Metal-Ceramic and All-Ceramic Restorations
Metal-Ceramic Restorations
The biggest advantage of the ceramic restoration is its permanent aesthetic quality. Since there is a solid connection between the masking ceramic and the metal, almost no changes of color will occur in the ceramic construction.
A metal-ceramic crown that has been correctly produced is also more stable and more durable than a regular jacket crown. However, a metal-ceramic bridge involving several teeth can fracture and chip the ceramic under stress due to its inferior flexure strength. Because of this, it is very important to consider occlusal conditions when such a bridge is made.
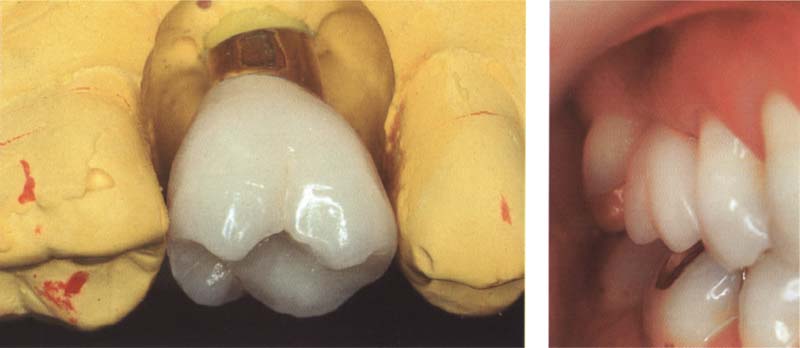
323 Metal-ceramic crown
Metal-ceramic is and remains the most important restorative material for crowns in the posterior regions. Metal-ceramic restorations are indispensible for implant-supported restorations.
Right: The same metal-ceramic crown on an implant in the mouth of the patient.
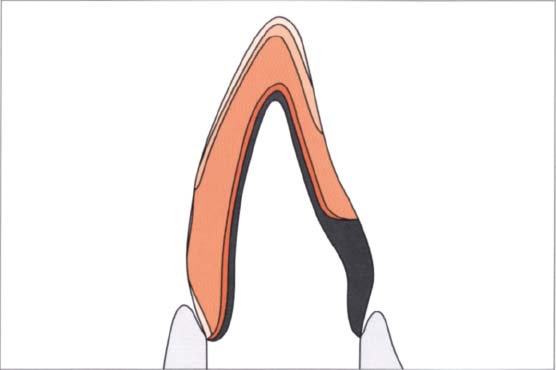
324 Structure of a metal-ceramic restoration
The metal framework of a metal-ceramic crown must support the sintered ceramic as much as possible. To prevent the dark metal framework from shining through, a layer of an opaquer must be placed and fired before the ceramic masses can be fired. This layer can lead to poor aesthetics if sufficient space is not available for the ceramic masses.
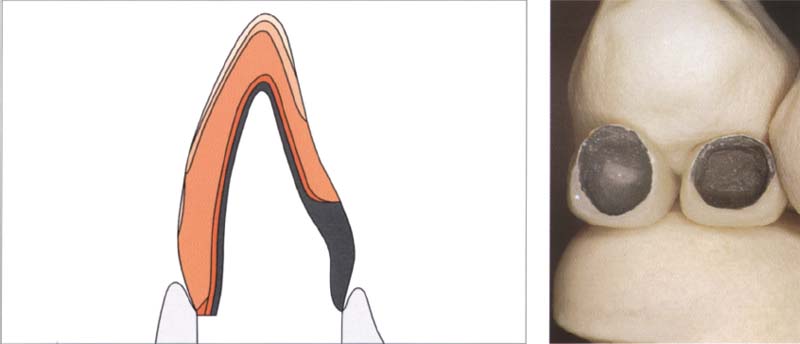
325 Structure of a metal-ceramic restoration with cervical ceramic margin
To improve gingival aesthetics (unsightly metal edges), the metal framework is shortened and an all-ceramic margin is made.
Right: lf aesthetic demands are greater, one can make a labial short ceramic shoulder.
Another advantage of metal-ceramic restorations, in contrast to all-ceramic restorations, is that less tooth substance needs to be removed to create enough space for the crown if metal is used to cover the occlusal and lingual as well as palatal surfaces.
The flexural strength needs to be optimized to prevent a fracture of the ceramic. The modulus of elasticity of ceramics is relatively high, while both tensile and shear strengths are low and as a result the flexibility is low.
Stresses exceeding the proportional limit result in brittle failures. Accordingly, the ceramic can only resist limited elastic deformation. The metal framework must be rigid in order to support the ceramic sufficiently. Since the anatomical form of the crown must not differ from that of the natural tooth, more tooth substance must be cut away in order to produce sufficient space for the crown.
Despite some disadvantages, metal-ceramic restorations belong to the most common treatments with fixed prostheses.
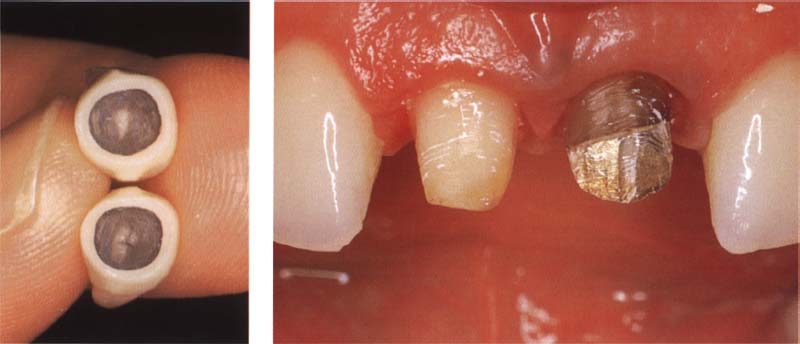
326 Metal-ceramic restorations with circular ceramic margin
Endodontically treated, strongly discolored teeth can best be treated with metal-ceramic restorations.
Left: For the highest aesthetic requirements, a circular, all-ceramic margin can also be constructed.
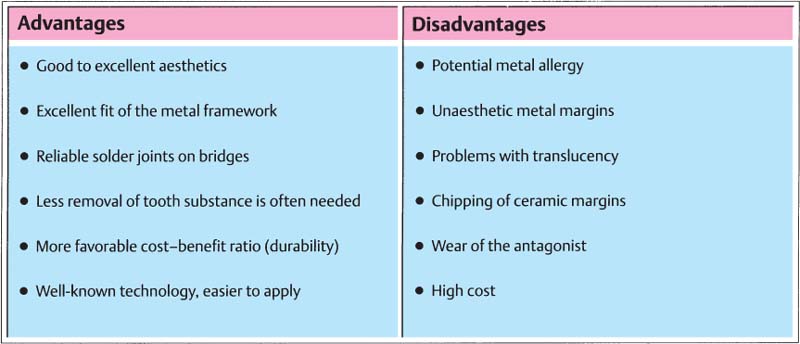
327 Advantages and disadvantages of metal-ceramics
328 All-ceramic and metal-ceramic restorations
Over the next few years, bridges will continue to be made in the posterior regions in metal-ceramics because no all-ceramic system is yet available on the market that has proved longevity for such treatments.
Left: If metal-ceramic crowns are directly adjacent to all-ceramic restorations (e.g., veneers), the different optical qualities of the two materials are obvious under certain lighting conditions.
The advantages and disadvantages of metal-ceramic restorations in comparison to all-ceramic systems are represented and summarized in Figure 327. An overview of the different all-ceramic systems as well as the advantages and disadvantages of all-ceramic crowns is presented on page 192.
Clinical Success of Metal-Ceramics
Metal-ceramic restorations have, compared with all-ceramic crowns and bridges, a much lower risk of fracture. Further advantages include excellent to outstanding aesthetic results and longevity. Metal-ceramic crowns and bridges show success rates of 97% during the first seven and a half years. After 10 years, 95% of the metal-ceramic restorations are still present in the oral cavity.
Established metal-ceramics systems can be applied successfully as long as two basic principles are fulfilled:
—compatibility of the materials (thermal compatibility and ceramic bonding ability) as well as
—proper framework-design and material processing
However, the success rate after seven and a half years refers only to noble metal alloys with a gold content of at least 40% as well as castings with conventional metal margins. Only a limited amount of information is available on crowns with ceramic shoulders. It may be possible that ceramic margins crack under high pressure and finally fracture. Data on metal-ceramic crowns that are built on titanium frameworks are not yet available. Therefore, the use of the latter crowns is recommended exclusively for the treatment of anterior teeth.
The Nature of Ceramic Tooth Restorations
Weak points in a ceramic construction include sharp angles, clear changes in the thickness of the ceramics, and high tensile stress levels in the ceramic framework. This is particularly true for ceramic bridges, which require the interdental spaces be carefully shaped and rounded. Tensile stresses are developed under occlusal force in these regions.
Ceramic shoulders improve the aesthetics in the gingival third of the metal-ceramic crown. However, the risk of fracture is increased, particularly with crowns in the molar regions.
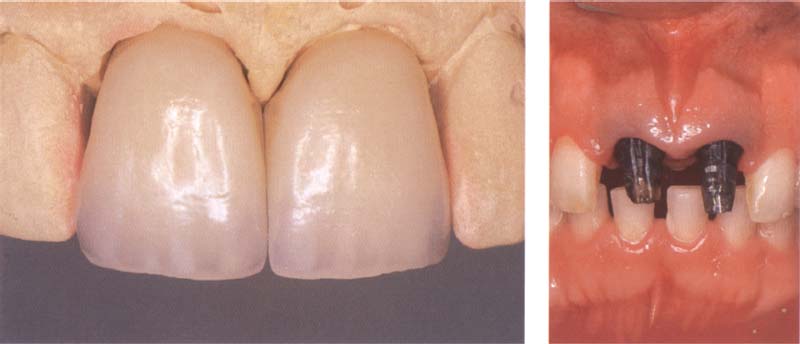
329 Advantages of metal-ceramic restorations used on strongly discolored teeth
Metal-ceramic crowns can cover strongly discolored teeth and can bring about an aesthetically appealing result.
Right: Initial situation consists of old metal post reconstructions that are very strongly discolored. These can no longer be removed. In this case a satisfactory result can only be achieved with metal-ceramic restorations.
Foil and electrochemically plated crowns enable improved aesthetics on the basis of the reduced metal thickness. However, they are also more susceptible to fracture than conventional metal-ceramic crowns. Commercial pure titanium, one of the most biocompatible metals, is also being used as the metal base. However, the latter procedure requires a ceramic with a very low melting point (850°C) and an extraordinarily low thermic expansion coefficient (≤8.5·10-6/oC).
Thermal Compatibility
To protect the ceramic from cracking or fracturing due to thermally induced tensile stresses, the ceramic should be in slight compression before the final adjustments and trimmings are made. To induce such compression during the firing process, the coefficient of thermal expansion of the metal should in principle be slightly higher than that of the ceramic during the cooling process to room temperature. Such a small difference causes a certain amount of pressure in the ceramic and thus strengthens the restoration.
In the case of metal-ceramic restorations, the coefficients of thermal expansion of the ceramic and the metal are of particular interest in the 600–625 °C or 500–525 °C temperature range. Metal-ceramic systems that have a difference in the coefficients of thermal expansion (metal minus ceramic) of 0.5 · 10-6/°C or less are acceptable. According to some experiments, it seems as if a somewhat higher difference up to 0.75 · 10-6/°C can be used without the risk of cracks or fractures in the ceramic.
The Ceramic-Metal Connection
One of the most important prerequisites for a successful metal-ceramic restoration is the lasting connection between the ceramic and the metal alloy and their thermal compatibility. The oxidation level of the alloy to a great extent determines its ability to form a lasting bond with the ceramic: alloys that produce a solid oxide layer during the degassing process can also produce a reliable bond to the ceramic. In contrast, alloys with a weak oxide layer form poor bonds with the ceramic.
Fractures of metal-ceramic restorations are rare. However, they may appear if one uses a new alloy, a new ceramic, or a new processing technique. Although there is a multiplicity of possible fracture line locations, below are three types that are particularly significant (Fig. 333):
—fracture lines in the opaque layer or between the opaque layer and the oxide layer
—fracture lines in the oxide layer
—fracture lines between metal and oxide layer
It is necessary to use 100 x magnification to recognize the location and cause of a fracture, since otherwise the fine transitions are not visible. Each of the above types of fracture line suggest material or processing failures. If the fracture is exclusively restricted to the ceramic, the failure is most likely due to high pressure, hyperocclusion, grinding of teeth, or larger defects present in the material structure.
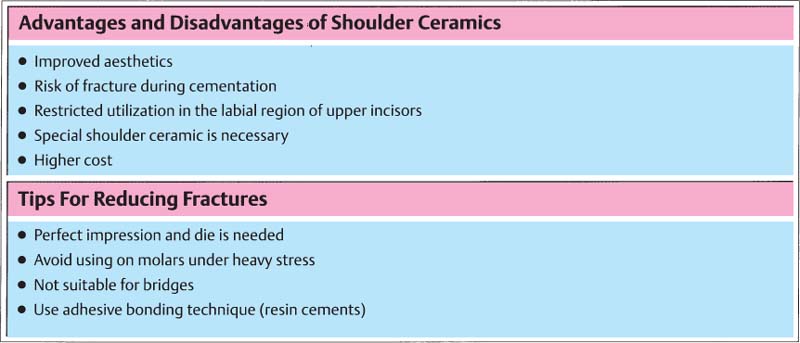
330 Advantages and disadvantages of shoulder ceramics used on metal-ceramic restorations (above) and tips on how to lower the risk of fracture of all-ceramic crowns and crowns with a ceramic shoulder (below)
To achieve a stable metal-ceramic bond on a cast metal framework, it is necessary to use an oxide generated by a basic metal component (e.g., indium or tin). This oxide layer is formed through a special thermal treatment method or during the first firing step of the opaquer.
Metal frameworks for foil and chemically plated crowns, such as the Captek System (Leach & Dillon), the Renaissance System (Unikorn), the Ceplatec System (Ceplatec) and other galvanic manufacturing processes, use a ceramic binder like the Capbond metal-ceramic agent for the Captek foil crown system (Leach & Dillon).
The gold-like color of the foil thimble improves the tone of the ceramic. A covering opaque layer is required with the normal dark oxides of the typical metal-ceramic alloys. Since one can vary the tone of gold from reddish-brown up to gray, this is an additional way of influencing the color of the ceramic.
Classification of Dental Ceramics
The following ceramics belong to the dental ceramic category: feldspar ceramics, leucite reinforced ceramics, ceramics with low melting point, glass ceramics, high-strength core masses (alumina), glass infiltrated alumina as well as CAD/CAM ceramics.
Dental ceramics are divided into different groups according to their chemical composition (feldspar, leucite, alumina, glass alumina, and glass ceramics), application (tooth reconstruction, ceramic-covering metals, veneers, inlays, crowns, and anterior bridges), the manufacturing procedure, or the structure of the material (cast metal, burnished metal foil, glass ceramics, CAD/CAM ceramic, and sintered ceramic core).
Sintering, pressing, casting, slip casting followed by glass infiltration, and machining (manually or computer-operated) are the different manufacturing methods that can be used for making ceramic restorations.
Classification of dental ceramics based on the firing temperature:
—high melting point: 1201–1450°C
—medium melting point: 1051–1200°C
—low melting point: 850–1050°C
—very low melting point: < 850°C
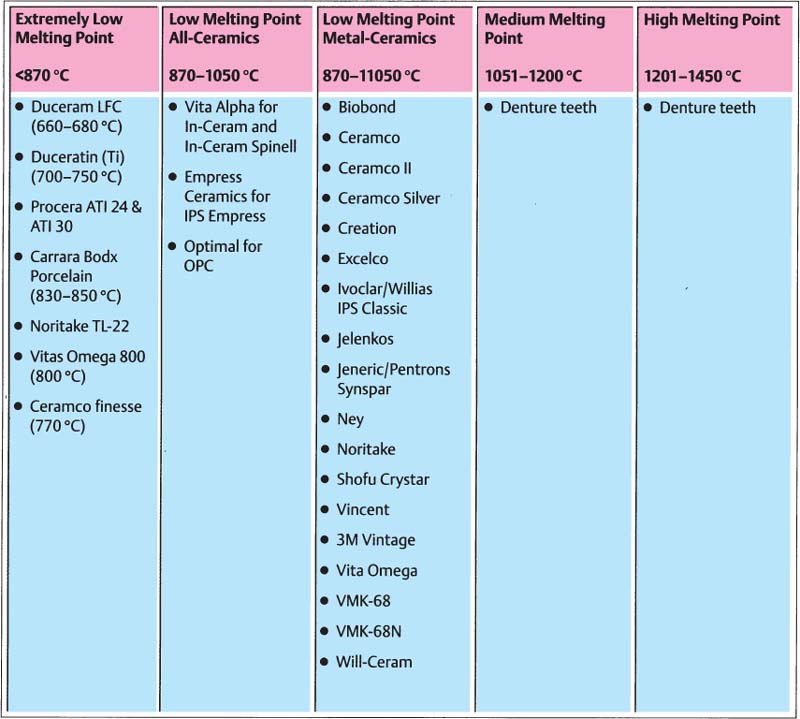
331 Classification of ceramic materials
Ceramics with medium to high melting point are used for making teeth used in dentures, while low and very low melting point ceramics are used for crown and bridge constructions.
Ceramics with especially low melting points are used to cover titanium frameworks (or titanium alloys), since their coefficient of thermal expansion is close to that of the metal. These ceramics can also be used to cover certain low melting type IV gold alloys. However, some of the ceramics with a low melting point can also be used for conventional metal-ceramic alloys (highly noble, noble, or nonnoble metals), since they have sufficiently high coefficients of thermal expansion.
Two of the very low-fusing ceramics (Duceram-LFC and Duceragold) are the so-called hydrothermal ceramics. They owe their improved resistance to a hydrolytic shrinkage that occurs when hydroxyl ions interact during heat and steam impact. The glazing temperature of conventional ceramics is lowered by the addition of alkali oxides.
However, the chemical and hydrolytic stability decreases with increased amount of alkali oxides (above a certain concentration). In addition, decreased fracture resistance occurs due to the presence of new inclusions. With low-fusing ceramics, the hydroxyl enriched surface makes the ceramic more flexible and allows the repair of surface inclusions.
Dental ceramics are classified into different types. A simple classification that is based on the application type is shown in Figure 331. Here, different brand names are also given for each group. The general qualities of these materials, their clinical advantages, and disadvantages as well as the steps in processing them will be reviewed in the following sections.
Ceramic products such as colors and glazes will here be classified as class 4 ceramics and will not be discussed further, since they do not have any significant effects on strength and fracture resistance of ceramic restorations.
As far as chemical durability is concerned, self-glazing ceramics are preferred over glazing masses. A higher concentration of glass modifiers reduces the resistance of the applied surface glaze compared to the normal surface glaze of the ceramic. During glazing a thin outer layer is formed. A certain temperature and treatment time leads to the formation of a softer glass phase and to the formation of crystalline particles within the surface region.
Water is another important glass modifier, although it is not one of the intended additives present in ceramics. In ceramics that contain glass modifiers, such as sodium or other metal ions, these ions can be replaced with hydronium ions. This phenomenon can result in slow crack propagation in the ceramic if it is exposed to tensile stresses or a moist surrounding. Also, this may be the reason why ceramic restorations fracture after some years without a recognizable reason.
Strength and Risk of Fracture of Ceramics
Ceramic materials do not show the strength one would expect from their molecular structure. Small defects, for example scratches, can be found on the surface of almost any material and these scratches are the reason for the lower strength. Such surface defects are comparable to sharp cuts with crack tips that can be as narrow as the distance between two atoms. The stress concentration that results from the defect results in a local increase in tensile stress. However, the theoretical strength of the material is based on the assumption that there is an even distribution of stresses in the entire structure. If the tensile stress exceeds the strength limit at the tip of a defect, the chemical bond breaks at this tip, and a crack starts propagating. The tensile stress at the tip of the crack remains until the crack has propagated through the entire material or has reached another crack, a pore, or a crystalline particle, so that compression occurs and the stresses are distributed. This phenomenon explains why some materials fail at pressures far below their theoretical strength values.
The failure of ceramics and their low tensile strength values can be explained on the basis of how stress concentrations are generated at surface inclusions. Under certain conditions, inclusions can also initiate cracks within the material. Since ceramics have no other stress distribution mechanism available to deal with tensile loads besides crack growth, cracks can continue to grow under low pressure conditions through the entire material. Therefore the flexure strength of ceramics and glass is essentially lower than their compressive strength.
Complex stresses develop in the mouth. The maximum pressure appears at the surface of the restoration. Therefore, surface inclusions are especially important in judging the strength of a ceramic. Removing or reducing the number of surface inclusions can result in a considerable improvement of the fracture resistance. This is one of the reasons why it is necessary to polish and glaze dental ceramics. The fracture strength of the material can be improved in two ways:
—through introducing a compressive stress within the material surface, and
—through interrupting crack propagation in the material.
Procedures For Strengthening Ceramics
Pressure Induced by Residual Compressive Stresses
A commonly used procedure for strengthening ceramics is to introduce compressive residual stresses within the material surface. To do so, one selects ceramics that have a smaller thermal expansion coefficient than the metal.
Ion exchange
Ion exchange is an effective method used to generate compressive stresses in the ceramic surface. The procedure is also called chemical hardening and includes sodium-ion exchange. Sodium is a common component in many glass types and has a relatively small ion radius. If one places glass which contains sodium into a bath containing melted potassium nitrate, sodium ions on the glass surface are replaced by potassium ions. Because the radius of the potassium ion is larger than the radius of the sodium ion by approximately 35%, the potassium ion will occupy a larger volume when it enters the surface and the material surface will increase its surface compression level.
GC Tuf-Coat (GC Corp.) is a pot/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


