43 Salivary Gland Transplantation
Physiology and Pathophysiology
Regulation of Secretory Function
Denervation of Salivary Glands
Warm Ischemic Tolerance of Salivary Tissue
Indications and Patient Selection
Submandibular Gland Transfer to Prevent Postradiation Xerostomia
Patient Information and Consent
Measures for Specific Complications
Introduction
The goal behind the idea of salivary gland transplantation is to permanently substitute any lost physiological lubrication by using secretion from one of the major salivary glands. The transplantation can be performed in an autologous or homologous fashion, as a vascularized or nonvascularized transfer. Only vascularized gland transfer is of clinical relevance, as experimental and clinical trials have shown that functioning vital salivary tissue was only found in a small percentage of cases after nonvascularized transfer even of small pieces of gland tissue.
Definition
Only autologous transfer of the submandibular gland is clinically established. This means transferring the denervated submandibular gland, including the afferent branches of the facial vessels, which are anastomosed in the recipient region in order to vascularize the gland. The Wharton duct has to be transferred with the gland and implanted into the area in which the secretion is to be replaced.
This method was pioneered by Murube del Castillo 25 years ago for the treatment of severe aqueous-deficient dry eyes,1 and has been modified and clinically established by others during the last 20 years.2–5 As severe cases of keratoconjunctivitis sicca are still the main indication for submandibular gland transfer, the following description of the surgical procedure, results, and outcome focuses on this procedure.
 Only autologous salivary gland transplantation is an established procedure.
Only autologous salivary gland transplantation is an established procedure.
Physiology and Pathophysiology
 Regulation of Secretory Function
Regulation of Secretory Function
The regulation of salivary gland function—the quality and quantity of saliva produced—primarily depends on complex synergistic and antagonistic influences in the parasympathetic and sympathetic autonomic innervation. Reflex salivary flow occurs at a low “resting” rate, and for short periods taste or chewing stimuli can evoke up to a tenfold increase in salivation.6 The parotid gland has a very low secretory rate in resting conditions in comparison with stimulation. By contrast, the submandibular and sublingual glands secrete relatively more saliva in resting conditions.7
All salivary glands are supplied by cholinergic parasympathetic nerves, which release acetylcholine that binds with muscarinic receptors (M3 and M1), evoking the secretion of saliva by acinar cells in the substance of the gland. Most glands also receive variable innervation from sympathetic nerves, which releases norepinephrine, promoting greater release of stored proteins, mostly from acinar cells but also from ductal cells.6 Water and electrolytes are actively transported by the acinar cells and the electrolyte content is modified, principally with the removal of sodium chloride, during passage through the ductal system to the mouth—converting saliva from an isotonic solution into a hypotonic solution.8
Parasympathetic and sympathetic nerves influence most cell types in the salivary glands, including the myoepithelial cells and blood vessels. Numerous other nonadrenergic, noncholinergic (NANC) transmitters have also been found in the nerves in salivary glands, all of which may play a role in the complex neuronal and humoral regulation of the secretion pattern.9
 Denervation of Salivary Glands
Denervation of Salivary Glands
Functional and structural long-term changes in surgically denervated salivary glands have been studied in numerous animal experiments for about 150 years.10,11 In the initial period after denervation, distinct secretion continues for a few days; this can be attributed to the release of neurotransmitters from degenerating postganglionic axons at neuroeffector terminals.12 Subsequent cessation of flow for several months coincides with a gross reduction in transmitter release, leading to some parenchymal atrophy and the development of hypersensitivity to humoral agents. Afterwards, increasing permanent “resting” secretion occurs. This paradoxical phenomenon was described and termed “paralytic secretion” by Claude Bernard in the 19th century. It was later attributed to circulating adrenal catecholamines and spontaneous leakage of acetylcholine from residual postganglionic parasympathetic axon terminals, both acting on the hypersensitive parenchymal cells.13,14
Clinical and experimental results have shown that there is long-lasting survival of functioning salivary tissue after denervation. Some reinnervation of submandibular glands due to sprouting from intact nerves has been described. Heterologous synaptic contacts may occur, with new adrenergic axons sprouting down existing parasym-pathetic trunks and in time developing functional connections with parasympathetic ganglionic cells.15,16
De novo parasympathetic innervation of regenerated acinar cells has also been demonstrated in rat submandibular glands after experimental atrophy of the gland caused by duct ligation.17
 Salivary gland transplantation leads to irreversible denervation of the original neural supply. Despite this, salivary secretion is restored by alternative mechanisms.
Salivary gland transplantation leads to irreversible denervation of the original neural supply. Despite this, salivary secretion is restored by alternative mechanisms.
 Warm Ischemic Tolerance of Salivary Tissue
Warm Ischemic Tolerance of Salivary Tissue
The warm ischemic tolerance of the tissue was examined in a rabbit model (of the submandibular and parotid glands) and showed increasing structural damage to the parenchyma, with an emphasis on the secretory cells, starting after 1.5 hours of ischemia. Islands of viable salivary tissue with marked predominance of remaining ductal elements were seen within areas of necrotic tissue and inflammatory cells. Cell proliferation in the remaining salivary gland tissue was significantly increased 14 days after the start of ischemia, as a sign of incipient regeneration of the gland tissue. After warm ischemia, a raised apoptotic rate was not detected. Consequently, genetic damage to the salivary gland cells inducing apoptosis did not occur. Accordingly, interruption of the blood supply to the gland for more than 1 hour during the transfer should be avoided, and this is in accordance with the surgical management described below.18
 The warm ischemia time should be less than 1 hour.
The warm ischemia time should be less than 1 hour.
Indications and Patient Selection
 Keratoconjunctivitis Sicca
Keratoconjunctivitis Sicca
Keratoconjunctivitis sicca is a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability, with potential damage to the ocular surface. Keratoconjunctivitis sicca, also known as dry eye disease, has an estimated prevalence of 5% to more than 30%, depending on the selection of study groups and the diagnostic criteria used. It has been shown that its development is associated with increasing age and female sex.19
Submandibular gland transfer to treat the condition is only indicated in patients who have had unsuccessful ophthalmological treatment using the full range of established procedures for treating keratoconjunctivitis sicca who have been suffering from progressive structural changes on the ocular surface for years. This small group of patients have an absolute aqueous tear deficiency (Schirmer test ≤ 1 mm) with persistent severe pain despite punctual occlusion, a trial of scleral or limbal fit rigid contact lenses, at least half-hourly application of unpreserved tear substitutes, and a conjunctivalized corneal surface.20,21
 Submandibular Gland Transfer to Prevent Postradiation Xerostomia
Submandibular Gland Transfer to Prevent Postradiation Xerostomia
To allay the symptoms of xerostomia caused by radiotherapy to the head and neck region, one submandibular gland can be preserved during the ablative oncological operation and moved to the submental space. During this procedure, the gland remains pedicled to the distal branches of the facial vessels, causing retrograde perfusion, and to the parasympathetic root and the duct. The gland is subsequently shielded from radiation.22,23 As this procedure simply involves moving the pedicled gland, the technique is not discussed further in this chapter.
 The most important indications for gland transplantation are keratoconjunctivitis sicca and prevention of postradiation xerostomia.
The most important indications for gland transplantation are keratoconjunctivitis sicca and prevention of postradiation xerostomia.
Diagnostic Work-Up
Before surgery, a detailed medical history regarding the etiology, course, and previous treatment of the keratoconjunctivitis sicca has to be taken. The present status of the disease is recorded, and the remaining options for conservative treatment are assessed by a consulting ophthalmologist.
To exclude destructive inflammatory processes of the salivary glands (e.g., in connection with systemic disease such as Sjögren syndrome), a biopsy is taken from the minor salivary glands of the lower lip and examined histologically. For functional evaluation, the secretory activity of the salivary glands of the head are documented scintigraphically using technetium Tc 99 m pertechnetate. As the results of this method correlate strongly not only with the clinicopathologic features of the salivary gland but also those of the lacrimal gland, this method can be used to exclude Sjögren syndrome preoperatively and to document the gland’ s viability postoperatively.24,25
Patient Information and Consent
Patients have to be informed about the expected course of the keratoconjunctivitis sicca using the remaining conservative treatment options, and about the expected course with or without submandibular gland transplantation. The patient consent procedure focuses on the expected course of keratoconjunctivitis sicca after submandibular gland transplantation (see Outcome, below) and possible complications due to the donor or recipient regions (see Complications, below).
Anesthesia and Positioning
The operation is carried out with the patient under general anesthesia, preferably with nasal intubation. The head should be placed in a straight position and covered in such a way that it can be turned.
Operative Steps
Harvesting the Submandibular Gland
The submandibular gland, including the Wharton duct and afferent branches of the facial vessels, is dissected via a conventional cervical access route to the submandibular triangle, protecting the submandibular division and the descending branch of the facial nerve. The gland can be taken from the ipsilateral or contralateral side to the recipient eye. If the procedure is indicated for the treatment of both eyes, the more severely affected eye is treated first and the second gland is transferred to the other side 3 months later at the time of the regular 3-monthly postoperative check-up.
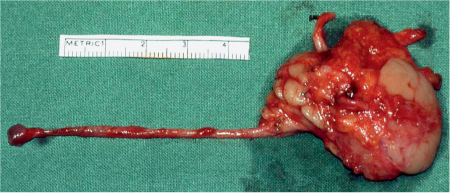
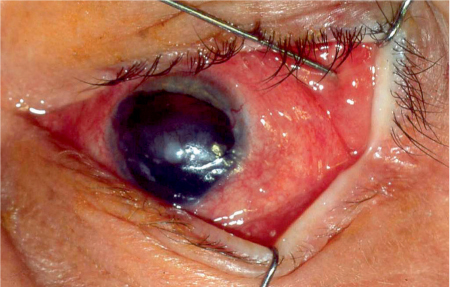
To harvest the whole length of the Wharton duct, the distal part is dissected using an intraoral approach after a blunt probe has been inserted into the orifice to label the duct. In patients with Stevens–Johnson syndrome or ocular pemphigoid, in which scarring has obliterated the conjunctival fornix, a cuff of mucosa from the floor of the mouth is included with the duct opening (Fig. 43.1). After the duct has been freed from both sides, it can be pulled out through the cervical access. The parasympathetic innervation of the gland—nerve fibers branching from the lingual nerve—is sacrificed, but the lingual nerve has to be carefully protected. Before the gland’s afferent vessels are severed, the recipient area should be prepared, including the recipient vessels, in order to shorten the ischemic period for the gland.
Preparation of the Recipient Temporal Area
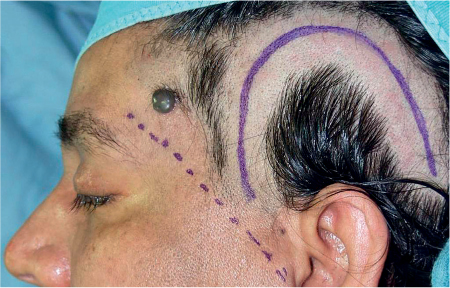
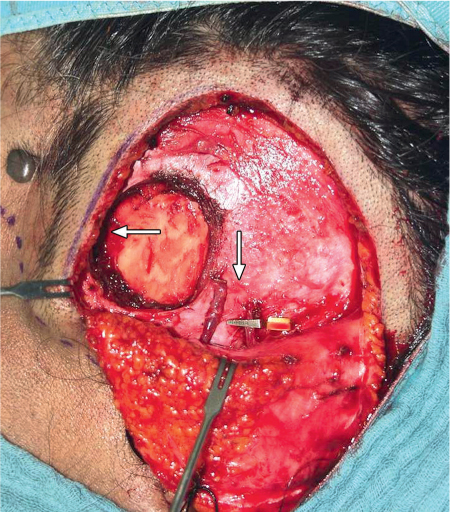
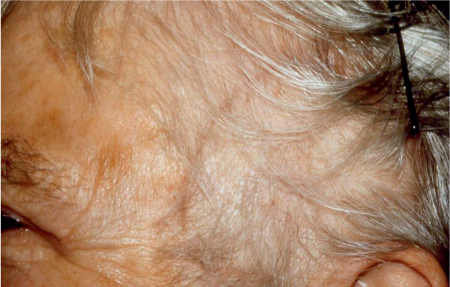
An enlarged, caudally based U–shaped temporal flap is elevated. The most ventral edge of the flap should be located close behind the landmarks for the course of the frontal branch of facial nerve (Fig. 43.2). This flap should be lifted in a layer below the superficial temporal vessels. When the flap has been raised, the vessels are carefully dissected free from the undersurface of the flap. Preoperative identification of the superficial temporal vessels using ultrasound will reassure the surgeon, but this is not mandatory as they are usually easy to identify. Alternatively, branches from the deep temporal vessels can be used if the superficial vessels are too small or have been sacrificed in previous operations. The deep temporalis fascia is incised and the temporalis muscle is fenestrated down to the underlying temporal bone to create a pocket to host the gland. This pocket should be dissected as far caudally and ventrally as possible, without risking the frontal branch of the facial nerve, to stay in a region in which the temporal muscle is thick enough to provide space for the gland and have a suitable distance to the conjunctival fornix that can be bridged with the Wharton duct.
Fig. 43.1 A submandibular gland with its vascular pedicle and the Wharton duct.
Fig. 43.2 The recipient area, with marking of the U–shaped flap behind the expected course of the frontal branch of the facial nerve (dotted line) and just behind the hairline.
Fig. 43.3 The exposed temple, with the recipient bed for the gland and dissected temporal vessels (horizontal arrow). A tunnel dissected subcutaneously leads to the upper conjunctival fold (vertical arrow).
A subcutaneous tunnel to the superotemporal conjunctival fornix is created using blunt dissection (Fig. 43.3).
Gland Transfer
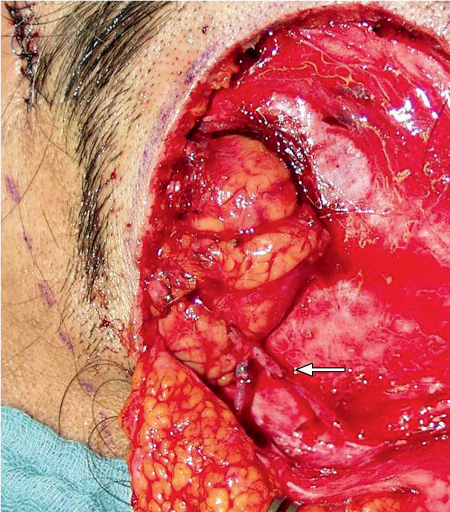
After the afferent vessels have been severed, the gland is removed, irrigated with heparinized saline, transferred to the new temporal bed, and anastomosed to the recipient vessels to reestablish the blood supply (Fig. 43.4). The Wharton duct is passed through the tunnel to the upper lateral conjunctival fornix. The distal end of the duct is sutured to an opening in the superotemporal conjunctival fornix in the same way as if marsupialization were being performed (Fig. 43.5).
Suction drains are applied at the donor and recipient sites before wound closure in layers. No occlusive wound dressing is used (Fig. 43.6).
Fig. 43.4 The transferred submandibular gland, connected to the superficial temporal vessels (the arrow indicates the anastomoses).
Fig. 43.5 Probe inside the implanted submandibular duct.
Fig. 43.6 The temporal area 3 months postoperatively, with the scar behind the hairline.
Fig. 43.7 Reduction procedure. The reexposed recipient region 1 year after submandibular gland transfer. Status post removal of the craniodorsal part of the transplant away from the vascular pedicle and the excretory duct. The arrow indicates the residual part of the graft, and the inset shows the resected part of the graft.
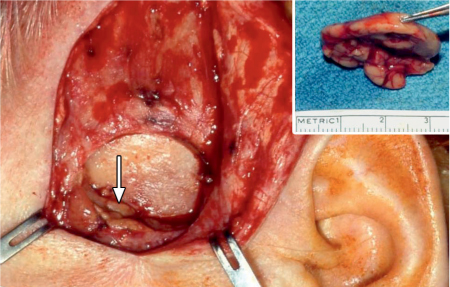
Gland Reduction
Due to inappropriate secretion, some patients develop excessive salivary epiphora (see Outcome, below), which can be managed with surgical reduction of the transplant (months to years after gland transfer). After the recipient area has been reexposed, about one-third to two-thirds of the gland tissue is excised, depending on the desired extent of functional reduction. The dorsocranial part of the gland should always be taken to avoid damage to the secretory duct or afferent vessels (Fig. 43.7). The cut surface of the remaining part of the transplant should be cauterized to avoid retrograde tearing before the wound is closed. A wound dressing is applied for about 3 days to apply some pressure to the reduction area.
 Tips and Tricks
Tips and Tricks
The Wharton duct is the most sensitive structure in the transplant. Any damage to it should be avoided, as scarring of the duct, which is only intermittently filled with saliva, could lead to obstruction. For the same reason, distortion of the duct should be excluded before it is implanted into the conjunctival fornix. The position of the recipient bed behind the course of the frontal branch of the facial nerve means that the full length of the secretory duct has to be used to bridge the distance to the superolateral conjunctival fornix without the duct being stretched too much.
Bleeding from paraductal veins may be problematic if it occurs after revascularization of the gland and implantation of the duct into its tunnel. Before the gland’s afferent vessels are severed in the donor area, therefore, the duct should be checked carefully for any bleeding in its vicinity.
After the transfer, the gland should be anastomosed immediately, as an ischemic period of more than 60 minutes could result in cellular damage to the susceptible glandular parenchyma.
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses