CHAPTER 34 Pituitary, Thyroid, and Parathyroid Pharmacology
HYPOTHALAMIC AND PITUITARY HORMONES
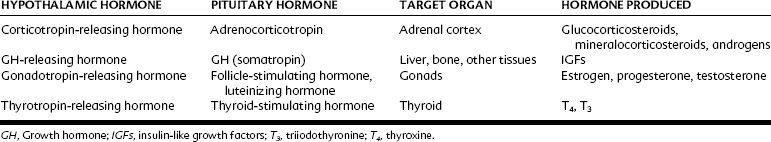
The pituitary gland consists of an anterior lobe (adenohypophysis) and a posterior lobe (neurohypophysis). It is connected to the hypothalamus, which lies above it, by the stalk that contains neurosecretory fibers and capillaries. The hypophyseal portal system drains the hypothalamus and perfuses the anterior pituitary. Numerous releasing factors or regulating hormones that are produced by the hypothalamus are carried to the anterior pituitary by this portal system. These hypothalamic releasing factors stimulate the anterior pituitary to produce and secrete numerous tropic hormones, which stimulate target glands to produce hormones. The hypothalamic releasing factors, anterior pituitary hormones produced, target glands, and target gland hormones are presented in Table 34-1. Pituitary hormone secretion is regulated by negative feedback. For anterior pituitary hormones, secretion of releasing factors from the hypothalamus is decreased when the concentration of target gland hormones is high and increased when it is low.
POSTERIOR PITUITARY HORMONES
Oxytocin
Therapeutic uses
Oxytocin is used intravenously for stimulation of labor and to induce postpartum lactation in cases of breast engorgement. Investigations are underway for its use in therapeutic interventions in a variety of conditions, especially those characterized by anxiety and aberrations in social function, such as autism.12
ANTERIOR PITUITARY HORMONES
Growth Hormone
Actions
In contrast to the direct effects of GH, the effects mediated by IGF-I are insulin-like. IGF-I acts through cell membrane receptors that resemble those of insulin. Insulin at high doses may act at IGF-I receptors and vice versa (see Chapter 36). In pharmacologic doses, GH causes an initial insulin-like effect followed by an effect antagonistic to that of insulin.
Therapeutic uses
GH (somatrem, somatropin) is used in the treatment of growth failure in children (pituitary dwarfism), wasting in acquired immunodeficiency syndrome (AIDS), and somatotropin deficiency syndrome. Short-term treatment of GH-deficient adults results in increased lean body mass, decreased fat mass, increased exercise tolerance, and improved psychological well-being. It is sometimes abused by athletes6 or used for its antiaging effect. GH is a potent anabolic agent and may have a role in clinical management of burn injuries. The GH-releasing hormone analogue sermorelin is used to treat GH deficiency in children who have growth retardation and diagnostically to determine the GH-releasing capacity of the pituitary. Octreotide, a somatostatin analogue that inhibits GH release, is approved for use in treating symptoms of vasoactive intestinal tumors, metastatic carcinoid tumors, and acromegaly. Other uses include AIDS-associated diarrhea and esophageal varices. Pegvisomant, a competitive antagonist of GH, is used to treat acromegaly.
Adverse effects
GH may induce relative insulin resistance. It has been documented to cause diabetes in AIDS patients16 and decreased insulin sensitivity that is dose-dependent, with a possible increase in type 2 diabetes in children.5 It may cause scoliosis in children. Arthralgia, especially in the hands and wrist, may occur. Patients may have headaches, especially in the first few months of therapy, and should be carefully observed (monitored) because of the possibility of intracranial hypertension.
Prolactin
Prolactin is an anterior pituitary hormone that is similar in structure to GH. Prolactin increases the growth of the secretory epithelium in the breast and stimulates the production of milk. Although prolactin is not used clinically, the secretion of prolactin can be altered by certain drugs. Because dopamine inhibits prolactin release (Table 34-2), drugs that affect dopamine levels or dopamine receptors in the pituitary affect prolactin release. Bromocriptine and cabergoline are dopamine-receptor agonists that are used to inhibit prolactin release and reduce the size of pituitary prolactin-releasing tumors.
TABLE 34-2 Hypothalamic Inhibitory Releasing Factors, Anterior Pituitary Hormones Inhibited, and Target Glands
| HYPOTHALAMIC HORMONE | PITUITARY HORMONE INHIBITED | TARGET ORGAN |
|---|---|---|
| Dopamine | Prolactin | Breast |
| Somatostatin | Growth hormone | Liver, bone, other |
THYROID HORMONES
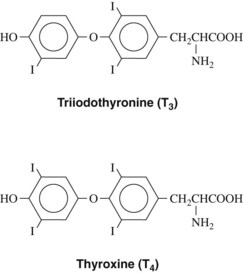
The active principles of the thyroid gland are iodine-containing amino acid derivatives of thyronine. They are formed from iodinated tyrosine residues. The structures are shown in Figure 34-1.
Synthesis of Thyroid Hormones
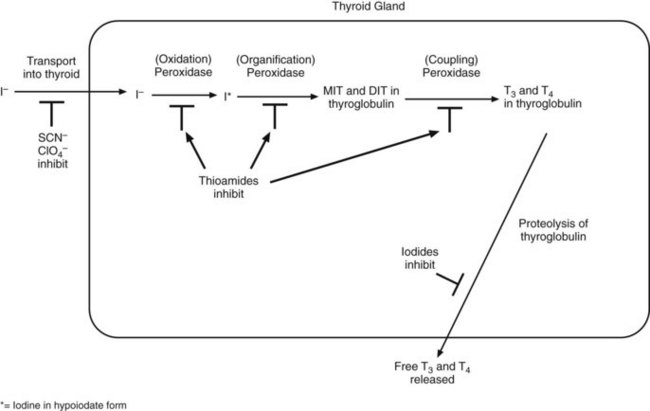
The synthesis of thyroid hormones is shown schematically in Figure 34-2. The first step is uptake of iodide by the thyroid gland. This step may be inhibited by ions of similar size and charge such as perchlorate. Iodide uptake is followed by oxidation of iodide to hypoiodite and iodination of tyrosyl groups of thyroglobulin to form iodotyrosyl groups. Tyrosine residues within the thyroglobulin molecule may be monoiodinated to monoiodotyrosine (MIT) or diiodinated to form diiodotyrosine (DIT). This step is catalyzed by thyroperoxidase and is rapid. Iodotyrosyl residues are coupled to form iodothyronyl residues within thyroglobulin. This may be either MIT plus DIT to form T3 or DIT plus DIT to form T4. The ratio of T4 to T3 formed is approximately 4 : 1. The coupling of iodotyrosyl groups is also catalyzed by peroxidase enzyme. Thyroid hormones are released by proteolysis of thyroglobulin. Most of the hormone released is T4, which is converted to T3 in peripheral tissues by iodothyronine deiodinases. T3 is about four times more potent than T4.
Actions of Thyroid Hormones
In addition, thyroid hormones are regulators of metabolism in most tissues. They increase basal metabolic rate and resting respiratory rate. Thyroid hormones stimulate the heart, resulting in the heart beating more rapidly and with greater force and an increase in cardiac output. Energy use in skeletal muscle, liver, and kidney is also markedly stimulated. T3 sensitizes the heart to the effects of circulating endogenous catecholamines by a direct effect on Ca++ channels,8 and thyroid hormones cause an increase in myocardial β-adrenergic receptors.17
THYROID DISORDERS
Hypothyroidism
Thyroid deficiency during development causes cretinism, which is characterized by gross retardation of growth and mental deficiency. In an adult, thyroid deficiency results in hypothyroidism and, in more severe cases, myxedema. Hypothyroidism is a common endocrine disorder affecting 1.4% to 2% of women and 0.1% to 0.2% of men. The prevalence of overt and subclinical hypothyroidism is significantly greater in women than in men and increases dramatically in women after age 40 years, affecting 5% to 10% of women older than 50 years.2 Subclinical hypothyroidism is common, especially among older women.15 It has been suggested that this condition may be associated with an increased mortality rate, particularly from cardiovascular disease and a subtle decrease in myocardial contractility.11 Subclinical hypothyroidism is associated with a small increase in low-density lipoprotein cholesterol and a decrease in high-density lipoprotein cholesterol, changes that increase risk of atherosclerosis and coronary artery disease.10 Cognitive impairment occurs in hypothyroidism, and attention, motor speed, memory, and visual spatial organization all are significantly impaired.4 In addition, hypothyroidism is an important risk factor for carpal tunnel syndrome.14
Replacement Therapy
Animal products include desiccated thyroid, which is composed of animal thyroid glands. Numerous preparations of levothyroxine sodium (T4) are available. Liothyronine sodium (T3) and liotrix, a mixture of T4 and T3 in a 4 : 1 ratio, are also available. Synthetic T4 has a uniform content and a long half-life and is the preferred and most widely used thyroid replacement medication. Because of its greater potential for cardiotoxicity and its shorter half-life, the use of T3 is controversial and much less frequent. Nevertheless, for some patients, the combination of T3 and T4 is better than T4 alone.3 Animal experiments have shown that in rats, only replacement of T3 and T4 ensures euthyroidism in all tissues.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses