The Role of Dental Calculus and Other Local Predisposing Factors
Calculus
Supragingival and Subgingival Calculus
The two most common locations for the development of supragingival calculus are the buccal surfaces of the maxillary molars (Figure 7-1) and the lingual surfaces of the mandibular anterior teeth (Figure 7-2).34 Saliva from the parotid gland flows over the facial surfaces of the upper molars via the parotid duct, whereas the submandibular duct and the lingual duct empty onto the lingual surfaces of the lower incisors from the submaxillary and sublingual glands, respectively. In extreme cases, calculus may form a bridgelike structure over the interdental papilla of adjacent teeth or cover the occlusal surface of teeth that are lacking functional antagonists.
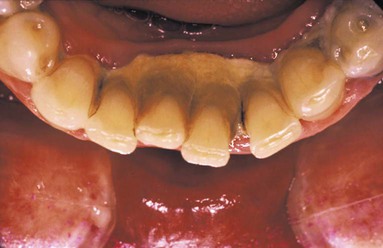
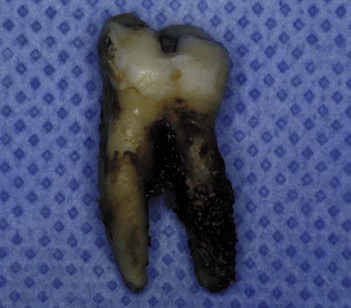
Subgingival calculus is located below the crest of the marginal gingiva and therefore is not visible on routine clinical examination. The location and extent of subgingival calculus may be evaluated by careful tactile perception with a delicate dental instrument such as an explorer. Clerehugh and colleagues31 used a World Health Organization no. 621 probe to detect and score subgingival calculus. Subsequently, these teeth were extracted and visually scored for subgingival calculus. An agreement of 80% was found between these two scoring methods. Subgingival calculus is typically hard and dense; it frequently appears to be dark brown or greenish black in color (Figure 7-3), and it is firmly attached to the tooth surface. Supragingival calculus and subgingival calculus generally occur together, but one may be present without the other. Microscopic studies demonstrate that deposits of subgingival calculus usually extend nearly to the base of periodontal pockets in individuals with chronic periodontitis but do not reach the junctional epithelium.
When the gingival tissues recede, subgingival calculus becomes exposed and is therefore reclassified as supragingival (Figure 7-4, A). Thus, supragingival calculus can be composed of both supragingival calculus and previous subgingival calculus. A reduction in gingival inflammation and probing depths with a gain in clinical attachment can be observed after the removal of subgingival plaque and calculus (Figure 7-4, B; see Chapter 46).
Prevalence
Anerud and colleagues4 observed the periodontal status of a group of Sri Lankan tea laborers and a group of Norwegian academicians for a 15-year period. The Norwegian population had ready access to preventive dental care throughout their lives, whereas the Sri Lankan tea laborers did not. The formation of supragingival calculus was observed early in life in the Sri Lankan individuals, probably shortly after the teeth erupted. The first areas to exhibit calculus deposits were the facial aspects of maxillary molars and the lingual surfaces of mandibular incisors. The deposition of supragingival calculus continued as individuals aged, and it reached a maximal calculus score when the affected individuals were around 25 to 30 years old. At this time, most of the teeth were covered by calculus, although the facial surfaces had less calculus than the lingual or palatal surfaces. Calculus accumulation appeared to be symmetric, and, by the age of 45 years, these individuals had only a few teeth (typically the premolars) without calculus. Subgingival calculus appeared first either independently or on the interproximal aspects of areas where supragingival calculus already existed.4 By the age of 30 years, all surfaces of all teeth had subgingival calculus without any pattern of predilection.
The Norwegian academicians received oral hygiene instructions and frequent preventive dental care throughout their lives. The Norwegians exhibited a marked reduction in the accumulation of calculus as compared with the Sri Lankan group. However, despite the fact that 80% of teenagers formed supragingival calculus on the facial surfaces of the upper molars and the lingual surfaces of lower incisors, no additional calculus formation occurred on other teeth, and its presence did not increase with the individual’s age.4
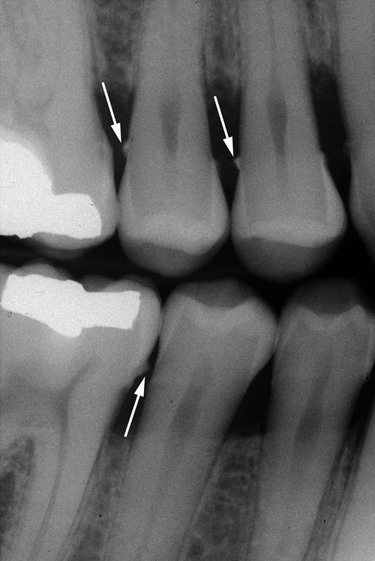
Both supragingival calculus and subgingival calculus may be seen on radiographs (see Chapter 32). Highly calcified interproximal calculus deposits are readily detectable as radiopaque projections that protrude into the interdental spaces (Figure 7-5). However, the sensitivity level of detecting calculus by radiographs is inconsistent.24 The location of calculus does not indicate the bottom of the periodontal pocket, because the most apical plaque is not sufficiently calcified to be visible on radiographs.
Composition
Inorganic Content.
Supragingival calculus consists of inorganic (70% to 90%52) and organic components. The major inorganic proportions of calculus have been reported as approximately 76% calcium phosphate, (Ca3[PO4]2); 3% calcium carbonate (CaCO3); and traces of magnesium phosphate (Mg3[PO4]2) and other metals.176 The percentage of inorganic constituents in calculus is similar to that of other calcified tissues of the body. The principal inorganic components have been reported as approximately 39% calcium, 19% phosphorus, 2% carbon dioxide, and 1% magnesium as well as trace amounts of sodium, zinc, strontium, bromine, copper, manganese, tungsten, gold, aluminum, silicon, iron, and fluorine.113
At least two thirds of the inorganic component is crystalline in structure.89 The four main crystal forms and their approximate percentages are as follows: hydroxyapatite, 58%; magnesium whitlockite, 21%; octacalcium phosphate, 12%; and brushite, 9%.
Two or more crystal forms are typically found in a sample of calculus. Hydroxyapatite and octacalcium phosphate are detected most frequently (i.e., in 97% to 100% of all supragingival calculus) and constitute the bulk of the specimen. Brushite is more common in the mandibular anterior region, and magnesium whitlockite is found in the posterior areas. The incidence of the four crystal forms varies with the age of the deposit.16
Organic Content.
The organic component of calculus consists of a mixture of protein–polysaccharide complexes, desquamated epithelial cells, leukocytes, and various types of microorganisms.99
Between 1.9% and 9.1% of the organic component is carbohydrate, which consists of galactose, glucose, rhamnose, mannose, glucuronic acid, galactosamine, and sometimes arabinose, galacturonic acid, and glucosamine.92,98,161 All of these organic components are present in salivary glycoprotein, with the exception of arabinose and rhamnose. Salivary proteins account for 5.9% to 8.2% of the organic component of calculus and include most amino acids. Lipids account for 0.2% of the organic content in the form of neutral fats, free fatty acids, cholesterol, cholesterol esters, and phospholipids.93
The composition of subgingival calculus is similar to that of supragingival calculus, with some differences. It has the same hydroxyapatite content, more magnesium whitlockite, and less brushite and octacalcium phosphate.142,165 The ratio of calcium to phosphate is higher subgingivally, and the sodium content increases with the depth of periodontal pockets.94 These altered compositions may be attributed to the origin of subgingival calculus being plasma, whereas supragingival calculus is partially composed of saliva constituents. Salivary proteins present in supragingival calculus are not found subgingivally.11 Dental calculus, salivary duct calculus, and calcified dental tissues are similar in inorganic composition.
Attachment to the Tooth Surface
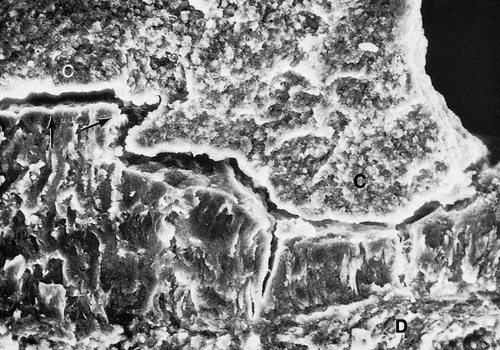
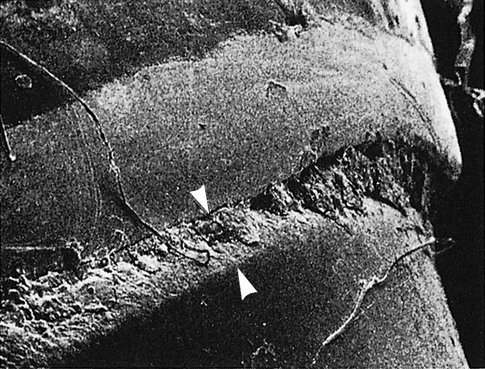
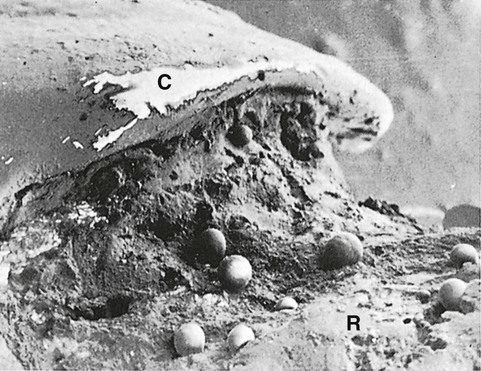
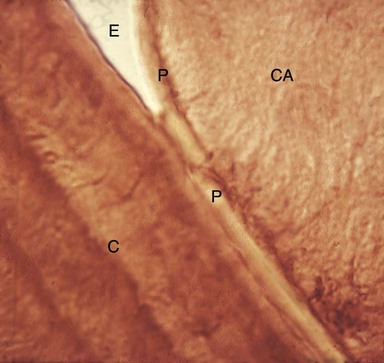
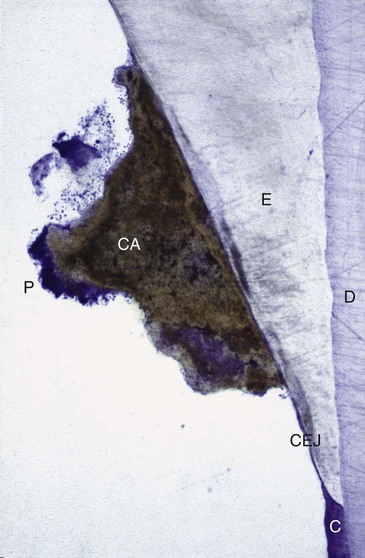
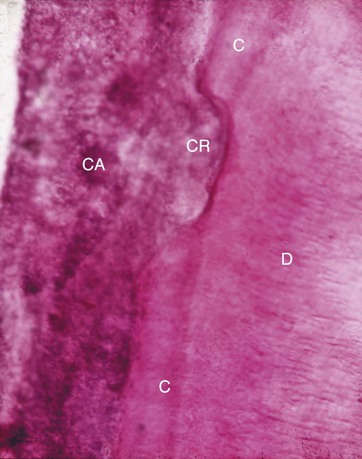
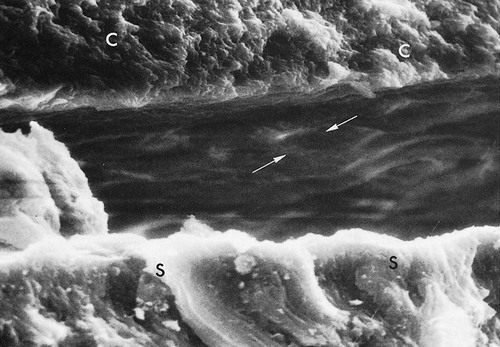
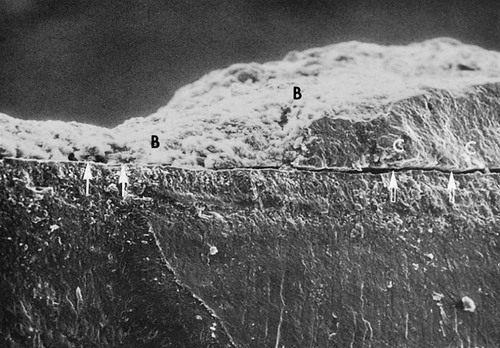
Differences in the manner in which calculus is attached to the tooth surface affect the relative ease or difficulty encountered during its removal. Four modes of attachment have been described.85,145,152,181 Attachment by means of an organic pellicle on cementum is depicted in Figure 7-6, and attachment on enamel is shown in Figure 7-7. Mechanical locking into surface irregularities, such as caries lesions or resorption lacunae, is illustrated in Figure 7-8. The fourth mode of attachment consists of the close adaptation of the undersurface of calculus to depressions or gently sloping mounds of the unaltered cementum surface,161 as shown in Figure 7-9, and the penetration of bacterial calculus into cementum, as shown in Figures 7-10 and 7-11.
Formation
Calculus is mineralized dental plaque. The soft plaque is hardened by the precipitation of mineral salts, which usually starts between the first and fourteenth days of plaque formation. Calcification has been reported to occur within as little as 4 to 8 hours.166 Calcifying plaques may become 50% mineralized in 2 days and 60% to 90% mineralized in 12 days.112,146,156 All plaque does not necessarily undergo calcification. Early plaque contains a small amount of inorganic material, which increases as the plaque develops into calculus. Plaque that does not develop into calculus reaches a plateau of maximal mineral content within 2 days.147 Microorganisms are not always essential in calculus formation, because calculus occurs readily in germ-free rodents.59
Saliva is the source of mineralization for supragingival calculus, whereas the serum transudate called gingival crevicular fluid furnishes the minerals for subgingival calculus.71,163 The calcium concentration/content in plaque is 2 to 20 times that found in saliva.16 Early plaque of heavy calculus formers contains more calcium, three times more phosphorus, and less potassium than that of noncalculus formers, thereby suggesting that phosphorus may be more critical than calcium for plaque mineralization.99 Calcification entails the binding of calcium ions to the carbohydrate–protein complexes of the organic matrix and the precipitation of crystalline calcium phosphate salts.97 Crystals form initially in the intercellular matrix and on the bacterial surfaces and finally within the bacteria.53,182
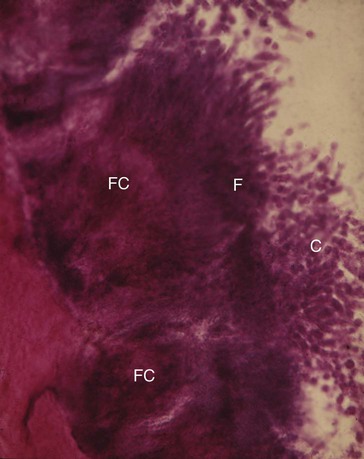
The calcification of supragingival plaque and in the attached component of subgingival plaque begins along the inner surface adjacent to the tooth. Separate foci of calcification increase in size and coalesce to form solid masses of calculus (see Figure 7-11). Calcification may be accompanied by alterations in the bacterial content and staining qualities of the plaque. As calcification progresses, the number of filamentous bacteria increases, and the foci of calcification change from basophilic to eosinophilic. There is a reduction in the staining intensity of groups that exhibit a positive periodic acid–Schiff reaction.
Sulfhydryl and amino groups are also reduced and instead stain with toluidine blue, which is initially orthochromatic but which becomes metachromatic and disappears.172 Calculus is formed in layers, which are often separated by a thin cuticle that becomes embedded in the calculus as calcification progresses.101
The initiation of calcification and the rate of calculus accumulation vary among individuals, among tooth variety in the same dentition, and at different times in the same person.113,168 On the basis of these differences, persons may be classified as heavy, moderate, or slight calculus formers or as noncalculus formers. The average daily increment in calculus formers is from 0.10% to 0.15% of dry weight calculus.156,168 Calculus formation continues until it reaches a maximum, after which it may be reduced in amount. The time required to reach the maximal level has been reported to be between 10 weeks32 and 6 months.170
Theories Regarding the Mineralization of Calculus.
The theoretical mechanisms by which plaque becomes mineralized can be stratified into two categories.114
1. Mineral precipitation results from a local rise in the degree of saturation of calcium and phosphate ions, which may be brought about in the following several ways:
• A rise in the pH of the saliva causes the precipitation of calcium phosphate salts by lowering the precipitation constant. The pH may be elevated by the loss of carbon dioxide and the formation of ammonia by dental plaque bacteria or by protein degradation during stagnation.15,66
• Colloidal proteins in saliva bind calcium and phosphate ions and maintain a supersaturated solution with respect to calcium phosphate salts. With the stagnation of saliva, colloids settle out, and the supersaturated state is no longer maintained, thereby leading to the precipitation of calcium phosphate salts.130,149
• Phosphatase liberated from dental plaque, desquamated epithelial cells, or bacteria precipitates calcium phosphate by hydrolyzing organic phosphates in saliva, thereby increasing the concentration of free phosphate ions.175 Esterase is another enzyme that is present in the cocci and filamentous organisms, leukocytes, macrophages, and desquamated epithelial cells of dental plaque.9 Esterase may initiate calcification by hydrolyzing fatty esters into free fatty acids. The fatty acids form soaps with calcium and magnesium that are later converted into the less-soluble calcium phosphate salts.
2. Seeding agents induce small foci of calcification that enlarge and coalesce to form a calcified mass.115 This concept has been referred to as the epitactic concept or, more appropriately, as heterogeneous nucleation. The seeding agents in calculus formation are not known, but it is suspected that the intercellular matrix of plaque plays an active role.100,112,182 The carbohydrate–protein complexes may initiate calcification by removing calcium from the saliva (chelation) and binding with it to form nuclei that induce the subsequent deposition of minerals.97,171
Role of Microorganisms in the Mineralization of Calculus.
Mineralization of plaque generally starts extracellularly around both gram-positive and gram-negative organisms, but it may also start intracellularly.87 Filamentous organisms, diphtheroids, and Bacterionema and Veillonella species have the ability to form intracellular apatite crystals (see Figure 7-11). Mineralization spreads until the matrix and the bacteria are calcified.53,183
Bacterial plaque may actively participate in the mineralization of calculus by forming phosphatases, which changes the pH of the plaque and induces mineralization,40,97 but the prevalent opinion is that these bacteria are only passively involved53,137,175 and are simply calcified with other plaque components. The occurrence of calculus-like deposits in germ-free animals supports this opinion.59 However, other experiments suggest that transmissible factors are involved in calculus formation and that penicillin in the diets of some of these animals reduces calculus formation.10
Etiologic Significance
Distinguishing between the effects of calculus and plaque on the gingiva is difficult, because calculus is always covered with a nonmineralized layer of plaque.147 A positive correlation between the presence of calculus and the prevalence of gingivitis exists,132 but this correlation is not as great as that between plaque and gingivitis.55 The initiation of periodontal disease in young people is closely related to plaque accumulation, whereas calculus accumulation is more prevalent in chronic periodontitis found in older adults.55,90
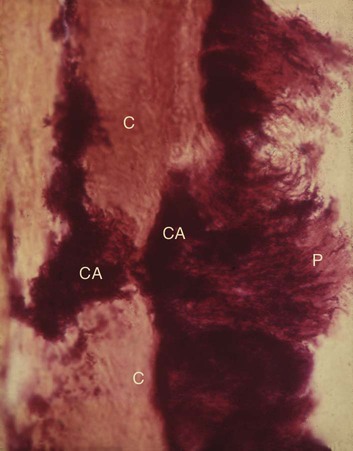
Calculus does not contribute directly to gingival inflammation, but it provides a fixed nidus for the continued accumulation of plaque and its retention in close proximity to the gingiva (Figure 7-12). Subgingival calculus is likely to be the product rather than the cause of periodontal pockets. Plaque initiates gingival inflammation, which leads to pocket formation, and the pocket in turn provides a sheltered area for plaque and bacterial accumulation. The increased flow of gingival fluid associated with gingival inflammation provides the minerals that mineralize the continually accumulating plaque that results in the formation of subgingival calculus (Figure 7-13). Over a 6-year period, Albandar and colleagues observed 156 teenagers with aggressive periodontitis.2 They noted that areas with detectable subgingival calculus at the initiation of the study were much more likely to experience a loss of periodontal attachment than sites that did not initially exhibit subgingival calculus.
Materia Alba, Food Debris, and Dental Stains
Materia alba is an accumulation of microorganisms, desquamated epithelial cells, leukocytes, and a mixture of salivary proteins and lipids, with few or no food particles; it lacks the regular internal pattern observed in plaque.148 It is a yellow or grayish-white, soft, sticky deposit, and it is somewhat less adherent than dental plaque. The irritating effect of materia alba on the gingiva is caused by bacteria and their products.
Most food debris is rapidly liquefied by bacterial enzymes and cleared from the oral cavity by salivary flow and the mechanical action of the tongue, cheeks, and lips. The rate of clearance from the oral cavity varies with the type of food and the individual. Aqueous solutions are typically cleared within 15 minutes, whereas sticky foods may adhere for more than 1 hour.88,169 Dental plaque is not a derivative of food debris, and food debris is not an important cause of gingivitis.39,75
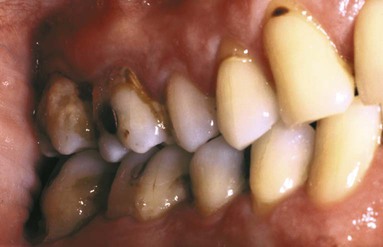
Pigmented deposits on the tooth surface are called dental stains. Stains are primarily an aesthetic problem and do not cause inflammation of the gingiva. The use of tobacco products (Figure 7-14), coffee, tea, certain mouthrinses, and pigments in foods can contribute to stain formation.95,162
Other Predisposing Factors
Iatrogenic Factors
Deficiencies in the quality of dental restorations or prostheses are contributing factors to gingival inflammation and periodontal destruction. Inadequate dental procedures that contribute to the deterioration of the periodontal tissues are referred to as iatrogenic factors. Iatrogenic endodontic complications that can adversely affect the periodontium include root perforations, vertical root fractures, and endodontic failures that may necessitate tooth extraction.180,181
Characteristics of dental restorations and removable partial dentures that are important to the maintenance of periodontal health include the location of the gingival margin for the restoration, the space between the margin of the restoration and the unprepared tooth, the contour of the restorations, the occlusion, the materials used in the restoration, the restorative procedure itself, and the design of the removable partial denture. These characteristics are described in this chapter as they relate to the etiology of periodontal disease. A more comprehensive review with special emphasis on the interrelationship between restorative procedures and the periodontal status is presented in Chapter 67.
Margins of Restorations.
Overhanging margins of dental restorations contribute to the development of periodontal disease by (1) changing the ecologic balance of the gingival sulcus to an area that favors the growth of disease-associated organisms (predominantly gram-negative anaerobic species) at the expense of the health-associated organisms (predominantly gram-positive facultative species)88 and (2) inhibiting the patient’s access to remove accumulated plaque.
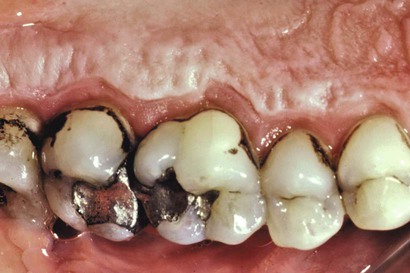
The frequency of overhanging margins on proximal restorations has varied in different studies from 16.5% to 75%.18,51,70,121 A highly significant statistical relationship has been reported between marginal defects and reduced bone height.18,60,70 The removal of overhangs allows for the more effective control of plaque, thereby resulting in a reduction of gingival inflammation and a small increase in radiographic alveolar bone support54,63,159 (Figure 7-15).
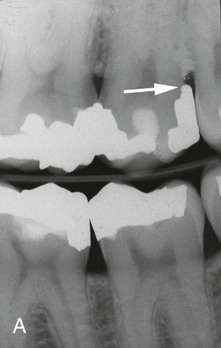
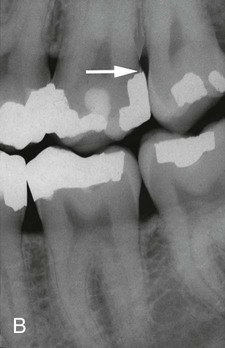
The location of the gingival margin of a restoration is directly related to the health status of the adjacent periodontal tissues.157 Numerous studies have shown a positive correlation between margins located apical to the marginal gingiva and the presence of gingival inflammation.51,67,74,135,159 Subgingival margins are associated with large amounts of plaque, more severe gingivitis, and deeper pockets. Even high-quality restorations, if placed subgingivally, will increase plaque accumulation, gingival inflammation,86,88,111,117,136,158 and the rate of gingival fluid flow.12,56 Margins placed at the level of the gingival crest induce less severe inflammation, whereas supragingival margins are associated with a degree of periodontal health similar to that seen with nonrestored interproximal surfaces.46,157
Roughness in the subgingival area is considered to be a major contributing factor to plaque buildup and subsequent gingival inflammation.157 The subgingival zone is composed of the margin of the restoration, the luting material, and the prepared and unprepared tooth surfaces. Sources of marginal roughness include grooves and scratches in the surface of acrylic resin, porcelain, or gold restorations (Figure 7-16); the separation of the restoration margin and the luting material from the cervical finish line, thereby exposing the rough surface of the prepared tooth (Figure 7-17); the dissolution and disintegration of the luting material between the preparation and the restoration, thereby leaving a space (Figure 7-18); and inadequate marginal fit of the restoration.157 Subgingival margins typically have a gap of 20 to 40 µm between the margin of the restoration and the unprepared tooth.155 Colonization of this gap by bacterial plaque undoubtedly contributes to the detrimental effect of margins placed in a subgingival environment.

Retained Cement/Periimplantitis.
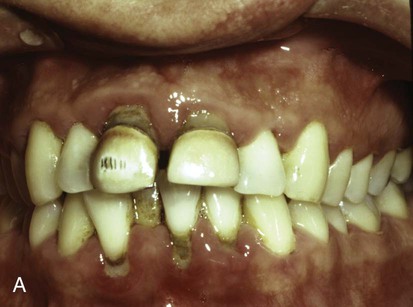
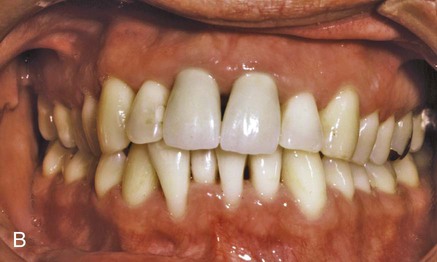
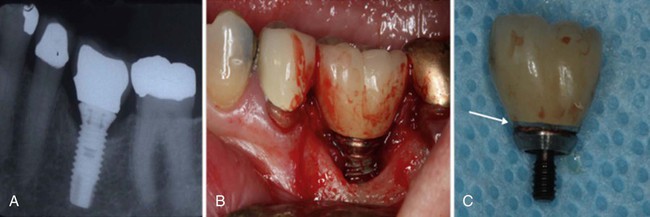
Periimplantitis is an inflammatory disease that affects the tissues around dental implants and that results in progressive bone loss (Figure 7-19, A and B).91 The prevalence of periimplantitis among implant supported prosthesis/restoration ranges from 28% to 56%.184 The early diagnosis of this condition and its proper management are critical to the longevity of the implant and supported prosthesis.
Classic studies that explored the composition of the periimplant biofilm in disease have shown that periimplantitis is an infection that is dominated by gram-negative bacteria with many similarities to the microbiota that is responsible for the development of periodontal and periimplant diseases, including Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, Tannerella forsythia, and Fusobacterium species. Recently, the application of new microbiologic approaches have allowed for a broader characterization of the periimplant microbiome, which has led to the identification of periimplant-specific microbial lineages such as Streptococcus mutans and Butyrivibrio fibrisolvens.84,108,144
A larger proportion of black-pigmented bacteria are found at periimplantitis sites in dentate patients as compared with edentulous patients. Presumably these periimplantitis infectious organisms arise from the sulcular microbiota of the natural dentition.7 Consequently, it is critical that practitioners evaluate the patient’s periodontal status and provide appropriate periodontal therapy in conjunction with implant rehabilitation.
Local factors such as retained residual cement or the inadequate seating of the implant abutment or prosthesis can lead to bone loss around an implant (
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses