Aging and the Periodontium
The evidence base is not without problems, many of which make it difficult to draw conclusions about the effects of aging. Some of these problems include inconsistency with regard to the definition of a true “older” group, the inadequate exclusion of adults with systemic diseases that can modify study findings, and attempts to extrapolate results from animal research. For the purposes of this chapter, the effects of aging will be limited to a narrow review of possible biologic and microbiologic changes. (For further reading about the effects of aging on the dental and periodontal patient, the reader should consult Lamster and Northridge’s Improving Oral Health for the Elderly. An Interdisciplinary Approach [Springer, 2008, New York].) The reader should be fully aware that this excludes many important age-associated phenomena, including reductions in an individual’s cognitive or motor function skills, which may have a direct impact on periodontal management. These will be discussed more fully in Chapter 39.
Since first writing this chapter 14 years ago, the volume of new research into aging and the periodontium has not increased substantially. Highly sensitive molecular and genomic techniques have been employed in related laboratory research, and these are starting to provide insight into age-associated effects. These approaches will likely be proven fruitful in the future. Despite the limited data directly related to the periodontium, much effort and many resources have been employed to research questions at least partially related to this topic. These include the effect of periodontal infection on general health (see Chapter 28) and the impact of osteoporosis on periodontal status (see Chapter 38).
Effects of Aging on the Periodontium
Gingival Epithelium
Thinning and decreased keratinization of the gingival epithelium have been reported with age.46 The significance of these findings could mean an increase in epithelial permeability to bacterial antigens, a decreased resistance to functional trauma, or both. If so, such changes may influence long-term periodontal outcomes. However, other studies have found no age-related differences in the gingival epithelium of humans or dogs.8,22 Other reported changes with aging include the flattening of rete pegs and altered cell density. Conflicting data regarding the surgical regeneration times for gingival epithelium have been ascribed to problems with research methodology.51
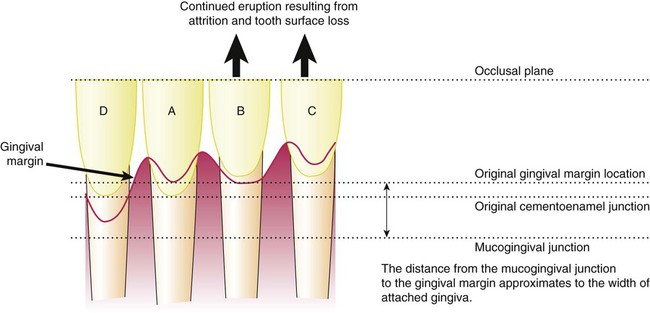
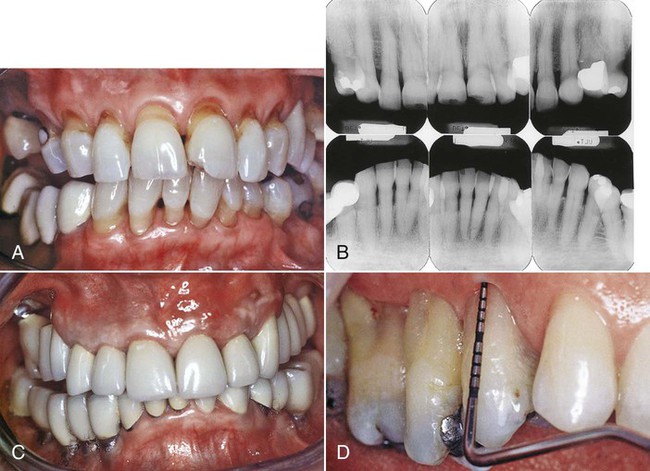
The effect of aging on the location of the junctional epithelium has been the subject of much speculation. Some reports show migration of the junctional epithelium from its position in healthy individuals (i.e., on the enamel) to a more apical position on the root surface with accompanying gingival recession.8 However, in other animal studies, no apical migration has been noted.25 With continuing gingival recession, the width of the attached gingiva would be expected to decrease with age, but the opposite appears to be true.2,3 Alternatively, the migration of the junctional epithelium to the root surface could be caused by the tooth erupting through the gingiva in an attempt to maintain occlusal contact with its opposing tooth (i.e., passive eruption) as a result of tooth surface loss from attrition (Fig. 2-1). The consensus is that gingival recession is not an inevitable physiologic process of aging but rather that it can be explained by the cumulative effects of inflammation or trauma on the periodontium6,8 (Fig. 2-2); this is discussed in more detail later in this chapter. Changes in the apoptotic gene expression of gingival tissue with age have recently been reported in nonhuman primates.17 This finding requires further research to understand its potential impact on gingival homeostasis and the pathogenesis of periodontal diseases.
Gingival Connective Tissue
Increasing age results in coarser and denser gingival connective tissues.54 Qualitative and quantitative changes to collagen have been reported. These include an increased rate of conversion of soluble to insoluble collagen, increased mechanical strength, and increased denaturing temperature. These results indicate increased collagen stabilization caused by changes in the macromolecular conformation.29 Not surprisingly, an increased collagen content has been found in the gingivae of older animals, despite a lower rate of collagen synthesis decreasing with age.8,29,51
Periodontal Ligament
Changes in the periodontal ligament that have been reported with aging include decreased numbers of fibroblasts and a more irregular structure, thus paralleling the changes seen in the gingival connective tissues.8,29,51 Other findings include decreased organic matrix production, epithelial cell rests, and increased amounts of elastic fiber.51 Conflicting results have been reported for changes in the width of the periodontal ligament in human and animal models. Although true variation may exist, this finding probably reflects the functional status of the teeth in the studies: the width of the space will decrease if the tooth is unopposed (i.e., hypofunction) or increase with excessive occlusal loading.29,51 Both scenarios can be anticipated as a result of tooth loss in this population. These effects may also explain the variability in studies that have reported qualitative changes within the periodontal ligament.
Recognition that the periodontal ligament has an important role in alveolar bone metabolism has led to an increased interest in investigating this role in relation to the maintenance of periodontal health and the pathogenesis of periodontitis. One approach to investigating this role is to examine mediators of bone homeostasis, such as receptor activator of nuclear factor-κB ligand (RANKL) and osteoprotegerin (OPG). RANKL is widely recognized for its role in activating osteoclasts, whereas OPG antagonizes RANKL binding, thus helping to maintain balance. The interplay of cytokines—and, in particular, the interleukin family—has also been extensively researched in periodontal pathogenesis. Some of the interleukins are potent mediators of inflammation (e.g., IL-1), whereas others have been shown to down-regulate this process (e.g., IL-4, IL-10). Therefore, evaluating the balance between these homeostatic processes would seem to be of great merit when evaluating possible negative changes in the periodontium. Such a strategy appears to be predictive of hard-tissue destruction among individuals with rheumatoid arthritis.16 Recent findings comparing periodontal ligament cells in older individuals (i.e., >60 years old) with those of younger individuals (i.e., 15 to 20 years old) have suggested greater gene expression for pro-inflammatory cytokines.7 This has been reported more widely as an age-associated change, although it is unclear whether it is a cause or an effect.38 However, in addition to increased IL-1 and IL-6 expression, OPG was also increased, which suggests that increased OPG may be a homeostatic response to the up-regulation of inflammation.7 If this homeostatic mechanism was effective, it may explain why an increase in inflammation did not result in greater tissue damage with age. Periodontal ligament cell proliferation was decreased with age, thereby suggesting an impairment of repair potential, although such an impact does not appear to be manifested clinically.53 Further research investigating periodontal ligament cells from individuals diagnosed with periodontitis will be important to understand the potentially important relationship between aging and periodontal ligament cells.
Cementum
Some consensus regarding the effect of aging on cementum exists. An increase in cemental width is a common finding; this increase may be 5 to 10 times with increasing age.8 This finding is not surprising, because deposition continues after tooth eruption. The increase in width is greater apically and lingually.51 Although cementum has limited capacity for remodeling, an accumulation of resorption bays explains the finding of increasing surface irregularity.18
Alveolar Bone
Reports of morphologic changes in alveolar bone mirror age-related changes in other bony sites. Specific to the periodontium are findings of a more irregular periodontal surface of bone and the less-regular insertion of collagen fibers.51 Although age is a risk factor for the bone mass reductions in individuals with osteoporosis, it is not causative and therefore should be distinguished from physiologic aging processes.21 Overriding the diverse observations of bony changes with age is the important finding that the healing rate of bone in extraction sockets appears to be unaffected by increasing age.4 Indeed, the success of osseointegrated dental implants, which relies on intact bone healing responses, does not appear to be age related.9 However, balancing/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


