http://evolve.elsevier.com/Haveles/pharmacology
Antiinfective agents play an important role in dentistry because infection, after pain management, is the dental problem for which drugs are most often prescribed. As the knowledge about the etiology of dental diseases is continually increasing and the involvement of microorganisms is becoming better understood, dental hygienists continue to better understand the proper place of antibiotics and their effect on microorganisms.
Dental infections can be divided into several types, as follows:
Dental infection “evolution”
Dental infections often follow similar pathways of evolution from beginning to end. In the beginning, the organisms responsible for a dental infection are primarily gram-positive cocci, such as Streptococcus viridans, or β-hemolytic streptococci. After a short time, the gram-positive infection begins to include a variety of both gram-positive and gram-negative anaerobic organisms, such as Peptostreptococcus (Peptococcus) and Bacteroides (Porphyromonas and Prevotella species). At this point, the infection is termed a mixed infection. Over time, the proportion of organisms that are anaerobic increases. With additional time and no treatment, the infection progresses until it consists of predominantly anaerobic flora. At this point, the anaerobic organisms coalesce into an abscess, often visible on radiograph (x-ray).
The choice of antibiotics for a dental patient’s infection depends on where it is in its evolution. If the infection is just beginning, the organisms most likely to be present are gram-positive cocci. Penicillin is the drug of choice, unless the patient has a penicillin allergy. Amoxicillin is most often used because it is less irritating to the stomach and can be taken with food or milk. In patients allergic to penicillin, alternatives include a macrolide antibiotic and clindamycin. When the infection is at the mixed stage, agents effective against both gram-positive organisms and anaerobic organisms may be successful. Treating gram-positive organisms is easier, and the drug of choice is penicillin/amoxicillin or, in the patient with a penicillin allergy, a macrolide antibiotic. For anaerobic organisms, metronidazole is effective. Eradicating one group of organisms, alters the balance between the two types of organisms, and the body can then resolve the infection. Clindamycin affects both gram-positive cocci and gram-positive and gram-negative anaerobes. To treat a dental infection, it is critical to know what organism(s) are likely to be involved and the sensitivity of those organisms to antibiotics. Decisions are based on the likelihood of certain infections and their sensitivities.
Definitions
A discussion of individual antimicrobial agents is preceded by definitions of the following terms:
The difference among the terms antibiotic, antiinfective, and antibacterial is that antibiotics are produced by microorganisms, whereas the other agents may be developed in a chemistry laboratory (not from a living organism). Antibacterial refers to a substance from any source that inhibits or kills bacteria. The term antiinfective refers to a substance from any source that inhibits or kills organisms that can produce infection, such as bacteria, protozoa, and viruses. This difference is largely ignored in general conversation, and antiinfectives are often referred to as “antibiotics.”
Whether an antibacterial agent is labeled bactericidal or bacteriostatic depends on variables such as the dose used and the organism being treated. Box 7-2 lists the most common antimicrobial agents and classifies them as bacteriostatic or bactericidal.
Infection
The factors that determine the likelihood that a microorganism will cause an infection are the following:
Resistance
There are a reported 2 million cases of antibiotic-resistant infections in the United States each year with approximately 23,000 reported deaths from these infections. It should also be noted that the presence of antibiotic-resistant bacteria is greatest during the month after completion of antibiotic therapy and can remain for up to 12 months after therapy.
Resistance (related to antibiotics) is the natural or acquired ability of an organism to be immune to or to withstand the effects of an antiinfective agent. Natural resistance occurs when an organism has always been resistant to an antimicrobial agent because of the bacteria’s normal properties, such as lipid structures in the cell wall. Acquired resistance occurs when an organism that was previously sensitive to an antimicrobial agent becomes resistant. This can occur by natural selection of a spontaneous mutation (“survival of the fittest”). An increase in the use of an antibiotic in a given population (e.g., a hospital) increases the proportion of resistant organisms in that population. Conversely, a decrease in the use of an antibiotic decreases the proportion of organisms resistant to that antibiotic in that given population. Another method by which resistance develops is by the transfer of DNA (deoxyribonucleic acid) genetic material from one organism to another via transduction, transformation, or bacterial conjugation. The first organism, which is resistant to one or more antibiotics, transfers its genetic material to a second organism. The second organism, which was not previously resistant, thus becomes resistant to the same antibiotic as the first organism without ever having been exposed to that antibiotic. This transfer of genetic material from one organism to another may occur among very different microorganisms, including transfer from nonpathogenic bacteria to pathogenic bacteria. The three most common mechanisms of acquired resistance are a decrease in bacterial permeability, the production of bacterial enzymes, and an alteration in the target site.
The misuse of antibiotics has contributed to one of the United States’ most pressing public health problems, antibiotic resistance. The number of antibiotic-resistant bacteria has increased dramatically over the past 10 years, and many bacterial infections are becoming resistant to some of the most commonly prescribed antibiotics. Every time a person uses an antibiotic, sensitive bacteria are killed off but resistant bacteria can thrive. Repeated and improper uses of antibiotics have jeopardized the usefulness of essential drugs. As a result of antibiotic resistance, some patients are experiencing longer-lasting illnesses, more doctor visits or hospital stays, and the need for more expensive antibiotic therapy.
Reducing the incidence of antibiotic-resistant bacteria can be achieved by reducing the number of prescriptions written annually as well as educating the patient and health professional about antibiotic-resistant bacteria. Health care professionals, including dentists and dental hygienists, can help reduce the incidence of resistance by prescribing antibiotics only when absolutely necessary. The correct antibiotic, with the correct dose and duration, should be prescribed. Only one antibiotic should be prescribed, and a second should be added only if it is clearly necessary. Patients should be educated about the correct use of antibiotics to include taking the antibiotic as prescribed and completing therapy. They should also be advised about the adverse effects of the antibiotic. If possible, the infection should be cultured and the continued need for antibiotic therapy should be assessed once the results of the culture are known. As always, patient education is the key.
Indications for antimicrobial agents
Considerable controversy exists regarding the need for antimicrobial agents in various situations. The two categories of indications are prophylactic and therapeutic.
Therapeutic Indications
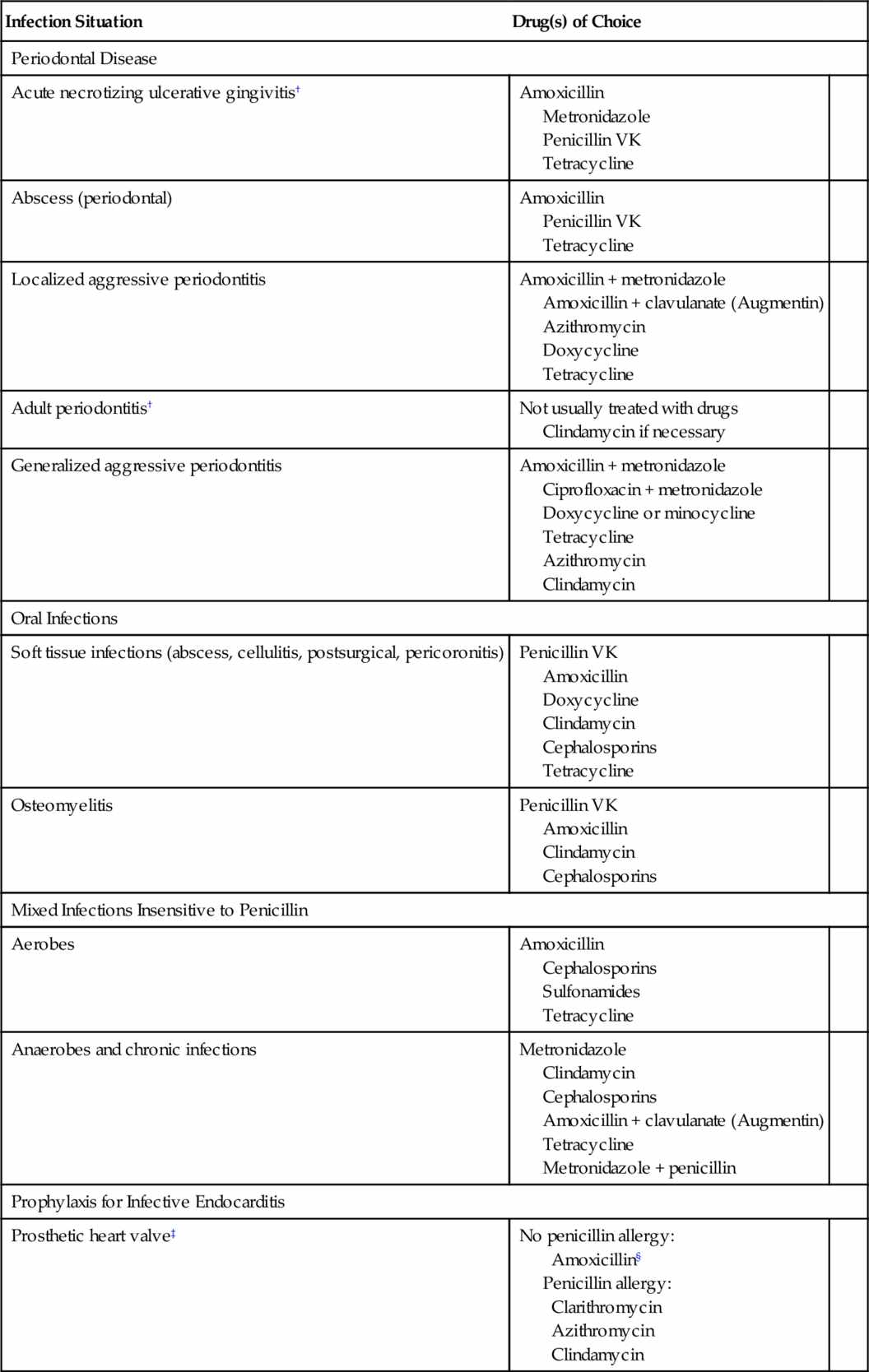
Although there is no simple rule to determine whether antimicrobial therapy is needed in dentistry, many infections do not require it. Most patients who do not have immune function deficiencies and in whom drainage can be used need no antibiotics to manage their dental infections. Table 7-1 lists the indications for treatment of dental infections along with the antibiotics of choice and their alternatives. If local resistance patterns vary from those found in the table, antibiotic choice should be based on that information. However, before a decision is made, several factors must be considered.
Patient
The best defense against a pathogen is the host response. A properly functioning defense mechanism is of primary importance. When this defense is lacking, the need for antimicrobial agents is more pressing.
Infection
The virulence and invasiveness of the microorganism are important in determining the acuteness, severity, and spreading tendency of an infection. An acute, severe, rapidly spreading infection should generally be treated with antimicrobial agents, whereas a mild, localized infection in which drainage can be established need not be treated. If the periodontal pocket (site) remains active despite repeated root planing, then the use of antibiotics to alter the flora may be considered.
When antimicrobial agents are to be used in the treatment of dental infections, the organisms likely to produce the infection and their susceptibility to antimicrobial agents must be considered. Table 7-1 lists the antimicrobials of choice for various dental situations (when culture and sensitivity testing are unavailable) and alternatives if the drug of choice cannot be used.
Prophylactic Indications
Few situations arise for which a definite indication for prophylactic antibiotic coverage exists. One clear-cut use of antibiotics for prophylaxis before a dental procedure (recommended by the American Heart Association and the American Dental Association) is a history of infective endocarditis, presence of a heart valve prosthesis, or specific types of congenital heart disease. The most current guidelines regarding antibiotic prophylaxis are discussed in detail at the end of this chapter.
General adverse reactions and disadvantages associated with antiinfective agents
Superinfection (Suprainfection)
All antiinfective agents can produce an overgrowth of an organism that is different from the original infecting organism and resistant to the agent being used. The wider the spectrum of the antiinfective agent and the longer the agent is administered, the greater the chance that superinfection will occur. This side effect can be minimized by use of the most specific antiinfective agent, the shortest effective course of therapy, and adequate doses.
Allergic Reactions
Like all drugs, all antiinfective agents have the potential to produce a variety of allergic reactions, ranging from a mild rash to fatal anaphylaxis. Some antiinfective agents, such as the penicillins and the cephalosporins, are more allergenic than other agents. Many antiinfective agents, such as erythromycin and clindamycin, have a low allergenic potential.
Drug Interactions
Antiinfective agents can interact with oral contraceptives, oral anticoagulants, and other antiinfectives (a bacteriostatic agent interferes with a bactericidal agent).
Oral Contraceptives
Some antibiotics have been found to decrease the efficacy of oral contraceptives by increasing their clearance from the body. This drug interaction, although unlikely, should be discussed with the patient whenever a patient using oral contraceptives receives a prescription for an antibiotic. Of those antibiotics used in dentistry, ampicillin and the tetracyclines are the most likely to have this effect. In certain patients, additional birth control measures should be used during antibiotic therapy.
Oral Anticoagulants
Antiinfective agents can potentiate the effect of oral anticoagulants. Oral anticoagulants are vitamin K inhibitors, so interfering with the production of vitamin K could increase the anticoagulant effect. Bacterial flora in the intestine produce most of the vitamin K in human bodies. Antiinfective agents (e.g., tetracycline) reduce the bacterial flora that produce vitamin K. With the vitamin K reduced, the oral anticoagulant’s effect is increased. Erythromycin and azithromycin inhibit the enzymes that metabolize warfarin, leading to an increase in warfarin levels. Prolongation of the international normalized ratio (INR) leading to bleeding or hemorrhage may result. The INR should be monitored more closely in patients on antiinfective therapy. Antiinfective agents interact with warfarin to varying degrees, depending on the specific agent.
Gastrointestinal Complaints
All antiinfective drugs can produce a variety of GI complaints. The complaints include stomach pain, increased motility, and diarrhea. The incidence varies greatly, depending on the particular agent used, the dose of the agent, and whether the patient takes the drug with food. Erythromycin has the highest incidence of GI complaints of any of the antibiotics. More serious GI complaints, such as pseudomembranous colitis, which has been historically linked with clindamycin, are now known to occur not only with a wide variety of antiinfective agents (cephalosporins, amoxicillin) but also in the absence of antimicrobial therapy.
Pregnancy Considerations
The antimicrobial agents that can be used during pregnancy to treat infections are limited. Although the risk-to-benefit ratio must be considered whenever pregnant women are given any medications, penicillin and erythromycin have not been associated with teratogenicity and are often used. The use of clindamycin is probably also acceptable, but before any antibiotics are used in the pregnant dental patient, the patient’s obstetrician should be contacted (this procedure also helps prevent medical-legal problems). Metronidazole is not usually used during pregnancy, but exceptions exist. The tetracyclines are contraindicated during pregnancy because of their effect on developing teeth and skeleton.
Dose Forms
Common adult dose forms of antibiotics are tablets and capsules. Children’s dose forms, including liquid and chewable antibiotic dose forms, contain sugar as the sweetening agent. After the dentition has erupted, the dental hygienist should encourage the parent or child to brush the child’s teeth after the use of these agents. The chewable tablets can stick to the teeth, especially in the pits. Long-term administration of antibiotics could increase the child’s caries rate.
Cost
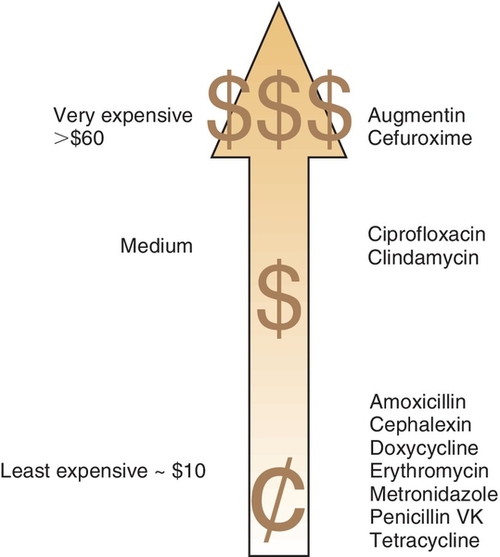
Cost is an important factor in choosing an antibiotic for a patient. If the perfect antibiotic is chosen and prescribed but the patient does not purchase the medication because it is too expensive, then poor results are likely. The best inexpensive antibiotic will be more effective than an expensive one that cannot be purchased. Figure 7-1 compares the costs of various antiinfective agents.
Penicillins
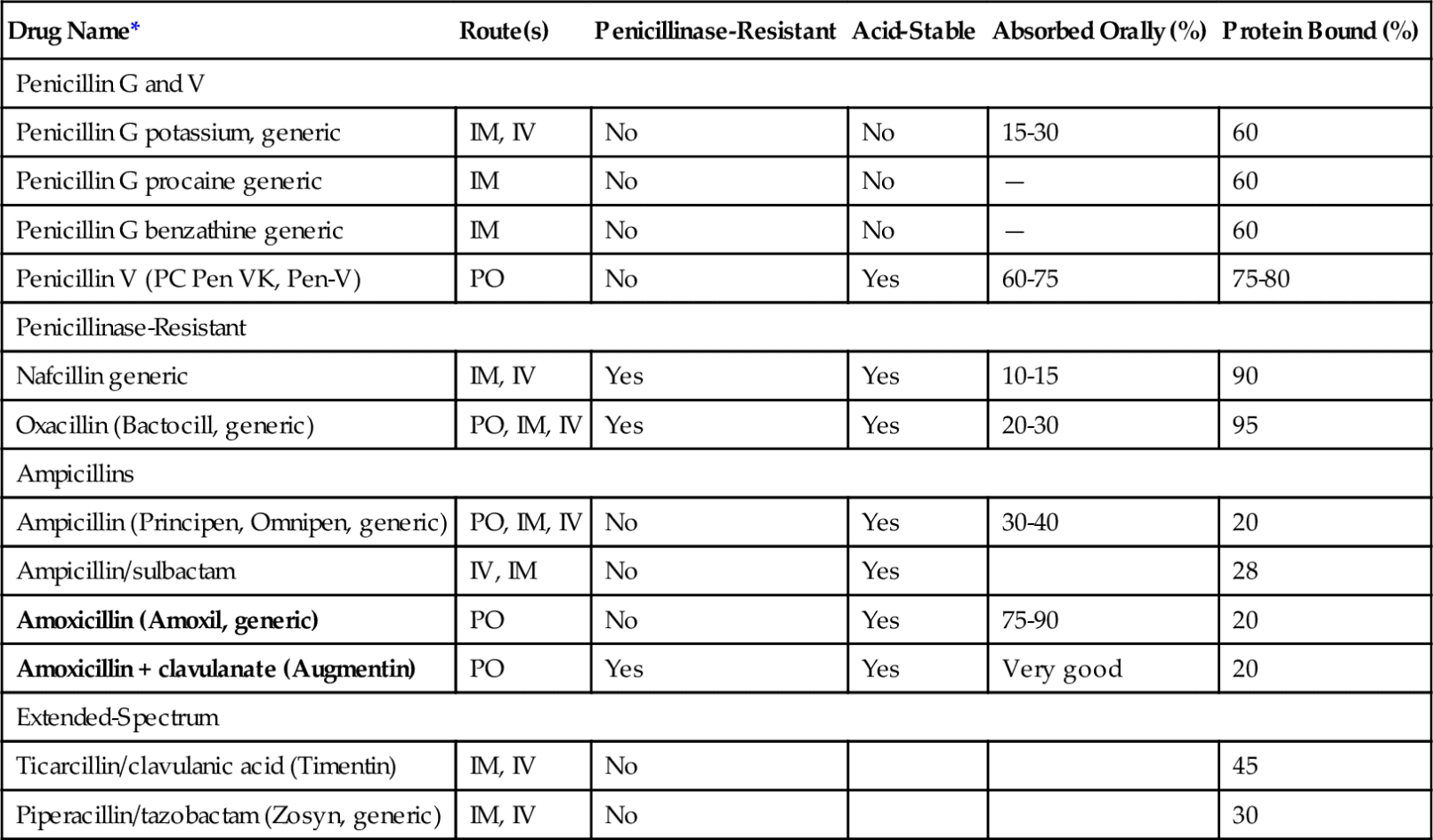
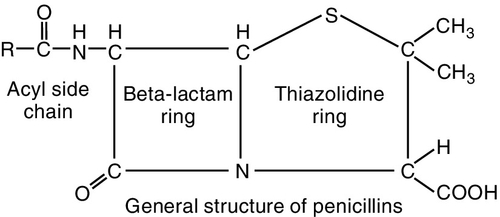
The penicillins (pen-i-SILL-ins) belong to the group of antibiotics known as β-lactam antibiotics. They are so named because of the β-lactam ring that is common to the molecular structure in all antibiotics in this group (Figure 7-2). The penicillins can be divided into four major groups (Table 7-2). The first group contains penicillin G and V, the second group is composed of the penicillinase-resistant penicillins, the third group contains amoxicillin, and the fourth group consists of extended-spectrum penicillins. Because the penicillins have many properties in common, their similarities are discussed first. In dentistry, the first and third groups are commonly used.
Pharmacokinetics
Penicillin can be administered either orally or parenterally but should not be applied topically because its allergenicity is greatest by that route. When penicillin is administered orally, the amount absorbed depends on the type of penicillin. The percentage can vary from 0% to more than 90% (see Table 7-2) and when its absorption from the oral route is too low, then it is only available via injecion. Penicillin V is better absorbed orally than penicillin G, so penicillin V is used for administration of oral penicillin.
The oral route provides the advantages of convenience and less likelihood of a life-threatening allergic reaction. The disadvantages of using the oral rather than the parenteral route are that the blood levels rise more slowly, the blood levels are less predictable because of variable absorption or lack of patient compliance (biggest problem), and some penicillins are degraded by gastric acid. The highest blood levels are obtained if the patient takes the penicillin orally at least 1 hour before or 2 hours after meals, but penicillin V and amoxicillin can be taken without regard to meals.
After absorption, penicillin is distributed throughout the body, with the exception of cerebrospinal fluid, bone, and abscesses. This distribution includes the tissue, saliva, and kidneys. Penicillin crosses the placenta and appears in breast milk.
Penicillin is metabolized by hydrolysis in the liver and undergoes tubular secretion in the kidney. The elimination half-life for both penicillin G and penicillin V is about 0.5 hour. In five half-lives, about 2.5 hours, these penicillins are virtually eliminated from the body.
Mechanism of Action
Penicillin is a very potent bactericidal agent that attaches to penicillin-binding proteins (PBPs) on the bacterial cell membrane. The PBPs are enzymes that are involved in the synthesis of the cell wall and the maintenance of the cell’s structural integrity. Penicillin acts as the structural analog of acyl-d-alanyl-d-alanine, inhibiting the formation of cross-linkages (transpeptidases). This destroys cell wall integrity and leads to lysis. The penicillins are more effective against rapidly growing organisms. Table 7-2 summarizes the types, routes of ingestion, and other properties of penicillins.
Spectrum
Penicillin G and V’s narrow spectrum of activity includes gram-positive cocci, such as Staphylococcus aureus, Staphylococcus pneumoniae, Streptococcus pyogenes, S. viridans, and certain gram-negative cocci, such as Neisseria gonorrhoeae (produces gonorrhea) and Neisseria meningitidis. Penicillin is also effective against spirochetes and anaerobes such as Actinomyces, Peptococcus, Peptostreptococcus, Bacteroides, Corynebacterium, and Clostridium species. The spectrum of activity of the penicillins matches the microbes responsible for many periodontal conditions. The other penicillins have somewhat different activity spectra that are discussed in each section.
Resistance
Resistance to penicillin can occur by several different mechanisms. Penicillinase-producing staphylococci are resistant because their enzymes destroy some penicillins. These penicillinases inactivate the penicillin moiety by cleaving the β-lactam ring.
In hospital environments, more than 95% of the population of staphylococci are penicillinase-producing organisms. Clavulanic acid serves as an inhibitor, which allows the use of amoxicillin to treat penicillinase-producing organisms. Certain bacteria have an outer cell membrane that prevents penicillin from reaching the PBPs.
Although most oral strains of S. viridans are sensitive to penicillin, an increasing number of strains are becoming resistant. The amount of bacterial resistance is proportional to the clinical use of the antibiotic; frequent use leads to increased resistance (and rarer use to decreased resistance).
Adverse Reactions
The untoward reactions to the penicillins can be divided into toxic reactions and allergic or hypersensitivity reactions. The penicillins are the most common cause of drug allergies.
Toxicity
Because penicillin’s toxicity is almost nonexistent, large doses have been tolerated without adverse effects. For this reason, there is a large margin of safety when penicillin is administered. With massive intravenous (IV) doses, direct central nervous system (CNS) irritation can result in convulsions. Large doses of penicillin G have been associated with renal damage manifested as fever, eosinophilia, rashes, albuminuria, and a rise in blood urea nitrogen (BUN). Hemolytic anemia and bone marrow depression have also been produced by penicillin. The penicillinase-resistant penicillins are significantly more toxic than penicillin G. Gastrointestinal irritation can manifest as nausea with or without vomiting. The irritation caused by injection of penicillin can produce sterile abscesses if given intramuscularly (IM) or thrombophlebitis if given intravenously.
Allergy and Hypersensitivity
Allergic reactions to penicillin always should be considered when penicillin is prescribed. Some studies indicate that 5% to 10% of patients receiving penicillin will have a reaction. Allergic reactions to oral penicillin are less common than to parenteral penicillin. Anaphylactic reactions are more frequent in patients first treated with β-blockers and subsequently given oral penicillin. Anaphylactic reactions in these patients have been reported to be difficult to treat.
The following are types of allergic reactions associated with the penicillins:
When reactions to penicillin occur, the consequences are often serious. It is estimated that an anaphylactic reaction occurs in up to 0.05% of penicillin-treated patients, with a mortality of 5% to 10%. It is estimated that 400 deaths occur annually in the United States because of an allergic reaction to penicillin. Although the chance of a serious allergic reaction to penicillin is greater after parenteral administration, anaphylactic shock and death after oral use have also been reported. Patients who have a history of any allergy are more likely to be allergic to penicillin.
Allergic reactions to penicillin of any type may be followed by more serious allergic reactions on subsequent exposure. Any history of an allergic reaction to penicillin contraindicates its use, and another antibiotic should be substituted. However, a history negative for penicillin allergy does not guarantee its lack. If a penicillin is prescribed and any question of a reaction remains, one should make sure that, after the first dose is taken, the patient is somewhere where help can be summoned if necessary.
Uses
Penicillin is an important antibiotic in medical and dental practice. Its use in dentistry results from its bactericidal potency, lack of toxicity, and spectrum of activity, which includes many oral flora. It is often used for the treatment of dental infections. Table 7-1 demonstrates the dental infections for which penicillin is the drug of choice if patients are not allergic to it. Amoxicillin, a close penicillin relative, is also used for specific prophylactic indications. It is the agent of choice for the prophylaxis of infective endocarditis in nonallergic patients who have cardiac conditions associated with the highest risk of adverse outcomes from endocarditis (see the discussion on antibiotic prophylaxis of infective endocarditis at the end of this chapter). Penicillin’s effectiveness in the treatment of dental infections is explained by its effectiveness against many aerobic and anaerobic bacteria.
Specific Penicillins
Penicillin G
Penicillin G, the prototype penicillin, is available as sodium, potassium, procaine, or benzathine salts. These salts differ in onset and duration of action and plasma level attained. One should note that the potassium salt given IV produces the most rapid and highest blood level, whereas the benzathine salt given IM produces the lowest and most sustained blood level. The potassium and procaine salts, given IM, produce intermediate blood levels and durations of action. The penicillin’s duration of action is inversely proportional to the solubility of the penicillin form: The least soluble is the longest acting.
The sodium salts of penicillin should be avoided in patients with a limited sodium intake such as those with cardiovascular problems. Patients with renal disease should not be given potassium salts, because doing so could result in hyperkalemia. Patients may be allergic to the procaine moiety in procaine penicillin G. Both procaine and benzathine penicillins are suspensions given IM, from which the penicillin is slowly released.
Penicillin V
Penicillin V has a spectrum of activity very similar to that of penicillin G. The potassium salt of penicillin V (K penicillin V or penicillin VK) is more soluble than the free acid and therefore is better absorbed when taken orally. Table 7-1 lists some situations in which penicillin is the drug of first choice if the patient is not allergic to it. The usual adult dose is 250-500 mg three (tid) to four times a day (qid) for treatment of an infection for a minimum of 5 days and preferably 7 to 10 days.
Penicillinase-Resistant Penicillins
Penicillinase-resistant penicillins should be reserved for use against penicillinase-producing staphylococci. Compared with penicillin G, the penicillinase-resistant penicillins are less effective against penicillin G–sensitive organisms. They also have more side effects, such as GI discomfort, bone marrow depression, and abnormal renal and hepatic function. Patients allergic to penicillin are also allergic to the penicillinase-resistant penicillins.
Because cloxacillin and dicloxacillin are better absorbed than the other penicillinase-resistant penicillins, they are the drugs of choice.
Ampicillins
Ampicillin (am-pi-SILL-in) and amoxicillin (a-mox-i-SILL-in) are most often used in medicine. These penicillinase-susceptible penicillins have a spectrum of activity that includes gram-positive cocci, Haemophilus influenzae, and enterococci such as Escherichia coli, Proteus mirabilis, and Salmonella and Shigella species.
Amoxicillin, a relative of ampicillin, is most often used to treat infections because it produces higher blood levels, is better absorbed, and requires less frequent dosing (three times daily versus four times daily for penicillin VK or ampicillin), and its absorption is not impaired by food. Amoxicillin is the drug of choice for prophylaxis for infective endocarditis before a dental procedure. Amoxicillin is used to treat upper respiratory tract infections (H. influenzae), urinary tract infection (E. coli), and meningitis (H. influenzae). Otitis media in children is often treated with amoxicillin. Amoxicillin is also available mixed with clavulanic acid, a β-lactamase inhibitor (Augmentin). Clavulanic acid combines with and inhibits the β-lactamases produced by bacteria. Therefore the amoxicillin is protected from enzymatic inactivation. This combination can be used with penicillin-producing organisms. It has had some use in the management of certain periodontal conditions (see Table 7-1).
Both ampicillin and amoxicillin can produce a variety of allergic reactions. Ampicillin is much more likely to cause rashes than other penicillins. There is strong evidence to suggest that the ampicillin rash is benign and not an allergic response. This unusual ampicillin-related rash is much more common in patients with mononucleosis (almost 100%) or those taking allopurinol. Cross-allergenicity between penicillin VK, amoxicillin, and ampicillin is complete (omitting the “weird” ampicillin rash).
Extended-Spectrum Penicillins
Ticarcillin and piperacillin have a wider spectrum of activity than penicillin G, with special activity against Pseudomonas aeruginosa and some strains of Proteus. They are not penicillinase resistant and are available parenterally to treat systemic infections.
Cephalosporins
The cephalosporin (sef-a-loe-SPOR-in) group of antibiotics is structurally related to the penicillins. Cephalosporins are active against a wide variety of both gram-positive and gram-negative organisms. The oral cephalosporin products, listed in Box 7-3, are divided into first-, second-, third-, and fourth-generation agents. Most third-generation cephalosporins are available for parenteral use. The orally active cephalosporins are discussed.
The source of the original cephalosporins was Cephalosporium acremonium, which was isolated from a sewer outlet near Sardinia in Italy. Because cephalosporins are true antibiotics, they were originally produced by organisms. Those available for oral use are relatively acid stable and highly resistant to penicillinase, but they are destroyed by cephalosporinase, an enzyme elaborated by some microorganisms.
Pharmacokinetics
The cephalosporins can be administered orally, intramuscularly, or intravenously. The agents that cannot be used orally are too poorly absorbed to provide adequate blood levels. The cephalosporins used orally are well absorbed. They are bound to the plasma proteins in a proportion between 10% and 65% (see Box 7-3). After absorption, they are widely distributed throughout the tissues. Like penicillin, the cephalosporins are excreted by glomerular filtration and tubular secretion into the urine. Their half-lives vary between 50 and 240 minutes.
Spectrum
The cephalosporins, which are bactericidal, are active against most gram-positive cocci, penicillinase-producing staphylococci, and some gram-negative bacteria. They inhibit most Salmonella and Klebsiella organisms, some paracolon strains, and E. coli. Serratia and Enterobacter species, H. influenzae, indole-positive Proteus, methicillin-resistant staphylococci, and most Pseudomonas strains are unaffected. The generation of the cephalosporin (first, second, or third) designates the width of antimicrobial action; the first-generation width is narrower (gram-positive, few gram-negative) than the second-generation width (gram-positive, more gram-negative and anaerobes), and the third-generation agents (gram-positive weaker, many gram-negative and anaerobes) have the broadest spectrum of activity.
Mechanism of Action
The mechanism of action of the cephalosporins is like that of the penicillins: inhibition of cell wall synthesis. They bind to enzymes in the cell membrane involved in cell wall synthesis. The cephalosporin acts as an analog of acyl-d-alanyl-d-alanine to produce a deficiency in the cell walls, leading to lysis. They are more effective against rapidly growing organisms (which explains the potential drug interaction between bacteriostatic and bactericidal antibiotics).
Adverse Reactions
In general, the cephalosporins have a low incidence of adverse reactions (excluding allergic reactions) and are well tolerated. They have more adverse reactions than penicillin VK. The following adverse reactions may occur.
Gastrointestinal Effects
The most common adverse reaction associated with the cephalosporins is gastrointestinal, including diarrhea, nausea, vomiting, abdominal pain, anorexia, dyspepsia, and stomatitis.
Nephrotoxicity
Evidence suggests that the cephalosporins may have nephrotoxic effects under certain conditions. Although some researchers have suggested that this is a toxic reaction, it may be an allergic reaction.
Superinfection
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses