9
Preparation of the Mouth
Preparation of the mouth is often necessary before dentures are made and may involve the elimination of pathology within the denture-bearing tissues or the creation of a more favourable anatomical environment.
The various conditions requiring treatment may be considered in two sections, those involving the oral mucosa and those involving bone.
1. Conditions involving the oral mucosa
- Denture stomatitis
- Inflammatory papillary hyperplasia of the palate
- Angular stomatitis (angular cheilitis)
- Shallow sulci
- Denture-induced hyperplasia
- Prominent frena
2. Conditions involving the bone
- Pathology within the bone
- Sharp and irregular bone
- Undercut ridges
- Prominent maxillary tuberosities
- Tori
Conditions involving the oral mucosa
Denture stomatitis
Clinical appearance
The clinical appearance of denture stomatitis varies from a patchy to a diffuse inflammation of the mucosa covered by a denture (Fig. 9.1). Newton (1962) classified denture stomatitis into three types on the basis of its clinical appearance:
Figure 9.1 Denture stomatitis. The characteristically sharp delineation of the diffuse palatal inflammation by the borders of the denture is seen at the junction of the hard and soft palate, where the line separating the inflamed from normal tissue corresponds to the posterior border of the denture.
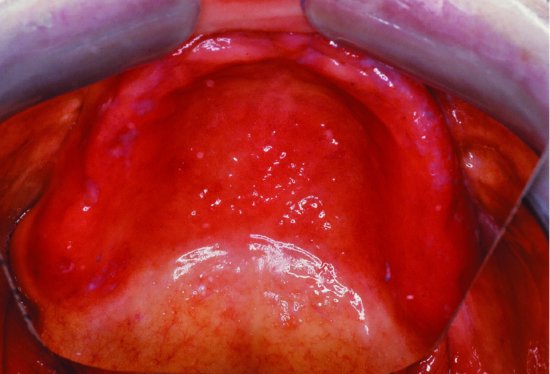
- Type I: Pinpoint hyperaemia
- Type II: Diffuse inflammation
- Type III: Granular (palatal inflammatory papillary hyperplasia (p. 118)
The condition typically involves the mucosa of the maxillary denture-bearing area and does not extend beyond the borders of the denture. It may occur alone but is sometimes seen with two associated conditions, inflammatory palatal papillary hyperplasia and angular stomatitis.
Nomenclature
In spite of the lesion’s rather angry appearance the patient rarely complains of soreness, therefore the term ‘denture sore mouth’, which has been used in the past to describe this condition, is inappropriate. Other terms currently used in the literature to describe the condition are:
- Denture-related stomatitis.
- Denture-induced stomatitis – however, it has been argued that as the denture is rarely the primary aetiological agent this term is seldom appropriate.
- Stomatitis prothetica.
- Chronic atrophic candidiasis – for this and the following term to be appropriate it is necessary that a microbiological investigation should have confirmed the presence of candida.
- Candida-related stomatitis
Prevalence
Denture stomatitis is a common condition, having been reported in 10–60% of patients wearing complete dentures. However, in the general complete denture-wearing population the prevalence of the condition is likely to be closer to the lower limit of this range than to the upper (Radford et al. 1999).
Predisposing factors
Age
Denture stomatitis has been found to be more common in the elderly, particularly if hyposalivation is present (Figueiral et al. 2007; Campisi et al. 2008).
Gender
Denture stomatitis occurs more frequently in females than in males, the ratio being approximately 4:1 (Davenport 1970; Pires et al. 2002). Although the cause of this predisposition to denture stomatitis in females is not known, possible explanations include endocrine imbalance, iron deficiency anaemia, vaginal carriage of candida, a higher oral carrier rate of candida and a greater inclination than males to wear dentures at night.
Night wearing of dentures
It appears that behavioural factors, such as wearing the dentures at night and poor denture hygiene, are of major importance in the aetiology of denture stomatitis (Jeganathan et al. 1997; Sakki et al. 1997; Saulman et al. 2006). Wearing the dentures at night can aggravate the effect of both denture plaque and denture trauma by increasing the exposure of the palatal mucosa to both aetiological factors.
Poor denture hygiene
Poor denture hygiene allows the build-up of denture plaque containing candida and other microorganisms. A strong association between poor denture hygiene and denture stomatitis has been identified (Khasawneh & Al-Wahadni 2002; Kulak-Ozkan et al. 2002).
High-carbohydrate diet
Dentures with a reduced masticatory efficiency may encourage a patient to adopt a relatively easily managed high-carbohydrate diet, which favours the growth of candida and increases the adhesion of the microorganism to the denture surface. An increase in the amount and frequency of intake of relatively inexpensive carbohydrates may also be encouraged by the patient’s economic circumstances.
Systemic factors
Systemic conditions which can predispose to denture stomatitis include immunological deficiencies (Golecka et al. 2006), hormonal imbalance, e.g. diabetes, and nutritional deficiencies, e.g. vitamin B complex, vitamin C and iron.
Aetiology
Many different local and systemic factors have been incriminated in the aetiology of denture stomatitis, which is now widely accepted to be multifactorial in origin. The relative importance of microorganisms, in particular Candida albicans, and trauma from the dentures is still debated. However, the bulk of the relevant literature supports the view that the presence of candida organisms within plaque on the impression surface of the denture is the key local factor.
In 1885, G.V. Black, commenting on the condition we now know as denture stomatitis, stated that, ‘Fungi grow readily under any plate irrespective of the material of what it is made. They produce acids which if the mouth and palate are not properly cleaned will cause sore mouth’. He sampled microorganisms from under the dentures of patients with this condition and expressed surprise at their abundance. He concluded that ‘plates are not cleaned often enough and that cleanliness is the chief preventive measure’. Thus, well over 100 years ago Black summarised the modern view of both the aetiology and management of denture stomatitis.
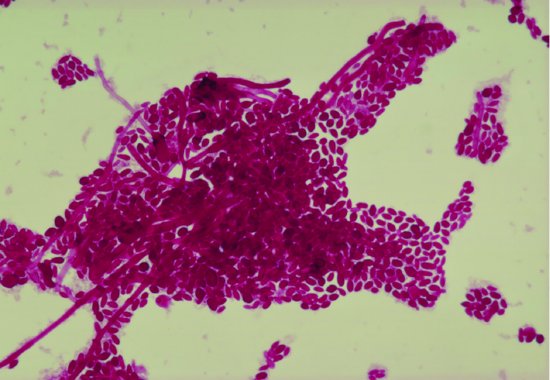
Figure 9.2 Mycelial and yeast forms of Candida albicans obtained from the impression surface of a denture.
In a study of 3450 patients wearing at least one removable denture the main risk factors associated with denture stomatitis were found to be wearing the dentures continuously, low levels of vitamin A and smoking (Shulman et al. 2005). The authors recommended improved denture cleaning and removing the dentures at night.
Candida albicans
The evidence suggests that the most important local causative factor of denture stomatitis is the presence of microorganisms within plaque on the impression surface of the denture (Davenport 1970; Budtz-Jørgensen 1974; Olsen 1974; Khasawneh et al. 2002; Ramage et al. 2004). The plaque is present in significant quantities because of inadequate denture hygiene and its effects are maximised if the patient wears the dentures at night. The fungus, Candida albicans (Fig. 9.2), is the organism most commonly associated with denture stomatitis and which has received the most attention in the literature, although other candida species may also be present (Marcos-Arias et al. 2009). This fungus is dimorphic, occurring as both yeast-like blastospores and filamentous pseudohyphae. In denture stomatitis, large numbers of both forms are usually found in plaque on the impression surface of the denture. Relatively few candida organisms are found on the mucosa and there is no evidence that candidal invasion of the mucosa occurs.
Bacteria
Although the evidence for a causal relationship between candida and denture stomatitis is strong, it has been suggested that a variety of bacteria may also play a part in the condition (Kulak et al. 1997).
Denture trauma
It has been proposed that trauma to the oral mucosa is a cause of denture stomatitis (Emami et al. 2008). Dentures can traumatise the mucosa either because of the presence of faults in the prostheses such as loss of fit or occlusal imbalance or because the patient exhibits parafunctional activity, such as bruxism, which overloads the tissues. It is sometimes argued that if trauma from the dentures was a major factor in the development of denture stomatitis then the condition would be more commonly seen under mandibular than maxillary dentures. This is because the area of mucosal support for the prosthesis is much less in the mandible than in the maxilla and therefore the load per unit area and potential degree of trauma is significantly greater in the mandible than in the maxilla. However, the widespread inflammation characteristic of denture stomatitis that is seen clinically is a feature of the maxillary, not the mandibular, denture-bearing mucosa.
Other factors
It should be remembered that palatal inflammation under dentures is a non-specific response to a variety of injurious agents. Therefore diffused inflammation of the denture-bearing mucosa may occasionally be seen, which is not the result of denture plaque or trauma. For example:
- Raised residual monomer. A faulty curing cycle when the dentures are processed can result in a residual monomer content high enough to produce mucosal inflammation (p. 116) (Austin & Basker 1980). This, unlike the classical denture stomatitis resulting from poor denture hygiene, is characteristically associated with discomfort from the inflamed mucosa.
- Self-medication. Palatal inflammation is sometimes caused by patients using topical agents in an inappropriate way. Certain mouthwashes, ointments, or other substances which are normally free of adverse effects in the mouth, can cause mucosal damage when applied beneath a denture. Examples of preparations that can behave in this way are chlorhexidine gel, salicylate ointments and even whisky – with which some more ingenious patients have been known to bathe their sore mucosa. The damage occurs because the exposure to the agents is increased by their being held against the mucosa for extended periods by the denture and because they are not diluted or washed away by the saliva. Also, a vicious cycle may be established in which the patient initially uses the preparation in a misguided attempt to relieve some discomfort; the preparation then causes further mucosal damage which increases the soreness and so the patient applies even more of the injurious agent.
Aetiological interactions
The possible interaction of the various predisposing and aetiological factors is complex and uncertain, but a possible scenario which is compatible with the bulk of the relevant literature is as follows (Fig. 9.3).
Figure 9.3 Possible aetiological interactions in denture stomatitis (see text for fuller explanation). (Reproduced with permission from Stephen Hancock. © 2000 British Dental Journal.)
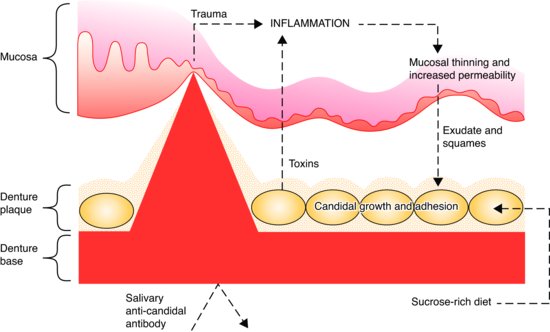
Toxins produced by the candida cells left on the denture surface by deficient hygiene measures, together with trauma from the denture initiate an inflammatory reaction. A resulting thinning of the epithelium results in increased permeability and escape of inflammatory exudates. The exudates, together with desquamated mucosal cells, form a favourable nutrient medium, which promotes the growth of Candida albicans. In addition, these exudates and the sucrose-rich diet which may result from the dietary selection sometimes associated with the wearing of dentures, may contribute to the condition by increasing the adhesiveness of the candidal cells, and thus encouraging the formation of denture plaque. As candidal proliferation occurs, the rate of production of potent toxins by the microorganisms increases. The passage of these toxins into the tissues is facilitated by the thinning and increased permeability of the mucosa. Aggravation of the inflammatory response occurs and so a vicious circle is set up. Anti-candidal antibody is secreted in parotid saliva but the denture base may restrict access of the antibody to the candida cells.
Diagnosis
The diagnosis of denture stomatitis is essentially a clinical one made on the basis of the appearance of the palatal mucosa. Identification of the aetiological factors responsible for a particular case may be achieved by noting the following.
Night-wearing of dentures
It should be noted routinely from the history whether or not the dentures are worn both day and night.
Denture hygiene
Details of the patient’s denture cleaning regime should be obtained, including the method and frequency of cleaning together with the type of immersion cleanser used. The amount of plaque on the impression surface of the denture should be recorded. This assessment of the quantity and distribution of denture plaque is made easier by applying a disclosing solution to the denture (Fig. 14.10). In those cases where only a little plaque is seen, it should be realised that a patient will sometimes have made an effort to clean the dentures in readiness for the visit to the dentist, which is atypical of their normal denture hygiene regimen.
Denture trauma
The degree of denture trauma should be assessed. This can be estimated from the relative functional adequacy of the dentures in terms of occlusion and fit, and from evidence of the presence or absence of parafunctional activity.
Other local factors
- Raised residual monomer. If palatal inflammation and discomfort occur shortly after a new denture has been fitted, or an existing denture repaired or relined, then a raised residual monomer content might be suspected.
- Self-medication. Questioning the patient abo-ut any current oral medication such as mouthwashes or ointments might indicate if the condition is iatrogenic, resulting from mucosal irritation by these preparations.
- Diet. A dietary assessment can be carried out if it is suspected that a high carbohydrate diet is contributing to the condition.
Systemic factors
The medical history may provide clues that systemic factors are playing a part. If this is the case, or if the condition subsequently fails to respond to local treatment, appropriate investigations should be arranged.
It is important to obtain details of drug therapy in case a particular medication is causing conditions such as a dry mouth (see Chapter 16).
Treatment
Although denture stomatitis is symptomless and the patient is often unaware of its presence, it should be treated before new dentures are constructed because of the following:
- Swelling of the oral mucosa will have occurred as a result of the inflammation. Producing a new denture from an impression of the mucosa in this condition will compromise the fit of the prosthesis, especially if the new well-fitting denture brings about some resolution of the swelling.
- The mouth may be the source of candida organisms and other microorganisms responsible for infection in other parts of the body, such as nailbeds, the pharynx and the larynx (Nikawa et al. 1998). In debilitated patients, systemic spread of candida from the mouth can occur with fatal consequences.
There is increasing evidence that the oral cavity, particularly in denture wearers, can be a reservoir for methicillin-resistant Staphylococcus aureus (MRSA) which clearly has serious implications for both patients and their carers (Lee et al. 2009). Commonly employed denture-cleaning solutions have been shown to be effective against MRSA in vitro (Maeda et al. 2007).
As behavioural factors are so important in the aetiology of denture stomatitis, appropriate modification of the patient’s behaviour is essential for long-term success. Therefore the primary objectives of local treatment of denture stomatitis (Lombardi & Budtz-Jørgensen 1993) are to improve denture hygiene and discourage the patient from wearing the dentures at night.
In addition, in those cases where denture faults are identified and which could be traumatising the denture-bearing tissues, correction of the faults should be undertaken.
Improve denture hygiene
- Motivating, instructing and monitoring the patient. It is vital that the clinician convinces the patient of the need for meticulous cleaning of the dentures. The methods for carrying it out should be discussed using appropriate language, clearly demonstrated, subsequently monitored and reinforced if necessary. Once a high level of denture hygiene has been achieved it is essential that it is maintained otherwise recurrence of the denture stomatitis is likely (Cross et al. 2004).
- Laboratory cleaning of the dentures. Where deposits are heavy and possibly partly calcified, and where the surface polish of the denture has deteriorated, it is recommended that laboratory cleaning and polishing of the denture is carried out before home care by the patient is instituted.
- Disinfection of the dentures. The dentures should be regularly immersed by the patient in a suitable disinfectant. Two solutions have been shown to be effective in controlling denture plaque: alkaline hypochlorite and aqueous chlorhexidine gluconate (Hutchins & Parker 1973; Altman et al. 1979; Budtz-Jørgensen 1979; Abelson 1985; Webb et al. 1998c; Barnabe et al. 2004). The former solution has been shown to be effective in removing denture plaque while the latter inhibits its formation. Overnight immersion is necessary if either a hypochlorite solution containing 0.08% available chlorine or 0.1% aqueous chlorhexidine gluconate is used. When it is impossible to persuade a patient to leave the denture out at night, immersion in a hypochlorite solution containing 0.16% available chlorine for 20 minutes daily or in 2% aqueous chlorhexidine gluconate for approximately 5 minutes daily are alternatives. Before immersion, the denture should be brushed thoroughly to remove most of the plaque and then, if chlorhexidine is to be used, rinsed carefully to remove any soap which would otherwise inactivate the chlorhexidine. Patches of brown staining usually appear on a denture that has been immersed in chlorhexidine solution. As a rule, the staining is not severe and can be removed by subsequent immersion in a hypochlorite cleaner. The presence of a metal denture base complicates matters because hypochlorite can cause corrosion of the base. However, the recommended short immersion period can be used with safety. Disinfection of dentures by short exposure to microwave irradiation has also been shown to be effective (Baysan et al. 1998; Webb et al. 1998c; Dixon et al. 1999; Webb et al. 2005). However the practicality of this approach remains uncertain: the procedure kills the organisms but does not remove the plaque, and there is the possibility that overexposure to the irradiation could have adverse effects on the denture materials.
Ultrasonic cleaning devices have been shown to be potentially useful for cleaning dentures (Hashiguchi et al. 2009; Arita et al. 2005).
Correction of denture faults
- Occlusal faults. An unbalanced occlusion should be corrected by occlusal adjustment or by the addition of cold-curing acrylic resin to the occlusal surfaces of the dentures (p. 95).
- Impression surface faults. Lack of fit in a denture can be corrected by applying a short-term soft lining material to the impression surface (p. 234). However, caution should be exercised in selecting this option as the presence of a temporary lining will make it much more difficult for the patient to maintain the all-important high level of de/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


