Introduction
The aims of this study were to measure changes in nasal minimum cross-sectional area and nasal airway resistance after surgically assisted rapid maxillary expansion and to explore a possible correlation with the subjective sensation of nasal obstruction.
Methods
Minimum cross-sectional area and nasal airway resistance were measured in 39 consecutive patients treated with surgically assisted rapid maxillary expansion. Subjective nasal obstruction was assessed by a questionnaire at pretreatment and at 3 and 18 months postoperatively.
Results
Subjective nasal obstruction had improved significantly by 3 months postoperatively. Minimum cross-sectional area increased and nasal airway resistance decreased. No correlations were found. In subjects with pretreatment subjective nasal obstruction and initially narrow anterior minimum cross-sectional area, there was a significant correlation between a moderate increase in anterior minimum cross-sectional area and improvement in perceived nasal obstruction. Eighteen months postoperatively, no changes were found from pretreatment values for subjective nasal obstruction, minimal cross-sectional area, or nasal airway resistance, and there were no correlations. Subjects with a sensation of nasal obstruction at treatment start reported a lasting significant subjective improvement.
Conclusions
The postoperative effects of surgically assisted rapid maxillary expansion did not persist in the long term. No correlation was found between objective and subjective findings. Subjects with pretreatment nasal obstruction, however, reported a lasting sensation of improved nasal function after surgically assisted rapid maxillary expansion.
Surgically assisted rapid maxillary expansion is a recognized approach for correction of skeletal transverse maxillary deficiencies in skeletally mature patients. In principle, the indications are the same as for conventional rapid maxillary expansion: ie, malocclusion associated with a narrow maxilla.
Candidates for rapid maxillary expansion often exhibit a typical vertical skeletal pattern, with long anterior lower facial height, high palatal vault, low tongue posture, incompetent lips, and mouth breathing. Similar dentofacial morphology was experimentally induced in rhesus monkeys: blocking the nasal passages led to the development of high palatal vaults and open bites. The vertical growth pattern has thus been related to the functional matrix theory of Moss and Salentijn : that growth in the craniofacial complex responds to specific functional demands—ie, there is a close relationship between dentofacial morphology and nasal breathing function.
It is generally accepted that orthopedic expansion (rapid maxillary expansion) and surgically assisted rapid maxillary expansion can affect the nasal passage. According to Wriedt et al and Warren et al, enlargement of the nasal valve can result in improvement of nasal breathing. More controversial is the question of whether rapid maxillary expansion can achieve a shift from oral to nasal breathing modes and change the subjective sensation of nasal obstruction. The clinical appearance of a patient might indicate difficulty in breathing through the nose but does not necessarily correlate with rhinometric measurements or the patient’s perception, and vice versa.
The effects of surgically assisted rapid maxillary expansion on the nasal airway might differ from those of rapid maxillary expansion. Although Babacan et al found no significant differences with respect to nasal volume, other parameters are also important in assessing nasal function: eg, cross-sectional area, nasal flow, nasal resistance, and subjective experience. Different methods of assessment capture different aspects and should perhaps be considered complementary. Widening of the nasal valve is important and might be different in surgically assisted rapid maxillary expansion. The surgical technique can also vary and influence the outcome with respect to changes in the hard and soft tissues as well as in subjective sensations. Age and growth are confounders with respect to the altered breathing pattern.
There is no gold standard for measuring the nasal airway. Acoustic rhinometry and rhinomanometry are 2 methods that can be categorized as objective measurements. Acoustic rhinometry is a noninvasive tool that reflects the anatomic profile along the length of the nasal cavity. This method is quite simple and has made it possible to estimate cross-sectional areas and volumes of the nasal cavity as a function of the distance from the nostril. Rhinomanometry is a physiologic method that measures the pressure-flow relationship during respiration and thus expresses nasal airway resistance. Which of these objective measurements best reflects nasal function is unclear.
A further common feature in candidates for surgically assisted rapid maxillary expansion is the subjective sensation of nasal obstruction. The subjective experience of postoperative nose breathing is difficult to measure and quantify, but nevertheless it is important.
Although many studies have evaluated the relationship between maxillary expansion and airway, none include the patient’s subjective experience.
The perception of obstruction can vary considerably, and correlations between objective and subjective findings are often contradictory.
The nasal obstruction symptom evaluation scale and a nasal obstruction visual analog scale are some examples of validated instruments for evaluating subjective experiences.
The purposes of this prospective longitudinal study were to use 2 objective methods, acoustic rhinometry and rhinomanometry, to evaluate short-term and long-term changes in the nasal airway after surgically assisted rapid maxillary expansion, and to compare and correlate these findings with the patient’s subjective sensation of nasal obstruction.
Material and methods
This study was conducted on skeletally mature subjects scheduled to undergo surgically assisted rapid maxillary expansion for treatment of a skeletal maxillary transverse discrepancy exceeding 5 mm ( Fig 1 ). The study was approved by the Research Ethics Committee, Faculty of Health Sciences, Linköping University, Sweden.
Forty subjects were recruited consecutively from the Department of Orthodontics at the Institute for Postgraduate Dental Education, Jönköping, Sweden, and from the Department of Dentofacial Orthopaedics, Maxillofacial Unit, University Hospital, Linköping, Sweden. Before treatment, all patients were examined by an otorhinolaryngologist (F.J.). One patient was excluded from the study because of a planned adenoidectomy. The sample thus comprised 39 patients (16 male, 23 female). The mean age at treatment start was 19.9 years (range, 15.9-43.9 years).
Measurements were taken before treatment and 3 and 18 months postoperatively. The treatment group was its own control group.
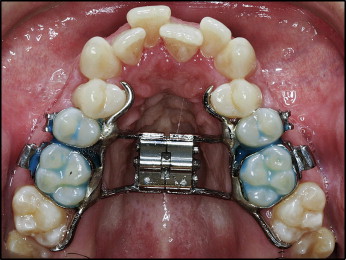
The orthodontic phase of treatment was undertaken at local orthodontic clinics under the supervision by the orthodontic departments in Jönköping and Linköping, Sweden. The maxillary expansion appliance consisted of a tooth-anchored device activated by means of a conventional hyrax expander (Hyrax II, Dentaurum, Ispringen, Germany) with a soldered framework and orthodontic bands ( Fig 2 ). No acrylic pads were used. The appliance was scheduled for insertion as close as possible to the date of surgery. The degree of expansion was calculated for each patient, including a general bilateral overexpansion of a half-cusp width. The patients were instructed to activate the jackscrew 1 turn (0.25 mm) twice a day after a postoperative latency period of 5 days. Postoperative control was scheduled 12 days after surgery and included an occlusal radiograph to ensure a clinically symmetrical opening of the midpalatal suture and a medial diastema. At that time, the amount of additional expansion was measured.
After a mean active expansion period of 15 days (range, 11-17 days), with overexpansion and buccal tipping, the appliance was used as a passive retainer for 90 days ( Fig 3 ). At that time, the hyrax expander was replaced by a modified transpalatal arch ( Fig 4 ), and fixed appliance treatment commenced.
On completion of the active treatment phase, the transpalatal arch was removed, and fixed appliance treatment continued with stiff rectangular archwires to adjust the transverse width and to control and correct the buccal root torque of the molars. All posterior crossbites were corrected by the end of treatment. A Hawley plate was used for retention. The orthodontic treatment period was then concluded.
Surgical treatment followed a technique similar to that described by Glassman et al and was undertaken by 2 experienced senior oral and maxillofacial surgeons (P.N.) in Jönköping and Linköping, Sweden.
The surgery was carried out under general anesthesia and nasotracheal intubation. A mucoperiosteal incision in the maxillary vestibule extended from the right second premolar to the left second premolar. The osteotomies were performed on the maxillary lateral walls from the piriform aperture to the pterygoid plates. The pterygomaxillary sutures were kept intact. The lining on the floor and lateral walls of the nasal passage was reflected. A vertical osteotomy at the anterior nasal spine and the median palatal suture was carried out to ensure separation of the maxillary halves. The hyrax expander was activated 12 turns (3 mm) to verify the success of the osteotomy and to ensure symmetrical separation, and then deactivated by the same amount. Depending on their physical condition and postoperative swelling, the patients were discharged from the hospital on the same day or the day after surgery.
The rhinologic examination was carried out by 2 otorhinolaryngologists (F.J.) at the Ear, Nose, and Throat Department, Ryhov County Hospital, in Jönköping, and at the Ear, Nose, and Throat Department, University Hospital, in Linköping, Sweden. The examinations were carried out before treatment (T1), and 3 and 18 months after the surgically assisted rapid maxillary expansion (T2 and T3, respectively). The examination comprised a clinical examination, acoustic rhinometry, and anterior rhinomanometry. For interexaminer calibration, the 2 otorhinolaryngologists were supplied with the same equipment (RhinoScan, RhinoStream; Interacoustics A/S, Assen, Denmark) and trained together.
The patient was allowed to rest for 15 minutes before the examinations and recordings. The presence of an adequate nasal cavity space was assessed by anterior rhinoscopic examination. The technical arrangements and data calculations were made in accordance with guidelines developed by the International Standardization Committee for Rhinomanometry.
Acoustic rhinometry measures the nasal airway dimensions by emitting wide-band noise into the nose. A standard probe for adults and tailored nose adapters to fit the right and left nostrils were used in accordance with the recommendations of the Standardization Committee on Acoustic Rhinometry. Acoustic rhinometry and rhinomanometry were performed with a digital platform, SRE 2000 Digital Signal Unit (Interacoustics A/S, Assens, Denmark). A RhinoScan software module was used for the acoustic rhinometry and RhinoStream for the rhinomanometry.
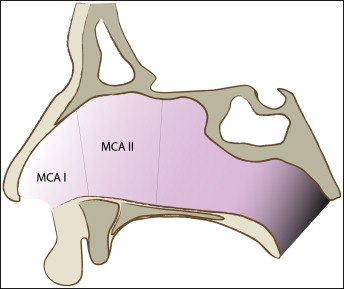
The front portion of the nasal cavity is the narrowest and most resistant area to nasal airflow and comprises the inner valve, the anterior part of the turbinate, and the isthmus nasi.
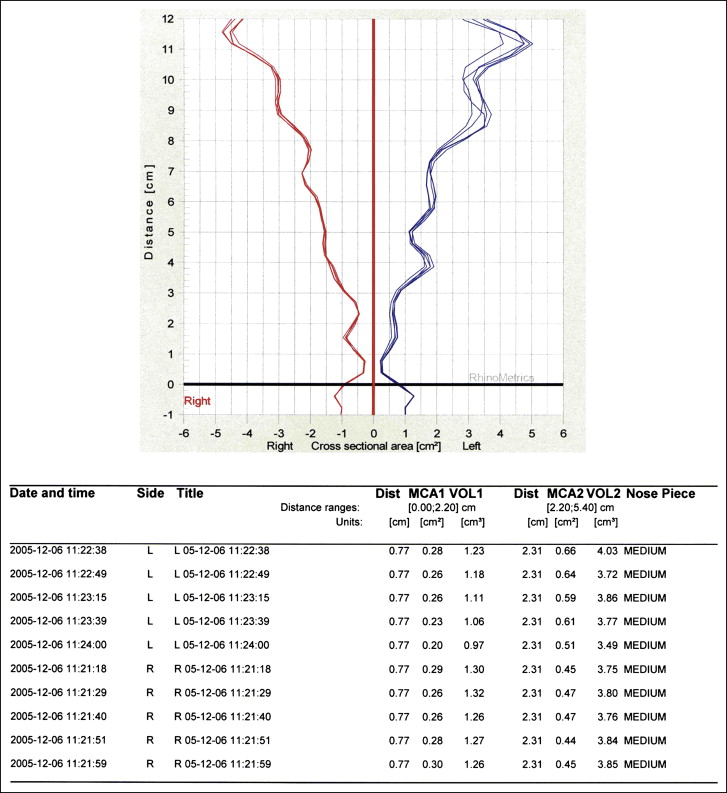
Acoustic rhinometry (RhinoScan) measures the minimum cross-sectional area at the front portion at 2 distances: minimum cross-sectional area I (the structural valve), from the nostril rim to approximately the anterior border of the inferior turbinate (0.0-2.2 cm) and minimum cross-sectional area II (the functional valve) from the anterior border of the inferior turbinate to the isthmus nasi (2.2-5.4 cm) ( Fig 5 ). The 5 recordings on each side were registered and displayed both graphically and as a chart ( Fig 6 ). The mean values for minimum cross-sectional areas I and II were calculated for the left and right sides, respectively.
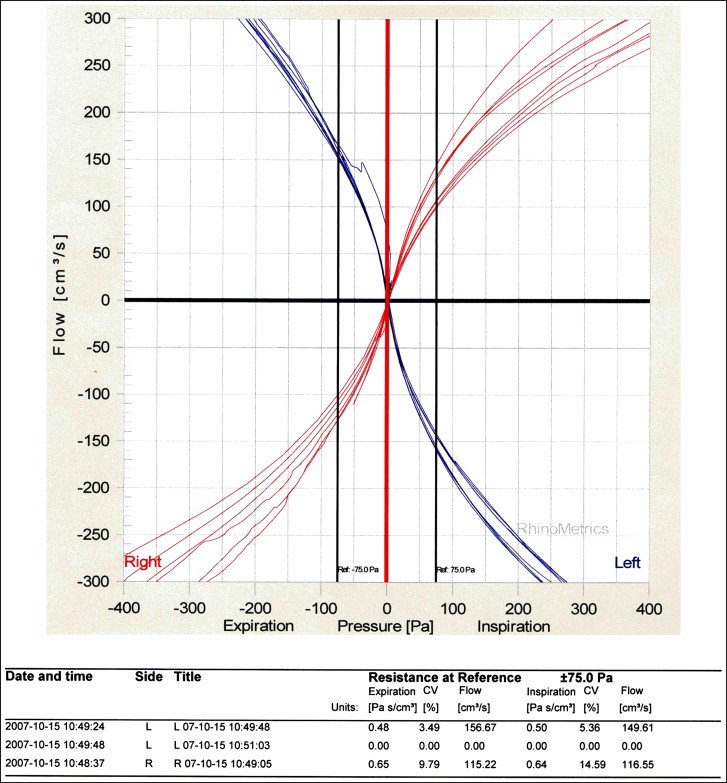
Rhinomanometry (RhinoStream) gives a dynamic assessment of nasal patency by measuring transnasal pressure and flow during respiration and on this basis calculates the nasal airway resistance. The RhinoStream module expresses nasal airway resistance at 75 Pa. The manometer probe attached to the tailored nose adapter registered values for inspiration and expiration on each side. The instrument was calibrated before each test, and each side was registered separately. The recordings were registered and displayed both graphically and as a chart ( Fig 7 ). The nasal airway resistance value ( R ) was calculated for inspiration ( insp ) and expiration ( exp ) according to the formula R insp/exp = R left ∗ R right /R left + R right . To reduce the mucosal swelling of the nasal valve, the registrations were made after administration of a decongestant (Otrivin, 1 mg/mL; Novartis Pharma AG, Basel, Switzerland).
The subjective parameters were assessed by a questionnaire according to Petruson et al (2001), approved by the Swedish Rhinologic Society. This questionnaire is a disability index used to evaluate the impact of nasal obstruction from the subject’s perspective. It is based on 10 anamnestic “yes” or “no” questions and 10 visual analog scales. The anamnestic questions were constructed to determine current medications, previous ear, nose, and throat illnesses and treatments, nasal obstructions, and expectations of the planned treatment.
The visual analog scale is useful in evaluating nasal blockage and discharge, facial pain or pressure, headache, and overall symptoms. The first 8 visual analog scales rated the subject’s symptoms on a scale of 0 to 10, with 0 as “never” and 10 as “constantly,” regarding nasal blockage, nasal congestion, running nose, snoring, facial pain, headache, nose breathing, and sense of smell. The last 2 from 0 to 10 were 0 as “good” and 10 as “bad,” regarding life quality in general and life quality from a rhinologic perspective. All subjects scored their subjective symptoms of nasal obstruction at the time points T1, T2, and T3.
The interexamination measurement errors for acoustic rhinometry and rhinomanometry were calculated by intraclass correlation coefficients, which are an expression of interobserver reliability. These were measured at 6 examinations and were above 0.75, indicating good reliability.
Statistical analysis
Statistical analyses were undertaken by using the Statistical Package for the Social Sciences III (version 13.0; SPSS, Chicago, Ill). The distribution of data was tested with the 1-sample Kolmogorov-Smirnov test. The nonnormal distribution of data and the sample size justified nonparametric tests such as the Wilcoxon signed rank test, sign test, and Spearman rho. Median values and percentiles were used to describe the changes and distributions. The Wilcoxon nonparametric test was used to evaluate the changes between T1, T2, and T3. The Spearman rho was applied to assess the correlation between subjective and objective findings.
Results
The pretreatment questionnaire showed that the main reason for seeking treatment was to correct a malocclusion, but 10 patients (1 male, 9 female) also expressed a need for treatment for esthetics, and 9 patients (3 male, 6 female) had expectations of improved nasal patency.
With respect to nasal obstruction, 16 subjects (6 male, 10 female) had never perceived any problems, 9 (3 male, 6 female) had occasional problems, 9 (4 male, 5 female) had frequent problems, and 5 (3 male, 2 female) had constant problems. Eight subjects (5 male, 3 female) had a mouth-breathing posture, 2 (both female) had a nose-breathing posture, and 29 (11 male, 18 female) had a mixed-breathing posture.
Table I shows the median values and percentiles for minimum cross-sectional areas, nasal airway resistance, and subjective sensation of nasal obstruction at T1, T2 and T3. Twenty-three subjects reported experiencing some degree of nasal obstruction before treatment ( Table II ). The minimum cross-sectional area, nasal airway resistance, and subjective sensation for this subgroup were analyzed separately and compared with the rest of the sample. There were no significant differences in minimum cross-sectional area or nasal airway resistance between subjects with and without a sensation of nasal obstruction before treatment.
| Total sample (n = 39) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T1-T2 | T3 | T1-T3 | |||||||
| Median | P 10 | P 90 | Median | P 10 | P 90 | Wilcoxon test | Median | P 10 | P 90 | Wilcoxon test | |
| Subject sensation (visual analog scale) | 2.0 | 0.0 | 8.2 | 0.9 | 0.0 | 6.2 | † | 1.5 | 0.0 | 6.2 | NS |
| MCA I (cm 2 ) | 0.36 | 0.23 | 0.49 | 0.41 | 0.26 | 0.55 | ∗ | 0.35 | 0.26 | 0.54 | NS |
| MCA II (cm 2 ) | 0.45 | 0.28 | 0.72 | 0.52 | 0.36 | 0.88 | ∗ | 0.50 | 0.34 | 0.70 | NS |
| NAR insp (Pa/cm 3 /s) | 0.19 | 0.14 | 0.36 | 0.15 | 0.11 | 0.20 | ∗ | 0.18 | 0.12 | 0.25 | NS |
| NAR exp (Pa/cm 3 /s) | 0.17 | 0.11 | 0.28 | 0.14 | 0.04 | 0.19 | ∗ | 0.17 | 0.10 | 0.25 | NS |
| Subjective nasal obstruction (n = 23) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T1-T2 | T3 | T1-T3 | |||||||
| Median | P 10 | P 90 | Median | P 10 | P 90 | Wilcoxon test | Median | P 10 | P 90 | Wilcoxon test | |
| Subject sensation (visual analog scale) | 5.1 | 1.3 | 8.5 | 2.0 | 0.0 | 6.5 | † | 2.0 | 0.0 | 7.0 | ∗ |
| MCA I (cm 2 ) | 0.35 | 0.23 | 0.48 | 0.41 | 0.27 | 0.52 | † | 0.35 | 0.28 | 0.54 | NS |
| MCA II (cm 2 ) | 0.41 | 0.26 | 0.70 | 0.54 | 0.36 | 0.89 | † | 0.52 | 0.34 | 0.75 | ∗ |
| NAR insp (Pa/cm 3 /s) | 0.20 | 0.13 | 0.40 | 0.14 | 0.11 | 0.23 | ∗ | 0.16 | 0.12 | 0.24 | NS |
| NAR exp (Pa/cm 3 /s) | 0.17 | 0.13 | 0.28 | 0.14 | 0.02 | 0.22 | ∗ | 0.16 | 0.01 | 0.25 | NS |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


