Inflammatory Disease
Linda Lee
Disease Mechanism
Inflammatory lesions are the most common pathologic condition of the jaws. The jaws are unique from other bones of the body in that the presence of teeth creates a direct pathway for infectious and inflammatory agents to invade bone by means of caries and periodontal disease. The body responds to chemical, physical, or microbiologic injury with inflammation. The inflammatory response destroys or walls off the injurious stimulus and sets up an environment for repair of the damaged tissue.
Under normal conditions, bone metabolism represents a balance of osteoclastic bone resorption and osteoblastic bone production. This is a complex, interdependent relationship in which osteoblasts mediate the resorptive activity of the osteoclasts. Mediators of inflammation (cytokines, prostaglandins, and many growth factors) tip this balance to favor either bone resorption or bone formation. For the purposes of this chapter, all inflammatory conditions of bone, regardless of the specific etiology, are considered to represent a spectrum or continuum of conditions with different clinical features (e.g., site, severity, duration).
When the initial source of inflammation is a necrotic pulp and the bony lesion is restricted to the region of the tooth, the condition is called a periapical inflammatory lesion. When the infection spreads in the bone marrow and is no longer contained to the vicinity of the tooth root apex, it is called osteomyelitis. Another source of inflammatory lesions in bone is extension of inflammation into bone from the overlying soft tissues; this type of lesion includes periodontal lesions (see Chapter 19) and pericoronitis, an inflammation that arises in the tissues surrounding the crown of a partially erupted tooth. The names of the various inflammatory lesions tend to describe their clinical and imaging presentations and behavior; however, all have the same underlying disease mechanism, including a common response of the bone to the injury.
General Clinical Features
The four cardinal signs of inflammation—redness, swelling, heat, and pain—may be observed in varying degrees with inflammation of the jaws. Acute lesions are lesions of recent onset. The onset typically is rapid, and these lesions cause pronounced pain, often accompanied by fever and swelling. Chronic lesions have a prolonged course with a longer insidious onset and pain that is less intense. Fever may be intermittent and low grade, and swelling may occur gradually. Some chronic, low-grade infections may not produce any significant clinical symptoms.
General Imaging Features
Location
With periapical inflammatory lesions, which are pathologic conditions of the pulp, the epicenter typically is located at the apex of a tooth. However, lesions of pulpal origin also may be located anywhere along the root surface because of accessory canals or perforations caused by root canal therapy or root fractures. Periodontal lesions have an epicenter that is located at the alveolar crest. If periodontal bone loss is severe, the bone inflammatory changes may extend to the root furcation level or to the root apex. Osteomyelitis, a diffuse, uncontained inflammation of the bone, most commonly is found in the posterior mandible. The maxilla rarely is involved.
Periphery
Most often the periphery is ill defined, with a gradual blending of normal trabecular pattern into a sclerotic pattern, or the normal trabecular pattern may gradually fade into a radiolucent region of bone loss.
Internal Structure
The internal structure of inflammatory lesions presents a spectrum of appearances. Cancellous bone may respond to an insult by tipping the bone metabolic balance either in favor of resorption (giving the area a radiolucent appearance) or in favor of bone formation (resulting in a radiopaque or sclerotic appearance). Usually there is a combination of these two reactions. The radiolucent regions may show no evidence of previous trabeculation or a very faint pattern of trabeculation. The increased radiopacity is caused by an increase in bone formation on existing trabeculae. Radiographically, these trabeculae appear thicker and more numerous, replacing marrow spaces. In acute disease, resorption typically predominates; with chronic disease, excessive bone formation leads to an overall radiopaque, sclerotic appearance. In cases of osteomyelitis, careful examination of the x-ray images may reveal sequestra, which appear as ill-defined areas of radiolucency containing a radiopaque island of nonvital bone.
Effects on Surrounding Structures
The effects of inflammation on surrounding cancellous bone include stimulation of bone formation, resulting in a sclerotic pattern, or bone resorption, resulting in radiolucency. The periodontal ligament space (PDL) involved in the lesion is widened; this widening is greatest at the source of the inflammation. For example, with periapical lesions, the widening is greatest around the apical region of the root; the widening is greatest at the alveolar crest in periodontal disease. With chronic infections, root resorption may occur, and cortical boundaries may be resorbed. The periosteal component of bone, whether on the surface of the jaws or lining the floor of the maxillary sinus, also responds to inflammation. The periosteum contains a layer of pluripotential lining cells that, under the right conditions, differentiate into osteoblasts and lay down new bone. Inflammatory exudate from infection within the bone can penetrate the cortex, lift up the periosteum from the surface of the bone, and stimulate the periosteum to produce new bone. Because inflammatory exudate is a fluid, the periosteum is lifted from the surface of bone in a manner that positions the periosteum almost parallel to the surface of the bone; thus, the layer of new bone is almost parallel to the bone surface.
Periapical Inflammatory Lesions
Synonyms
Periapical inflammatory lesions have been called acute apical periodontitis, chronic apical periodontitis, periapical abscess, and periapical granuloma. Radiolucent presentations have been called rarefying osteitis, whereas radiopaque presentations have been called sclerosing osteitis, condensing osteitis, and focal sclerosing osteitis. Chapter 21 discusses periapical cysts of inflammatory origin (radicular cysts).
Disease Mechanism
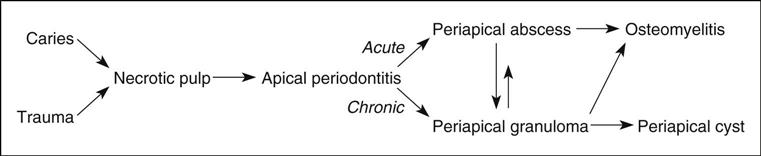
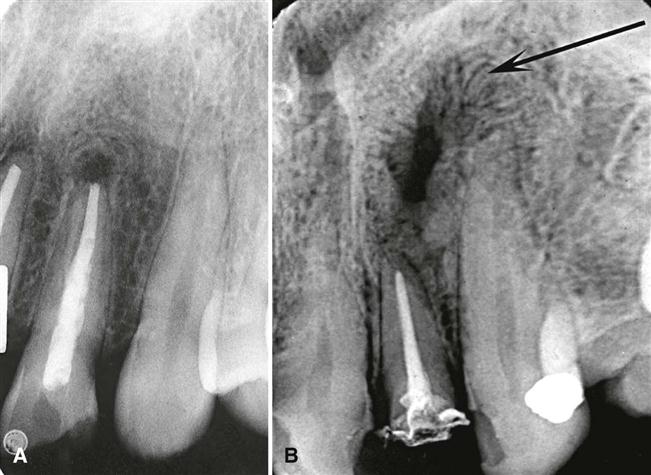
A periapical inflammatory lesion is defined as a local response of the bone around the apex of a tooth that occurs as a result of necrosis of the pulp or through destruction of the periapical tissues by extensive periodontal disease (Fig. 20-1). The pulpal necrosis may occur as a result of pulpal invasion of bacteria through caries or trauma. In Figure 20-1, the periapical inflammatory lesion is characterized by apical periodontitis, an inflammatory process that may histologically represent either a periapical abscess or a periapical granuloma. Toxic metabolites from the necrotic pulp exit the root apex to incite an inflammatory reaction in the periapical periodontal ligament and surrounding bone (apical periodontitis). This reaction is characterized histologically by an inflammatory infiltrate composed predominantly of lymphocytes mixed with polymorphonuclear neutrophils. Depending on the severity of the response, the neutrophils may collect to form pus, resulting in an apical abscess. This result is categorized as acute inflammation. Alternatively, in an attempt to heal from apical periodontitis, the body stimulates the formation of granulation tissue mixed with a chronic inflammatory infiltrate composed predominantly of lymphocytes, plasma cells, and histiocytes, giving rise to periapical granuloma. Entrapped epithelium (the rests of Malassez) may proliferate to form a radicular or apical cyst. Acute exacerbations of the chronic lesions may occur intermittently.
If the surrounding bone marrow becomes involved with the inflammatory reaction through the spread of pyogenic organisms (bacteria that stimulate an inflammatory response), the localized periapical abscess may transform into osteomyelitis. The exact point at which a periapical inflammatory lesion becomes osteomyelitis is not easily determined or defined. The size of the area of inflammation is not as important as the severity of the reaction. However, considering the size of the lesion, periapical inflammatory lesions usually involve only the local bone adjacent to the apex of the tooth, whereas osteomyelitis involves a larger volume of bone. Periapical lesions occasionally may be large, but the epicenter of the lesion remains in the vicinity of the tooth apex. If the periapical lesion extends farther, so that the lesion no longer is centered on the tooth apex, osteomyelitis may be considered as a possible diagnosis. The distinction between periapical inflammation and osteomyelitis can be made if sequestra are detected in x-ray images. Progression from periapical inflammation to osteomyelitis is relatively rare, and other factors play a role in its development, such as a decrease in the host defenses and an increase in the virulence of pathogenic microorganisms.
Clinical Features
The symptoms of periapical inflammatory lesions can range across a broad spectrum, from being asymptomatic to an occasional toothache to severe pain with or without facial swelling, fever, and lymphadenopathy. A periapical abscess usually manifests with severe pain, mobility, and sometimes elevation of the involved tooth, swelling, and tenderness to percussion. Palpation of the apical region elicits pain. Spontaneous drainage into the oral cavity through a fistula (parulis) may relieve the acute pain. In rare cases, a dental abscess may manifest with systemic symptoms (e.g., fever, facial swelling, lymphadenopathy) along with pain. The acute lesion may evolve into a chronic one (periapical granuloma or cyst), which may be asymptomatic except for intermittent flare-ups of “toothache” pain, which mark the acute exacerbation of the chronic lesion. Patients often give a history of intermittent pain. The associated tooth may be asymptomatic, or it may be mobile or sensitive to percussion. More often, however, the periapical lesion arises in the chronic form de novo; in this case, it may be asymptomatic. The clinical presentation does not necessarily correlate with the histologic or imaging findings.
Imaging Features
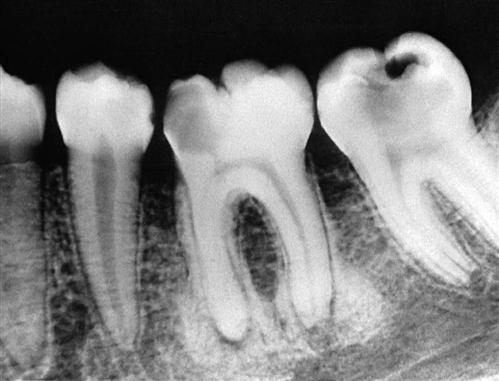
The features of periapical inflammatory lesions vary depending on the time course of the lesion. Because very early lesions may not show any changes in the image, diagnosis of these lesions relies solely on the clinical symptoms (Fig. 20-2). More chronic lesions may show lytic (radiolucent) or sclerotic (radiopaque) changes, or both.
Location
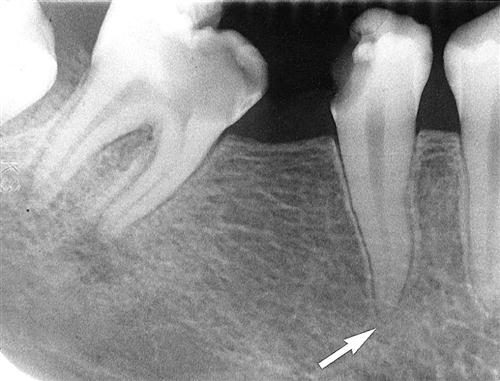
In most cases, the epicenter of periapical inflammatory lesions is found at the apex of the involved tooth (Fig. 20-3). The lesion usually starts within the apical portion of the periodontal ligament space. Less often, such lesions are centered about another region of the tooth root; this may occur because of accessory pulpal canals, perforation of the root structure from instrumentation of the pulp canal, and root fracture.
Periphery
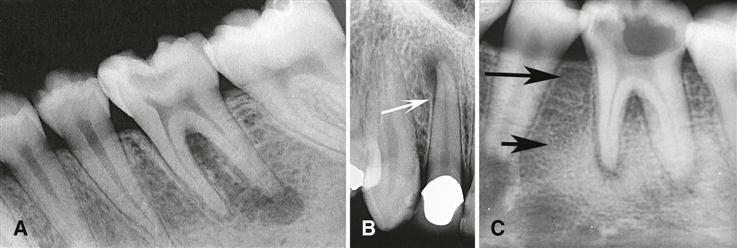
In most instances, the periphery of periapical inflammatory lesions is ill defined, showing a gradual transition from the surrounding normal trabecular pattern into the abnormal bone pattern of the lesion (Fig. 20-4; see Fig. 20-3, C). Rarely, the periphery may be well defined, with a sharp transition zone and an appearance suggesting a cortical boundary.
Internal Structure
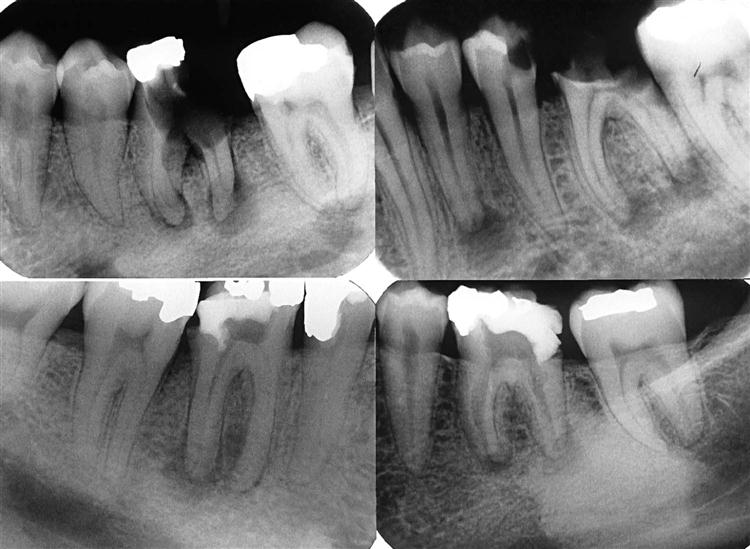
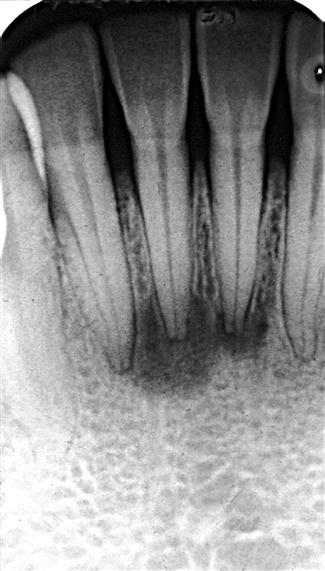
Early periapical inflammatory lesions may show no apparent change in the normal bone pattern. The earliest detectable change is loss of bone density, which usually results in widening of the periodontal ligament space at the apex of the tooth and later involves a larger diameter of surrounding bone. At this early stage, no evidence may be seen of a sclerotic bone reaction (see Fig. 20-2). Later in the evolution of the disease, a mixture of sclerosis and rarefaction (loss of bone giving a radiolucent appearance) of normal bone occurs (see Fig. 20-4). The percentage of these two bone reactions varies. When most of the lesion consists of increased bone formation, the term periapical sclerosing osteitis is used (Fig. 20-5), and when most of the lesion is undergoing bone resorption, the term periapical rarefying osteitis is used (see Fig. 20-3, A). The area of greatest bone resorption usually is centered on the apex of the tooth, with the sclerotic pattern located at the periphery. The radiolucent regions may be bereft of any bone structure or may have a faint outline of trabeculae. Close inspection of sclerotic regions reveals thicker than normal trabeculae and sometimes an increase in the number of trabeculae per unit area. In chronic cases, the new bone formation may result in a very dense sclerotic region of bone, obscuring individual trabeculae. Occasionally, the lesion may appear to be composed entirely of sclerotic bone (sclerosing osteitis), but usually some evidence exists of widening of the apical portion of the periodontal membrane space (see Fig. 20-5).
Effects on Surrounding Structures
As mentioned previously, periapical inflammatory lesions may stimulate either the resorption of bone or the manufacture of new bone. The lamina dura around the apex of the tooth usually is lost. The sclerotic reaction of the cancellous bone may be limited to a small region around the tooth apex or may be extensive in some cases. In rare instances in the mandible, the sclerotic reaction may extend to the inferior cortex. In chronic cases, external resorption of the apical region of the root may occur. If the lesion is long-standing, the pulp canal may appear wider than adjacent teeth. This is a result of the death of odontoblasts and subsequent cessation of the formation of secondary dentin, which occurs naturally with time to diminish the caliber of the pulp canal slowly.
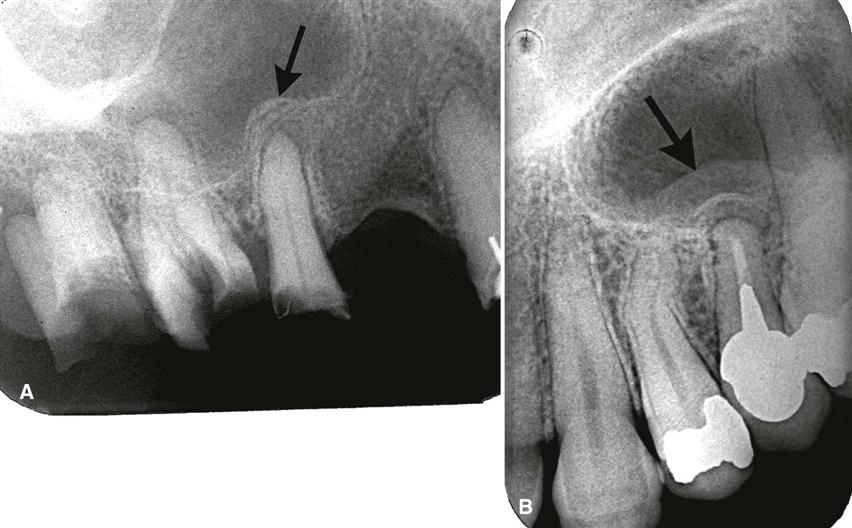
Nearby cortical boundaries may be destroyed, such as a segment of the floor of the maxillary antrum, the floor of the nasal fossa, or the buccal or lingual plates of the alveolar process immediately adjacent to the root apex. These lesions are capable of producing an inflammatory periosteal reaction, most notably in the adjacent floor of the maxillary antrum. This usually results in a thin layer of new bone produced by the inflamed periosteum within the maxillary antrum, sometimes referred to as a “halo shadow” (Fig. 20-6). A regional mucositis may be present within the adjacent segment of the maxillary antrum. Periosteal reaction may also occur on the buccal or lingual surfaces of the alveolar process and in rare cases on the inferior aspect of the mandible.
Differential Diagnosis
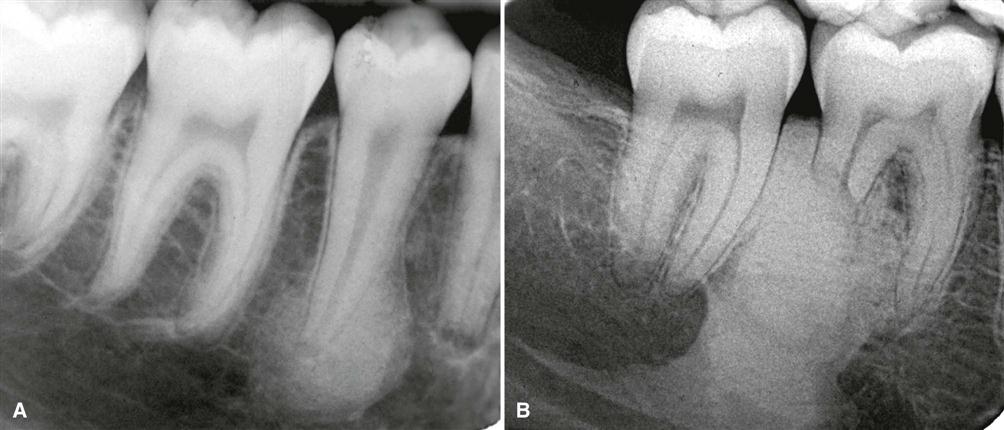
The two types of lesions that most often must be differentiated from periapical inflammatory lesions are periapical osseous dysplasia (POD) and a dense bone island (DBI) (enostosis, osteosclerosis) at the apex of a tooth. In the early radiolucent phase of POD, the imaging characteristics may not reliably differentiate this lesion from a periapical inflammatory lesion (Fig. 20-7). The diagnosis may rely solely on the clinical examination, including a test of tooth vitality. With long-standing periapical inflammatory lesions, the pulp chamber of the involved tooth may be wider than the adjacent teeth. More mature POD lesions may show evidence of a dense, radiopaque structure within the radiolucency, which helps in the differential diagnosis. Also, a common site for POD is associated with the apical region of the mandibular anterior teeth. External root resorption is more common with inflammatory lesions than with POD. When a DBI is centered on the root apex, it may mimic an inflammatory lesion. However, the periodontal ligament space around the apex of the tooth has a normal uniform width (Fig. 20-8). Also, the periphery of a DBI usually is well defined and does not blend gradually with surrounding trabeculae.
Small, radiolucent periapical lesions with a well-defined periphery simulating a cortex may be either periapical granulomas or cysts (radicular cysts). Differentiation may be impossible unless other characteristics of a cyst, such as displacement of adjacent structures and expansion of the outer cortical boundaries of the jaw, are present. Lesions larger than 1 cm in diameter usually are radicular cysts. If the patient has had endodontic treatment or apical surgery, a periapical radiolucency may remain that may look like periapical rarefying osteitis (Fig. 20-9). In either case, the destroyed bone may not be replaced with normal-appearing bone but with dense fibrous scar tissue. The differential diagnosis cannot be made on radiologic grounds alone; thus, the clinical signs and symptoms must take precedence.
In rare cases, metastatic lesions and malignancies such as leukemia may grow in the periapical segment of the periodontal membrane space. Close inspection of the surrounding bone may reveal other small regions of malignant bone destruction.
Management
Standard dental treatment of periapical lesions includes root canal therapy or extraction with the intention of eliminating the necrotic material in the root canal and the source of inflammation. If left untreated, the tooth may become asymptomatic because of drainage established through the carious lesion or a parulis. However, the possibility always exists that the lesion will spread to involve a larger area of bone, resulting in osteomyelitis, or into the surrounding soft tissue, which may result in a space infection or cellulitis.
Post–Endodontic Treatment Complications
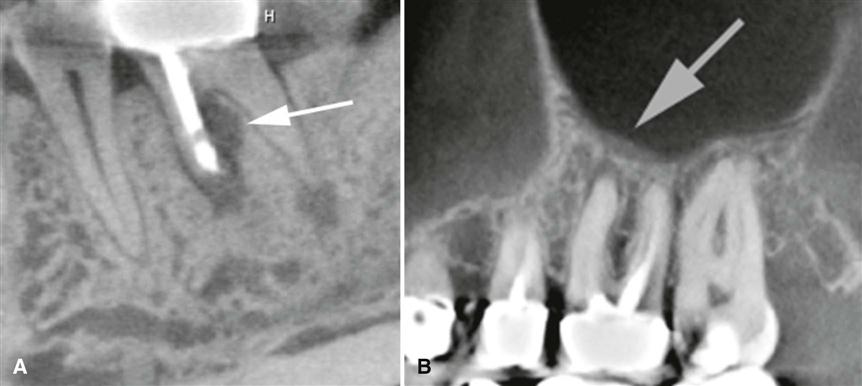
Recurrence or persistent inflammatory lesions after endodontic treatment can occur. Possible etiologies include inadequate root canal filling, instrumentation perforation of the outer root surface, unusual morphology of the root canal, unusual accessory canal, or root fracture. The application of high-resolution, small field-of-view cone-beam computed tomographic (CBCT) images has been very useful in the determination of the etiology of these complications (Fig. 20-10).
Osteomyelitis
Disease Mechanism
Osteomyelitis is an inflammation of bone. The inflammatory process may spread through the bone to involve the marrow, cortex, cancellous portion, and periosteum. In the jaws, pyogenic organisms that reach the bone marrow from abscessed teeth or postsurgical infection usually cause osteomyelitis. However, in some instances, no source of infection can be identified, and hematogenous spread is presumed to be the origin. In some patients, no infectious organisms can be identified, possibly because of previous antibiotic therapy or inadequate methods of bacterial isolation. Bacterial colonies also may be present in small, isolated pockets of bone that may be missed during sampling.
In patients with osteomyelitis, the bacteria and their products stimulate an inflammatory reaction in bone, causing resorption of the endosteal surface of the cortical bone. This resorption may progress through the cortical bone to the outer periosteum. In young patients, in whom the periosteum is more loosely attached to the outer cortex of bone than it is in adults, the periosteum is lifted up by inflamm/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses