Mandibular Fractures
Location of Mandibular Fractures
Facial Fractures Associated With Mandibular Fractures
Number of Fractures Per Mandible
Nonmaxillofacial Trauma Associated With Mandibular Fractures
Classification of Mandibular Fractures
Diagnosis of Mandibular Fractures
General Principles of Treatment
Historical Considerations
Management of mandibular fractures has continued to evolve since on of the earliest descriptions in Egyptian literature. The first described case in 1650 BC, discussed the examination, diagnosis, and treatment of mandibular fractures and other surgical complications. Treatment options were limited and quite often the condition led to the demise of the patient.1,2
It was not until the advent of the Hippocratic concept for reapproximation and immobilization that management of mandibular fractures was revolutionized. Hippocrates described direct reapproximation of the fracture segments with the use of circumdental wiress, similar to today’s bridle wire. He advocated wiring of the adjacent teeth with external bandaging to immobilize the fracture.3,4 Hippocrates had the insight to realize that reapproximation and immobilization are paramount in the treatment of mandibular fractures.
In 1180, a textbook written in Salerno, Italy, described the importance of establishing a proper occlusion.5 In 1492, an edition of the book Cirurgia, printed in Lyons, France, first mentioned the use of maxillomandibular fixation in the treatment of mandibular fractures.6 In 1795, Chopart and Desault described the effects of elevator and depressor muscles on mandible fragments.7 Chopart was also the first to use dental prosthetic devices in an attempt to immobilize fracture segments (Fig. 14-1).
History of Treatment
Bandages and External Appliances
Bandages, first mentioned by Hippocrates,3 gained notoriety and acceptance as a standard of care when John Rhea Barton described his Barton bandage15 (Fig. 14-2). This bandage provided posterior directional forces on the fractured mandible, resulting in deformities such as the “bird face” and malunions.16,17
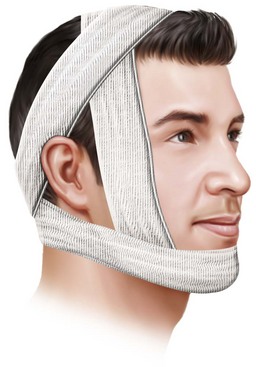
FIGURE 14-2 The Barton bandage, named for John Rhea Barton.
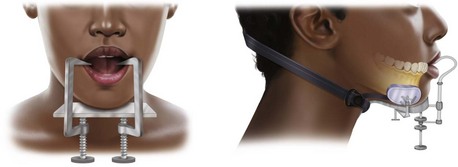
External or extraoral appliances also failed to provide stable reduction of fractures. These appliances had chin cups and an assortment of straps and external supports that like the bandages, produced an undesirable posterior directional force.18–25
Extraoral and Intraoral Appliances
A primitive approach combined a rigid splint on the occlusal surface of the teeth and one on the undersurface of the mandible. This viselike device was then used to apply pressure to the two splints, theoretically immobilizing and fixating the fractured segments. Dorrance and Bransfield26 wrote about the multiple disadvantages of this method, however, including the inability to gain proper occlusion, its inapplicability for fractures of the posterior mandible and for bilateral fractures, its promotion of drooling, and the lack of immobilization necessary for proper union.
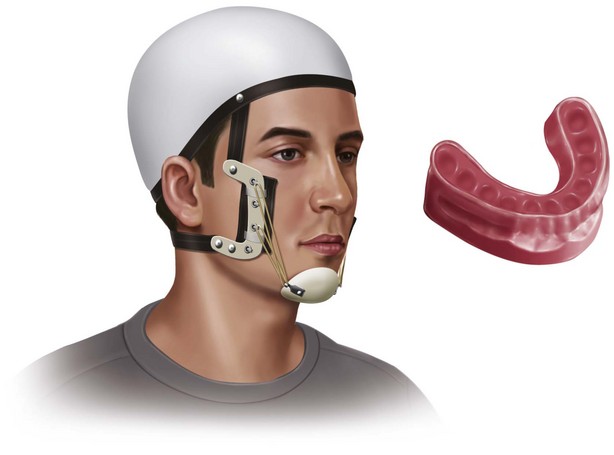
Gunning27 was the first to use a custom-fitted intraoral dental splint for immobilization. He used the splint in conjunction with an external head appliance, and these splints could also be applied to the maxilla and mandible, resulting in IMF (Fig. 14-3). An anterior space was provided for nourishment.27–29 This basic principle, of using splints for intermaxillary fixation, although modified, is still used routinely in the treatment of edentulous and partially edentulous mandibular fractures.
Monomaxillary Wiring, Bars, Arches, And Splints
Originally advocated by Hippocrates,3 and then later supported in the writings of numerous authors,30–33 monomaxillary wiring was popular in the treatment of mandibular fractures. It was similar in concept to today’s bridle wire and the Risdon wire of the early 1920s, and monomaxillary wiring afforded some element of reduction, but without supplemental fixation, fractures were inadequately immobilized.
Splints became popular in the middle of the nineteenth century and were usually cast of metal and custom-fitted for the patient. Splints were fabricated with the use of models made after reduction of the fragments. Some were cemented to the teeth and others involved sophisticated clamps that would tie buccal and lingual segments together.17,34–43 The use of monomaxillary wiring, bars, arches, and splints is limited to fractures containing stable teeth on both sides of the fracture.
Intermaxillary Wiring
Guglielmo Salicetti (William of Saliceto)44 has been credited with being the first to use intermaxillary fixation. Gilmer,45 also credited with being the first to use this technique, passed wires around individual teeth of both arches and then ligated these opposing wires.
Orthodontic bands and arches were used in patients with loose, fractured, or missing teeth. The famous American orthodontist Angle46 described many methods of IMF that used bands and other orthodontic techniques. A recent study by Cousin revisited this methodology and noted success using nonwire fixation for IMF.
Sauer47 has described the ligation of an iron wire to the teeth with fine ligature wires. However, it was Gilmer48 who took this idea a step further and fixated full arch bars on both the mandible and maxilla, revolutionizing the treatment of fractures.
Open Reduction and Internal Fixation Using Sutures
Historically, mandibular body, ramus, or angle fractures were treated with closed reduction and intermaxillary fixation (IMF). Currently, screw and plate osteosynthesis is standard procedure to stabilize these fractures by open reduction and fixation.8–10 A fracture of the condyle can be treated conservatively with IMF, surgically by an open reduction using a submandibular, preauricular, retroauricular, or transparotid approach, or by a transoral approach or endoscopically.11–14
Transosseous wires
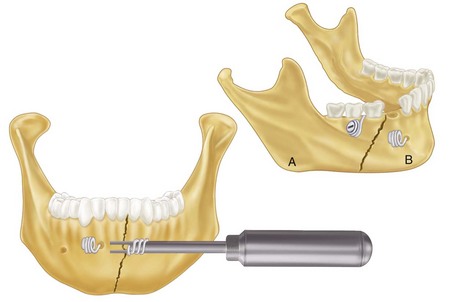
In the middle of the nineteenth century, Buck and Kinlock have described the use of wire ligature for the immobilization of mandibular fractures.17,49 Using this method, one would drill a hole on both sides of the fracture site and then pass a wire. The wire, which was iron or silver, was then tightened periodically by creating pigtails on each end of it (Fig. 14-4). This method was hampered by a high incidence of infection.26
Bone Plates
In 1881, Gilmer36 described a method of mandibular fracture fixation that used two heavy rods placed on either side of the fracture and wired together. The rods were pushed through skin, mucous membranes, and bone and were wired on both the mouth and skin sides. Dorrance and Bransfield stated that the earliest reference to the use of true bone plates was that of Schede, who, around 1888, used a solid steel plate held by four screws.26 During World War I, Kazanjian50 used wire sutures through bone fragments and tied the wire to an arch bar for fixation. In 1900, Mahé51 used multiple plates to secure multiple mandibular fragments after applying a monomaxillary splint. A similarity to the plating systems used today can be noted.
In 1915, Ivy52 attempted the use of Sherman’s steel plates but abandoned the method because of infection and necrosis. In 1917, Cole53 used silver plates and screws on each side of the fracture and then silver wires attached to the plates to immobilize the fracture. In 1934, Vorschutz introduced two long screws through the skin into the bone, reduced the fracture, and held the screws in position with the use of a plaster of Paris bandage (similar to the Joe Hall Morris appliance of today).54
Biodegradable Plates and Plating Systems
Internal rigid fixation of the facial skeleton is a reliable method for obtaining osteosynthesis. Historically, metallic plates and screws have been used to allow for early passive and active function. To date, titanium plating remains the reference standard of mandibular fracture treatment because of its high biocompatibility, stability55, resistance to corrosion56,57, and cost-effectiveness (in comparison to resorbable plating systems).58 However, these titanium plating systems have certain disadvantages, including migration from the original position, infection, palpability, and exposure, all which might require revision surgery, and can cause distortion in magnetic resonance imaging (MRI) and computed tomography (CT) scanning.59 It may become necessary to remove plates because of stress protection–induced osteopenia in the cortex beneath the plate, causing a reduction in cortical thickness and shaft caliper. Others have reported that metallic alloys containing nickel, cobalt, and chromium may sensitize the patient and lead to allergic reactions. Physicians must take backscatter into consideration in patients in whom reconstruction with metallic implants has been performed following tumor resection and who will require radiation therapy. The same population will also have artifacts on future scans.
Resorbable plating systems or biodegradable fixation devices have been used in orthopedics and craniomaxillofacial fields for over 25 years.60 These materials not only reduce the risk of complications and mitigate the need for implant removal, but add the benefit of rigid fixation with biodegradation. Most of the studies included polymers (e.g., poly-L-lactide, polyglycolide, polyglycolic acid, polylactic acid, and dioxanone). Although reports of the use of resorbable plates in the mandible have been sparse, a prospective study of 50 fractures has reported success in achieving union with 2.5-mm resorbable plates and screws.61 Resorbable plates now consist of amorphous molded copolymer of L-lactide–D-lactide and trimethylene carbonate for malleability and stability.61 Initially, the load is carried entirely by the resorbable plate and ultimately by the healed biological union.62 In 1991, Wittenberg et al63 found that poly-L-lactide plates were successful in areas of low stress and no compression.
The dimensions of biodegradable plates were usually large compared with miniplates. This would limit their use in the maxillofacial region. These materials have less strength, which would limit their use in loaded and functional bone, such as the mandible. The stability, strength, and elasticity of resorbable osteosynthesis have been extensively investigated in vitro.63,64 Araujo et al have reported that elastic stiffness and surgical stability of bioresorbable plates may change after surgery, however, and the reduced stiffness could permit relapse after surgery.64 Polymer chemists and clinicians continue to evaluate biodegradable materials in the hope of developing implants with clinical potential equal to that of metallic implants.
The cases of plate fixation after World War II were sporadic. Before the 1960s, the biomechanical knowledge of internal rigid fixation for the facial skeleton was adapted from the orthopedic literature. In the mid-1960s, Luhr65 pursued research in rigid fixation for the facial skeleton and developed the Vitallium mandibular compression plate using glide screw principles. Throughout the 1960s, Luhr continued research on rigid fixation and also contributed the self-threading screw. In the 1970s, several investigators, including Spiessl,67 studied AO-ASIF principles. They found that adaptation of a compression plate on the lateral cortex, at the inferior border of the mandible, gave excellent compression and adaptation at the inferior border but the superior border (tension zone) splayed. Many investigators thought that a second tension zone plate would be necessary, whereas others believed that arch bars in tooth-bearing areas were sufficient to limit tension zone splaying. In 1973, Schmocker and Speissl68 developed the eccentric dynamic compression plate, which provided compression at the tension and compression zones of the mandible. When the screws closest to the fracture were tightened, the fracture line would be placed under compression. When the eccentric terminals were tightened, the alveolar segment would be reduced.
Further development of the this plate fixation technique introduced reconstruction plates. These were also called load-bearing plates because of their rigidity and application over complex comminuted fractures. Further Innovation by Raveh et al introduced a system that allowed the heads of screws to lock into the titanium plate holes.69–71 The advantage of this system was that the vascularity of periosteum was not endangered by plate pressure. In the early 1970s, research was initiated to evaluate the use of smaller plating systems. In 1973, Michelet et al72 placed bendable monocortical miniplates to treat mandibular fractures. The advantages of miniplates were their thinness and the fact that they could be placed through intraoral incisions. Also known as load-sharing plates, miniplates transferred the path of static compression to one of dynamic compression. Fixation to the alveolar ridge allows tensile stress placed on the miniplate to cause an increase in dynamic compression and stability.
Edentulous Mandibular Fractures
Treatment of the fractured edentulous jaw began with a report by Baudens73 (1844), who used circumferential wiring to reduce and fix the bone. Gunning used splints, as described earlier.27–29 In 1851, Robert74 used silver wire passed circumferentially around the mandible with a needle and tied the wire around a piece of lead that had been molded to the edentulous mandible. Gilmer36 (1881) described a gutta-percha or vulcanite splint used by Black that was maintained by circumferential wires of silver or platinum. Pickerill75 (1913) passed screws into the bone on either side of the fracture and maintained them with a connecting steel bar. Circummandibular wiring to maintain a denture splint was often described by Ivy and Curtis in the early 1900s.39,40
Another technique, described by Partridge41 in 1976, involves the use of a nylon circumferential strap wrapped around the body of the mandible and tightened. Two or more of these straps are placed on the fracture and tightened. This method can be applied only to fractures that have extreme obliquity, but the amount of periosteal stripping involved may outweigh any potential benefit.
Demographics and Epidemiology
Causes
The causes of maxillofacial fractures have changed over the past decades and will continue. Different societies and cultures show different patterns of facial trauma. Varying socioeconomic conditions combined with behavioral differences, however, make the aggregate comparison of mandibular fractures difficult. Obtaining analysis data from various regions can increase the understanding of facial trauma and allow for optimization of treatment.76,77 Demographic information on maxillofacial injuries has changed with the onset of motor vehicle seat belt and airbag laws, reduced speed limits, and increasing urban violence. Many countries, including Brazil, India, and China, are contributing to this demographic data, along with reports from the United States,42,43,78–87 England,88,89 Germany,90,91 the Netherlands,92,93 and the Scandinavian countries.94,98
The main causes of mandibular fractures worldwide include motor vehicle accidents (MVAs), interpersonal violence (IPV), falls, and sports-related injuries. Past data from industrialized or developed nations with large numbers of vehicles indicated that multiple mandibular fractures were occurring with severe concomitant facial fractures and associated nonmaxillofacial injuries, situations that required extensive treatment. In contrast, assaults and falls have become the predominant mechanism for facial injuries.99–101 Statistics from smaller developing countries have indicated that MVAs remain the most frequent cause. Ogundare et al102 retrospectively analyzed mandibular fractures seen in a U.S. urban setting and found that a striking 79% of 1267 mandibular fractures were caused by IPV, whereas Chrcanovic et al103 found that 44% of mandibular fractures were caused by MVAs in Brazil. It is important to note, however, that local laws and socioeconomic conditions in developed versus developing countries create mixed results for case by case studies.
Despite the many variables associated with the causes of mandibular fractures, MVAs and assaults are undoubtedly the primary causes of mandibular fractures throughout the world. Work-related accidents, firearms, and pathologic conditions are also causative factors. Reports have shown that on average, more than 75% of mandibular fractures are caused by MVAs and IPV, whereas 7% are work-related, 7% occur as the result of a fall, 4% occur in sports-related accidents, and the remainder have miscellaneous causes. Sports-related maxillofacial injuries were especially evaluated by Roccia et al; of 138 patients, 27% sustained a fractured mandible and 6.5% had dental and alveolar fractures. Sane and Ylipaavalniemi101 studied 8640 accidents that occurred in Finnish soccer players. They found that 6.4% of the injuries occurred in the maxillofacial and dental regions. Of these, 81% affected the teeth or alveolar process and 11% were fractures of the mandible and the middle third of the face. Linn et al89 have reported on 319 patients treated for sports-related accidents in the Netherlands; 15% of them sustained a mandibular fracture and 5.5% fractured the mandibular alveolar process, had luxated teeth, or both.
Studies of firearm-related mandibular fractures in civilians are limited. Peleg and Sawatari evaluated 92 patients who sustained gunshot wounds to the mandible. In France, Vaillant and Benoist107 evaluated 14 cases of bullet wounds of the mandible. The age of the patients ranged from 6 to 68 years. Two children were victims of accidents and the adults were suicide or assault victims. The degree of injury was proportionate to the velocity of the projectile. Civilian injuries were not as life-threatening when compared with their high-velocity combat counterparts. Handguns caused 72 injuries, followed by assault rifles and shotguns. Their surgical approach involved fabrication of an occlusal splint, IMF, aggressive débridement of hard and soft tissues, and immediate reconstruction with a titanium plate, which they believed could restore the appropriate function and contour of the patient.
Dental implants have revolutionized the restorative treatment of edentulous patients over the last decade. Unfortunately, a sequela of implant placement and loading is fracture. As implantation procedures have become the norm, fractures of the mandible and other maxillofacial bones are more common. The amount of osseous resorption secondary to long-term use of a prosthesis and the higher incidence of metabolic abnormalities in older adults result in a lower grade of recipient bone. Therefore, patient selection is paramount, and adhering to good osseointegration principles allow for better results. Greenstein et al103 have stated several predisposing factors for mandibular fracture, such as osteoporosis (reduction of bone mass), stress at the implant location, and trauma (tensile forces placed on the bone during mandibular function). Laskin has suggested that the degree of mobility or displacement determines ultimate treatment; thus, implants that have undergone osseointegration and are involved in the line of fracture should not be removed.104 Implants that are infected should be removed. The use of open reduction and internal fixation (ORIF) should be avoided because of the amount of periosteal stripping and the lack of bone in the atrophic mandible. Thus, conservative (closed) treatment of these fractures is the best option.
Location of Mandibular Fractures
In the cases evaluated for fracture location, the incidences were as follows: angle (30%), condyle (23%), symphyses (22%), body (18%), ramus (2%), and coronoid process (1%).105–111 As noted, there are many variables, but generalization fractures that occur in the symphyses, condyle, and angle do not differ much in incidence, and fractures of the ramus and coronoid process are rare.
Individual studies demonstrate how the cause plays a role in fracture location. Chrcanovic et al100 have shown that when fractures caused by MVAs are considered, the body region is the most common site. When motorcycle accidents are considered, the condyle is the area most often affected. When assault is considered, the body demonstrated the highest incidence of fracture. In another case study, Boole et al have found that assaults and fights lead to a higher percentage of angle fractures, whereas MVAs and motorcycle accidents result in more ramus fractures.107
Nonmaxillofacial Trauma Associated with Mandibular Fractures
The literature dealing with concomitant nonmaxillofacial injuries associated with mandibular fractures is difficult to interpret with the wide variation in reported injuries. The incidence of facial injuries occurring in conjunction with major trauma was shown in several surveys to range from 15% of 1,088 trauma patients in Liverpool112 to 34% of 87,174 trauma patients in a North American Database on Trauma.113 In the study of Ellis et al,110 90% of patients had no other injuries, probably because the cause was primarily assault. Olson et al84 have reported associated injuries in 46.6% of all patients treated, most of whom were involved in MVAs. Mulligan and Mahabir113 have reported associated cervical spine injuries in 6.5% of patients, whereas Elahi et al114 noted such injuries in only 2.6%. Kapoor and Srivastava115 noted other (nonfacial) fractures in 40% of patients and Adekeye116 found other fractures in 9% of patients.
Classification of Mandibular Fractures
Mandibular fractures have been classified in several ways, with the use of terminology that has not been standardized. For example, the term simple fracture has been defined by Assael and Tucker as “… a complete transection of the bone with minimal fragmentation of the fracture site.”117 Rowe and Killey defined simple fractures as “…linear fractures which are not in communication with the exterior.”85 Kruger has stated that a simple fracture “… is one in which the overlying integument is intact. The bone has been broken completely but it is not exposed to air. It may or may not be displaced. It may be comminuted.”118 Whitestone and Raley have defined a simple fracture as follows: “…the overlying mucosa is intact with no potential sources of direct communication.”119
Dictionary Classification
For the sake of this discussion, the following fracture terms have been adopted from Dorland’s Illustrated Medical Dictionary120:
1. Simple or closed: A fracture that does not produce a wound open to the external environment, whether it be through the skin, mucosa, or periodontal membrane
2. Compound or open: A fracture in which an external wound, involving skin, mucosa, or periodontal membrane, communicates with the break in the bone
3. Comminuted: A fracture in which the bone is splintered or crushed
4. Greenstick: A fracture in which one cortex of the bone is broken, the other cortex being bent
5. Pathologic: A fracture occurring from mild injury because of preexisting bone disease
6. Multiple: A variety in which there are two or more lines of fracture on the same bone not communicating with one another
7. Impacted: A fracture in which one fragment is firmly driven into the other
8. Atrophic: A spontaneous fracture resulting from atrophy of the bone, as in edentulous mandibles
9. Indirect: A fracture at a point distant from the site of injury
10. Complicated or complex: A fracture in which there is considerable injury to the adjacent soft tissue or adjacent parts; may be simple or compound
Classification by Anatomic Region
Mandibular fractures are also classified by the anatomic areas involved, as follows: symphysis, body, angle, ramus, condylar process, coronoid process, and alveolar process. Dingman and Natvig78 have defined these regions as follows:
1. Midline: Fractures between central incisors
2. Parasymphyseal: Fractures occurring within the area of the symphysis
3. Symphysis: Bounded by vertical lines distal to the canine teeth
4. Body: From the distal symphysis to a line coinciding with the alveolar border of the masseter muscle (usually including the third molar)
5. Angle: Triangular region bounded by the anterior border of the masseter muscle to the posterosuperior attachment of the masseter muscle (usually distal to the third molar)
6. Ramus: Bounded by the superior aspect of the angle to two lines forming an apex at the sigmoid notch
7. Condylar process: Area of the condylar process superior to the ramus region
8. Coronoid process: Includes the coronoid process of the mandible superior to the ramus region
9. Alveolar process: The region that would normally contain teeth
Kazanjian and Converse120 have classified mandibular fractures by the presence or absence of serviceable teeth in relation to the line of fracture. They thought that their classification was helpful in determining treatment. Three classes were defined:
Class I: Teeth are present on both sides of the fracture line.
Class II: Teeth are present on only one side of the fracture line.
Rowe and Killey85 have divided mandibular fractures into two classes: (1) those not involving basal bone; and (2) those involving basal bone. The first class primarily is comprised of alveolar process fractures. The second class is divided into single unilateral, double unilateral, bilateral, and multiple.
Kruger118 has classified mandibular fractures into simple, compound, and comminuted. Kruger and Schilli121 took into account many of the aforementioned classifications described and developed four categories of mandibular fractures:
1. Relation to the external environment
3. Dentition of the jaw with reference to the use of splints
a. Fractures of the symphysis region between the canines
b. Fractures of the canine region
c. Fractures of the body of the mandible between the canine and angle of the mandible
d. Fractures of the angle of the mandible in the third molar region
e. Fractures of the mandibular ramus between the angle of the mandible and sigmoid notch
Shetty et al122 combined six significant injury criteria to create the acronym FLOSID, which essentially allowed for ease of assessment and defined fracture characteristics. They assessed mandibular fractures using the taxonomy described and added weighting factors to address severity (mandible injury severity score):
An important classification of mandibular angle and body fractures relates to the direction of the fracture line and the effect of muscle action on the fracture fragments. Angle fractures may be classified as (1) vertically favorable or unfavorable or (2) horizontally favorable or unfavorable. Figures 14-5 through 14-8 demonstrate the various types.
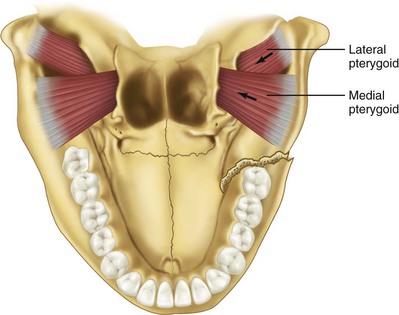
FIGURE 14-5 Vertically unfavorable fracture.
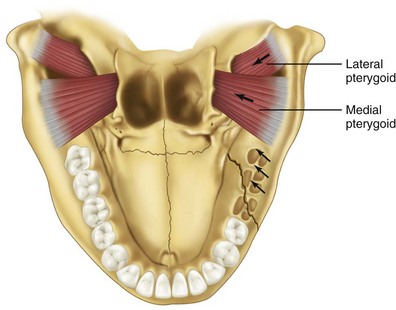
FIGURE 14-6 Vertically favorable fracture.
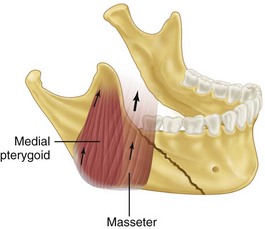
FIGURE 14-7 Horizontally favorable fracture.
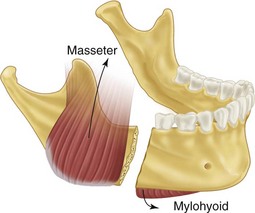
FIGURE 14-8 Horizontally unfavorable fracture.
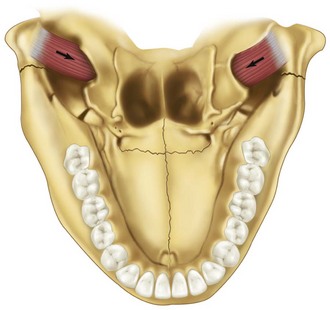
In fractures of the angle of the mandible, the muscles attached to the ramus (masseter, temporal, and medial pterygoid) displace the proximal segment upward and medially when the fractures are vertically and horizontally unfavorable (Fig. 14-9). Conversely, these same muscles tend to impact the bone, minimizing displacement in horizontally and vertically favorable fractures. The farther forward the fracture occurs in the body of the mandible, the more the upward displacement of those muscles is counteracted by the downward pull of the mylohyoid muscles. In bilateral fractures in the canines areas, the symphysis of the mandible is displaced inferiorly and posteriorly by the pull of the digastric, geniohyoid, and genioglossus muscles (Fig. 14-10).
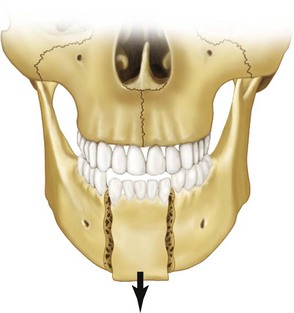
FIGURE 14-10 Inferior and posterior displacement of the symphysis of the mandible in bilateral fractures in the canine area.
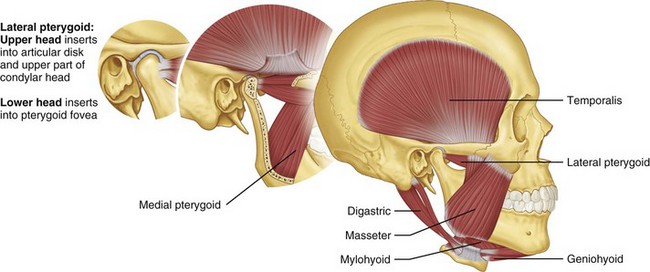
Condylar fractures are generally classified as extracapsular, subcondylar, or intracapsular. Condylar fractures are influenced by location and muscle action. The lateral pterygoid muscle has a tendency to cause anterior and medial displacement of the condylar head, depending on the location, severity of the fracture, and effect of the supporting capsule (Fig. 14-11). In 1934, Wassmund has described five types of condylar fractures.17 Type I is defined as a fracture of the neck of the condyle, with relatively slight displacement of the head. The angle between the head and axis of the ramus varies from 10 to 45 degrees. These fractures tend to reduce spontaneously. Type II fractures produce an angle from 45 to 90 degrees, resulting in tearing of the medial portion of the joint capsule. In type III fractures, the fragments are not in contact and the head is displaced mesially and forward because of traction of the lateral pterygoid muscle. The fragments are generally confined within the area of the glenoid fossa. The capsule is torn and the head is outside the capsule. An open reduction is recommended for this fracture type. Type IV fractures of the condylar head articulate on, or in a forward position with regard to, the articular eminence. The type V group consists of vertical or oblique fractures through the head of the condyle, and a bone graft is suggested to reconstitute the condylar head when considerable displacement of the fragments has occurred.17
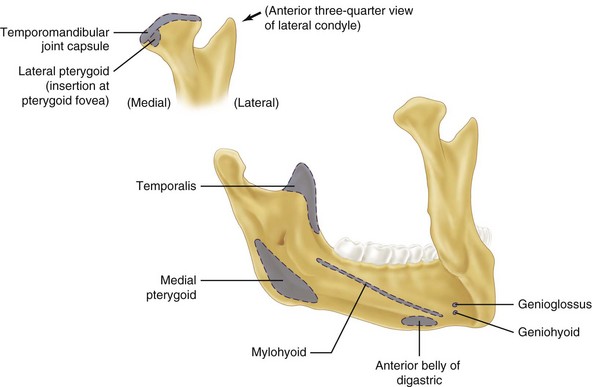
FIGURE 14-11 The lateral pterygoid muscle tends to cause anterior and medial displacement of the condylar head.
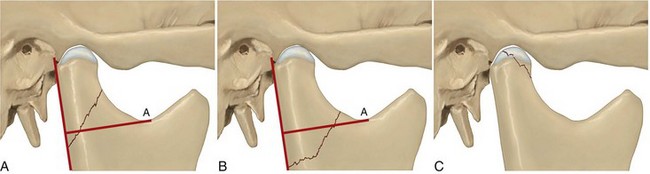
As a means of simplifying the classification of high and low condylar fractures, Loukota et al have proposed a three-part system.123 The classification system revolves around a reference line, which is a linear line that extends from the posterior border of the condylar neck through the sigmoid notch to the tangent of the ramus. First, the diacapitular type describes a fracture through the head of the condyle, because it may start on the articular surface and extend outside the capsule. Second, a condylar neck type describes a fracture that is at minimum over 50% above the reference line. Finally, a condylar base type refers to a fracture line that runs behind the mandibular foramen and is at minimum over 50% below the reference line (Fig. 14-12).
Diagnosis of Mandibular Fractures
Patient History
Knowing the direction of force can help the clinician diagnose concomitant fractures. An anterior blow directly to the chin can result in bilateral condylar fractures (Fig. 14-13) and an angled blow to the parasymphysis may cause a contralateral condylar or angle fracture. A patient with teeth clenched together at the moment of impact is more likely to have dental and alveolar process fractures than basal bone fractures. Even knowing where the patient was sitting in an automobile may aid in the diagnosis of mandibular or other injuries. Chest injuries caused by noncollapsible steering wheels, facial fractures caused by striking unpadded dashboards, and facial lacerations from non–safety glass are a few examples of predictable injuries that have been eliminated by the use of seat belts and more effective automotive safety engineering.124,125
Clinical Examination
Change in Occlusion.
Any change in occlusion is highly suggestive of mandibular fracture. The clinician should ask the patient whether his or her bite feels different. A change in occlusion can result from fractured teeth, fractured alveolar process, fractured mandible at any location, and trauma to the TMJ and muscles of mastication. Post-traumatic premature posterior dental contact or an anterior open bite may result from bilateral fractures of the mandibular condyle or angle (Fig. 14-14) and from maxillary fractures with inferior displacement of the posterior maxilla. A posterior open bite may occur with fractures of the anterior alveolar process or parasymphyseal fractures. A unilateral open bite may occur because of ipsilateral angle and parasymphyseal fractures (Fig. 14-15). A posterior crossbite can result from midline symphyseal and condylar fractures, with splaying of the posterior mandibular segments.
FIGURE 14-14 Bilateral angle fractures can cause an open bite.
Lacerations, Hematoma, and Ecchymosis.
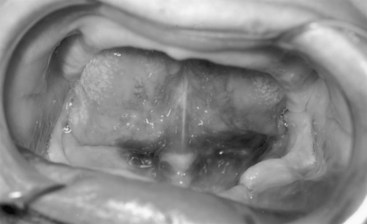
Trauma significant enough to cause loss of skin or mucosal continuity, or subcutaneous-submucosal bleeding, can result in trauma to the underlying mandible. Lacerations should be carefully inspected before closure. The direction and type of fracture may be visualized directly through the laceration, with the clinician thereby obtaining diagnostic information that might be impossible to ascertain clinically or radiographically. The common practice of closing facial lacerations before treating underlying fractures should be discouraged from diagnostic and treatment standpoints. The diagnostic sign of ecchymosis in the floor of the mouth indicates a mandibular body or symphyseal fracture (Fig. 14-16).
Radiologic Examination
The following radiologic studies are helpful in the diagnosis of mandibular fractures:
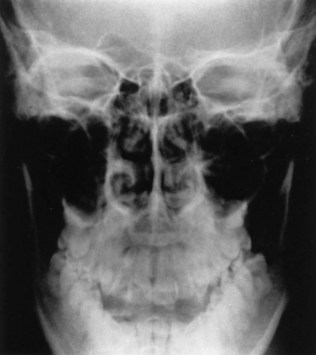
The single most informative radiologic study used in diagnosing mandibular fractures is the panoramic radiograph, showing the entire mandible, including condyles (Fig. 14-17).126 The advantages are simplicity of technique, ability to visualize the entire mandible in one radiograph, and generally good detail, but there are several disadvantages: (1) the technique usually requires the patient to be upright (machines that allow the patient to be prone are available), which may make it impractical in the severely traumatized patient; (2) it is difficult to appreciate buccal-lingual bone displacement or medial condylar displacement; and (3) fine detail is lacking in the TMJ, symphysis (depending on the type of equipment), and dental and alveolar process regions. A secondary but important disadvantage is that panoramic radiographic equipment is not available in all hospital radiology departments. In addition, because of the sensitivity of the panoramic radiographic technique, superimposition of structures can lead to interpretation errors.127
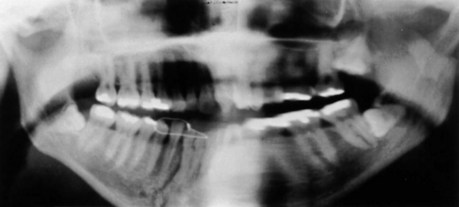
FIGURE 14-17 Panoramic radiograph, the single most informative radiograph for the diagnosis of mandibular fractures.
The lateral oblique view of the mandible can be helpful in the diagnosis of ramus, angle, and posterior body fractures (Fig. 14-18). The technique is simple and can be done in any radiology department. The condyle region is often unclear, as are the premolar and symphysis regions. The Caldwell posteroanterior (PA) view demonstrates any medial or lateral displacement of fractures of the ramus, angle, body, and symphysis (Fig. 14-19). The condylar region is not well demonstrated on this view, but midline or symphyseal fractures can be well visualized. The anteroposterior view is occasionally used for patients who cannot be placed in the supine position; however, considerable magnification and distortion occur with this view. The mandibular occlusal view demonstrates discrepancies in the medial and lateral position of body fractures and also shows anteroposterior (AP) displacement in the symphysis region (Fig. 14-20). The reverse Towne view is ideal for showing medial displacement of condyle and condylar neck fractures (Fig. 14-21). Transcranial lateral views of the TMJ are helpful in detecting condylar fractures and anterior displacement of the condylar head. Periapical dental films show the most detail and can be used for nondisplaced linear fractures of the body and alveolar process and dental trauma. Plain tomograms can be used in an AP and lateral direction when greater detail is necessary. The traditional CT scan is ideal for condylar fractures that are difficult to visualize; however, greater expense and radiation exposure limit its use to cases that cannot be diagnosed with plain films and panoramic tomography (Fig. 14-22).128 With the advent of newer technology, helical CT (HCT) is faster, more accurate, and less expensive and delivers decreased radiation exposure as compared with conventional CT scans. According to Roth et al, it also provides enhanced resolution because of less motion misregistration. Hence, the trend toward making HCT the current gold standard is underway.128
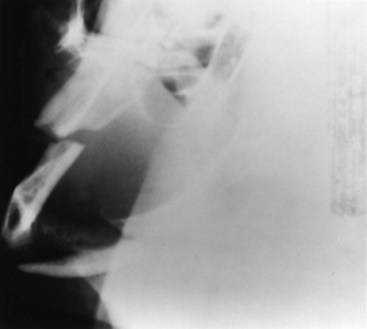
FIGURE 14-18 The lateral oblique view of the mandible is helpful in diagnosing ramus, angle, and posterior body fractures.
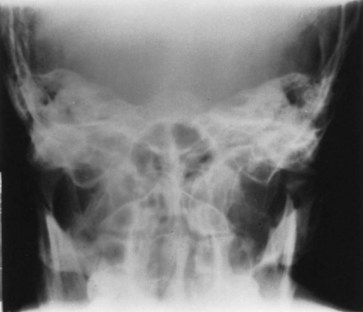
FIGURE 14-21 The reverse Towne view is most helpful for showing medial displacement of the condyle and condylar neck fractures.
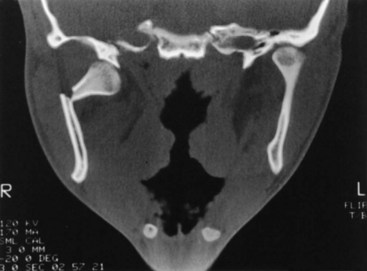
FIGURE 14-22 CT scanning is ideal for condylar fractures.
In summary, as with most other imaging procedures, it is usually optimal to have views of the mandible in two planes oriented at 90 degrees to each another. Ideal imaging includes sections in the axial and coronal planes but practical considerations of time, the patient’s condition, and cost mandate selectivity. Thus, the history, physical examination, and understanding which structures are best shown by certain imaging methods dictate the proper radiologic technique.129,130
General Principles of Treatment
1. The patient’s general physical status should be carefully evaluated and monitored before any consideration of treating mandibular fractures.
The literature is replete with case reports of isolated fractures of the mandible in which life-threatening medical conditions became apparent after treatment was instituted. Post-traumatic thrombotic occlusion of the internal carotid artery associated with mandibular fractures (and no other apparent injuries) was diagnosed 24 hours after injury in one case and 9 hours after injury in another.131 Banna131 has also reported a case and reviewed the literature on post-traumatic thrombotic occlusion of the internal carotid artery after minimal neck trauma. A basal skull fracture associated with an undisplaced body fracture of the mandible became apparent 48 hours after injury.132 Gordon et al132 have described a patient with a unilateral body fracture of the mandible who had symptoms of a ruptured spleen 5 days after injury and 3 days after arch bars had been placed. Bertolami and Kaban133 have reported on a case of a 20-year-old woman involved in an MVA who sustained a 3-cm chin laceration without malocclusion or limitation of mandibular movement. After 8 hours, radiographs were reviewed and showed a nondisplaced fracture of the angle of the mandible and a fracture of the second cervical vertebra. Minton and Tu134 have noted a case of bilateral cervical subcutaneous emphysema, pneumothorax, and pneumomediastinum secondary to a bilateral fractured mandible without other apparent injuries.
2. Diagnosis and treatment of mandibular fractures should be approached methodically, not with an emergency-type of mentality.
3. Dental injuries should be evaluated and treated concurrently with treatment of mandibular fractures.
a. Fractured teeth can become infected and jeopardize bone union; however, an intact tooth in the line of fracture that is maintaining bone fragments can be protected with antibiotic coverage (Fig. 14-23).
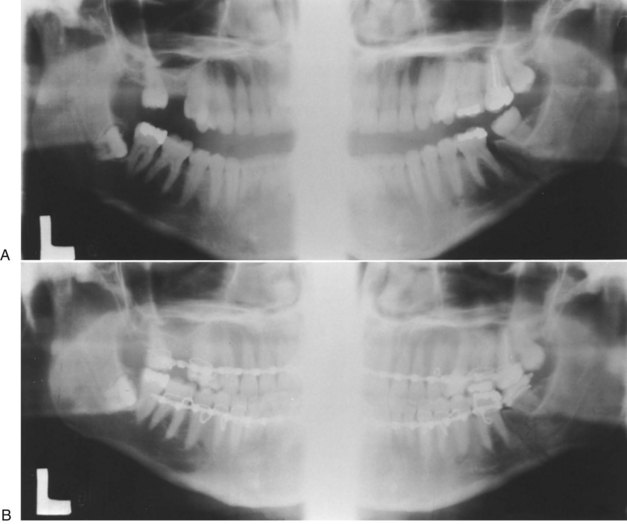
FIGURE 14-23 An intact tooth in the line of fracture can be maintained to prevent displacement of the segment.
b. A second molar on an otherwise edentulous posterior fracture segment should be maintained to prevent superior displacement of the fragment in intermaxillary fixation.
c. Mandibular canines are the cornerstone of occlusion and should be maintained at all costs.
d. Some teeth are not critical to restoration and can be removed when their prognosis is doubtful and when maintenance may adversely affect fracture treatment. For example, a lone mandibular incisor adds little to future bridge or partial denture construction; however, a single molar tooth in an otherwise edentulous posterior quadrant can be critical to dental rehabilitation.
e. Some fractured teeth cannot be salvaged, no matter how critical they may be. For example, a molar tooth may be split mesially and distally, so reconstruction would be impossible. Maintenance of this tooth during intermaxillary fixation could result in severe discomfort and perhaps infection.
4. Reestablishment of occlusion is the primary goal in the treatment of mandibular fractures.
5. With multiple facial fractures, mandibular fractures should be treated first.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses