http://evolve.elsevier.com/Haveles/pharmacology
Epilepsy
Epilepsy comprises a group of disorders that involve a chronic stereotyped recurrent attack of involuntary behavior or experience or changes in neurologic function caused by electrical activity in the brain that can be recorded via an electroencephalography (EEG). This activity can be localized or generalized. Each episode is termed a seizure. The seizure may be accompanied by motor activity, such as convulsions, or by other neurologic changes (e.g., sensory or emotional).
Because seizure disorders are estimated to affect approximately 1% of the population, the dental team is likely to encounter a patient with epilepsy. The antiepileptic agents are used long term, so potential adverse reactions that might alter dental treatment must be considered.
There are many etiologies for epilepsy, including infection, trauma, toxicity to exogenous agents, genetic or birth influences, circulatory disturbances, metabolic or nutritional alterations, neoplasms, hereditary factors, fevers, and degenerative diseases. The majority of epileptic patients have idiopathic epilepsy; this term is used when the cause is unknown.
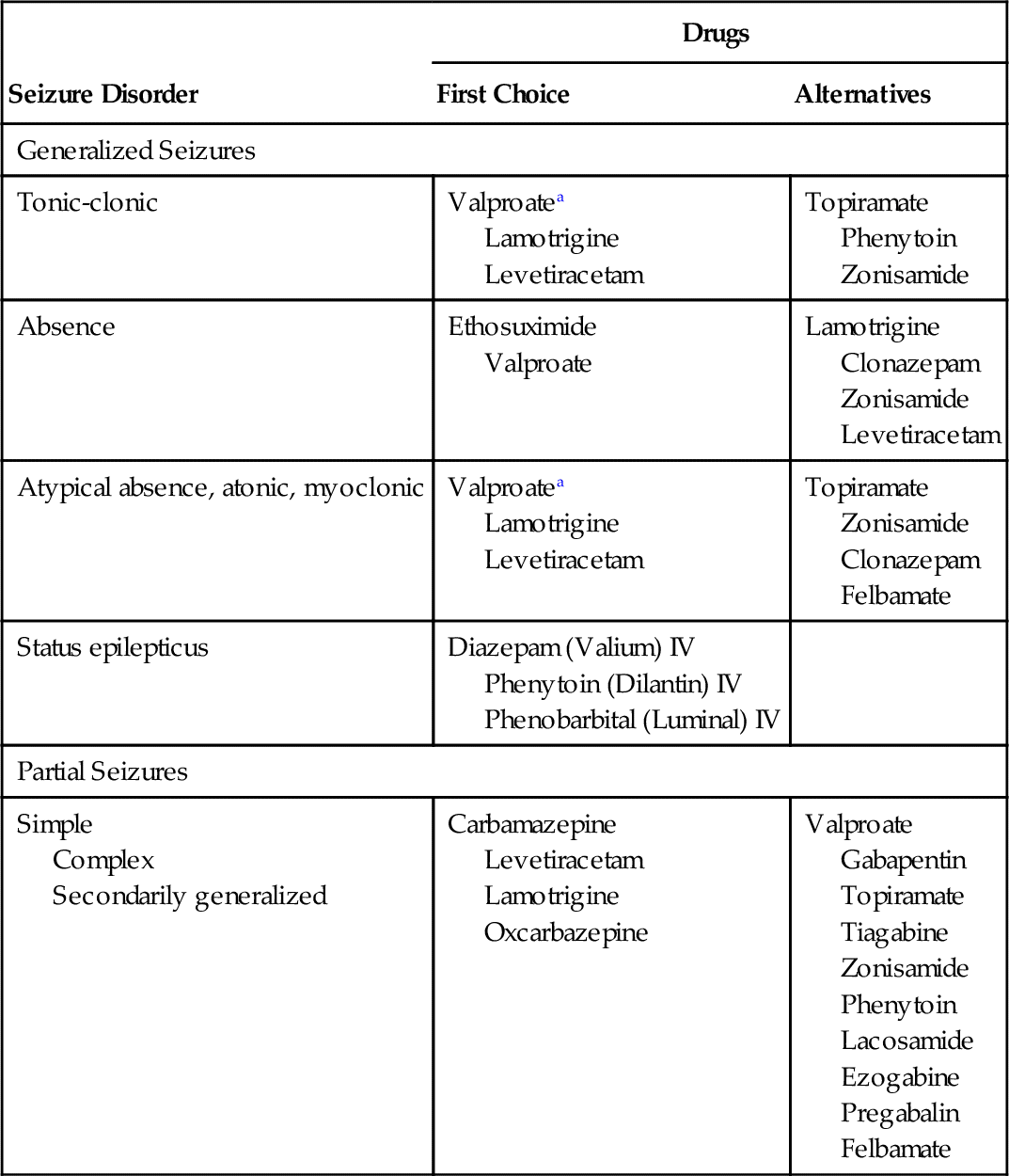
Epilepsy has been classified based on causes, symptoms, duration, precipitating factors, postictal state (postseizure), and aura. Currently, The International Classification of Epileptic Seizures divides seizures into two major groups and a miscellaneous group (Table 16-1). The two major groups are partial seizures and generalized seizures. Partial seizures are divided into simple and complex attacks. The most common generalized seizures are (1) tonic-clonic and (2) absence.
Generalized Seizures
Generalized seizures are divided into two large groups: (1) absence and (2) tonic-clonic types. Consciousness is lost in both types. Little movement occurs in absence seizures, but in tonic-clonic seizures, major movement of large muscle groups occurs. Often, the patient may experience an aura (a brief period of heightened sensory activity) before the onset of the seizure. It may be characterized by numbness, nausea, or unusual sensitivity to light, odor, or sound.
Absence Seizures
The symptoms of absence seizures include a brief (few seconds) loss of consciousness with characteristic EEG waves and little movement. Absence seizures usually begin during childhood and disappear in middle age. The patient is usually unaware that these seizures are occurring, and body tone is not lost. There is no aura or postictal state, and the patient quickly returns to normal activity. The drug of choice in the treatment of typical absence seizures is ethosuximide.
Management of absence seizures poses no problems for the dental team. The team’s main concern when treating patients with absence seizures is the adverse reactions that can occur from long-term administration of the drugs used to treat the disease.
Tonic-Clonic Seizures
The generalized tonic-clonic seizures include longer periods of loss of consciousness and major motor activity of the large muscles of the body. As the seizure begins, the body becomes rigid and the patient falls to the floor. Urination, apnea, and a cry may be present. Tonic rigidity is followed by clonic jerking of the face, limbs, and body. The patient may bite the cheek or tongue. Finally, the patient becomes limp and comatose. Consciousness returns gradually, with postictal confusion, headache, and drowsiness. Some patients experience prodromal periods of varying durations, but a true aura does not occur. Because this seizure type involves the violent movement of major muscle groups, it is more likely to result in serious injury to the patient. Valproate, lamotrigine, and levetiracetam are considered the drugs of choice for treating generalized tonic-clonic seizures (Table 16-2).
Status Epilepticus
Status epilepticus seizures are continuous tonic-clonic seizures that last longer than 30 minutes or recur before the end of the postictal period of the previous seizure. This is an emergency situation, and rapid therapy is required, especially if the seizure activity has produced hypoxia. Parenteral benzodiazepines, such as diazepam (Valium), are the drugs of choice to control this seizure type (see Chapter 11).
Partial (Focal) Seizures
Partial seizures involve activation of only part of the brain, and the location of the activity determines the clinical manifestation. When consciousness is not impaired, the attack is called an simple partial attack. When consciousness is impaired, the attack is termed a complex partial attack. In contrast to absence seizures, which last a few seconds, complex partial seizures last several minutes. Some patients with complex partial seizures have an aura, and full consciousness is slow to return. For the partial seizures, lamotrigine, carbamazepine, levetiracetam and oxcarbazepine are used (see Table 16-2).
Drug therapy of patients with epilepsy
Drug therapy of the patient with epilepsy has variable efficacy, from complete control of all seizures to reduction in the frequency of seizures. Antiepileptic agents may be used singly or in combination. The goal is to control seizures and minimize potential adverse reactions. Some newer antiepileptics are able to treat previously untreatable seizures, but more serious side effects can accompany them. General principles on the management of the dental patient taking any antiepileptic agents are listed in Box 16-1.
The antiepileptic drug used to treat a specific patient depends on the type of seizures the patient has. Because these agents are usually taken for life, their long-term toxicity becomes an important consideration in choosing a particular antiepileptic agent and determining the drug’s dental implications.
General Adverse Reactions of Antiepileptic Agents
Central Nervous System Depression
Depressed central nervous system (CNS) function is a common side effect of the antiepileptic agents. Tolerance often develops to these sedative effects while the antiepileptic effect persists. Impairment of learning and cognitive abilities occurs in some patients. Behavior alterations reported include both hyperactivity and sedation. Another CNS side effect is exacerbation of a seizure type that is not being treated. This CNS depression is additive with other CNS depressants such as the opioids. If another CNS drug is given to the patient, the dose should be reduced.
Gastrointestinal Distress
Gastrointestinal distress, including anorexia, nausea, and vomiting, can occur with most antiepileptics. These effects can be minimized by taking the drugs with food. Agents with adverse reactions related to the gastrointestinal tract, for example, nonsteroidal antiinflammatory drugs (NSAIDs) or opioids, should be prescribed cautiously.
Drug Interactions
Many drug interactions can occur with the antiepileptics. They may interact with themselves, with each other, or with other drugs. The mechanisms of drug interactions include altering absorption or renal excretion and inducing or inhibiting metabolism. The outcome may alter the levels of the inducing drug itself, another concomitant antiepileptic, or some other drug that is extensively metabolized by the liver microsomal enzymes.
The most important drug interaction of the antiepileptics involves stimulation of the hepatic microsomal enzymes. Inducing these enzymes results in a reduction in the blood level of the affected drugs (those metabolized by the liver enzymes). Figure 2- 11A shows the normal, unaffected enzyme situation. When the enzymes are stimulated (Figure 2- 11B), the level of the affected drug (D) is reduced because it is being metabolized more quickly, producing its metabolite (Dm).
Drug interactions with the older antiepileptics, such as phenytoin, carbamazepine, and valproate, are more significant than those with other drug groups because of these agents’ narrow therapeutic indexes. If the level of an older antiepileptic drug is altered sufficiently by a drug interaction, either toxicity (level too high) or loss of seizure control (level too low) can result. Before any changes or additions are made to a patient’s therapy, the possibility of drug interactions should be considered.
Idiosyncratic reactions
A wide range of idiosyncratic reactions occurs with the antiepileptics. Dermatologic side effects include rash, Stevens-Johnson syndrome, exfoliative dermatitis, and erythema multiforme. Drug-induced systemic lupus erythematosus and hematologic effects have also been reported with most of these agents.
Teratogenicity/growth
Reports have associated the antiepileptic agents with alteration in growth, profound effects being seen on fetal development and in children receiving antiepileptic medications during growth and development. The teratogenic potential of the antiepileptics has been documented. Several have been implicated in the production of fetal anomalies. However, antiseizure therapy may be necessary. In some instances the mother’s seizures may be more damaging to fetal development than the drug itself. In this case, the risk-to-benefit benefit ratio must be considered.
Withdrawal
Abrupt withdrawal of any antiepileptic medication can precipitate seizures. Although many patients require medication for life, certain seizure types tend to disappear as patients grow older. In these patients, gradual withdrawal of their seizure medications under controlled conditions can be undertaken after an appropriate interval of drug use.
Valproate
A group of antiepileptic agents that are not structurally related to any other antiepileptics are the valproates, which include valproic (val-PRO-ik) acid, valproate (val-PRO-ate) sodium, and divalproex (dye-VAL-pro-ex) sodium. The term valproate is used here to refer to all of these agents. Divalproex sodium is a 1:1 ratio of valproic acid and valproate sodium. The mechanism of action of valproate may be its effect on sodium or potassium channels, a reduction in aspartate levels, or an increase in the inhibitory neurotransmitter γ-aminobutyric acid (GABA). Clinical trials have shown that it is effective and usually well tolerated in patients being treated for myoclonic and atonic seizures. It is often used first in treating patients with generalized tonic-clonic seizures on the basis of results presented in the medical literature, although this use is not approved by the FDA.
Other uses
Valproate is FDA-approved for migraine prophylaxis and bipolar disorder.
Adverse Reactions
Gastrointestinal effects
Indigestion, nausea, and vomiting are the most frequent adverse effects associated with valproate. They can be minimized by giving the drug with meals or increasing the dose very gradually. Divalproex sodium may have fewer adverse gastrointestinal effects than its components. Other gastrointestinal side effects are hypersalivation, anorexia, increased appetite, cramping, diarrhea, and constipation.
Central nervous system effects
Sedation and drowsiness have been reported with valproate. Rarely, ataxia, headache, and nystagmus have been noted. Some children exhibit hyperactivity, aggression, and other behavioral disturbances. Weight gain and an increase in appetite have been reported.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses