http://evolve.elsevier.com/Haveles/pharmacology
The term adrenocorticosteroids (a-dree-noe-KOR-ti-KO-ster-oids) (adrenal corticosteroids, adrenocorticoids, corticosteroids, or steroids) refers to a group of agents secreted by the adrenal cortex. The dental team should be aware of the effects, adverse reactions, and dental implications of these agents for at least the following reasons:
Mechanism of release
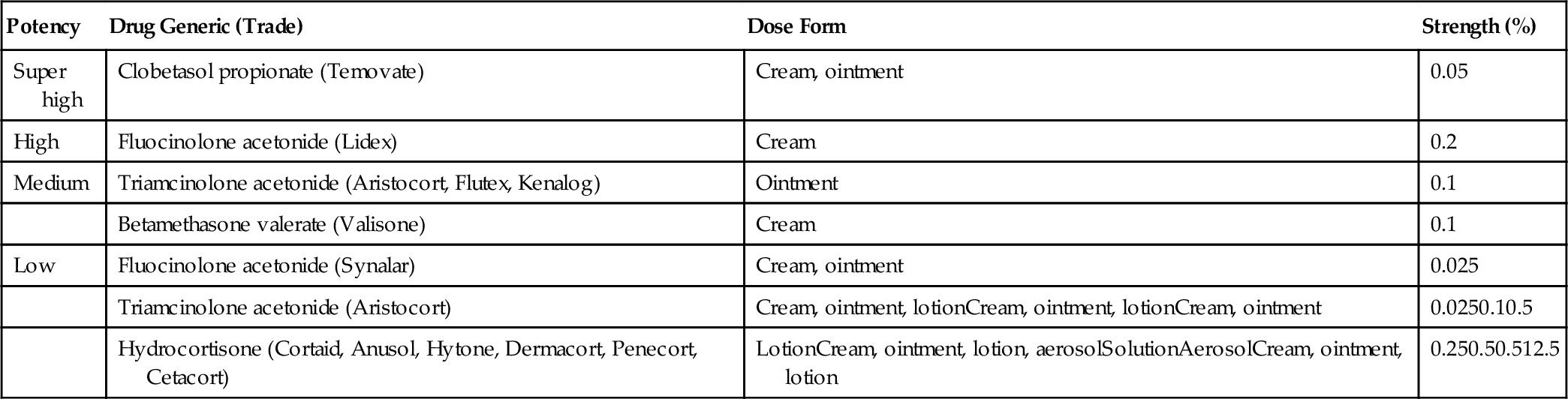
The adrenocorticosteroids are naturally occurring compounds secreted by the adrenal cortex. Their release is triggered by a series of events (Figure 18-1). First, a stimulus such as stress (1) causes the hypothalamus (2) to release corticotropin-releasing hormone (CRH) (3), which acts on the pituitary gland (4). Under the influence of CRH, the pituitary gland secretes adrenocorticotropic hormone (ACTH) (5), which stimulates the adrenal cortex (6) to release hydrocortisone (7). Hydrocortisone then acts on both the pituitary (8) and the hypothalamus (9) to inhibit the release of CRH and ACTH, respectively. This mechanism is called negative feedback. Exogenous steroids act in the same way as hydrocortisone (10); that is, they inhibit the release of CRH and ACTH. With long-term administration of steroids, ACTH release is suppressed and the adrenal gland atrophies. If the administration of the exogenous steroid is then abruptly stopped, a relative steroid deficiency results. This can cause severe problems, including adrenal crisis.
Classification
The adrenocorticosteroids can be divided into two major groups: the glucocorticoids, which affect intermediate carbohydrate metabolism, and the mineralocorticoids, which affect the water and electrolyte composition of the body. The major glucocorticoid present in the body is cortisol (hydrocortisone). Without stress, the normal adult secretes about 20 mg of hydrocortisone daily. A tenfold increase can occur with stress. Maximal secretion occurs between 4 am and 8 am in people with a normal schedule. The chemical structures of the synthetic agents and the naturally occurring adrenocorticosteroids, such as hydrocortisone, are similar. Many chemical modifications have been made in an attempt to produce synthetic glucocorticoids with fewer adverse reactions and more specific activity.
Although the term adrenocorticosteroids refers to those steroids secreted by the adrenal cortex and includes both the glucocorticoids and the mineralocorticoids, this chapter discusses primarily the action of glucocorticoids because of their more common use.
Definitions
The following terms are used in this chapter:
Routes of administration
Glucocorticoids are available in a wide variety of dose forms. They are routinely used topically, orally, intramuscularly, and intravenously. Systemic effects are commonly obtained when the drug is administered orally or parenterally, but topical administration may rarely cause systemic effects. If a large quantity of a steroid is applied topically, especially if the skin is denuded or an occlusive dressing such as plastic wrap is applied, systemic effects can occur. Table 18-1 shows the relative potencies of selected topical corticosteroid products.
Mechanism of action
The mechanism of action of the steroids involves binding to a specific receptor and forming a steroid-receptor complex. The complex then translocates into the nucleus and alters gene expression (turns genes on or off), resulting in the regulation of many cellular processes. Because of the mechanism, a lag time exists in the action of the steroids, and the relationship between their effects and blood levels is poor. Other effects of glucocorticoids are mediated by catecholamines producing vasodilation or bronchodilation.
The antiinflammatory action of glucocorticoids results from their profound effects on the number, distribution, and function of peripheral leukocytes and from their inhibition of phospholipase A. The use of steroids results in an increase in the concentration of neutrophils and a decrease in the lymphocytes (T and B cells), monocytes, eosinophils, and basophils. Steroids induce the synthesis of a protein that inhibits phospholipase A, decreasing the production of both prostaglandins and leukotrienes from arachidonic acid.
These agents, responsible for the delayed phase of acute inflammation, act synergistically. Steroids also inhibit interleukin-2, migration inhibition factor, and macrophage inhibition factor.
Pharmacologic effects
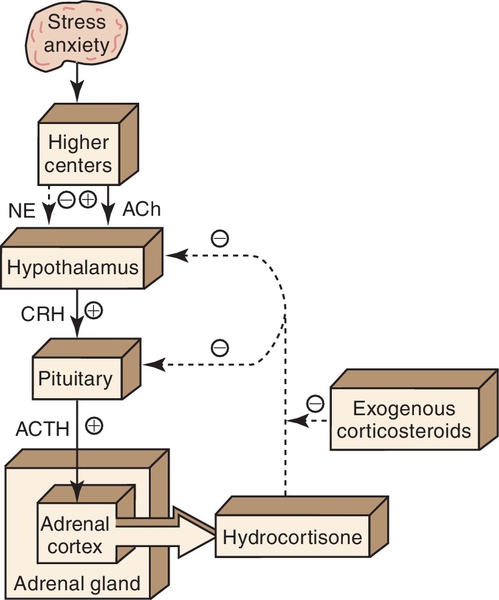
The pharmacologic effects and the adverse reactions of corticosteroids are closely related (Figure 18-2). The effects for which they are used include their antiinflammatory action and suppression of allergic reactions. They also suppress the immune response. Corticosteroids are palliative rather than curative. The glucocorticoid effects and the mineralocorticoid effects are listed in Box 18-1. Many of these effects produce adverse reactions and are discussed in the following section.
Adverse reactions
The adverse reactions of corticosteroids are proportional to the dose, frequency and time of administration, and duration of treatment. With prolonged therapy and sufficiently high doses, the following side effects occur.
Metabolic Changes
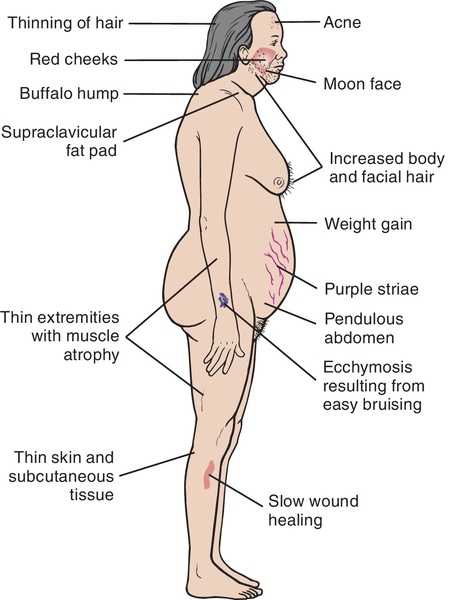
Moon face (round), buffalo hump (fat deposited on back of the neck), truncal obesity, weight gain, and muscle wasting give patients the Cushing’s syndrome appearance (Figure 18-3). Hyperglycemia (diabetes-like) may be aggravated or initiated, especially in prediabetic patients. More antidiabetic medication may be required.
Infections
Corticosteroids decrease resistance to infection. Because of their antiinflammatory action, they may also mask its symptoms. Patients taking long-term glucocorticoid therapy are given isoniazid, an antituberculosis agent, to prevent tuberculosis.
Central Nervous System Effects
Changes in behavior and personality, including euphoria (with increasing dose), agitation, psychoses, and depression (with decreasing dose), can occur.
Peptic Ulcer
Because corticosteroids stimulate an increase in production of stomach acid and pepsin, they may exacerbate peptic ulcers. Healing is impaired, and the ulcer may perforate.
Impaired Wound Healing and Osteoporosis
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses