Results of Implant Treatment
The landmark Göteborg study, as well as the replica study at the University of Toronto, set the stage for success and predictability expectations of root-form dental implants. The study, conducted over a 15-year period at the University of Göteborg, Sweden, by P. I. Brånemark and coworkers, began in 1965 and concluded in 1980. The results, reported in several articles, defined the concept of osseointegration, described protocols for success, and shared clinical experiences. Perhaps the most significant article from the study, published in 1981, was a paper on osseointegrated implants in the treatment of the edentulous jaw.2 Briefly, the Göteborg study included 2768 root-form implants placed into 410 edentulous jaws in 371 consecutive patients. The data were most often reported in subsets according to the three study phases (initial, developmental, and routine). Those cases treated in the routine period with standardized procedures and an observation time of 5 to 9 years were thought to reflect the potential of the method and were the basis of data reported in that historic publication. This subset consisted of 895 implants placed in 130 jaws. Implant survival was 81% in the maxilla and 91% in mandible. Prosthesis survival (continuous stability) was 89% in the maxilla and 100% in the mandible. The replica study, conducted at the University of Toronto, demonstrated that comparable results could be predictably achieved using the same implant design and treatment protocols.5,73–75 Together, these studies demonstrated that implant survival of 81% or more and prosthesis survival of 89% or more could be expected in the edentulous patient.
In the decades since the landmark discovery of osseointegration and the documentation of its clinical effectiveness, clinicians have experienced tremendous success replacing missing teeth with endosseous, root-form dental implants in partially edentulous, as well as edentulous, patients.1,46 Yet, despite the high level of success and long-term predictability, achieving success all the time, in every patient, and in each situation is not absolute (100%). Complications and implant failures do occur.2,12 Some implants fail to achieve osseointegration, some achieve osseointegration only to lose bone progressively over time leading to failure, and other implants rapidly lose bone and fail in a short time. Just as important, some implants may achieve and maintain osseointegration but fail because they do not meet the patient’s or clinician’s esthetic expectations.
Defining Implant Outcomes
Implant success is defined by specific criteria used to evaluate the condition and function of the implant. Criteria for implant success have been proposed in the literature but not used consistently. The problem is that a universally accepted definition of implant success has not been established. In the classic definition, Albrektsson et al3 defined implant success as an implant with no pain, no mobility, no radiolucent periimplant areas, and less than 0.2 mm of bone loss annually after the first year of loading.3 Bone loss in the first year was recognized but it was not defined or quantified as part of the success criteria until later in a separate definition by Roos.57 The real challenge in comparing reported data from study to study is that authors use different criteria of success in their work. As a result, it is difficult or impossible to make comparisons between studies and drawing conclusions about implant success or failure from data reported in different studies is tenuous.
It is important to recognize that success rates are dramatically affected by variations in the criteria used to define them. In strict terms, if one considers “implant success” to be the outcome without any adverse effects or problems, then the treatment would be performed as planned, implants would remain stable and functioning without problems, periimplant tissues would be stable and healthy, and both the patient and the treating clinician(s) would be pleased with the outcome (Figure 84-1). If such strict criteria are used, implant success rates would be undoubtedly lower than those determined using less stringent criteria. Table 84-1 illustrates the powerful effect that small changes in success criteria have on the reported success rates. The data demonstrates that simply changing the criteria of success to include probing pocket depth (PPD) ≤5 mm to ≤6 mm changed the implant success rates from 52.4% to 62% and from 79.1% to 81.3% in patients with and without a history of periodontitis, respectively.40 It also shows that implant survival (last column) is quite different than implant success.
TABLE 84-1
Effect of Slight Modifications in Implant Success Criteria on Implant Success Rate Outcomes*
| PPD ≤5 mm No BOP BL <0.2 mm/year (%) | PPD ≤6 mm No BOP BL <0.2 mm/year (%) | PPD ≤5 mm No BOP (%) | PPD ≤6 mm No BOP (%) | Implant Survival (%) | |
| Group A (history of periodontitis) | 52.4 | 62 | 71.4 | 81 | 90.5 |
| Group B (periodontal health) | 79.1 | 81.3 | 94.5 | 96.7 | 96.5 |

PPD, Probing pocket depth; BOP, bleeding on probing; BL, bone loss.
*The initial success criteria at 10 years were set at PPD ≤5 mm, no BOP, and BL <0.2 mm annually. The implant success rates for group A (patients with a history of periodontitis) and group B (patients with periodontal health) using this initial criteria is listed in the first column. Notice how dramatically the success rates change when the criteria used to define success are modified to include PPD ≤6 mm (second column). The next two columns show the success rates for each group using PPD ≤5 mm and PPD ≤6 mm when the criteria are modified to omit bone loss as a determinant. The last column shows the implant survival rate for each group, which clearly demonstrates that survival rate is different than success rate.
Based on data from Karoussis IK, Salvi GE, Heitz-Mayfield LJ, et al: Clin Oral Implant Res 14:329-339, 2003.
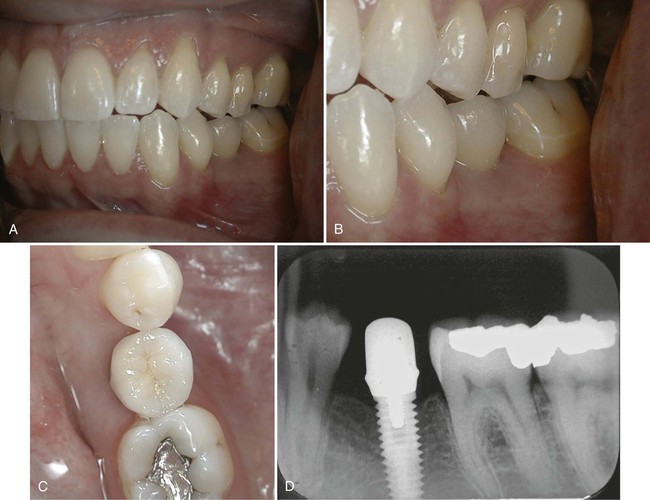
“Implant survival,” conversely, is simply defined as any implant that remains in place at the time of evaluation, regardless of any untoward signs, symptoms, or history of problems. Clearly, there is a difference between implants that are present, healthy, and functioning under an implant-retained restoration, implants that are present but suffering from periimplant bone loss, or implants that are present but not connected to any restoration and not functioning (Figure 84-2). Implants that are osseointegrated but not restored into function are referred to as “sleepers” and should not be considered successful merely because they are present and osseointegrated. To reiterate, implant survival and implant success are distinctly different outcome measures. As an example, consider the dramatic difference between survival and success rate for implants reported in a systematic review (21 studies included) of implants supporting fixed partial dentures; the reported 5-year implant survival rate was 95.4%, whereas the 5-year patient success rate,* defined as free of any complications, was only 61.3%.55 In a 10-year retrospective study of 397 fixed implant reconstructions in 300 patients, the observed mechanical complication rate was 24.7%. The most frequent complication was ceramic chipping (20.31%) followed by occlusal screw loosening (2.57%) and loss of retention (2.06%).72 Although relatively infrequent, occlusal screw loosening can result in a subgingival gap at the implant-abutment junction that retains plaque and stimulates an inflammatory reaction in both the soft and hard tissue leading to bone loss around the implant. These implants could be considered surviving but would fail to meet optimal success criteria.
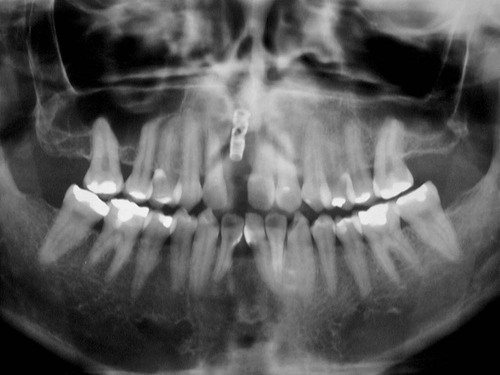
Factors That Influence Implant Outcomes
Anatomic Location
Osseointegration depends on the availability of an adequate quantity and quality of bone at the implant site(s). Areas with abundant bone volume in the desired location will be better than areas with deficient bone volume. Likewise, areas with good bone density provide more predictable outcomes than areas with poor bone density. The bone classification system described by Lekholm and Zarb47 was intended to define bone sites that would have different levels of bone support for implants and that would likely impact survival and success. The quality of bone support is greatly influenced by the anatomic location, thus implant outcomes are sometimes categorized according to anatomic location. Extreme examples are the anterior mandible and the posterior maxilla. The anterior mandible typically consists of dense cortical bone, which offers great support and high bone-to-implant contact, whereas the posterior maxilla is often limited in volume because of alveolar resorption and sinus pneumatization and typically consists of a loose, trabecular structure and a thin cortical bone shell.
Implants placed in the posterior maxilla are less well supported compared to implants placed in the anterior mandible. Jaffin and Berman34 demonstrated the significance of bone quality on implant survival in a report of 1054 Brånemark implants. Ninety percent of these implants were placed in type I, II, and III bone with only 3% failures. However, of the 10% that were placed in type IV bone, 35% failed.34 Excluding those placed in the mandible, 23/52 implants placed in type IV bone in the maxilla failed for a dismal 56% survival rate (44% failed). They concluded that bone quality stood out as the single greatest determinant of implant loss.
Implant Design
What are the best implant design characteristics? Is there an implant design that performs better than the rest? Actually, it is nearly impossible to determine which implant design characteristics are important because there are many variables to consider and implants are successful most of the time. Given the generally high success, it seems that subtle differences in implant design are probably not significant for the majority of patients and situations.22,26 However, in compromised patients, inadequate sites, or challenging circumstances, certain implant designs may perform better than others. A prime example is the effect of implant surface characteristics. Implants with an altered surface microtopography (i.e., acid-etched, blasted) have been shown to enhance the bone-to-implant interface42,43 and are thought to improve implant outcomes, especially in compromised sites.66,67 Lower success rates have been associated with smooth surface (i.e., machined) implants.35,56,60 Implant length is another consideration. Implant length is a factor that has long been held as important for success (i.e., longer is better), and many studies have supported this dogma,61,62 but recent studies are challenging it.29
Placement and Loading Protocols
The traditional implant placement protocol required a healed edentulous ridge into which implants were placed and allowed to osseointegrate for a period of time without occlusal loading (see Chapter 75). The nonloading period after implant placement was empirically determined to be 3 to 4 months in the mandible and 6 months in the maxilla.10 A strongly held belief at the time was that early loading would lead to higher failure rates.11 In contrast to these early standards, some current protocols advocate dramatically different approaches, including implant placement immediately after tooth extraction and implant occlusal loading immediately or shortly after placement. Each of these approaches ha/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


