Fig. 5.1
Root dentine surface of debonded specimens with different dentine pretreatments, in which Ketac Endo sealer had been used. (a) Scanning electron micrograph (SEM) (×800) of dentine surface completely covered with sealer (citric acid pretreatment). (b) SEM (×200) of gutta-percha surface (citric acid pretreatment). (c) EDS analysis of gutta-percha surface revealing traces of sealer. Gutta-percha is mostly organic with zinc making up most of its inorganic component. The calcium and silicon in the EDS analysis are derived from the sealer. (d) SEM (×800) of dentine surface partly covered with sealer (S). Open dentinal tubules (D) with no sealer tags are seen in most of the figure elsewhere (EDTA pretreatment). (e) EDS analysis of the area indicated within the white square in (d), showing that this area appears to be clear dentine, i.e. without any remnants of sealer. Dentine is composed of calcium and phosphorus as seen in the EDS analysis. (f) SEM (×800) of gutta-percha surface partly covered with sealer (S) (EDTA pretreatment) (Reprinted from Saleh et al. (2003). With permission from Elsevier)
The removal of the smear layer with ethylenediaminetetracetic acid (EDTA) did not appear to have any effect on the bond strength of a glass-ionomer sealer (Ketac Endo) (Najar et al. 2003). In fact, when EDTA was used to remove the smear layer from root canal walls, no penetration of glass-ionomer sealer was shown inside the dentinal tubules as indicated in Fig. 5.1 (Saleh et al. 2003). Ketac Endo exhibited continuous penetration when phosphoric acid and citric acid were used to pretreat the dentine (Saleh et al. 2003). The bond strengths of a glass-ionomer sealer to gutta-percha were weaker than other test sealers (Saleh et al. 2002). The bond strength of glass-ionomer to gutta-percha was also shown to be low when compared to other classes of sealer cements (Lee et al. 2002). It has been suggested that pretreatment with phosphoric acid or citric acid should be done in association with glass-ionomer root canal sealer to achieve the most effective removal of the smear layer and to provide better adhesion (Timpawat et al. 2001; Saleh et al. 2003). On the other hand, the use of 2.6 % sodium hypochlorite solution used as a dentine conditioner improved the sealer adhesion, and the presence of a hybrid layer was noted (Lalh et al. 1999b). The adhesion of glass-ionomer sealers to root canal walls is also affected by the presence of calcium hydroxide and formocresol medicaments used in the root canal during root canal treatment (Chung et al. 2001). Although bonding of glass-ionomer cement to dentine walls can potentially minimize microleakage, no correlation between microleakage and adhesive properties was found when four commonly used sealers including glass-ionomer cement were evaluated (Pommel et al. 2003).
Testing for adhesive properties has been mostly conducted by performing a shear bond strength test (Lalh et al. 1999a, b; Chung et al. 2001; Gogos et al. 2004) and tensile testing (Tagger et al. 2002; Lee et al. 2002; Saleh et al. 2002, 2003). The push-out bond strength test has also been used to test the sealing ability and bonding using different sealers including a glass-ionomer sealer (Nagas et al. 2009). Push-out bond strength testing is currently the most popular test to assess adhesive properties of materials used within the root canal.
5.2.2 Fluoride Release
Fluoride ion release is one of the properties of glass-ionomer cements. The action of polyacrylic acid on the glass particle results in ion dissolution over time. In the early stages, calcium and aluminium ions are preferentially released in solution, while sodium and fluorine are leached in the long term. While the fluoride ion release has been shown to result in remineralization of tooth tissue (Arends et al. 1990; Almqvist and Lagerlöf 1993), this property has not been extensively researched for glass-ionomer cements used for endodontic applications. There is no literature on the effect of fluoride released from glass-ionomers used for endodontic applications and its effect on root dentine remineralization.
The acidic environs of glass-ionomer-based materials are not known to promote bioactivity and remineralization when compared to the local bioactivity of the tricalcium silicate-based materials like mineral trioxide aggregate (MTA) and Biodentine (Septodont, Saint-Maur-des-Fossés, France). Materials based on tricalcium silicate release calcium hydroxide on hydration; this in turn results in an alkalinizing medium that has been shown to produce mineralization within the adjacent dentine substrate, extending deep within the tissues. This suggests that the local ion-rich alkaline environment may be more favourable to mineral repair and reconstruction (Tay and Pashley 2008; Dong et al. 2011), compared with glass-ionomers (Watson et al. 2014). To this effect, a remineralization medium consisting of a Portland cement/phosphate-containing fluid that included polyacrylic acid and polyvinylphosphonic acid biomimetic analogues resulted in both interfibrillar and intrafibrillar apatites becoming readily discernible within the hybrid layers after 2–4 months (Tay and Pashley 2009). Thus, the calcium ion release from the tricalcium silicates may have a significant effect on remineralization of dentine.
Root canals are often irrigated with solutions such as EDTA and citric acid that remove the smear layer. The best combination for effective removal of a smear layer has been found to be alternate use of sodium hypochlorite (NaOCI) and EDTA (Baumgartner and Mader 1987; Fig. 5.2). Removal of the smear layer allows the sealers to enter some of the dentinal tubules. The use of a fluoride-containing sealer potentially may enhance dentine remineralization. The concentration of fluoride in the dentine in the coronal part of the root canal was found to be increased after obturation with gutta-percha and a glass-ionomer sealer (Saunders et al. 1992). In another study assessing a silver-reinforced glass-ionomer cement used as a root-end filling material, fluoride ion release was also reported, but its effect on dentine was not assessed; the corrosion potential of the silver was however investigated (Roth 1991). It was shown that there was no difference in dye leakage of the different glass-ionomers investigated when using vertically condensed gutta-percha. It was thus concluded that glass-ionomer cement is possibly a clinical alternative for the sealing of retrograde cavities. The silver-reinforced materials, however, may cause tissue irritation from release of silver ions and their corrosion products (Roth 1991).
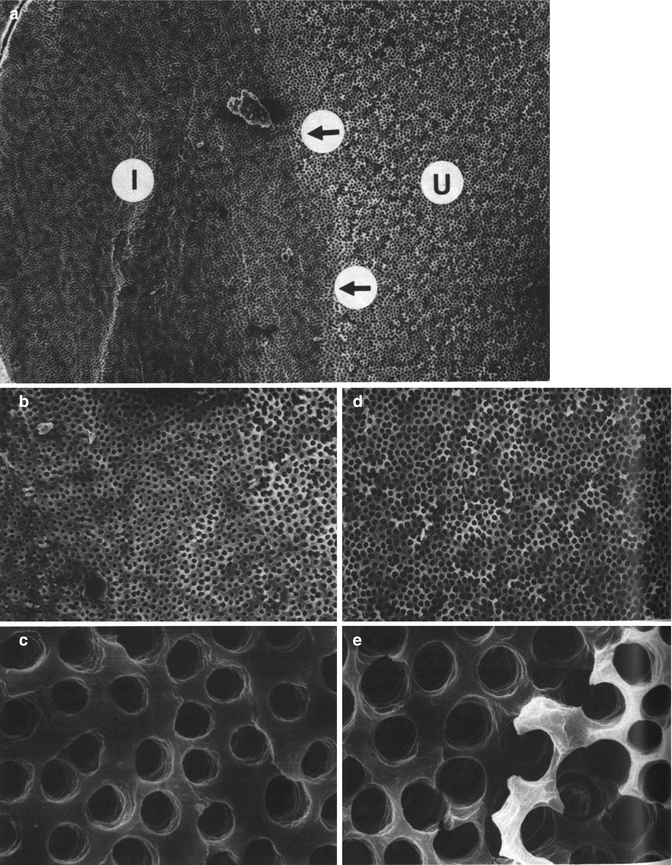

Fig. 5.2
SEMs of the root canal wall showing the effects of removal of the smear layer by using a regimen of NaOCI and EDTA alternately. (a) SEM (×148) of the junction (arrowed) between the instrumented (I) and un-instrumented (U) halves of the canal wall. (b) SEM of the instrumented half of the canal wall at a lower magnification (×504) and (c) at higher magnification (×4,040). Note the clean, smoothly planed appearance of the canal wall and the sharply defined orifices of the dentinal tubules. (d) Lower magnification (×506) and higher magnification (e) (×4,000) micrographs of the un-instrumented half of the canal wall. Note the erosion of the globular surface of the calcospherites and enlargement of the orifices of the dentinal tubules (Reprinted from Baumgartner and Mader (1987). With permission from Elsevier)
Fluoride-releasing glass-ionomer cement seems to be an appropriate material to seal the root canals of overdenture abutments, because it has an inhibiting effect on demineralization at the cavity wall in vitro (Ikebe et al. 2001). When glass-ionomer was used over carious root surfaces in one study, fluoride concentrations varied markedly along the outer edge of both normal and carious root tissues (Shu et al. 1998). However, fluoride increase at the edge of the lesion was found to be a result of remineralization events during caries (Shu et al. 1998). A sealer composed of pre-reacted glass-ionomer filler particles leaching aluminium, boron, fluoride, sodium, silicon, strontium and zinc showed that the incorporation of fluoride, strontium, silicon and boron along the outermost dentine of the sealer-dentine interface and the incorporation depth increased with prolonged immersion (Han and Okiji 2011). This provides evidence that fluoride causes remineralization of root dentine and hence, glass-ionomer cements may offer some potential benefits in this regard.
5.2.3 Biocompatibility
The biocompatibility of glass-ionomer materials is an important property when the materials are used as root-end fillers and to a certain extent when used as sealers which come into contact with the tissues at the apex. The biocompatibility of glass-ionomer cements has been assessed mostly at the cellular level, with osteosarcoma cells (Peltola et al. 1992; Lee et al. 2012) and gingival fibroblasts (Peltola et al. 1992; Koulaouzidou et al. 2005; Vajrabhaya et al. 2006; Al-Hiyasat et al. 2012; Subbarao et al. 2012) being the preferred cell lines. Animal models have also been used (Kolokuris et al. 1996; Tassery et al. 1999; Ogasawara et al. 2003) with intraosseous implantation in rabbit mandible being more reliable than implantation in femurs (Tassery et al. 1999). Subcutaneous implantation was also used for assessment of biocompatibility of glass-ionomers (Kolokuris et al. 1996). Mild inflammatory reaction was observed with Ketac Endo on the 5th day (Fig. 5.3a); the connective tissue was infiltrated with plasma cells as well as lymphocytes and macrophages were observed. The intensity of the reaction diminished by the 15th day (Fig. 5.3b), and this reduction continued progressively through the 60th and 120th days (Fig. 5.3c). The control material, a zinc oxide-eugenol-based sealer, remained irritating even after long-term implantation periods (Kolokuris et al. 1996).
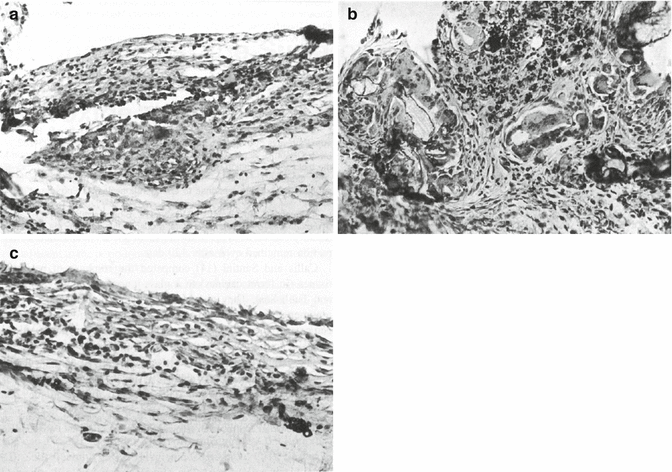

Fig. 5.3
Histological assessment of tissue biocompatibility of Ketac Endo sealer in rat connective tissue showing microscopic appearance on the (a) 5th day: mild inflammation with the presence of mononuclear cells. (b) 15th day: presence of giant cells and macrophages. Note also the presence of connective tissue. (H ×215). (c) 60th day: presence of a few mononuclear cells. (H ×325) (Reprinted from Kolokuris et al. (1996). With permission from Elsevier)
The biocompatibility of glass-ionomer cement used as a root-end filling material was previously compared to zinc oxide-eugenol cement, where in every assessment, the glass-ionomer outperformed the zinc oxide. It was found that the zinc oxide-eugenol sealer was largely absorbed and surrounded by fibrous tissue with many macrophages. The disintegrated material was completely engulfed by macrophages, whereas the glass-ionomer cement remained in the bone cavity with no inflammatory reaction (Ogasawara et al. 2003). The short-term biocompatibility of glass-ionomer cement was similar to zinc oxide-eugenol, but after 12 weeks, the former had better performance when assessed by intraosseous implantation (Tassery et al. 1999). Subcutaneous implantation of glass-ionomer cement (Fig. 5.3) and zinc oxide-eugenol showed connective tissue infiltration with plasma cells for glass-ionomers, while necrosis was evident with zinc oxide-eugenol (Kolokuris et al. 1996). Thus, glass-ionomer cement appears to have at least similar but probably better biocompatibility than zinc oxide-eugenol.
Further studies on glass-ionomer biocompatibility were performed to compare glass-ionomers to MTA. Glass-ionomer cements were comparable to MTA in one study (Lee et al. 2012), but the biocompatibility of glass-ionomers was generally lower than that of MTA in several other reports (Koulaouzidou et al. 2005; Vajrabhaya et al. 2006; Al-Hiyasat et al. 2012). The surface of a glass-ionomer restorative material (Ketac Fil, 3M ESPE, St Paul, MN, USA) showed very sparse cellular growth, with cells of round shape, and a rough surface due to the extending processes, namely, microvilli. While on the root surface, cells demonstrated excellent attachment features including a spindle shape, lamellipodia, filopodia and microvilli (Fig. 5.4). This was in contrast to MTA where cell attachment to the material was similar to the attachment on the dentine surface (Al-Hiyasat et al. 2012). Glass-ionomer was less biocompatible than composite resin, titanium and amalgam (Peltola et al. 1992). The biocompatibility of glass-ionomer cement to fibroblasts appeared to be improved by the addition of bioactive glass than by the addition of collagen. The glass-ionomer and modified glass-ionomer still exhibited reduced biocompatibility when compared to MTA (Subbarao et al. 2012).


Fig. 5.4
Scanning electron micrograph of Ketac Fil: (a) material surface (×6,000) showing limited cell attachment. (b) Root surface (×6,000) showing very good cell attachment. (Arrows indicate cells.) (Reprinted from Al-Hiyasat et al. (2012). With permission from Journal of Applied Oral Sciences (JAOS))
5.2.4 Antibacterial Properties
The antibacterial effect of glass-ionomer cement is related to its acidity (Vermeersch et al. 2005); adjustment of the liquid to pH 5 results in a loss of antibacterial activity (DeSchepper et al. 1989). Furthermore, the fluoride concentration has been linked with the antimicrobial activity, with no antimicrobial activity reported when the fluoride release is lowered (DeSchepper et al. 1989). The effect of fluoride does not have a great impact on glass-ionomers used as sealers, root-end filling materials and materials used in perforation repair sites as fluoride recharge is necessary for sustained fluoride release by glass-ionomers, and these sites are relatively inaccessible for recharge. In fact, the antibacterial activity of glass-ionomers is increased with topical applications of fluoride toothpaste and gels (Seppä et al. 1993).
Glass-ionomer sealers exhibit antibacterial activity initially, but this effect reduces drastically after 7 days according to some studies (Shalhav et al. 1997; Anumula et al. 2012). In other reports, it exhibits no antimicrobial activity at all (Heling and Chandler 1996). However, the technique used to test antibacterial activity, time of incubation and the ingredients of the materials tested can affect the results of the microbiological studies (Cobankara et al. 2004). This should be taken into consideration when reviewing conflicting results which prevent firm conclusions to be drawn.
5.3 Uses of Glass-Ionomer Cements in Endodontics
Glass-ionomers are used in endodontics for the following:
-
Nonsurgical applications: for temporary inter-appointment restoration during root canal treatment, as intra-orifice barrier materials, as root canal sealers and for nonsurgical perforation repair
-
Surgical applications: as root-end filling materials and for surgical perforation repair
There are no specifications for root-end filling and perforation repair materials although root canal sealer properties have to comply with ANSI/ADA Specification 57 (2000) and ISO 6876 (2012).
5.3.1 Root Canal Sealers
Root canal sealer properties have to comply with ANSI/ADA Specification 57 (2000) and ISO 6876 (2012). Regardless of the extensive use of glass-ionomers as root canal sealers, there is very limited research on the material properties and compliance to the specifications for root canal sealers. Only the compliance of glass-ionomer materials for radiopacity (Shah et al. 1996; Tanomaru-Filho et al. 2008) has been reported. Unfortunately, glass-ionomer sealers have also shown high solubility and disintegration (Carvalho-Júnior et al. 2003), which is not in compliance with the ADA specifications for root canal sealers (ADA Specifications No. 57, 2000).
The main scope of a root canal sealer is to bind the gutta-percha to the dentine walls. Thus, the ideal sealer should adhere to both the gutta-percha and also to the dentine. Glass-ionomer sealers have shown poor adhesive properties to both root dentine and gutta-percha. This lack of adhesion would imply a potential for increased microleakage if glass-ionomer is used as a sealer.
Studies evaluating microleakage of glass-ionomer sealers show that they consistently perform worse than epoxy-based resin sealers and were comparable or even worse than zinc oxide-based root canal sealers. These evaluations were performed using dye leakage with methylene blue (Smith and Steiman 1994; Rohde et al. 1996; Oliver and Abbott 1998; Kumar and Shruthi 2012), India ink (Goldberg et al. 1995; Leonard et al. 1996), dye leakage with three-dimensional reconstruction (Lyroudia et al. 2000), salivary bacterial leakage (Malone and Donnelly 1997) and the fluid transport model (Cobankara et al. 2002; Miletić et al. 2002; Economides et al. 2005).
Glass-ionomer sealer provided adequate sealing of the root canal and was comparable to a number of commercial sealers (Miletić et al. 1999). The apical seal exhibited by glass-ionomer cement was not significantly different from that provided by zinc oxide-eugenol cement regardless of the presence or absence of a smear layer (Timpawat and Sripanaratanakul 1998), but inferior to that of a resin-based sealer used in conjunction with a dentine bonding agent (Leonard et al. 1996) and a polydimethylsiloxane-based root canal sealer (Cobankara et al. 2002). Epoxy resin-based sealers invariably exhibited a better seal than glass-ionomer sealers (De Almeida et al. 2000). In a previous study, both a glass-ionomer sealer (Ketac Endo) and a zinc oxide-eugenol-based sealer prevented coronal microleakage in the absence of a coronal restoration (Malone and Donnelly 1997). In the Oliver and Abbott (1998) study, interestingly enough, coronal gutta-percha was removed and the coronal 3 mm filled with Ketac Endo only but not in the AH 26 (Dentsply, DeTrey, Konstanz, Germany) group which served as the control. The study found comparable coronal leakage of Ketac Endo specimens to the control group (AH 26), regardless of the different methodologies used to obturate the root canals (Oliver and Abbott 1998). In another study, the area of adhesive failure was 88 % for the glass-ionomer sealer compared to 15 % for the epoxy resin-based root canal sealer (De Gee et al. 1994). In general, the thickness of the sealer and sealer type has been shown to affect the adequacy of the obturation and subsequent microleakage (Georgopoulou et al. 1995). Glass-ionomer sealer exhibited a better seal when used in very thin sections than other sealer types but did not perform as well when thicker amounts of materials were used (Wu et al. 1994, 1997).
Regardless of the poor outcomes of the results of leakage studies, it is now widely believed that there is little value in using in vitro models as they are not totally adept at quantifying the seal of root canals. In addition, the major limitations of in vitro leakage studies are their lack of reproducibility, relatively small sample size and inadequate statistical power, lack of standardization as well as lack of correlation among different leakage models (Wu and Wesselink 1993; Schuurs et al. 1993; Lucena et al. 2013). The clinical implications for experimental laboratory-based sealability models are unclear. The ideal assessment of endodontic treatment outcome is still clinical evaluation.
5.3.2 Intra-orifice Barrier and Restorative Material
The use of glass-ionomer cement as an intra-orifice barrier material and as an interim dressing relies on the material’s sealing ability, antimicrobial properties and adhesion. The use of glass-ionomer cements to block the root canal orifice used in conjunction with gutta-percha obturations is shown diagrammatically in Fig. 5.5. Glass-ionomer significantly reduced coronal microleakage compared to a previously available thermoplastic synthetic polymer-based root canal filling, Resilon (resin-based solid cones) (Epiphany, Pentron Clinical Technologies, Wallingford, CT, USA), used without sealer and the glass-ionomer intra-orifice barrier (Jack and Goodell 2008). When used as a coronal barrier in cases of nonvital bleaching, glass-ionomer provided adequate seal comparable to that of white MTA (Vosoughhosseini et al. 2011). However, another research has shown a high degree of leakage when glass-ionomer was used as an intra-canal orifice barrier during tooth bleaching with sodium perborate and peroxide gel (Canoglu et al. 2012).
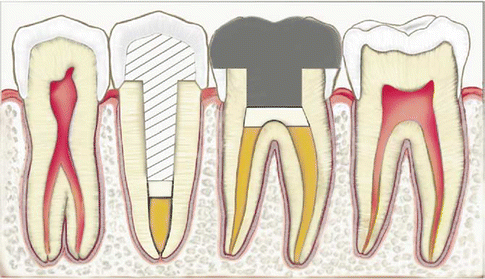

Fig. 5.5
Intra-canal glass-ionomer barrier illustrated under the post in the second premolar. Intra-orifice glass-ionomer barriers over the canals and over the pulpal floor are illustrated in the first molar (Reprinted from Mavec et al. (2006). With permission from Elsevier)
Glass-ionomer used as a restorative material after root canal treatment is not affected by the presence of other endodontic materials used for temporary filling or sealers present on dentine which are left from previous endodontic visits. The bond strength of dentine with and without the presence of other endodontic materials was the same (Capurro et al. 1993). A clinical picture of glass-ionomer used as a temporary dressing is shown in Fig. 5.6.

Fig. 5.6
Glass-ionomer cement used as a temporary filling material (Courtesy of Ms. Chiara Brincat and Dr. Matthew Cachia, University Dental Clinic, Faculty of Dental Surgery, University of Malta, Malta)
When glass-ionomer cement is used over MTA, as in cases of pulpotomy, the glass-ionomer appeared to exhibit a high degree of microcracking and some porosity in the interfacial region with MTA (Camilleri 2011). In addition, strontium from the glass-ionomer was detected at 200 μm within the MTA (Camilleri 2011). Hence, it appears that the pH disparity between MTA and glass-ionomer cement affects the properties of both materials and their interaction.
5.3.3 Root-End Filling Material
Root-end (retrograde) filling materials should exhibit an adequate seal and also encourage cell growth and differentiation particularly with periodontal ligament cells. The sealing ability of glass-ionomer root-end filling materials was reported to be comparable to that of amalgam, and it was preferable to use a root-end filling material rather than leave the tooth unfilled at the root-end (Olson et al. 1990). In several studies, conventional glass-ionomer cement used as a root-end filling material exhibited better sealing ability; it was shown to be better than amalgam (Chong et al. 1991; Ozata et al. 1993), silver-reinforced glass-ionomer (Ozata et al. 1993), amalgam applied with varnish or amalgam applied with a dentine bonding agent (Sutimuntanakul et al. 2000
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


