
16

Facial Paralysis Rehabilitation
Patients with facial paralysis suffer significant functional, aesthetic, and psychosocial sequelae. Even single branch weakness or paralysis may leave patients with significant functional disability and aesthetic deformity. Spontaneous recovery from facial paralysis is rare except in some inflammatory conditions. Consequently, treatment of underlying conditions must be augmented with nerve repair and reconstruction of associated deficits to achieve optimal rehabilitation. This chapter reviews repair and reconstructive options for rehabilitation of patients with facial paralysis resulting from congenital, traumatic, inflammatory, idiopathic, and neoplastic conditions. The diagnosis of these congenital, inflammatory, idiopathic, and neoplastic processes, assessment of trauma, and the role of decompression and pharmacological management are, however, not the focus of this discussion and are not reviewed here. This discussion begins with an overview of physician–patient communication. The signs and symptoms of facial paralysis are then reviewed, followed by an outline of physical examination and diagnostic evaluation of facial paralysis. Planning surgically based rehabilitation follows, with discussion of surgical options, technique, and associated nonsurgical rehabilitative care and detailed analysis of primary options.
 Physician Patient Communication
Physician Patient Communication
Communication is the single most important aspect of successful surgically based rehabilitation for facial paralysis. Communication between the physician and the patient forms the foundation for therapeutic intervention most likely to result in optimal outcomes. Surgical repair and reconstruction have inherent risk to facial nerve function. Nuances of restoration and degrees of dysfunction are difficult for many patients to appreciate and accept. The range of recovery and potential for synkinesis and mass motion are not easily instilled even with detailed patient and family teaching. Both patient satisfaction and risk management issues are apparent in the context of repair for existing deficits, risk of less than full functional return, and possible complications. Thus building rapport with the patient and family and leading frank discussions that include realistic descriptions of the plan, the risks, and the expected course are essential. Finally, professional, empathic bedside manner and patient rapport are invaluable in the postoperative phase of rehabilitation.
 Facial Paralysis
Facial Paralysis
Signs and Symptoms
Facial paralysis is a condition made complex by the anatomical and functional intricacy of the facial nerve. A range of partial to total facial nerve injuries can manifest in a single branch or the entire nerve as facial paresis or paralysis. The branches of the facial nerve, as described in Chapter 1, comprise arborizations of the upper (temporofacial) and lower (cervicofacial) divisions. These include the temporal, zygomatic, buccal, marginal mandibular and cervical branches. Table 16-1 outlines signs and symptoms by facial nerve branch.
Table 16-1 Branch Facial Nerve Deficits with Associated Signs and Symptoms
| Branch deficit | Sign and symptom |
|---|---|
| Upper frontal | Brow ptosis |
| Upper zygomatic | Lagophthalmus |
| Ectropion | |
| Corneal and conjunctival exposure | |
| Zygomatic/bucca | Malar flattening |
| Loss of nasolabial fold | |
| Nasal obstruction alar displacement | |
| Nasal twisting | |
| Lip commissure ptosis | |
| Lower buccal/marginal | Oral commissure incompetence |
| Lower lip ptosis | |
| Dysarthria | |
| Drooling |
Prominent physical signs found in facial paralysis affect the brow and eyelid complex, the midface, and the oral complex. Brow and eyelid signs include brow ptosis, lagophthalmus, ectropion, and corneal and conjunctival exposure. The midface may show paresis or paralysis, nasal deviation, alar base drift, and nasal valve collapse. Oral complex signs include oral commissure ptosis, lip laxity, and oral incompetence. The effect of adjacent anesthesia and hypesthesia are often underestimated, especially in facial paresis as opposed to paralysis. Findings of twitches, tics, subtle asymmetries, and numbness may not be primary complaints for patients but are nonetheless important to elicit in a thorough assessment.
Patients complain of a variety of disabling symptoms associated with these signs. Potential loss of vision or visual acuity is common with corneal and conjunctival exposure. Nasal obstructive symptoms and malposition of the nasal base accompanies midface paralysis. Ptosis begets dysarthria and oral incompetence from oral commissure drift inferiorly and oral tone loss due to denervation of the orbicularis oris. Malposition of the lower lip, stemming from marginal mandibular branch weakness or combined inferior and buccal facial weakness, is also common. Aesthetic asymmetry is also disturbing to patients who see brow ptosis, nasolabial fold flattening, asymmetric faces, and exaggerated asymmetry with smile or other emotionally expressive states.
 Facial Aesthetics
Facial Aesthetics
Facial aesthetics mandates a more qualitative approach to assessment. The smile is a central element of facial aesthetics in facial paralysis. Rubin1 posits that smile pattern can be studied and classified as one of three types. The “Mona Lisa” smile is the most common at 67%; it is a product of the dominant action of the zygomaticus major. The corners of the mouth move laterally and superiorly, and the upper lip moves minimally.1 The canine smile (31%) is a vertical elevation of the upper lip and then lateral lift of the corners of the mouth.1 The toothy or full denture smile is rare at 2%.1 It is created by simultaneous contracture of the depressors and elevators of the lips and commissure of the mouth.
The combined effects of dysfunction and aesthetic asymmetry generate significant psychological impact and social stigma. Often these conditions are the sequelae of the treatment of a potentially life-threatening carcinoma. Physical and mental functional status, along with the complex issues of age, identity, body image, and familial and social role, influences patient and family response and coping strategies during rehabilitation. Optimistic patients seem to transcend the symptoms of paralysis and participate more effectively in the rehabilitation plan. Factors such as grossly dysfunctional interpersonal or family dynamics, suspected substance use, and a significant psychiatric history are likely to influence coping and participation in rehabilitation and may augur behavioral complications during recovery or worsening perception of symptoms. Presence of these or similar factors in the history or assessment warrant appropriate psychosocial or psychiatric consultation. These consultations may be extremely helpful as the reconstructive plan is designed and implemented.
 Patient Assessment
Patient Assessment
Patient assessment in facial paralysis is necessarily detailed and must include both systemic and focal considerations.2 Patients typically present with a personal impression of acute onset facial paralysis. The history or exam may reveal that this acuity may or may not be the case; often, facial paralysis was subtly present and ultimately became visible to the patient and family. The medical history, as a highly important aspect of assessment, generally illuminates the underlying etiology and may offer significant prognostic implications. For example, ~ 80% of patients with Bell’s palsy will recover complete function in the subsequent 6 months.2 A history of varicella infection can be associated with later development of herpes zoster oticus resulting in facial paralysis. The rare schwannoma and neurofibroma can also manifest with facial paralysis as the presenting symptom. More ominously, a history of a malignant neoplasm with high distant metastatic potential, such as breast, lung, and renal cancers, may result in facial nerve involvement and subsequent paralysis. Similarly, history of local temporal or cheek skin cancers or parotid tumors with high potential for locoregional recurrence and perineural invasion in the absence of a significant mass may create facial nerve pathology and consequent paralysis.
In the presence of known facial nerve invasion by primary neoplasms or metastases, tumor status, adjacent nerve and muscle status, and radiotherapy history and future treatment plans including radiotherapy should be assessed for their influence on the rehabilitation plan and effect on the surgical bed. The impact of radiation has been argued in both pre-and postoperative contexts. Conley3 felt that the impact of radiation was minimal, and this was confirmed by McGuirt and McCabe4 experimentally. Fisch5 studied 42 grafted patients and reported that the function of nerve grafts could be diminished by 75–25% in patients undergoing post-operative radiotherapy.5 The theoretical impact is on neovascularization of the surgical bed, though the nerve grafts per se are felt to be radio-resistant. We successfully utilize grafts for patients who will undergo radiotherapy.
A careful general history, including constitutional factors, is complemented by a detailed otorhinolaryngologic assessment along with the patient’s story of the paralysis and associated symptoms. Major points to elicit in history taking are outlined in Table 16-2.
 Physical Examination
Physical Examination
Physical examination begins with a complete head and neck and cranial nerve examination. Cranial nerve examination should emphasize the facial, trigeminal, hypoglossal, and glossopharyngeal nerves. Further, special attention should be focused on temporalis and masseter function and tone, as these muscles may be used in the reconstruction. Otologic evaluation must also be included, as should oral cavity and mandibular assessment. The facial exam includes assessment of resting position and tone, symmetry, and partial or total weakness. Previous scars and incisions should be fully evaluated, and photographs or line drawings of scars and related findings may be helpful in diagnosis and follow-up.
Specific facial features to assess include brow position, eyelid position, and ectropion. The position of the lacrimal puncta should be carefully evaluated. An ophthalmology consultation may be indicated if any concerns about corneal protection or integrity arise or if further assessment of these structures is required. The appearance and position of the malar fat pad, suborbicularis oculi fat pad (SOOF), and nasolabial fold are important to note. Position of the nasal ala and caudal septum is often altered with inferior and medial drift of the ala and twisting of the nose to the nonparalyzed side. Lip tone, commissure position, lip length, and ability to purse the lips are generally altered. The associated gross functions of oral competence, speech, and mastication should be assessed concurrently. These functions are generally assessed qualitatively rather than quantitatively.
Clinical facial nerve function should be graded by standardized scales and refined by descriptors to further assist in the assessment of the deficit. The House-Brackmann scale is the current standard grading system for facial nerve function.6 The Facial Nerve Disorders Committee of the American Academy of Otolaryngology–Head and Neck Surgery adopted the scale in 1985 as a reproducible, easily used clinical tool.6 The House-Brackmann scale affords a general quantification of facial nerve function. Nonetheless, initial trauma or rehabilitative procedures may result in each branch or facial region having a different grade of function. Thus each branch or region should also be considered individually. Most easily, the face may be divided into the horizontal thirds, as in aesthetic surgery, and graded in separate exams.
 Diagnostic Testing
Diagnostic Testing
The evaluation of facial paralysis should include imaging and other diagnostic studies as appropriate to the patient’s history and current condition. Cross-sectional imaging, including magnetic resonance imaging (MRI) and computed tomography (CT) of the neck and often the temporal bone, is often undertaken as an initial step.7 If the facial nerve is dysfunctional but CT is negative, MRI may help localize the site of facial nerve abnormality.7 It will show abnormal enhancement at the site of the nerve pathology. Abnormal enhancement on MRI has been the only positive finding in some patients who have facial paralysis secondary to perineural invasion of previously resected skin neoplasms.8–10 Positron emission tomography (PET) may also be performed if primary imaging does not reveal a gross lesion invading the facial nerve. Additionally, PET affords the diagnostic asset of the ability to rule out distant metastasis.
Functional testing is equally essential to complete evaluation. Often assessment of the visual anatomical and functional sequelae (including corneal anatomical and functional protective mechanisms and lacrimation) is necessary. Schirmer testing or visual and corneal assessments are important to protect the health and vision of the eye, especially for patients with xerophthalmia and/or corneal anesthesia from multiple neuropathy. Auditory and neurovestibular assessments may be indicated for internal auditory canal lesions. If a history of skin cancer is identified, workup may include biopsy of scar or other lesions in the field. Patients with newly diagnosed or concurrently evaluated parotid or other neoplasia require imaging pursuant to the mass itself, as well as excellent examination of the facial nerve. At baseline, neurological function of the parotid compartment and ear and skull base region function may be normal, impaired, or paralyzed for the preoperative patient undergoing facial resection.
Neuromuscular diagnostic testing evaluates nerve function and injury. Electromyography quantifies the potential of the facial nerve to reenervate the muscle. Electroneuronography comparatively qualifies the function of the neuromuscular unit. Hence, neuromuscular testing is critical to a successful rehabilitation plan.
Electromyography (EMG) helps define the ability of the muscle to be reinnervated.11 EMG aids in identification of denervation atrophy or subclinical innervation, particularly if facial paralysis is long-standing (e. g., a duration of a year or longer). The normally innervated muscle will have functional motor axons; which will stimulate the motor end units of the muscles and generate normal voluntary action potentials. Facial muscles undergoing reinnervation will be identifiable as they generate polyphasic potentials. These may precede visible facial motion or tone. The normal muscle that is denervated will give the EMG pattern called denervation or fibrillation potentials. The denervated muscle will be electrically silent, reflecting denervation atrophy or in congenital cases potential absence of the muscle. The EMG technician or neurologist should indicate which muscles need to be assessed if not all regions of the face are involved.
Electroneuronography (ENoG) (also known as electroneurography or neuromyography) is helpful in quantifying the type of injury that the facial nerve has sustained, particularly in acute nontraumatic or surgical facial paralysis when the integrity of the nerve is not in question. EnoG assesses the integrity of the nerve–muscle unit through comparison with the unaffected side.12 As a comparative technique, ENoG is useful in judging the need for decompression for inflammatory or traumatic conditions and monitoring nerve recovery. In surgical cases, neuropraxia with an intact nerve is most often treated expectantly with supportive management of sequelae. Electrical testing with ENoG will assist in postoperative counseling. The plan for rehabilitation after resection of intracranial lesions involving resection of the facial nerve will be addressed based on how much of the facial nerve root remains.
 Surgical Rehabilitation
Surgical Rehabilitation
Rehabilitation Planp
Developing a plan for rehabilitation requires careful consideration of the individual needs of the patient, including comorbid disease and other relevant medical concerns, extent of paralysis and rehabilitation potential, and patient and family capacities and expectations, along with the technical capabilities of the surgeon. Discrete physical factors encompassing but not limited to the surgical bed and the length and other characteristics of planned procedures must also be considered. Immediate needs are balanced against potential and capacity to create achievable short-and long-term rehabilitative goals in partnership with a well-informed patient and family. The postoperative plan and resultant expectations for patient and family participation should be clearly laid out in that partnership. Incorporation of physical therapy and other rehabilitative modalities including biofeedback are best programmed into the initial rehabilitative plan as optional or actual interventions.
The ideal repair and reconstruction plan results in normal facial appearance at rest and symmetric spontaneous emotional function, and symmetric voluntary function with no significant surgical scarring or delay in return of function. Ideal medical management limits side effects and results in full function without deficit. Unfortunately, if the facial paralysis results from removal or transection of the nerve, this is usually a goal that is not attainable. The goals and expectations should then be realistically reviewed by the patient and surgical team, to define the plan and achieve the best results possible. If the nerve is paralyzed from an inflammatory etiology, the clinical decision will be whether to decompress the facial nerve, or the middle ear and mastoid in cases of otomastoiditis. In cases of Lyme disease and viral neuronitis, for example, the treatment plan will involve consultation with an infectious disease specialist and the selection of an appropriate antibiotic or antiviral, most often with concomitant pulsed corticosteroids.
Ideal surgical management—the focus of this chapter—involves minimal or imperceptible donor site morbidity for nerve or muscle grafts with optimal return of function after reinnervation and engraftment occur. At its core, the plan for surgical repair and rehabilitation is based simply on the functional status and anatomical integrity of the facial nerve or its branches and muscle end organs. Nerve growth is the rate-limiting factor in reinnervation, and the quality of reinnervation affects overall rehabilitation. Muscle motor end-plate status is tied to reinnervation and thus is equally important when given that innervation, at the rate of 1.0 to 1.5 mm growth per day, could take 6 to 9 months for most grafts. Full restoration of static and dynamic facial aesthetics is, in a sense, the “holy grail” of facial nerve paralysis rehabilitation. Functional return is never complete with the current armamentarium of surgical and allied procedures; in measures of both subtle and, often, gross performance status.
 Surgical Repair
Surgical Repair
The nature of paralysis (i. e., complete vs partial and paralysis vs paresis) and neuromuscular status, as well as the etiology, influences prognosis for functional return and options for rehabilitation. Factors affecting the surgical bed, such as a history or plan for radiation therapy, previous surgeries in the field, and related anatomical and tissue changes like fibrosis further narrow the rehabilitative surgical options. Repair and overall rehabilitation are dictated by status of the nerve, whether intact, transected, resected, or congenitally absent; muscle status, as having reinnervation (polyphasic), denervation (fibrillation potentials), or denervation atrophy as measured by EMG; and prognosis with expectation of likely, ambiguous, or absent functional return. The partially or completely intact nerve may be treated with a combination of drug therapy, surgical decompression, and monitoring. The completely transected or resected nerve with functional neuromuscular potential, however, warrants primary anastomosis or nerve grafting for repair.
Surgical repair of the facial nerve is accomplished through end-to-end anastomosis or nerve or neuromuscular grafts. Anastomosis is indicated in acute traumatic or surgical transaction as the anatomy of nerve ends allows. Nerve grafts are employed when the nerve cannot be approximated and the neuromuscular unit has sufficient functional innervation potential as judged by EMG and ENoG. Further, surgical support for the intact motor end plate may be accomplished with temporary or “babysitter” nerve grafts from adjacent cranial motor nerves. Neuromuscular flaps and grafts are necessitated by absent innervation potential.
Patients with persistent facial weakness after reinnervation with either nerve grafts or neuromuscular flaps present an interesting challenge. If they do not show denervation potentials on EMG, then a new interposition, cross-face nerve, or transposition hypoglossal nerve graft can be performed. These patients are not benefited by neurotization procedures such as nerve–muscle pedicles or nerve to muscle–innervated muscle grafts into the muscle. This type of procedure will not work unless the nerve supply to the muscle is transected. Moreover, the issue of graft take in preexisting facial paralysis has been debated; nonetheless, we undertake graft procedures as the opportunity cost is low. Grafting may be delayed, or both graft and static procedures may be utilized in the context of this clinically equivocal debate.
Anastomosis or primary repair; nerve grafts including interposition graft, cross-face nerve graft, trigeminal to facial nerve graft, and hypoglossal to facial nerve graft; dynamic sling procedures; static or adynamic sling procedures; and adjunctive aesthetic procedures are outlined in the following section. Summary for each type of procedure is offered, along with discussion of indication, procedure, and technique for selected operations. Nerve graft donor sites are also delineated.
Anastomosis or Primary Repair of Nerves
Indication
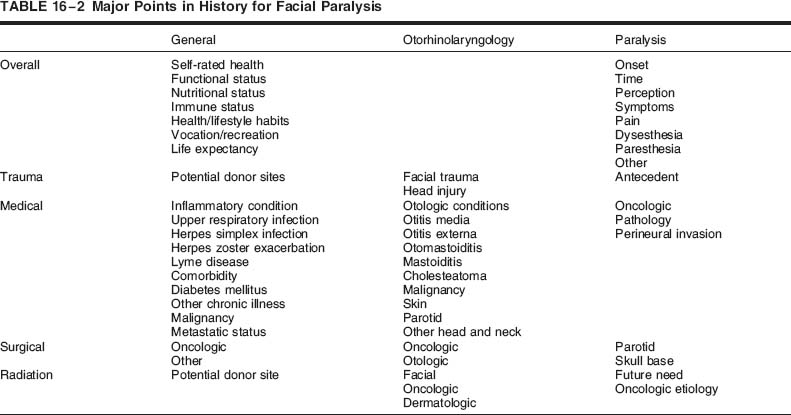
Transection of the facial nerve or its branches is best treated with primary repair (Fig. 16–1). Sharp lacerating transection is most easily repaired at the time of presentation or within 48 and, at the latest, 72 hours so the distal nerve can be identified with nerve stimulators. If a surgical delay is warranted for other reasons, such as physiological instability, then nerve repair becomes more difficult after 24 to 72 hours. Importantly, nerve stimulation with neuromuscular response is lost after this period. Further, local inflammatory changes make the distal nerve end difficult to identify. The nerve may still be identified anatomically, confirmed with frozen section histologic assessment, and then repaired. In all cases, the surgical bed should be well vascularized.
Procedure and Technique
The nerve ends are isolated and cleared of connective tissue from the nerve sheath for the first millimeter or two from the edge of the transection. In delayed repair for deficits with no loss of nerve length, primary repair is preferred, with freshening of the nerve and epineurium, followed by primary neurorrhaphy. The nerve may be mobilized if resection is required. However, mobilization can further alter blood supply, and one must weigh the extent of mobilization with the potential harm versus benefit. The effects of tension and the benefit of eliminating tension in peripheral nerve repair suggest that an interposition nerve graft is a better option than mobilization.13–16 Though the facial nerve is thought to have topographic orientation, the orientation cannot be identified even with magnification and is thus clinically insignificant in repair.17–21 The epineurium is anastamosed in an end-to-end or end-to-side fashion with monofilament microsutures.22 In delayed cases the edges of the nerve ends should be freshened. The nerve fascicles (endoneurium) may extend beyond the retracted epineurium but will usually be overcome through anastomosis.
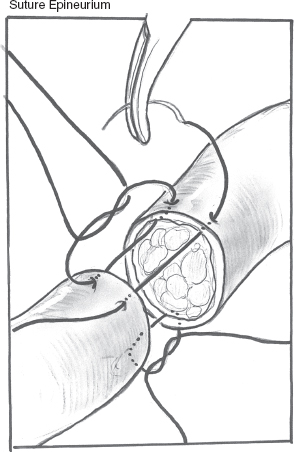
FIGURE 16-1 End-to-end anastomosis of epineurium in primary nerve repair.
Fine monofilament sutures (e. g., 8.0, 9.0, or 10.0) are placed through the epineurium. The fewest number of sutures are used to minimize inflammation, although the actual number varies based on size of the nerve. For example, distal anastomosis to facial branches 6 to 8 cm from the stylomastoid foramen usually requires one or two sutures. The facial nerve trunk at the level of the pre-or post-stylomastoid foramen may need 3 to 5 sutures. Needle size is at the discretion of the operator. Loop or microscope magnification is required, as are jeweler’s microforceps and a micro-needle driver, preferably of the nonlocking type. Adhesive, bioabsorbable and Silastic couplers and tube guides have also been used.23
Nerve Grafts (Interposition Nerve Grafts)
Indication
Segmental defects of the main trunk of the facial nerve, pes, or individual branches are best treated with interposition nerve graft. This technique allows for multi-segment rehabilitation to end-plate intact muscle. The direction of the nerve graft is debated as being of variable clinical importance. If multiple branches are needed, then orientation is based on anatomy of the graft. In simple segment branch grafts, some authors have postulated that reversing graft direction facilitates neural regeneration.22
Procedure and Technique
A nerve graft placed in the mastoid to graft to the vertical segment of the facial nerve can be secured with one, two, or often no sutures depending on the “fit” in the canal.
Segmental Defects in the Field of Soft Tissue Injury
For facial nerve injuries in the area of significant tissue necrosis such as blast injury, avulsion, or crush injury, the primary goal is to identify the distal and proximal ends of the nerves. These should be tagged with monofilament sutures. Most often these cases will require nerve grafting when the bed is suitable. If the soft tissue envelope is absent, then the area will require flap reconstruction of the skin and envelope. This may be a local fasciocutaneous flap or a remote tissue transfer such as a pedicled or microvascular flap. For patients with acute loss of facial nerve continuity due to laceration, tumor resection, or other process causing anatomical disruption of facial nerve continuity, the primary reconstructive goal is reestablishment of facial nerve continuity with primary or interposition graft repair.
For patients with preexisting paralysis and the presence of neoplasm, muscle status may be the most important element of repair. Electromyography must be used to assess the status of the muscle in cases of segmental nerve defect presenting for delayed repair or patients with prolonged duration of tumor and paralysis. The muscle loses motor action plates if it has been denervated for more than 12 to 18 months. The nerve graft cannot cause activation of denervated muscle, and hence the use of muscle replacement with muscle-free flap or use of adjacent muscles driven by other motor cranial nerves (i. e., trigeminal) must be considered.
Nerve Graft Donor Sites
Sural Nerve
The sural nerve allows for the longest, largest caliber nerve graft (Fig. 16–2). This graft offers a distant harvest/donor site option that allows for two-team harvest and ablation. It is contraindicated in patients with neuropathy from chemotherapy, diabetes, or trauma, with venous stasis disease, and with arterial insufficiency, as the normal patient will experience some anesthesia over the lateral inferior foot as a result of harvest.
Clinical Pearls
- The nerve is adjacent to the small saphenous vein/sural artery vein.
- The nerve can be harvested through a linear vertical incision or multiple stair-step (~ 1 cm) incisions with intervening subcutaneous dissection along the course of the nerve.
- A vein stripper may be used to harvest the nerve.
- The nerve graft obtained can be up to 25 to 40 cm in length, contingent upon the height of the patient.
Cervical Sensory Nerves
Cervical sensory nerves make an ideal graft with sufficient length and branching to meet the needs of a complex multisegment facial nerve defect. The great auricular nerve, given its terminal two or three branches, can serve as the major graft branch (Fig. 16–3). The great auricular nerve can be traced to Erb’s point at the posterior border of the sternocleidomastoid. From Erb’s point, the adjacent sensory nerves, such as the transverse cervical nerve and posteriorly directed sensory branches, can be followed peripherally and inferiorly into levels IV and V. This technique yields grafts up to 14 cm long with four or five branches as the nerves fuse at Erb’s point to a single branch and are followed deep to the root of the neck posteriorly to the sternocleidomastoid muscle. This graft should be harvested prior to starting a neck dissection if it is clinically indicated and not prohibited oncologically.
Medial Antebrachial Cutaneous Nerve
The medial antebrachial cutaneous nerve from the volar forearm offers a graft option with low donor site morbidity. The volar forearm is subsequently anesthetic, but most patients do not perceive this as a significant deficit. The graft is of appropriate size and length (at least 10–15 cm), and arborizations allow for three to five branches if needed. Further, this nerve graft can be harvested with and separated from a radial forearm flap in the case of microvascular reconstruction of the ablative defect. The bulk of the graft can also be kept embedded in the flap and the distal ends isolated for anastomosis, providing a vascularized nerve graft. The length of the graft is substantial if followed to distal arborizations.
Clinical Pearl
- The sural nerve site is preferable if a radial forearm flap is not being harvested simultaneously.
Cross-Face Nerve Grafts
Cross-face nerve grafts provide the most precise rehabilitative option for patients who have complex multi-segmental facial nerve defects where resection ablates the proximal facial nerve. Contralateral facial nerve branches are used to drive affected facial muscles. One of the branches from the unaffected side is employed in this technique, thereby weakening somewhat the function of that contralateral face as a result of the graft. However, this weakness affords secondary benefit in helping to restore overall facial symmetry. The cross-face nerve graft can “drive” innervate muscle on the injured facial nerve side. The muscle must have intact motor end plates. This technique provides the optimal option for these difficult cases, as very specific nerve branches from the contralateral facial nerve are used as the motor nerve for driving the contralateral matching muscle unit. This procedure should provide the most near symmetry in cases of successful engraftment. The nerve grafts are most often harvested from the sural nerve site, which allows for appropriately lengthy nerve grafts.
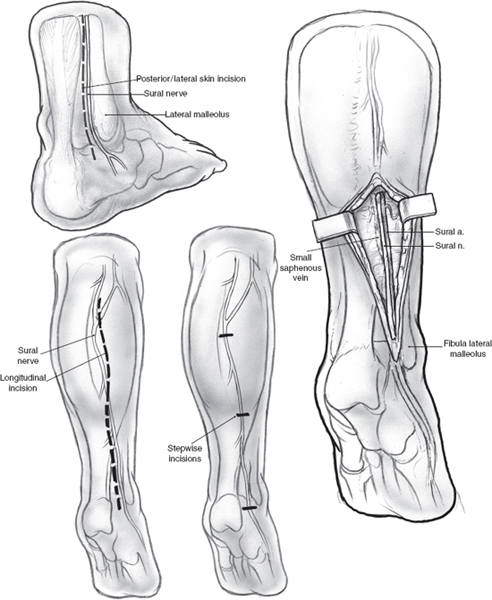
FIGURE 16-2 The sural nerve provides the longest, largest caliber graft. Stepwise or longitudinal incisions are utilized for harvest.
Cross-Face Nerve Graft to Intact Muscle
Multisegment rehabilitation to end-plate intact muscle with cross-face nerve grafts is required for patients who have no remnant proximal facial nerve. For example, this technique is useful for patients with internal auditory canal lesions with no proximal nerve available for interposition grafting procedures.
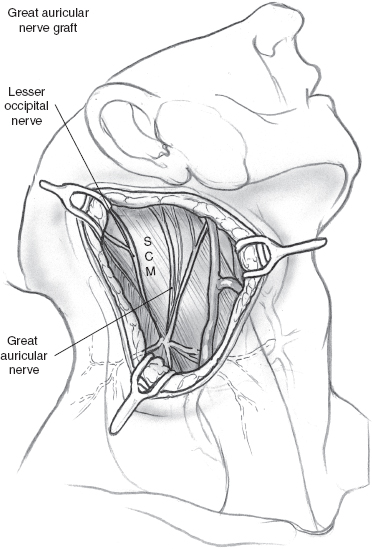
FIGURE 16-3 The great auricular nerve, when traced to Erb’s point, allows for harvest of a multibranched graft. SCM, sternocleidomastoid muscle.
Cross-Face Nerve Grafts to Neuromuscular Free Flaps
Multisegment rehabilitation to muscle without intact motor end plates is necessary in cases where there has either been muscle dennervation or muscle tissue loss from surgical resection, trauma, or another condition. The procedure provides muscle that can be driven by the facial nerve from the contralateral face. These candidates may be equally well served by the dynamic solution of ipsilateral temporalis or masseter flaps driven by the trigeminal nerve. This is, however, not an appropriate procedure for patients with high-risk tumors and high likelihood of mortality in the near term.
Transposition Hypoglossal Nerve Graft
Indication
Skull base surgery patients with known transection of facial nerve at the internal auditory canal (IAC) or brainstem, with no suitable proximal remnant for grafting, are primary candidates for a hypglossal nerve graft (Fig. 16–4). This procedure is also an option for patients who have an intact injured nerve with paralysis and denervation. However, the donor deficit from hijacking the trigeminal or hypoglossal nerves can be significant and hence deserves judicious consideration. The option of hypoglossal or trigeminal facial anastomosis may serve as definitive rehabilitation or as a “babysitter” with concurrent cross-face nerve grafts to the affected branches.24
Procedure
The procedure for hypoglossal to facial nerve transfer has evolved to avoid the need for a secondary interposition graft. Instead of segmental resection, ~ 40% of the ipsilateral, hypoglossal nerve is separated from the fascicle. The portion of the nerve is then mobilized and anastmosed to the facial nerve stump. Reinnervation ensues over the next 3 to 4 months.
Technique
Most commonly, this anastomosis uses a short interposition graft to the cut end of the hypoglossal nerve. Alternatively, a less morbid approach is to partially transect one third to just less than half or ~ 40% of the hypoglossal nerve cross section. This portion is then anastomosed to the interposition nerve graft.25,26 An alternative, which has the least morbidity, is the end-to-side anastomosis of an interpostion graft through epineural windows.22,27,28 If the facial nerve is absent, the ansa hypoglossi innervated strap muscle can be used to neurotize the facial mimetic muscles (Fig. 16–5).
Transposition Mandibular Branch of Trigeminal Nerve Graft
Indication
Indications for transposition hypoglossal nerve graft apply to use of the mandibular branch of the trigeminal nerve.
Procedure
The nerve can be mobilized to anastomose to a common facial trunk or upper/lower division as definitive or “babysitter” prior to cross-face nerve grafts.
Technique
The muscle or a portion can be mobilized on the neurovascular pedicle to dynamically animate the oral commissure and lips. The mobilized strip of muscle will be sutured to the oral commissure–upper/lower lip at the vermilion-cutaneous junction.
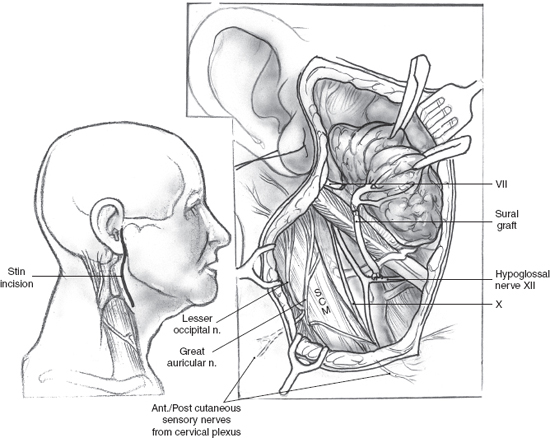
FIGURE 16-4 In a hypoglossal jump graft, the sural or great auricular is anastomosed to hypoglossal epineurium in an end-to-side fashion. SCM, sternocleidomastoid muscle.
Dynamic Slings
Dynamic support of involved facial musculature is achieved through a variety of means. The masseter muscle and trigeminal nerve may be transferred to address the midface and oral complex. The temporalis muscle can be employed to restore the lip and oral complex.
Masseter Muscle Transposition
Indication
Masseter muscle transposition offers definitive dynamic support to the affected midface, lips, and oral commis sure (Fig. 16–6). Further, its use can afford temporary support to patients with oral incompetence and nerve grafts or paresis who are expected to regain function or to supplement function in patients with incomplete recovery. The masseter flap can also be used in combination with an interposition graft to the upper face. The vector of pull of the temporalis muscle flap overall reconstructs a better smile because the temporalis muscle is thinner, longer, and has a greater contraction excursion. In addition, the masseter flap will create a bulge in the corner of the mouth and a concavity over the mandible. The masseter flap can be rotated, however, following special situations such as after radical cancer surgery to the parotid. The lateral lip and oral commissure will undergo myoneurotization after fresh denervation of the respective facial muscles.
Technique
The neural innervation to the masseter muscle arises deep to the zygomatic arch and deep to the masseter muscle running from posterosuperior to anteroinferior and must be preserved. The periosteum of the mandible must be elevated along with the masseter muscle, as sutures will not hold to muscle alone. The entire muscle is used.
Temporalis Muscle to Lip Complex Transfer
Indication
This procedure dynamically restores position and tone to the lateral lip and oral commissure, and function to the midface lip and lateral nose with good outcomes (Fig. 16–7). It provides temporary support for patients with oral incompetence and nerve grafts or paresis who are expected to regain function and supplements function in patients with incomplete recovery. The superficial temporoparietal fascia can be used to minimize scaphoid defect from mobilizing a portion of the temporalis muscle. Bulk and bulge are generally minimized through atrophy over time after careful appraisal of the initial volume of tissue transferred. The expected amount of excursion of the lip that can be achieved in this transfer is ~ 1.5 to 2.0 cm.
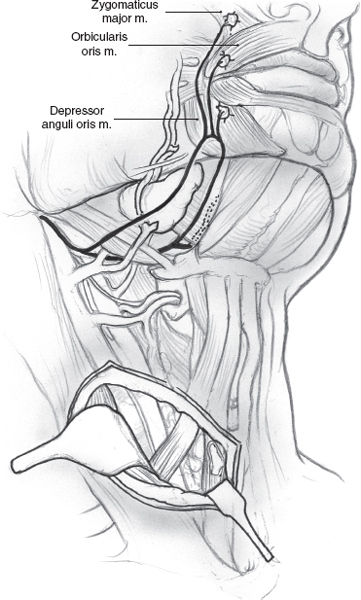
FIGURE 16-5 Ansa hypoglossi innervated strap muscle can be employed for neurotization if the facial nerve is absent.
Technique
The neurovascular supply to the temporalis muscle is from the deep surface of the muscle. Previous radical parotidectomy or mandibulectomy may have damaged the blood supply and should be considered in planning the use of this procedure. The middle one third of the muscle is used, after the overlying and posterior temporoparietal fasciae are mobilized and reflected out of the way. To help further refine the choice of the temporalis muscle segment, the vector of pull can be diagrammed at the oral commissure and extended out to the temporalis.
Procedure
- The temporalis muscle flap is designed to be ~ 1.5 to 2.0 cm at the base (zygoma). It can be made wider at the distal portion.
- Dissect the flap off the temporoparietal bone to the zygomatic arch; the arch will serve as the fulcrum. The neurovascular pedicle is on the deep surface. Just above the level of the zygomatic arch one should be careful not to divide the fibers prior to assessing the pedicle.
- The distal dissection into the cheek inferiorly to the lip should be in a subcutaneous plane not only to avoid intact nerve or nerve grafts, for patients who may be able to recover nerve function, but also because this is the plane of insertion of the facial mimetic muscles.
- The distal end of the muscle is then split into two or three segments. Most often two are ideal. They are then sutured to the surface of the orbicularis oris at the mucocutaneous junction of the vermilion.
- The repair is performed with permanent suture with buried knots. Braided or monofilament permanent suture, usually 3.0 or 4.0 size, is suitable.
- Muscle strips should be drawn taut, with an intraoperative lip position that of a casual smile, essentially overcorrected from baseline symmetric commissure position.
- The length of the muscle strips should not be augmented with temporoparietal fascia or other material, as it may fibrose and prevent direct transmission of muscle force to the orbicularis oris and commissure. If necessary, the lip or midface subunit should be mobilized to reach the graft by superficial and/or subperiosteal dissection.
- The temporoparietal fascia is then mobilized into the defect.
- If autologous fat or dermal allografts are used, longer-term antibiotic coverage may be beneficial, though I personally do not augment with allograft.
- The grafts should be sutured into position.
- A short-term drain, cool compresses, and analgesics are helpful in wound healing and pain management.
- After a period of early postoperative healing, physical therapy for muscular training through mimed mastication and other exercises can be instituted for training and restoration of symmetry.
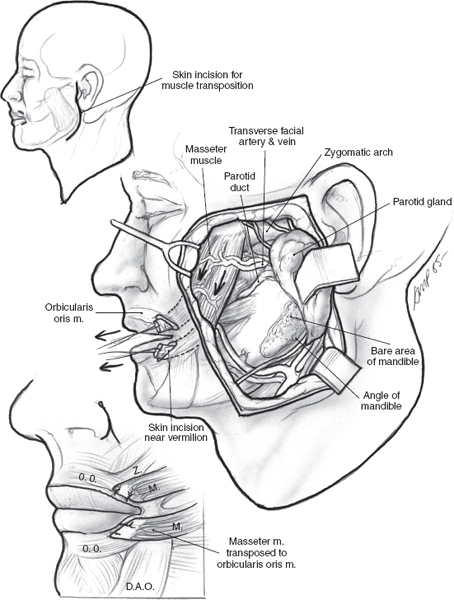
FIGURE 16-6 The masseter muscle is mobilized, divided into two branches, and sutured to the surface of the orbicularis oris to achieve definitive dynamic support. Overcorrection is recommended.
Neuromuscular Grafts
Indication
Reconstruction of defects with intact nerve but absent muscle (e. g., cases of midface or lip tumors with resection of the facial mimetic muscle as the end functional unit) are addressed through application of neuromuscular grafts. Serratus, gracilis, and pectoralis minor muscles are all suitable donor sites.
Technique
Basic technique includes transplantation of muscle and nerve with engraftment to native facial nerve branch stump. Options encompass the serratus for multiple strips or the gracilis for single strip of muscle. Serratus can be harvested with low donor site morbidity with a pedicle based on a branch of the thoracodorsal artery and vein. The long thoracic nerve is parallel to the pedicle. The proximal first through approximately fourth strips of the serratus must be preserved to prevent winging of the scapula. The gracilis vascular supply is a branch of the profunda femoris artery and vein and occasionally from the medial circumflex femoris vessels. The motor nerve supply is the anterior branch of the obturator nerve. The pectoralis minor was pioneered by Terzis and colleagues to rehabilitate facial mimetic and periorbital muscles.29,30 Its harvest results in minimal donor site morbidity. The vascular supply is variable, stemming from the lateral thoracic artery (predominant presentation), direct branch of the axillary artery, or pectoral branch of the thoracoacromial artery. Venous drainage is similarly variable and includes venae comitantes and a direct vein. Innervation is predominantly provided by a major branch of the median pectoral nerve. This graft has a short pedicle. All of these muscles are revascularized, based on their pedicles, to the superficial temporal, facial, or adjacent arterial and venous vessels. Use of these reconstructive options in rehabilitation of facial paralysis requires skill in microvascular techniques.
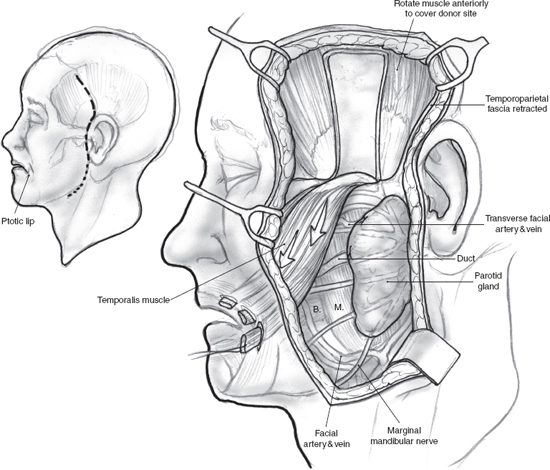
FIGURE 16-7 The middle third of the temporalis muscle is mobilized, divided into three branches, and sutured to the surface of the orbicularis oris to achieve definitive dynamic support. Overcorrection is recommended.
 Complex Challenge: Functionally Incomplete Innervation
Complex Challenge: Functionally Incomplete Innervation
The challenge of incomplete facial nerve or graft function is clinically difficult to address. Electrophysiologically, the involved facial muscle has an intact nerve motor unit but is incompletely innervated or is an injured muscle. Hence, function will be compromised. Possible solutions include contralateral partial chemical denervation with botulinum toxin (Botox) or contralateral nerve transection or division of the partially functional nerve graft with reanastomosis. The option of augmenting midface position with a dynamic temporalis muscle sling or facial static suspensions or lip/lid shortening procedures is a useful adjunct. The solution that is not an option is a nerve muscle pedicle from the nonfacial nerve muscle donors. This neurotization procedure cannot be performed if muscle is even partially innervated.
Adjunctive Aesthetic Procedures
Most broadly, rehabilitation of functional and aesthetic signs and symptoms associated with affected branches of the facial nerve are addressed through an array of reconstructive surgical options. Table 16-3 outlines facial nerve branch deficits, associated signs and symptoms, and options for surgical nerve, functional, and aesthetic repair and reconstruction within the rehabilitation plan. Repair of Stensen’s duct is often necessary, given its anatomical proximity to the facial nerve, to complete reconstruction in cases of trauma.
Targeted aesthetic procedures offer reconstructive refinements to address specific needs of individual patients. Brow ptosis is addressed through brow lifts, either endoscopic or direct midforehead (Fig. 16–8). Techniques from aesthetic surgery should be adopted, within the surgeon’s skill base, and targeted to meet the patient’s needs. In the case of nasal valve collapse, alar battens offer an effective reconstructive option. Lagophthalmos can be addressed with gold weight placement and canthoplasty levator release. Ectropion repair may be achieved with canthoplasty and wedge resection or classic tarsorrhaphy to fuse the eyelid closed for temporary or permanent protection of the cornea/eye. Canthoplasty is important to augment the treatment of the paralyzed eye. Temporal and zygomatic grafts often are difficult to perform for advanced skin cancers invading into the parotid in this area. If this is the case, the lateral canthoplasty will correct the ectropion. Patients and physicians should be aware that the effects of gravity and aging will usually lead to further laxity and may require intermittent revisions of this procedure. In aesthetic treatment of ocular signs of facial paralysis, eye care, including emollient ointment and artificial tears, is necessary to ensure eye health.
Gold Weights
Indication
Lagophthalmos, inadequate eyelid closure, with resultant risk for exposure keratitis
Procedure
Placement of a gold, or alternatively titanium, weight into the eyelid allows passive closure of the eyelid to correct the Bell’s phenomenon. The effect of concurrent canthoplasty or brow lift should be considered if this procedure is undertaken. The weight should then be test fitted with the new brow and lid position. The eyelid canthal spring is not addressed here, as it requires significant surgical experience to minimize incidence of complications such as device extrusion.
Technique
- The gold, or titanium, weight should be test fitted on the eyelid prior to starting the procedure.
a. With the patient sitting up, the weight should be held to the lid with a small portion of Steri-Strip or cutaneous adhesive, such as benzoin or mastisol. The weight may amplify or cause ptosis, triggering assessment of the lid–limbus relationship. The patient’s perception of field of view should also be elicited.
i. The most commonly used weights are those of 0.8 to 1.0 g.
ii. The patient can be assessed prior to surgery in the office; many companies offer trial weights for office use.
- Local anesthesia with or without sedation provides adequate anesthesia for placement.
- The incision is placed in the supratarsal crease, ~ 1.5 cm or adequate length to allow satisfactory dissection and exposure of the tarsus.
- Dissection extends, in the submuscular plane, to the tarsal plate.
- The gold or titanium weight is placed in the dissected pocket.
- The weight is secured to the tarsus with permanent monofilament, ~ 5.0, preferably clear suture.
- The orbicularis is coapted with a fine Vicryl 5.0 suture.
- Cutaneous closure is achieved with absorbable or monofilament.
- Cool compresses can be provided for the first 24 hours postoperatively to minimize swelling and pain.
Table 16-3 Branch Facial Nerve Deficits, Associated Signs and Symptoms, and Rehabilitative Options
| Branch deficit | Sign/symptom | Procedures |
|---|---|---|
| Upper frontal | Brow ptosis | Brow lifts Endoscopic Direct midforehead |
| Upper zygomatic | Lagophthalmus Ectropion Corneal and conjunctival exposure |
Gold weight Levator procedures Canthoplasty Lateral Medial Tarsorrhaphy Tarsal strip |
| Zygomatic/buccal | Malar flattening Loss of nasolabial fold Nasal obstruction alar displacement Nasal twisting Lip commissure ptosis |
Fascial suspension Midface and SOOF lifts Microvascular neuromuscular grafts Nasal valve spreader “batten” grafts |
| Lower buccal/marginal | Oral commissure incompetence Lower lip ptosis Dysarthria Drooling |
Fascial slings Lip excisions |
SOOF, suborbicular oculi fat
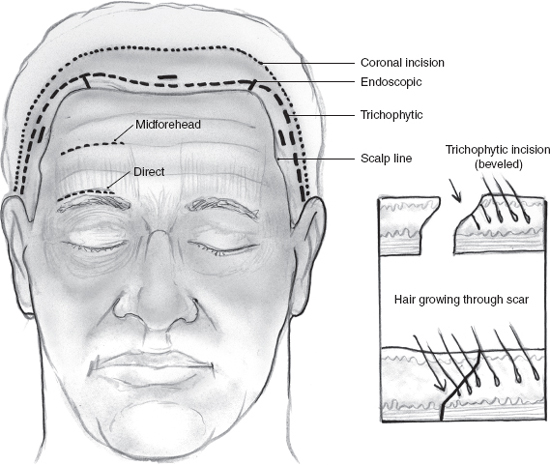
FIGURE 16-8 The multiple approaches and incisions for brow and forehead lifts are delineated in schematization.
Static slings offer the option of fine-tuned static repositioning of the face. The slings are positioned to elevate the oral commissure, re-create a nasolabial fold, and lateralize and lift the nasal alar base. Fascia lata, Alloderm (Life Cell Corporation, Branchburg, NJ), suture material, Gortex (W. L. Gore and Associates, Flagstaff, AZ), and barbed sutures or sutures with studded alloplastic pledgets have been used. The material for the sling most often used is the autologous fascia lata. The fascia lata can be harvested from the lateral thigh under local anesthetic if the procedure is being done under local anesthetic with sedation. The fascia lata can be retrieved with a fascia lata stripper, which is often available in most operating rooms. If not, two or more small incisions on the lateral thigh can be used to harvest the fascia. The fascia is classically suspended to the zygomatic arch or temporalis fascia. Alternatively, it can be attached to the infraorbital rim periosteum or the bone itself for a more secure position. Permanent sutures may be used.
Face lift and suspension techniques may be borrowed from aesthetic surgery as part of the rehabilitation planning for facial nerve paralysis. For example, the subperiosteal face lift can be used to augment midface rehabilitation through suspension techniques to address the nasolabial fold and oral commissure with simultaneous midface subperiosteal lifting.
Lip resections target signs and symptoms of facial paralysis stemming from anesthetic, adynamic tissue in the oral complex. Resection of the lower lip can help restore position and decrease the amount of anesthetic and adynamic tissue in the lower face. Fascial strips or allografts can also be used to create a cerclage that will tighten and can also be used to suspend the lower lip. This procedure can combat lip ptosis and oral incompetence with drooling. The typical lip wedge resection techniques with multilayer muscular mucosal and cutaneous repair are performed to shorten the adynamic portion of the lip. This resection should be performed as laterally as possible to avoid injuring any partially innervated muscle from the contralateral orbicularis innervation. The cerclage with autograft fascial strip or allograft (i. e., Alloderm or donor fascia or pericardium) is placed in a plane between the orbicularis oris and skin. This technique is not one that is often required, unless there is bilateral injury. It can be traumatic to the neurologically intact side and therefore is of limited benefit.
Stensen’s Duct Repairp
Indication
Traumatic injury to the duct generally necessitates simple or complex repair.
Procedure
Repair of the duct is achieved with stenting and repair of adjacent injury to the gland.
Technique
- The distal segment edge of Stensen’s duct can be identified by retrograde cannulation through the ampulla intraorally. Loop magnification and lacrimal duct probes can be utilized. An imaginary line drawn through the tragus and the mid-upper lip defines the duct trajectory in the buccal subunit and thereby aids in its identification.31
- If suitable stents are not available, angiocatheters, large-caliber suture, or other small-caliber silicone tubes are a reasonable and appropriate substitute to aid in identification of the duct.
- The proximal portion of the duct can be more difficult to identify, and use of magnification will be helpful. Milking the gland can often lead to expression of saliva from the cut end of the proximal duct, making identification possible.
- In the case of a double transection, the intervening segment may be anatomically identifiable. Apposition of the adjacent parotid tissue by traction of the parotid fascia will aid in anatomical identification. If an intervening segment of the gland is absent, a small-caliber vein graft can be used as a stent.
- Anastomosis of the duct is achieved most precisely with magnification. Use of monofilament microsutures, ranging in size from 7.0 to 9.0, is the most common technique.
- Suitable stents include silicone catheters, small-gauge angiocatheters, and lacrimal stents. The stent is brought out of the ampulla and secured to the adjacent buccal mucosa with a suture. The stent can be left in situ for 10 to 14 days and is most important in cases of significant trauma to the duct.31
- Tension on the repair is minimized by coapting adjacent parotid tissue. This strategy is also important in adjacent neurorrhaphy of the buccal branches, which are most often injured concurrently.
- Antibiotic coverage should cover the gram-positive flora and other indicated flora based on the type of injury and the extent of oral contamination.
- Postoperative management should include and encourage hydration, possible massage, and pain management.
- For the patients who have inadequate structural duct remnants to repair, there are two options. One is to marsupialize the remnant of the duct that is attached to the body of the gland to the buccal mucosa, if possible; the other is to tie off the duct and allow the gland to undergo fibrosis and atrophy.
REFERENCES
1. Rubin, LR. The anatomy of a smile: its importance in the treatment of facial paralysis. Plast Reconstr Surg 1974; 53 (4): 384–387
2. Roob, G Fazekas, F Hartung, HP. Peripheral facial palsy: etiology, diagnosis and treatment. Eur Neurol 1999; 41 (1): 3–9
3. Conley, JJ. Facial nerve engrafting. Arch Otolaryngol 1961; 73: 322–327
4. McGuirt, W McCabe, BF. Effects of radiation therapy on facial nerve cable grafts. Laryngoscope 1977; 87: 415–428
5. Fisch, U. Cross-face grafting in facial paralysis. Arch Otolaryngol 1976; 102 (8): 453–457
6. House, JW Brackmann, DE. Facial nerve grading system. Otolaryngol Head Neck Surg 1985; 93 (2): 146–147
7. Yetiser, S Kazkayas, M Altinok, D Karadeniz, Y. Magnetic resonance imaging of the intratemporal facial nerve in idiopathic peripheral facial palsy. Clin Imaging 2003; 27 (2): 77–81
8. Selesnick, SH Burt, BM. Regional spread of nonneurogenic tumors to the skull base via the facial nerve. Otol Neurotol 2003; 24 (2): 326–333
9. Caldemeyer, KS Mathews, VP Righi, PD Smith, RR. Imaging features and clinical significance of perineural spread or extension of head and neck tumors. Radiographics 1998; 18 (1): 97–110; quiz 147
10. Gulya, AJ Scher, R Schwartz, A Wilson, WR. Facial and trigeminal neural dysfunction by a primary cutaneous squamous cell carcinoma: MRI and clinicopathologic correlates. Am J Otol 1992; 13 (6): 587–590
11. Sittel, C Stennert, E. Prognostic value of electromyography in acute peripheral facial nerve palsy [comment]. Otol Neurotol 2001; 22 (1): 100–104
12. Aimoni, C Lombardi, L Gastaldo, E Stacchini, M Pastore, A. Preoperative and postoperative electroneurographic facial nerve monitoring in patients with parotid tumors. Arch Otolaryngol Head Neck Surg 2003; 129 (9): 940–943
13. Highet, E Saunders, FK. The effects of stretching nerves after suture. Br J Surg 1943; 30: 355–369
14. Denny-Brown, D Doherty, MM. Effects of transient stretching of peripheral nerve. Arch Neurol Psychiatr 1945; 54: 116–129
15. Salvi, V. Problems connected with the repair of nerve sections. Hand 1973; 5 (1): 26–32
16. Terzis, J Faibisoff, B Willaims, HB. The nerve gap: suture under tension vs graft. Plast Reconstr Surg 1975; 56: 166–170
17. Gacek, RR Radpour, S. Fiber orientation of the facial nerve: an experimental study in the cat. Laryngoscope 1982; 92 (5): 547–556
18. Radpour, S. Organization of the facial nerve nucleus in the cat. Laryngoscope 1977; 87 (4, Pt 1): 557–574
19. Radpour, S Gacek, RR. Facial nerve nucleus in the cat: further study. Laryngoscope 1980; 90 (4): 685–692
20. Radpour, S Gacek, RR. Anatomic organization of the cat facial nerve. Otolaryngol Head Neck Surg 1985; 93 (5): 591–596
21. Radpour, S Gacek, RR. Facial nerve fiber orientation, linkage between central nervous organization and muscular function. Eur Arch Otorhinolaryngol 1994; Dec: S10–S15
22. Kakibuchi, M Tuji, K Fukuda, K, et al. End-to-side nerve graft for facial nerve reconstruction. Ann Plast Surg 2004; 53 (5): 496–500
23. Terris, DJ Fee, WE. Current issues in nerve repair. Arch Otolaryngol Head Neck Surg 1993; 119: 725–731
24. Kalantarian, B Rice, DC Tiangco, DA Terzis, JK. Gains and losses of the XII–VII component of the “baby-sitter” procedure: a morphometric analysis. J Reconstr Microsurg 1998; 14 (7): 459–471
25. Atlas, MD Lowinger, DS. A new technique for hypoglossal-facial nerve repair. Laryngoscope 1997; 107 (7): 984–991
26. Cusimano, MD Sekhar, L. Partial hypoglossal to facial nerve anastomosis for reinnervation of the paralyzed face in patients with lower cranial nerve palsies: technical note. Neurosurgery 1994; 35 (3): 532–534
27. May, M Sobol, SM Mester, SJ. Hypoglossal-facial nerve interpositional- jump graft for facial reanimation without tongue atrophy. Otolaryngol Head Neck Surg 1991; 104: 818–825
28. Yoleri, L Songur, E Yoleri, O Vural, T Cagdas, A. Reanimation of early facial paralysis with hypoglossal/facial end-to-side neurography: a new approach. J Reconstr Microsurg 2000; 16 (5): 347–355, discussion 355, 356
29. Terzis, JK. Pectoralis minor: a unique muscle for correction of facial palsy [comment]. Plast Reconstr Surg 1989; 83 (5): 767–776
30. Terzis, JK Noah, ME. Analysis of 100 cases of free-muscle transplantation for facial paralysis. Plast Reconstr Surg 1997; 99 (7): 1905–1921
31. Steinberg, MJ Herrera, AF. Management of parotid duct injuries. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2005; 99 (2): 136–141
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


