Chest trauma can cause mortality from airway obstruction, tracheobronchial injuries, pulmonary secretions, hemorrhage, loss of oxygenation, ventilation, exsanguination, or cardiac failure. Blunt forces inflicted on the chest wall will cause injury by three mechanisms—rapid deceleration, direct impact, and compression.11 Rapid deceleration is usually associated with MVAs and falls from heights. It is important to recognize that serious intrathoracic injury can occur without obvious external chest injuries or rib fractures. The degree of injury to the chest wall is usually not indicative of the severity of the injury to the internal tissue and/or organs. The possibility of injury to the heart, lungs, vessels, and other tissue should be taken into account (Box 6-1). Deceleration produces traction on the great vessels and trachea, with disruption and tears near points of anatomic fixation, such as the ligamentum arteriosum with the aorta and the carina with the trachea.9
When the chest wall is exposed to a significant enough force that can fracture ribs, the semirigid chest wall of older adults absorbs energy that would otherwise be transmitted to the lung parenchyma and great vessels. However, the flexible chest wall of the younger patient transmits essentially all the kinetic energy to the thoracic contents. Thus, the absence of rib fractures does not exclude the possibility of life-threatening injuries because injuries to the lung parenchyma, cardiac contusion, or pneumothorax are possible without external signs. Almost 50% of all patients with traumatic aortic injury12–14 and up to one third of patients with myocardial contusion15,16 have no external signs of trauma. If the injury is not recognized early, death frequently results. Those caring for patients who have sustained blunt trauma to the chest must not be deceived by initial clinical stability and must always be alert for the likelihood of later deterioration. Some patients with life-threatening injuries may appear clinically well on arrival to the emergency room.
Pulmonary Dysfunction
Most of the pulmonary dysfunction associated with nonpenetrating chest trauma is secondary to the underlying pulmonary contusion or lung injury rather than disruption of chest wall expansion and respiratory mechanics caused by fractured ribs and/or sternum, pain, or muscle splinting.17–19 Compression of the chest wall by heavy objects (e.g., automobiles, rocks, with a trapped victim) prevents respiration and may result in significant increase in pressure within the veins of the upper thorax and traumatic asphyxia. Frequent observation of any patient with blunt chest injury is as important as the initial evaluation to detect progressive changes in function. A pneumothorax may develop several hours after an injury and a small hemothorax may continue to increase in size over a period of several hours. A flail segment not apparent on an initial examination may become obvious from 1 to 10 days after an injury.11,20
Extrathoracic Injuries
Patients with thoracic injuries frequently sustain multiple extrathoracic injuries. The physician’s attention must not be diverted from thoracic injuries by obvious extrathoracic trauma, such as abdominal, maxillofacial, or central nervous system (CNS) injuries. Associated injuries occur in 66% to 80% of patients with chest trauma and usually involve the head, abdomen, pelvis, and extremities.21
Process for Treating Chest Wall Injuries
After any immediate life-threatening emergencies have been treated, the physician should undertake a careful history, if possible, and should methodically examine all systems of the injured patient. Knowledge of the patient’s past medical history, allergies, and medications may influence subsequent therapy. Clues to likely injuries may be gained from knowledge of the circumstances under which the injuries occurred. The chest wall should be inspected for wounds, lacerations, contusions, abrasions, or other evidence of trauma and splinting, paradoxical movement of the chest wall with breathing caused by rib or sternum fractures. The neck should be evaluated for distention of the veins or tracheal deviation. The physician should listen to the lung fields and heart for the presence, absence, or alteration of breath or cardiac sounds, and heart murmurs or bruits that may be indicative of a developing cardiac, lung, or vessel injury. A nasogastric tube should be placed, unless contraindicated because of injury, to prevent acute gastric dilation, which can exacerbate respiratory compromise in injured patients.11
Arterial Blood Gas
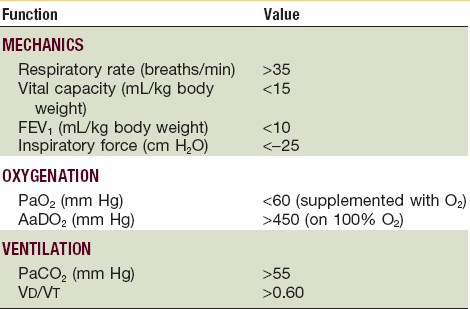
Arterial blood gas (ABG) determinations are a means of indirect measurement and serial assessment of the functional impairment associated with pulmonary injury. All patients with suspected flail chest or pulmonary contusion should be observed, including taking serial blood gas measurements. Often, the first sign of impending respiratory failure will be marked hypoxia (Table 6-1). The ratio of arterial oxygen tension to the fraction of inspired oxygen (PaO2/FiO2 ratio) yields an estimate of the extent of an intrapulmonary shunt and may be used as a parameter indicating the need for mechanical ventilation in patients with pulmonary contusion and flail chest.22 Serial ABG determinations, compared with baseline data obtained while the patient breathes room air, may be useful in predicting trends in respiratory function (Table 6-2). Using a fractional concentration of inspired oxygen (FiO2) of 0.21 permits consistent serial comparisons and gives a more accurate estimation of the severity of the respiratory impairment. Patients should be followed initially with ABG determinations every 4 hours for 48 hours to detect subtle changes that might prompt modification of treatment. Noninvasive measurement of oxygen saturation by pulse oximetry can be used as an indicator of the degree of functional pulmonary injury and response to treatment.
TABLE 6-2
| Condition | pH | PaCO2 | HCO3 |
| Respiratory acidosis | Decreased | Increased | Normal |
| Respiratory alkalosis | Increased | Decreased | Normal |
| Metabolic acidosis | Decreased | Normal | Decreased |
| Metabolic alkalosis | Increased | Normal | Increased |
| Parameter | Definition | Normal Range |
| pH | Measurement of acidity or alkalinity based on the hydrogen (H+) ions present | 7.35-7.45 |
| PaO2 | Partial pressure of oxygen that is dissolved in arterial blood | 80-100 mm Hg |
| SaO2 | Arterial oxygen saturation | 95%-100% |
| PaCO2 | Partial pressure of carbon dioxide in arterial blood | 35-45mm Hg |
| HCO3 | Amount of bicarbonate in the bloodstream | 22-26 mEq/liter |
| Base excess | Indicates the amount of excess or insufficient level of bicarbonate in the system | −2 to +2 mEq/liter |
Radiographic Examination
Radiographic examination using standard supine chest radiographs, with inspiratory and expiratory views and a lateral projection, should be obtained early in the examination of patients with nonpenetrating chest wall trauma to evaluate the sternum, spine, ribs, heart, lungs, and other structures of the neck, thorax, and abdomen for the presence of a pneumothorax, hemothorax, widened mediastinum, subcutaneous air, pulmonary contusion, ruptured diaphragm, and other abnormal findings (see Box 6-1). It is important to obtain quality chest x-rays quickly in the initial management of patients with blunt chest trauma, even when the patient has serious associated injuries, such as head trauma or compound fractures, because there may be developing life-threatening injuries that have not been previously detected or clinically apparent. It is also important to obtain baseline radiographs during the initial examination of the patient to evaluate the patient’s clinical progress or changes in clinical signs and symptoms further. Chest films should be delayed only if obvious therapeutic measures are immediately required, such as for significant bleeding, shock, and airway and/or ventilatory problems.
Chest computed tomography (CT) has proven to be of great use in distinguishing chest wall injury from parenchymal or mediastinal injury. Also, by CT scanning, more injuries are detected and consequently treated. Therefore, every patient who has sustained severe thoracic trauma and is hemodynamically stable should get a CT scan of the chest on admission to a hospital.23–26
Pneumothorax
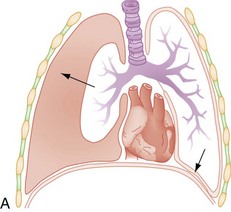
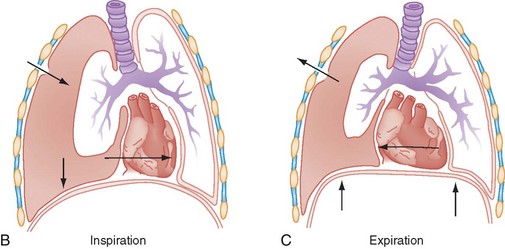
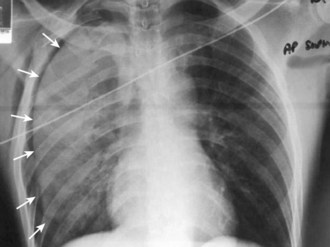
The thoracic cavity is normally filled by the expanded lung. A potential space exists between visceral and parietal pleura, blood, or air in the space is pathologic. If there is air present in the pleural space, one of two events has occurred: (1) communication between alveolar spaces and pleura; or (2) direct or indirect communication between the atmosphere and pleural space.27 Pneumothorax is a common complication of penetrating chest trauma caused by a defect in the chest wall that allows air to be moved in and out of the pleural cavity with each respiration or a sudden increase in intrathoracic pressure (Fig. 6-2). An equilibrium develops between the intrathoracic pressure and atmospheric pressure, which affects the ability of the lung to remain inflated. The involved lung collapses on inspiration and slightly expands on expiration. This causes the air to be drawn out of the wound in the chest wall with an open pneumothorax or from a wound in the lung from a fractured rib. Inspired air escapes into the pleural cavity in a closed pneumothorax injury (Fig. 6-3). Pneumothorax is usually unilateral; however, it may be bilateral in 10% to 15% of cases.8 Most of these injuries are associated with MVAs, but blast injuries, falls, direct blows to the chest, or automobile-pedestrian collisions may also be associated with a pneumothorax. Pneumothoraces are categorized as open or closed and are further defined as simple or complicated. A simple pneumothorax is air in the pleural space that does not communicate with the atmosphere or distort the mediastinum. The two possible mechanisms include laceration of the pleura or lung parenchyma caused by a fractured rib and compression of the chest against a closed glottis that ruptures the alveoli.28
Closed or Noncommunicating Pneumothorax
A closed pneumothorax, the most common type, usually results from a rib fracture, causing a parenchymal laceration (Fig. 6-4). It may also occur without an accompanying rib fracture. In general, this type of pneumothorax tends to be self-limited because the resultant pulmonary collapse usually seals the air leak (simple pneumothorax). However, if a large parenchymal disruption exists, air from the damaged lung continuously is allowed to enter the pleural cavity. Because the air cannot escape from the pleural cavity, it tends to accumulate under pressure, producing a tension pneumothorax. Air from the lung to the pleural space equalizes the pressure within the chest cavity and the lung collapses. A ventilation-perfusion deficit occurs because the blood circulated to the affected lung is not oxygenated. With a pneumothorax, percussion of the chest shows hyperresonance and breath sounds are usually distant or absent.
Tension Pneumothorax
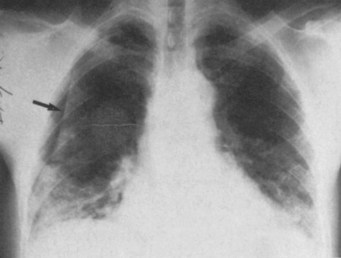
A tension pneumothorax that develops as a result of trauma exists when the tissues surrounding the opening into the pleural cavity act as a valve, allowing air to enter the pleural cavity but not to escape (Fig. 6-5). This results in a shift of the mediastinum, with compression of the opposite lung and resultant hypoxia. The positive intrapleural pressure produces compression of the vena cava, thereby interfering with diastolic filling of the heart and resulting in low cardiac output.
Positive pressure ventilation is the major cause of tension pneumothorax; however, a tension pneumothorax may be associated with invasive medical procedures such as central line placement. The development of a tension pneumothorax occasionally occurs with spontaneous breathing, but is more likely in patients who undergo bag-valve-mask or mechanical ventilation. Positive-pressure ventilation may also convert a simple pneumothorax to a tension pneumothorax. As the intrapleural pressure increases, the mediastinum shifts and the venous return to the superior vena cava is impaired. Tension pneumothorax causes hypotension, increases the central venous pressure, decreases cardiac output, and ultimately produces cardiovascular collapse.29,30
Simple Pneumothorax
Treatment
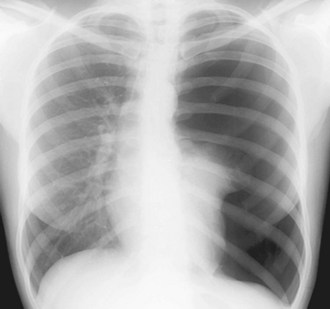
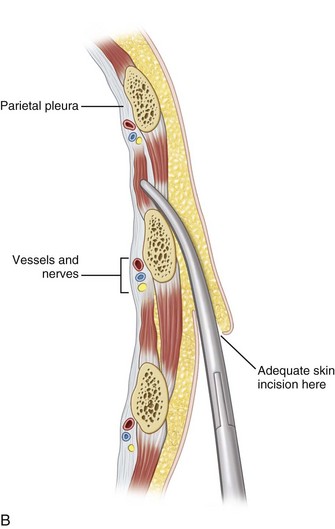
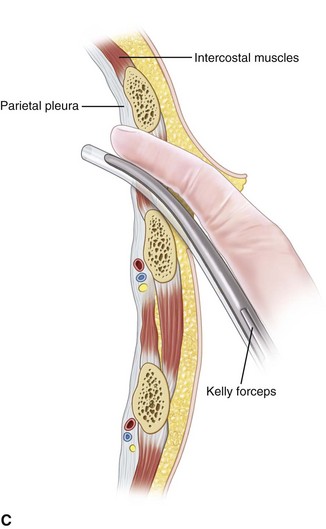
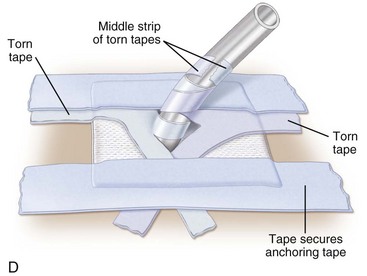
The indications for closed tube thoracostomy (Fig. 6-6) include the following: (1) pneumothorax of moderate to large size; (2) presence of respiratory symptoms regardless of the size of the pneumothorax; (3) increase in size of the pneumothorax that initially was treated conservatively; (4) recurrence of pneumothorax after the initial chest tube was removed; (5) any pneumothorax in a patient who requires ventilatory support; (6) any pneumothorax in a patient who is undergoing general anesthetic; (7) associated hemothorax; (8) bilateral pneumothoraces; and (9) tension pneumothorax.31
Open Pneumothorax
Sucking chest wounds demand immediate surgical treatment. Patients may require intubation and ventilatory support. The wound should be covered with a sterile occlusive dressing secured on three sides of the dressing to the chest. The unsecured side of the dressing acts as a one-way valve, allowing air to escape the pleural cavity on expiration. Secure taping of all edges of the dressing results in an accumulation of air within the thoracic cavity and a subsequent tension pneumothorax. Occlusive dressings such as petrolatum gauze may be used as a temporary measure during initial examination or over large defects. A chest tube must be placed in a distant site on the affected chest wall to prevent development of a tension pneumothorax, and the wound must eventually be closed in the operating room. If the lung does not expand after closure of the defect or if signs of poor ventilation persist, the patient should be placed on a ventilator with positive end-expiratory pressure (PEEP) to expand the lung. The patient should be carefully monitored and have a chest tube in place to prevent the development of a tension pneumothorax caused by a tear in one of the bronchi or in the lung parenchyma (Fig. 6-7). Signs of a tension pneumothorax in patients on ventilators include increased airway resistance and diminished tidal volume.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses