Easily manipulated with ample working time
Radiopaque and easily discernible on radiographs
Dimensionally stable with no shrinkage once inserted
Seals the canals laterally and apically conforming to the canal anatomy
Impervious to moisture and non-porous
Inhibits bacterial growth
Non-irritating to periapical tissues
Unaffected by tissue fluids (no corrosion or oxidation)
Does not stain tooth structure
Easily removed from canal if necessary
Sterile
Table 7.2
Overview of core materials and chemical composition
|
Function
|
Material
|
Main composition
|
|---|---|---|
|
Core materials
|
Gutta-percha
|
Gutta-percha, zinc oxide, metallic salts, waxes, colouring agents
antioxidants
|
|
Silver point
|
Silver, nickel
|
|
|
Resin-coated gutta-percha
|
Resin, gutta-percha, zinc oxide
|
|
|
Thermoplastic polymer
|
Polyester, bioactive glass, bismuth oxychloride, barium sulphate
|
|
|
Mineral trioxide aggregate (MTA)
|
Tricalcium silicate
Dicalcium silicate
Bismuth oxide
Calcium sulphate
Tetracalcium aluminoferrite
|
Table 7.3
Overview of sealers, chemical composition and various brands
|
Type
|
Brand
|
Main composition
|
|---|---|---|
|
Zinc oxide eugenol
|
Roth
|
Zinc oxide eugenol, colophony, bismuth and barium
|
|
Kerr PCS
|
Zinc oxide eugenol, thymol, silver
|
|
|
ProcoSol
|
Zinc oxide eugenol, colophony, bismuth and barium
|
|
|
Endomethasone
|
Zinc oxide eugenol, paraformaldehyde
|
|
|
Resin
|
AH Plus
|
Epoxy-bis-phenol resin, adamantine
|
|
Epiphany
|
BisGMA, UDMA and hydrophilic methacrylates
|
|
|
EndoREZ
|
UDMA
|
|
|
Russian Red
|
Recorcin-formaldehyde
|
|
|
Calcium hydroxide
|
Sealapex
|
Toluene salicylate, calcium oxide
|
|
Apexit
|
Salicylates, calcium hydroxide
|
|
|
Silicone
|
RoekoSeal
|
Polydimethylsiloxane, silicone oil, zirconium oxide, catalyst
|
|
GuttaFlow
|
Polydimethylsiloxane, silicone oil, zirconium oxide, gutta-percha
|
|
|
Glass ionomer
|
Ketac Endo
|
Polyalkenoate cement
|
|
Calcium silicate
|
iRoot SP
|
Calcium phosphate, calcium silicate, zirconium oxide, calcium hydroxide
|
Jasper introduced silver cones and due to their inherent rigidity made them easy to place. However, their inability to fill the irregularly shaped root canal system permitted leakage and resulted in corrosion products that were found to be highly cytotoxic. Silver cones are no longer utilised for root canal obturation [9, 10].
Sargenti and Richter proposed the principle of mummification and fixation of pulp tissues using highly toxic paraformaldehyde pastes (N2) in view of its antimicrobial activity. Other paste fills containing formaldehyde include SPAD and Endomethasone. Inadvertent overextension often resulted in biologically unacceptable outcomes or worse still with accompanying neural damage. Coupled with treatment difficulties encountered when attempting to retreat such cases, their use is frowned upon within the endodontic community [11–15].
Gutta-percha has proven to be the material of choice worldwide for successful obturation when used in combination with a root canal sealer. Gutta-percha points alone cannot completely seal the space between the dentinal walls, root canal irregularities, lateral and accessory canals and spaces between gutta-percha points used in lateral condensation. So a sealer is necessary and volumes should be minimal to counteract possible shrinkage and dissolution over time. Gutta-percha can be made to flow using heat or using solvents, such as chloroform or eucalyptus, as well as by ultrasonics and vibration. It is a trans-isomer of polyisoprene and exists in two crystalline forms alpha and beta, with differing properties. The use of alpha phase gutta-percha has increased as thermoplastic techniques have become more popular [16–18].
Gutta-percha cones consist of approximately 20 % gutta-percha, 65 % zinc oxide, 10 % radiopacifiers and 5 % plasticisers. Gutta-percha points are manufactured in various forms including standardised points that match the ISO sizes and have a 2 % taper. Accessory points have fine tips and variable taper to facilitate and improve lateral compaction. Greater taper points are available in 4 % and 6 % taper to match modern rotary preparation techniques. Feather-tipped points permit individual and specific cone fitting to the prepared root canal. As clinicians we expect that the diameter and tapers of GP cones to be accurate with no variability. However, even if the manufacturers are following the current standards, the accepted diameter tolerance levels vary from 0.05 to 0.07 mm, depending on the cone size. Such tolerance means that cones of one size can theoretically span more than one size above and/or below the stated size. For example, a size #30 cone has an allowable tip diameter of 0.23–0.37 mm. Therefore, the nominal diameter and taper values as listed by the manufacturer may vary greatly and still be in accordance with the ‘standards’. Gutta-percha points cannot be sterilised by traditional means, but rapid disinfection before use by placing the cones with 70 % isopropyl alcohol, 2 % chlorhexidine or 1–5 % NaOCl is effective [16, 19, 20].
Several techniques have been developed for placing gutta-percha into root canals including single-cone techniques [21, 22], cold lateral compaction [23], warm lateral compaction using ultrasonics (energised spreading) [24], chloroform customization of gutta-percha [25], thermomechanical compaction technique [26], hybrid thermomechanical compaction technique [27], warm vertical compaction [28], continuous wave compaction [29], thermoplasticised injectable gutta-percha obturation [30] and solid-core carry insertion techniques [31, 32] (see Figs. 7.1 and 7.2).
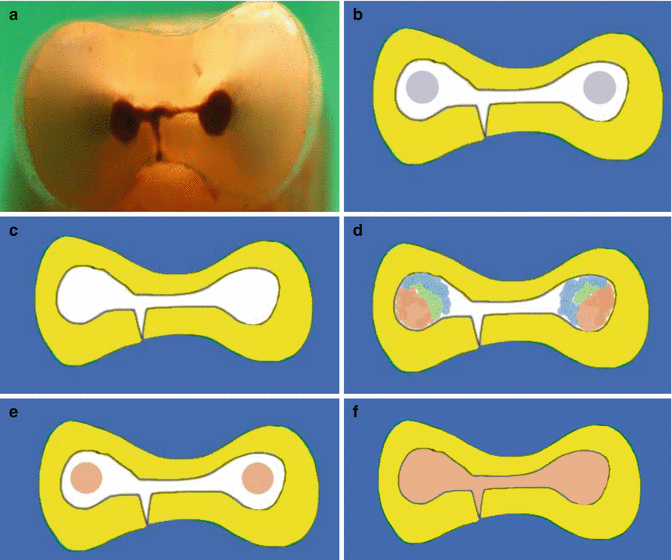
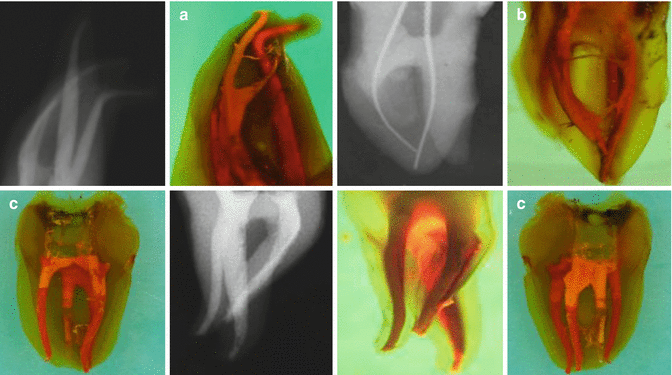

Fig. 7.1
Diagrams representing different methods of obturation used and how these techniques fill the apical portion of the tooth. Note (a) sectioned mesial root of a lower first molar demonstrating apical anatomy 1 mm from the apex. Obturation methods using (b) silver points, (c) paste fill, (d) single-cone obturation, (e) cold lateral compaction, and (f) warm lateral compaction, and warm vertical compaction, thermomechanical compaction, and injection techniques (Adapted from Professor John Whitworth)

Fig. 7.2
Clinical radiographs and photographs demonstrating different methods of obturating root canals. Extracted teeth were obturated using different techniques and cleared. Note (a) obturation using McSpadden technique resulting in filling of MB2 canal and lateral anatomy. (b) Extracted upper first molar tooth with two palatal canals. Obturation technique using warm vertical compaction. (b, c) The importance of disinfection is highlighted by the fins and isthmuses between the two roots that cannot be prepared using mechanical preparation alone
As no single approach can unequivocally boast superior evidence of healing success, the choice depends on the canal anatomy, unique objectives of treatment in each case and other factors such as speed, simplicity and economics [33].
A shortcoming of gutta-percha-based root filling materials is their lack of adhesiveness to the canal walls relying on a sealer or cement interface to fill the voids and irregularities in the root canal, lateral and accessory canals and spaces between gutta-percha and dentinal walls and within the homogenous mass of gutta-percha created using lateral condensation techniques. A number of different type of sealers are commercially available including zinc oxide eugenol [34, 35] and non-eugenol paraformaldehyde-containing sealers [15, 36], calcium hydroxide sealers [37], methacrylate resin-based sealers [38, 39], silicone sealers [40, 41] and calcium silicate sealers [42] (Table 7.3).
Resin-based obturation systems have been introduced as an alternative to gutta-percha in an attempt to seal the root canal system more effectively by creating an intra-canal adhesive monoblock and perhaps strengthening the root. Resilon is a biodegradable polycaprolactone-based thermoplastic root filling material bondable to self-etch or self-adhesive root canal sealer. It resembles gutta-percha and can be manipulated similarly, consisting of a resin core material, available in conventional/standardised cones or pellets, and a resin sealer. However, the desire to create a three-dimensional impervious seal has been unattainable due to ineffective bonding challenges due to root canal anatomy and limitations in the physical and mechanical properties of current adhesive materials [43, 44].
Calcium hydroxide, dentine plugs and hydroxyapatite have been advocated for placement as barriers in canals exhibiting an open apex to permit obturation, whilst minimising extrusion of traditional materials such as gutta-percha into the periradicular tissues. Placement of an apical barrier and immediate obturation is an alternative to apexification. Mineral trioxide aggregate (MTA) has been suggested as an apical barrier material. MTA is sterile, biocompatible and capable of inducing hard tissue formation. The material is compacted into the apical portion of the root and allowed to be set, and then gutta-percha can be compacted without extrusion. The technique is quick and clinically successful and eliminates the need for numerous visits and possible recontamination during apexification or theoretical root fracture [45, 46].
The apical limit of root canal instrumentation and obturation has been contested with most clinicians preferring to end biomechanical preparation at the apical constriction. All endodontic materials including relatively inert gutta-percha can induce a foreign body-type reaction when extruded beyond the confines of the root canal system. A small puff of sealer through apical ramifications or lateral canals, although viewed by some clinicians as a sign of optimal cleaning and shaping, can elicit an inflammatory response resulting in on-going pain for the patient [47].
Overfilling of root canals (beyond the radiographic apex) can occur as a result of inflammatory apical root resorption, an incompletely formed root apex or over-instrumentation through the apical foramen resulting from inaccurate measurement of the working length. In such cases, creating an apical stop becomes more difficult, thus leading to overfilling. There is evidence to suggest that overfilling has a negative effect on the long-term prognosis for endodontic therapy whereby the filling material might act as a foreign body, causing irritation of the periradicular tissues, localised inflammation and a delay in periradicular healing [48].
Gross overextension of root canal filling material and/or sealer is potentially harmful, particularly when in contact with vital structures such as the inferior alveolar nerve or maxillary sinuses. The use of toxic materials should be avoided, and even common sealers that do not release formaldehyde can result in paraesthesia when in contact with surrounding neural tissues. Mechanical compression, chemical neurotoxicity and local infection as a direct result of overfilling may cause irreversible nerve damage. Neural injuries, most commonly classified using the Seddon classification, include neurotmesis (the most severe), axonotmesis or neuropraxia. If neurological complaints appear after root filling in the lower jaw, a nerve injury due to root filling material should be ruled out. In cases of overfilling, an urgent referral may be warranted to consider immediate apicectomy and decompression of the nerve with conservation of the tooth if possible, thereby providing the best chance of avoiding permanent nerve damage. Even if nerve damage is not suspected, the clinician must bear in mind that the worst prognosis for endodontics is instrumentation and filling beyond the confines of the canal and this should be avoided in every case [4, 6, 7, 11, 13, 14, 49–51].
Root canal-treated teeth with overextension of the root canal sealer or solid materials such as gutta-percha or silver points into the sinus may be the main aetiological factor for aspergillosis of the maxillary sinus in healthy patients. Root filling materials such as zinc oxide eugenol are considered to be a growth factor for Aspergillus. Aspergillus fumigatus need heavy metals such as zinc oxide for proliferation and metabolism. If the sinus has been breached with extrusion of either sealer or solid materials, then mechanical irritation can ensue resulting in an inflammatory reaction. Management of extruded obturation material into the maxillary sinus may require surgical endodontic therapy with the need for a subsequent Caldwell-Luc approach to remove the foreign material from within the sinus cavity [52–54].
Underfilling or incomplete filling of the root canals (more than 2 mm short of the radiographic apex) often occurs as the result of incomplete instrumentation, ledge formation or loss of working length due to inadequate irrigation and recapitulation of canal patency. The latter leads to the accumulation of dentine filings and debris resulting in canal blockage. Consequently, the clinician does not remove the infected necrotic tissue remaining in the apical portion of the root canal leading to persistent bacterial infection that may initiate or perpetuate periradicular inflammation after endodontic therapy. There is evidence to suggest that instrumentation and filling canals as near to the apical terminus as possible (‘canal terminus patency’ and ‘extension of canal cleaning as close as possible to its apical terminus’) and prevention of overfilling (‘extruded root filling’) are important prognostic factors for the long-term survival of endodontically treated teeth [55–59].
The life of an endodontically treated tooth depends on the accuracy of the diagnosis and planning, excellence of disinfection, instrumentation and filling procedures (antimicrobial strategies, root canal shaping and coronal and apical seal) and finally the rehabilitation management [60].
The final obturation is a culmination of all these factors giving us an insight into the likely success of treatment provided where due diligence and optimal treatment were performed at all stages. Choice of obturation technique and materials used is based both on sound scientific evidence and also the concept of what works best in your hands. No single approach can unquestionably be relied upon to successfully manage all cases. Instead the clinician should be aware of several different approaches that can be applied according to individual tooth requirements (see Fig. 7.3).
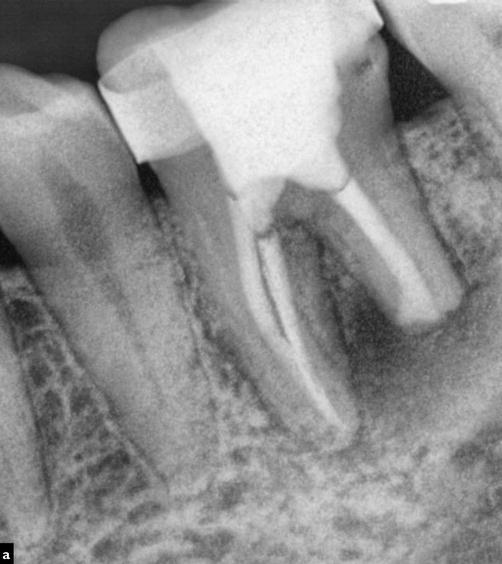

Fig. 7.3
Clinical radiograph demonstrating the use of different obturation techniques in a single case to achieve the desired outcome of a correctly cleaned, shaped, and obturated case. The distal canal had a very unusual apical irregular shape as a result of extensive apical root resorption that had occurred. The mesial canals were obturated using a warm vertical compaction technique. The distal canal was filled using MTA in the apical 1/3 followed by Obtura back fill. The use of conventional techniques would have increased the risk of a gross overfill which may only have been rectified by further surgery. This was avoided due to an understanding of techniques available including inherent limitations, thereby selecting the most appropriate technique which would achieve the necessary result with minimal complications
7.2 Sealers
Sealers are used between dentine surfaces and core materials to seal the space, between core and filling materials in lateral condensation techniques, to seal complex canal anatomical irregularities such as lateral canals and dentinal tubules, to lubricate and facilitate seating of the master cone and provide antimicrobial properties within the final obturation material. Traditionally desirable characteristics were to adhere to dentine and the core material as well as to have adequate cohesive strength. Newer-generation sealers are being engineered to improve their ability to penetrate into dentinal tubules and bond to, instead of just adhering to, both the dentine and core material surfaces. Although no sealer meets all the properties of Grossman’s ideal sealer (Table 7.1), there are many sealers available that are clinically acceptable and widely used.
Zinc oxide eugenol based
The materials are produced in a powder and liquid form (e.g. Roth Root Canal cement, Roth International Ltd., Chicago, USA) or a two-paste preparation (e.g. Tubli-Seal Kerr, Orange, USA). The components of the powder, consisting of zinc oxide, bismuth subcarbonate, barium sulphate and sodium borate, are mixed with the eugenol to form the sealer. These sealers are radiopaque (due to the addition of silver, bismuth and barium), antimicrobial and toxic to vital tissues. The setting reaction of the sealer is due to the formation of zinc eugenolate crystals embedded with zinc oxide. Free eugenol is present as the material sets, decreasing over setting time and is responsible for most of the cytotoxicity when unset. Setting time and working time are dependent on the proportion of bismuth subcarbonate to sodium borate and are affected by temperature and humidity. Paraformaldehyde was added to the controversial N2 paste and in Endomethasone for added antibacterial activity.
Resin based
AH Plus (Dentsply Detrey, GmbH, Konstanz, Germany) is an epoxy resin-based sealer, which was produced as an alternative to its predecessor AH26 that released formaldehyde during the setting reaction. AH Plus exhibits acceptable biocompatibility, dimensional stability and insolubility.
Epiphany (Pentron Clinical Technologies, Wallingford, Conn.) consists of a dual cured sealer based on BisGMA, UDMA (urethane dimethacrylate) and hydrophilic methacrylates with radiopaque fillers that can coat the dentinal walls. Prior to application of the sealer, a primer must be applied to the dentine surface after a chelator such as EDTA has removed the smear layer. The sealer may then form a secondary monoblock bonding effectively to both dentines via the primer and with a chemical integration with the core material (Resilon).
EndoREZ is a hydrophilic UMDA resin capable of flowing into dentinal tubules with good biocompatibility. The sealer is used in combination with resin-coated gutta-percha points allowing the possibility of dual curing with both dentine and core material even in the presence of moisture.
Russian Red sealer is a variant of the phenol-formaldehyde resin, which sets very hard resulting in an insoluble mass within the root canal system. The sealer is strongly antibacterial and on setting results in shrinkage that leaves a reddish hue in the surrounding tooth structure. This type of paste filling is not advocated and can be very difficult to retreat.
Calcium hydroxide based
Sealapex (SybronEndo, Orange, USA) and Apexit (Ivoclar Vivadent, Schaan, Liechtenstein) are well-known sealers based on calcium hydroxide that when used in conjunction with gutta-percha core material provides biocompatibility with the possible advantage of bioactivity of calcium hydroxide when placed adjacent to vital tissue. However, for this to occur, calcium hydroxide must dissociate into calcium and hydroxyl ions requiring the sealer to break down or dissolve compromising the seal. Questions remain regarding the long-term stability of these sealers.
Silicone based
Roekoseal (Coltene Whaledent, Altstätten, Switzerland) is a polydimethylsiloxane-based root canal sealer that polymerises with minimal shrinkage and shows good biocompatibility. The material is mixed with auto-mix tips similar to impression materials producing a white fluid paste that has low viscosity to flow within the root canal system. GuttaFlow (Coltene Whaledent Ltd., Sussex, UK) is a cold gutta-percha filling system that utilises a silicone matrix and powdered gutta-percha that is triturated before carriage to the canal on either a gutta-percha cone or by passive injection using a plastic cannula.
Glass ionomer based
Ketac Endo (3M ESPE, St. Paul, MN, USA) is an adhesive root canal sealer with good biocompatibility and claims of root reinforcement due to adhesion to dentine. These sealers have been found to be more soluble and less antimicrobial with in vitro findings of leakage and disintegration over time. Due to its potential chemical bonding with dentine, issues about insolubility when using traditional gutta-percha solvents potentially make retreatment difficult.
Calcium silicate based
A new category of root canal sealers based on MTA such as iRoot SP (Innovative BioCeramix, Vancouver, Canada) has been introduced in an attempt to address the inherent problems with existing sealers and biocompatibility particularly when extruded beyond the confines of the root canal. Calcium silicate sealers include some of the same hydraulic compounds found in Portland cement including tricalcium silicate and dicalcium silicate powder that reacts with water to form a highly alkaline cement (pH 12) to create a rigid matrix of calcium silicate hydrates and calcium hydroxide. When these sealers set, the dimensional change is less than 0.1 % expansion, which helps create a barrier that has superior sealing ability. The combination of a hydrophilic sealer that is biocompatible with excellent dimensional stability seems ideal, particularly with the potential of obturation materials coming into contact with fluid either apically or coronally potentially salvaging a compromised seal.
7.3 Core Materials Used for Obturation
Gutta-percha
Gutta-percha has been the most popular core material used for obturation. It is derived from the Taban tree (Isonandra perchas) and has been used for various purposes such as coating the first trans-Atlantic cable and for the cores of golf balls. It can be made to flow using heat or using solvents, such as chloroform or eucalyptus, as well as by ultrasonics and vibration. It is a trans-isomer of polyisoprene and exists in two crystalline forms (alpha and beta) with differing properties. Gutta-percha undergoes phase transitions when heated from beta to alpha phase at around 115°F (46°C). At a range between 130 and 140°F (54–60°C), an amorphous phase is reached. When cooled at an extremely slow rate, the material will recrystallise to the alpha phase.
Traditional gutta-percha points or cones are produced in the beta phase and contains a mixture of zinc oxide, gutta-percha, radiopacifiers and plasticisers. Cones are manufactured as standardised 0.02 taper ISO cones and non-standardised 0.04, 0.06, 0.10 and 0.12 taper with specified tips or feathered tips according to manufacturing brand. Accessory points are also available from various manufacturers sized according to specified matched spreaders (e.g. SybronEndo system, SybronEndo, Orange, USA – XF, FF, MF, F, FM and M) (see Fig. 7.4).


Fig. 7.4
Clinical photographs demonstrating a variety of tapered gutta-percha cones available for obturation. Selection of cone is dependent on preparation technique and canal anatomy. Note (a) standardized 2 % gutta-percha cones available in corresponding sizes to match 2 % stainless steel files. (b) ProTaper-matched gutta-percha cones corresponding to final master apical size (F1, F2, F3, F4, or F5). (c) Accessory gutta-percha cones selected to match corresponding finger spreaders. (d) Four percent gutta-percha cones specific to rotary file system selected
An important characteristic of gutta-percha and of clinical importance is the fact that when it is exposed to air and light over time it becomes more brittle. Storage of gutta-percha in a refrigerator extends the shelf life of the material.
Gutta-percha is unable to provide a hermetic seal on its own and requires the use of an appropriate sealer. Gutta-percha is often applied to the canal using either a lateral condensing and/or vertical condensing force using spreader or pluggers to ensure adaptation occurs within the anatomical intricacies of the root canal system.
Cones can be sterilised chair side by immersion in sodium hypochlorite solution for 1 min. Gutta-percha is easily dissolvable by organic solvents such as chloroform, halothane or xylene making retreatment possible. The biocompatibility of gutta-percha has been tested thoroughly using cytotoxic, implantation, mutagenicity and usage tests.
Resilon
Resilon, a new, synthetic resin-based polycaprolactone polymer has been developed as a gutta-percha substitute to be used with Epiphany, a new resin sealer in an attempt to form an adhesive bond at the interface of the synthetic polymer-based core material, the canal wall and the sealer, otherwise known as a secondary ‘monoblock’. It is manufactured in standardised ISO sizes and shapes, conforms to the configuration of the various nickel-titanium rotary instruments and is available in pellet form for injection devices. The manufacturer states that its handling properties are similar to those of gutta-percha, and therefore it can be used with any obturation technique. Resilon contains polymers of polyester, bioactive glass and radiopaque fillers (bismuth oxychloride and barium sulphate) with a filler content of approximately 65 %. It can be softened with heat or dissolved with solvents like chloroform.
Like traditional dentine bonding systems, etching and priming of the dentine surface is required to achieve a chemical bond. The root canal system must first be rinsed with 17 % ethylene-diamine-tetra-acetic acid (EDTA) to remove the smear layer. The Epiphany, a self-etch primer, is then applied to the canal and Resilon core material used to fill the root canal system. Light curing for 40 s will cure the coronal 2 mm of the canal, and self-curing of the remainder of the canal will occur within 15–30 min. This creates a secondary ‘monoblock’ in which the Resilon bonds to the Epiphany sealer, which in turn bonds to the dentine wall.
The ability of Resilon to effectively bond to tooth structure and thereby preventing leakage has been unproven to date. The reality of achieving an adhesive ‘monoblock’ in the apical portion of the canal is unpredictable with current available materials. The literature also suggests that Resilon is not stiff enough to strengthen residual root structure. Concerns remain regarding the biocompatibility and cytotoxicity of the Epiphany sealer. Evidence-based randomised controlled trials are lacking comparing Resilon to current gold standard gutta-percha techniques that have a proven track record at this time.
Coated cones
Another strategy to achieve an intra-canal monoblock utilising traditional gutta-percha was to chemically bind or mechanically impregnate a coating over the surface of gutta-percha cones creating a similar chemistry to the sealer. Ultradent Corporation has surface coated their gutta-percha cones with a resin (functional methacrylate groups) in attempt to create a bond between the canal wall, the core material and sealer (hydrophilic self-priming methacrylate-resin based) (Ultradent, South Jordan, Utah). A bond is formed when the resin sealer (EndoREZ) contacts the resin-coated gutta-percha cone creating a hypothetical tertiary ‘monoblock’ within the root canal system with three adhesive interfaces (dentine-sealer, sealer-coating and coating-gutta-percha).
Paste fills
Since the inception of the cement material generally known by its trade name ‘Mineral Trioxide Aggregate’ in 1993 by Torabinejad, it has found many applications in dentistry including direct pulp capping, pulpotomy, root-end filling, apexification and apexogenesis, treatment of horizontal root fractures, repair of internal and external resorption defects, perforation repair and obturation of the root canal system. It is a hydraulic cement (e.g. sets and is stable under water) relying primarily on a hydration reaction for setting to occur. Apical barriers are important for the obturation of canals with immature roots with large open apices. Traditional techniques employed have been to use chloroform customization of gutta-percha that can result in extrusion with either long-term biocompatibility issues or the need for surgical revision. Alternative barriers to prevent extrusion of material have included the use of calcium hydroxide, dentine plugs and hydroxyapatite, which permit obturation, whilst minimising potential extrusion of material beyond the confines of the canal. Calcium hydroxide apical barriers are often time-consuming repeated treatments that have been associated with the potential of root fractures when used for prolonged periods of time. Placement of an apical barrier and immediate obturation is an alternative to apexification using MTA. The material is compacted into the apical portion of the root canal and allowed to set. Gutta-percha is then compacted against this apical barrier without risk of extrusion. The technique is quick, reliable and clinically successful, eliminating the need for numerous visits and possible recontamination during traditional calcium hydroxide-based apexification procedures. It is worth noting that when a root canal system is fully obturated with MTA, re-entry for retreatment or the ability to create a post space can be very difficult using ultrasonic or rotary nickel-titanium instruments due to the inherent hardness and strength of the material.
Silver points
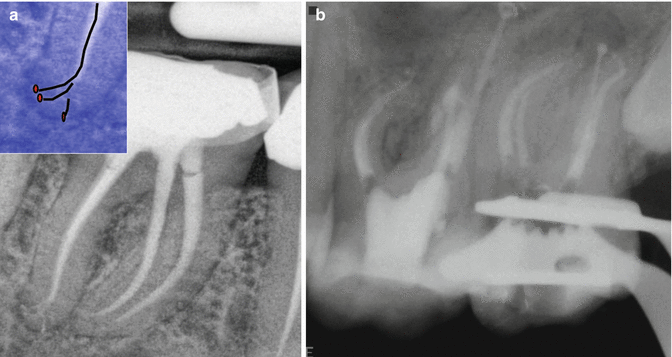
Silver points were historically introduced as root canal obturation materials that were easily placed due to their inherent rigidity. However, their inability to three-dimensionally flow and conform to irregularly shaped root canal systems permitted leakage and the potential for corrosion. Corrosion by-products can cause irreversible staining and the potential for cytotoxicity inducing persistent periapical inflammation. Other potential shortcomings including complications encountered during apical surgery and the difficulties when needing to create a post space have made these materials redundant, particularly in the face of modern techniques and improved materials that are available today (Fig. 7.5).
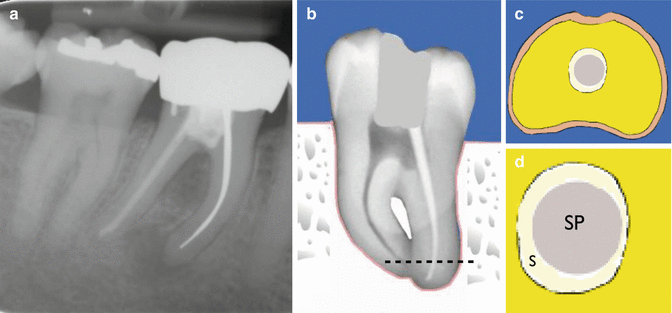

Fig. 7.5
Clinical radiograph and diagrams demonstrating silver cone obturation of a tooth. Note (a) tooth 46 with silver cone root filling in mesial root with obvious periradicular infection. (b) Silver cone placement in a prepared canal resulting in voids which are filled using sealer (dashed line represents cross-section of root taken at this level. (c, d) Apical cross section demonstrating silver cone (SP) and sealer (S) obturation. Over time, the sealer is likely to wash away, and in combination with silver cone corrosion products increase the probability of leakage and failure
7.4 Cold Lateral Compaction
A master cone corresponding to the final instrumentation size and length of the canal is coated with sealer, inserted into the canal, laterally compacted with spreaders and filled with additional accessory cones (see Figs. 7.6, 7.7 and 7.8). This technique has been the ‘gold standard’ to which other techniques have been compared. Lateral cold condensation has the advantage of excellent length control and can be accomplished with any of the acceptable sealers. The disadvantage of this technique is that it may not fill canal irregularities as well as the warm vertical compaction technique. The technique is as follows:
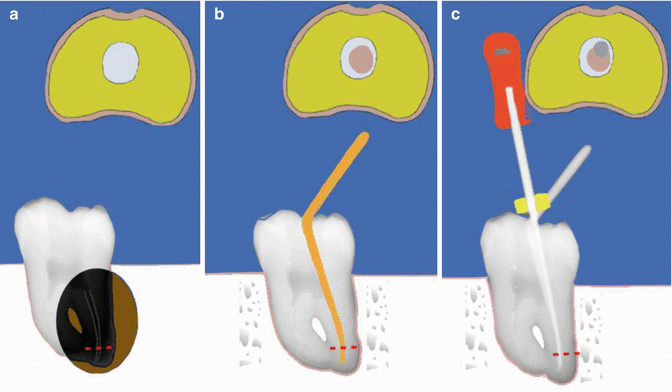
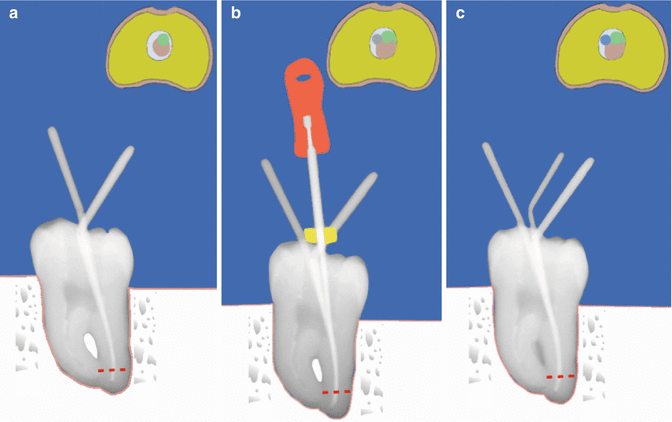
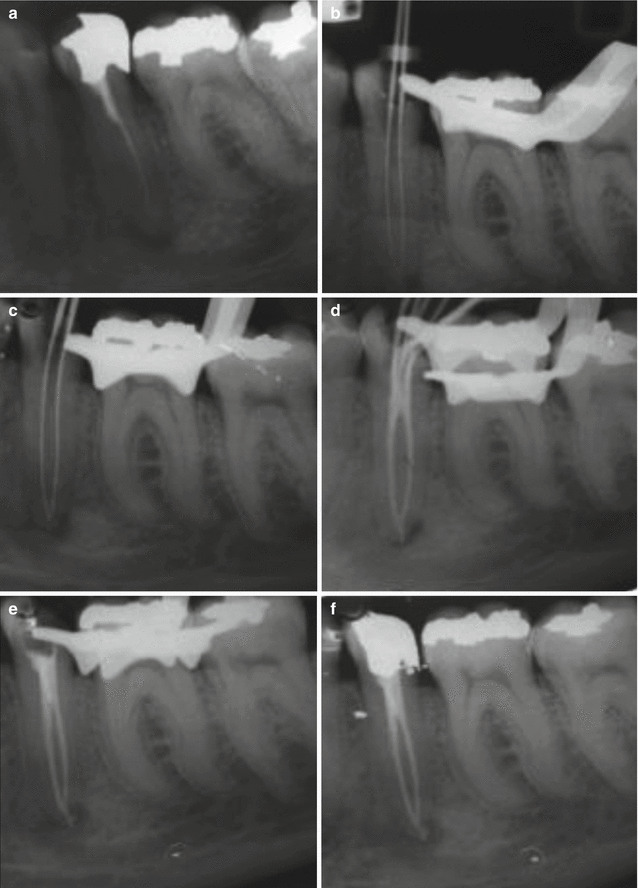

Fig. 7.6
Clinical diagrams demonstrating cold lateral obturation technique. Note (a) preoperative view, (b) master gutta-percha cone matching master apical file size placed to length with sealer, and (c) finger spreader placed to within 1 mm of working length. Cross sections of the distal canal taken at 1 mm from the working length (red dashed line) have been diagrammatically represented showing unfilled/filled canal space in relation to each step

Fig. 7.7
Clinical diagrams demonstrating cold lateral obturation technique. Note (a) matched accessory cone (green) placed in space created by finger spreader. (b) Process of placing finger spreader repeated to create space for placement of (c) another accessory cone (blue). The process is repeated until no more cones can be placed. The key is to select a finger spreader that reaches to within 1 mm of the working length and select matched accessory cones that are repeatedly placed to the level of where the finger spreader has been placed. Cross sections of the distal canal taken in the apical 1/3 (red dashed line) have been diagrammatically represented showing unfilled/filled canal space in relation to each step

Fig. 7.8
Clinical radiographs demonstrating nonsurgical root canal re-treatment of tooth 35. Note (a) preoperative view demonstrating off-centered root filling with untreated additional anatomy. (b) Following removal of previous gutta-percha, two canals were located and working lengths established. (c) Master apical file radiograph demonstrating confluence of canals. Preparation completed using 2 % stainless steel files using a 1 mm step-back technique. (d) Cold lateral compaction using ZnOE sealer. Note the “Christmas tree” effect with placement of additional accessory cones (e, f) posttreatment completed case
- 1.
A standardised gutta-percha cone is selected with a diameter consistent with the largest file used at working length. Standardised cones (0.02) generally have less taper than conventional cones and permit deeper spreader penetration. This ‘master cone’ is measured and grasped with a forceps so that the distance from cone tip to forceps is equal to the prepared length. The cone is placed in the canal to working length, and if an appropriate size has been selected, there will be resistance to displacement or ‘tug back’. If there is no resistance, modifications are required.
- 2.
Master cone placement can be confirmed with a periapical radiograph. Sealer is applied to the canal walls via the last-sized file used at working length. A finger spreader is selected that matches the taper of the canal. Appropriate accessory points are selected according to the taper of the spreader. The spreader should fit within 1–2 mm of the prepared length, and when introduced into the canal with the master cone in place, it should be within 2 mm of the working length.
- 3.
After placement, the spreader is removed by rotating it back and forth as it is withdrawn, whilst being careful not to spear the master point. An accessory cone is placed in the space made by the instrument, and the process is repeated until the spreader no longer penetrates beyond the coronal third of the canal. Excessive forces should be avoided to prevent potential root fracture.
- 4.
Excess gutta-percha in the chamber is then seared off and vertically compacted with a heated plugger at the canal orifice.
7.5 Chloroform Customization
Solvents such as chloroform, eucalyptol or halothane are used to soften the outer surface of the cone as if making an impression of the apical portion of the canal. The canal must be wet with sodium hypochlorite solution prior to ‘impression’ taking to ensure that the gutta-percha master cone does not stick to the canal walls. The chloroform cone-dipped master gutta-percha cone is then removed from the canal and allowed to dry for 1 min to ensure that all solvent has evaporated and shrinkage is minimal. The cone is reinserted into the canal ensuring it follows the same path of insertion as withdrawal previously. The cone must be coated with sealer and laterally condensed with spreaders and accessory cones (same as cold lateral compaction) (Fig. 7.9).
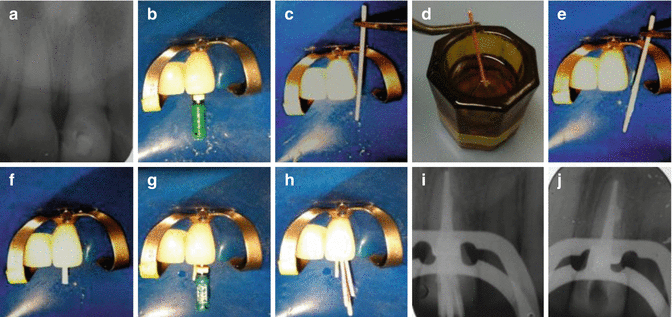

Fig. 7.9
Clinical radiographs and photographs demonstrating nonsurgical root canal treatment of tooth 21 using cold lateral compaction and chloroform dip technique. Note (a) preoperative view of tooth 21. No obvious external root resorption is noted. (b) Master apical file size verified to working length. (c) Trial insertion of master gutta-percha cone. (d) Cone is dipped in chloroform to soften apical 1/3. (e) Cone is placed in the canal making sure that the canal is flooded with sodium hypochlorite solution. The cone is then removed with an impression of the apical anatomy. The canal is subsequently dried before seating of the master cone. (f) Placement of dipped cone coated with sealer to length. (g) Placement of finger spreader. (h) Cold lateral compaction technique. (i, j) Radiograph demonstrating obturated canal space. Note lateral defect has been obturated with sealer
7.6 Warm Lateral Compaction
Ultrasonics has also been advocated for obturation of the root canal system using a warm lateral condensation technique. Lateral condensation can be used by alternating heat after the placement of an accessory gutta-percha cone. Heat is transferred to the gutta-percha using an ultrasonically vibrated K-file, which softens the mass of gutta-percha allowing penetration of the finger spreader and matched accessory cone. This technique allows for better condensation and homogeneity of both sealer and gutta-percha (see Figs. 7.10, 7.11, 7.12 and 7.13).