Dentin-Pulp Complex
Dentin and pulp have been treated separately in textbooks on dental histology largely because dentin is a hard connective tissue and pulp is a soft one. However, as explained in Chapter 1, dentin and pulp are related embryologically, histologically, and functionally; therefore, they are described together in this chapter.
Basic Structure of Dentin
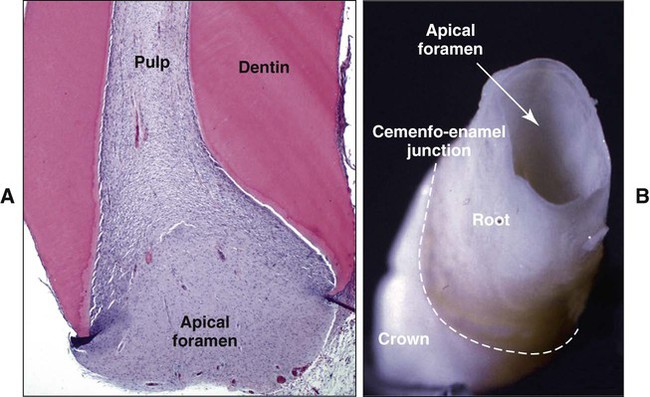
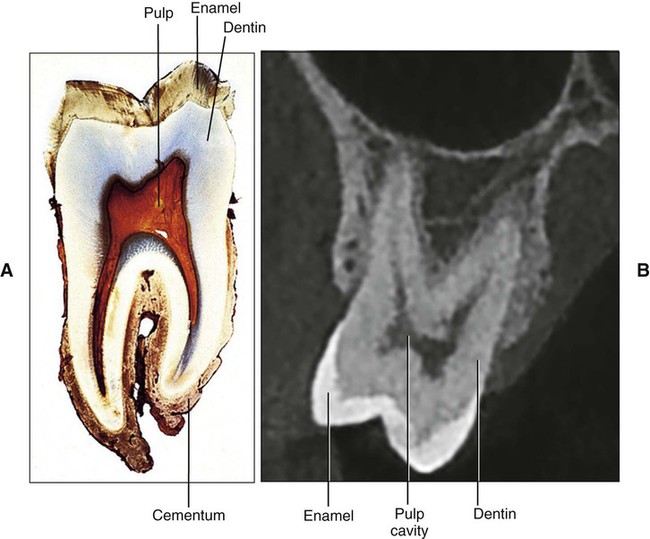
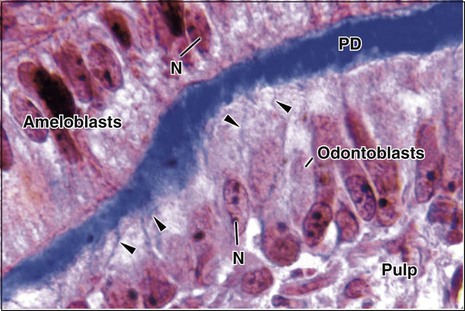
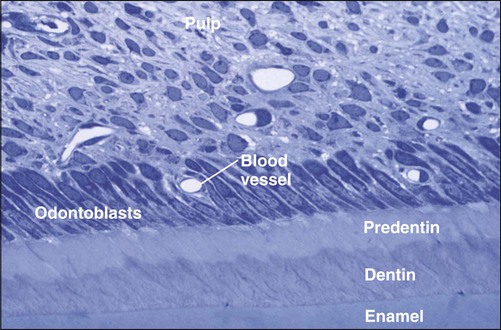
Dentin is the hard tissue portion of the pulp-dentin complex and forms the bulk of the tooth (Figure 8-1). Dentin is a bonelike matrix characterized by multiple closely packed dentinal tubules that traverse its entire thickness and contain the cytoplasmic extensions of odontoblasts that once formed the dentin and then maintain it. The cell bodies of the odontoblasts are aligned along the inner aspect of the dentin, against a layer of predentin, where they also form the peripheral boundary of the dental pulp.
The root canal (or root canal system, as it is called in multirooted teeth) terminates at the apical foramen, where the pulp and periodontal ligament meet and the main nerves and vessels enter and leave the tooth. In the developing tooth the apical foramen is wide and centrally located (Figure 8-2). As the tooth completes its development, the apical foramen becomes smaller in diameter and more eccentric in position. Sizes from 0.3 to 0.6 μm, with the larger diameter occurring in the palatal root of maxillary molars and the distal root of mandibular molars, are typical of the completed foramen. The foramen may be located at the very end, or anatomic apex, of the root but usually is located slightly more occlusally (0.5 to 0.75 mm) from the apex. If more than one foramen is present on a root, the largest is designated the apical foramen and the others the accessory foramina.
Composition, Formation, and Structure of Dentin
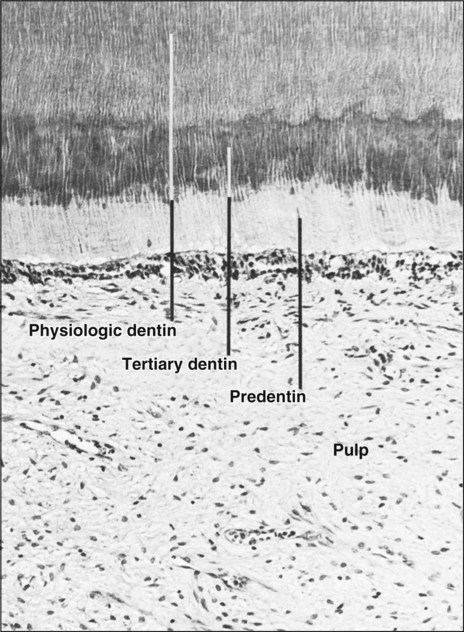
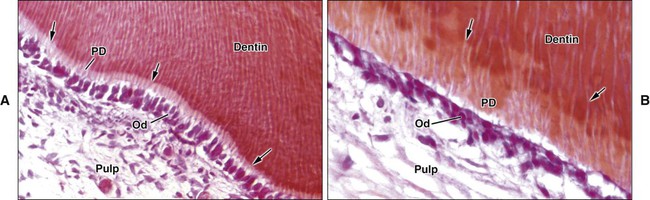
Dentin is first deposited as a layer of unmineralized matrix called predentin that varies in thickness (10 to 50 μm) and lines its innermost (pulpal) portion. Predentin consists principally of collagen and is similar to osteoid in bone; it is easy to identify in histologic sections because it stains less intensely than mineralized dentin (Figure 8-3). Predentin gradually mineralizes into dentin as various noncollagenous matrix proteins are incorporated at the mineralization front. The thickness of predentin remains constant because the amount that calcifies is balanced by the addition of new unmineralized matrix. Predentin is thickest at times when active dentinogenesis is occurring and diminishes in thickness with age.
Mature dentin is made up of approximately 70% inorganic material, 20% organic material and 10% of water. The inorganic component of dentin consists of substituted hydroxyapatite in the form of small plates. The organic phase is about 90% collagen (mainly type I with small amounts of types III and V) with fractional inclusions of various noncollagenous matrix proteins and lipids. Although studies have for a long time focused on identifying proteins specific to bone or dentin, it is now clear that bone matrix proteins can be found in dentin and that dentin matrix proteins also are present in bone (see Table 1-1).
Collagen type I acts as a scaffold that accommodates a large proportion (estimated at 56%) of the mineral in the holes and pores of fibrils. The noncollagenous matrix proteins regulate mineral deposition and can act as inhibitors, promoters, and/or stabilizers; their distribution is suggestive of their role. For instance, intact proteoglycans appear to be more concentrated in predentin and thus are believed to prevent the premature mineralization of the organic matrix while collagen fibrils mature and attain the correct dimension. DPP is an unusual phosphoprotein. It has an isoelectric point of 1 and has numerous aspartic acid-serine-serine residues and many of its serine residues are phosphorylated. Having a high negative charge, DPP binds large amounts of calcium. In vitro studies show that DPP binds to collagen and is able to initiate hydroxyapatite formation. DSP and DMP1 are predominantly immunodetected in peritubular dentin (discussed later in the chapter), where they may inhibit its growth and thus prevent occlusion of the tubule. In addition to their codistribution, DSP and DMP1 exhibit similarities in biochemical features; they thus may have redundant or synergistic functions. DSPP mutations result in a variety of dental phenotypes, including dentin dysplasia and dentinogenesis imperfecta that affect both the primary and permanent dentition. There are three types of dentinogenesis imperfecta; type I is also associated with osteogenesis imperfecta. In both type I and II, the pulp chamber is no longer visible because abnormal dentin deposits in it (Figure 8-4). Mice that do not express DSPP or DMP1 show enlarged pulp chambers (as seen in type III dentinogenesis imperfecta), an increase in the thickness of predentin, and hypomineralization, indicating additional functions to the control of peritubular dentin. Noteworthy is that DSPP and DMP1 are present in bone and dentin as processed fragments and that absence of DMP1 has profound effects on bone.
Dentin is slightly harder than bone and softer than enamel. This difference can be distinguished readily on radiographs on which the dentin appears more radiolucent (darker) than enamel and more radiopaque (lighter) than pulp (see Figure 8-6, B). Because light can pass readily through the thin, highly mineralized enamel and can be reflected by the underlying yellowish dentin, the crown of a tooth also assumes such coloration. The thicker enamel does not permit light to pass through as readily, and in such teeth the crown appears whiter. Teeth with pulp disease or without a dental pulp often show discoloration of the dentin, which causes a darkening of the clinical crown.
Physically, dentin has an elastic quality that is important for the proper functioning of the tooth because the elasticity provides flexibility and prevents fracture of the overlying brittle enamel. Dentin and enamel are bound firmly at the dentinoenamel junction that appears microscopically, as seen in the previous chapter, as a well-defined scalloped border (see Figure 7-58). In the root of the tooth, the dentin is covered by cementum, and the junction between these two tissues is less distinct because, in the human being, they intermingle.
Types of Dentin
Primary Dentin
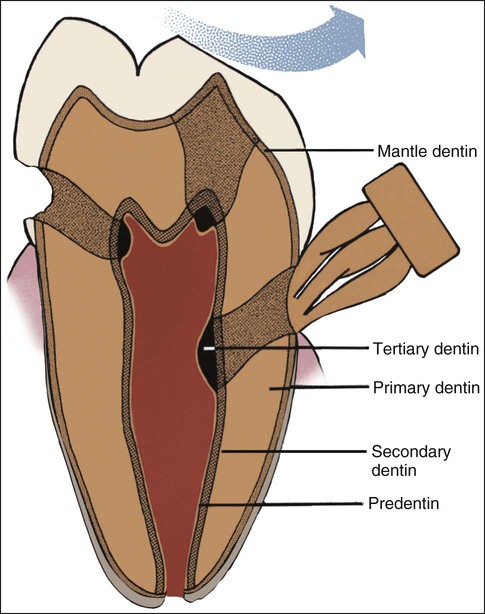
Most of the tooth is formed by primary dentin, which outlines the pulp chamber and is referred to as circumpulpal dentin (see Figure 8-1). The outer layer, near enamel or cementum, differs from the rest of the primary dentin in the way it is mineralized and in the structural interrelation between the collagenous and noncollagenous matrix components. This outer layer is called mantle dentin; the term, however, generally is used to refer to the outer layer in coronal dentin.
Secondary Dentin
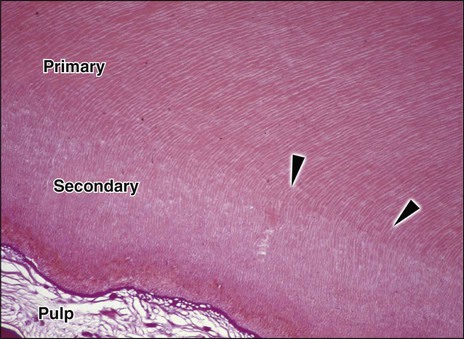
Secondary dentin develops after root formation has been completed and represents the continuing, but much slower, deposition of dentin by odontoblasts (Figure 8-5). Secondary dentin has a tubular structure that, though less regular, is for the most part continuous with that of the primary dentin. The ratio of mineral to organic material is the same as for primary dentin. Secondary dentin is not deposited evenly around the periphery of the pulp chamber, especially in the molar teeth. The greater deposition of secondary dentin on the roof and floor of the chamber leads to an asymmetrical reduction in its size and shape (Figure 8-6). These changes in the pulp space, clinically referred to as pulp recession, can be detected readily on histologic sections and radiographs (see Figure 8-6), and are important in determining the form of cavity preparation for certain dental restorative procedures. For example, preparation of the tooth for a full crown in a young patient presents a substantial risk of involving the dental pulp by mechanically exposing a pulp horn. In an older patient the pulp horn has receded and presents less danger. Some evidence suggests that the tubules of secondary dentin sclerose (fill with calcified material) more readily than those of primary dentin. This process tends to reduce the overall permeability of the dentin, thereby protecting the pulp.
Tertiary Dentin
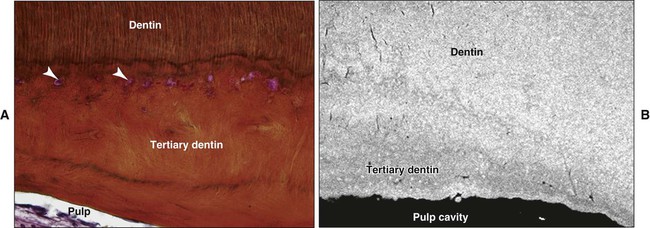
Tertiary dentin (also referred to as reactive or reparative dentin) is produced in reaction to various stimuli, such as attrition, caries, or a restorative dental procedure. Unlike primary or secondary dentin that forms along the entire pulp-dentin border, tertiary dentin is produced only by those cells directly affected by the stimulus. The quality (or architecture) and the quantity of tertiary dentin produced are related to the cellular response initiated, which depends on the intensity and duration of the stimulus. Tertiary dentin may have tubules continuous with those of secondary dentin, tubules sparse in number and irregularly arranged, or no tubules at all (Figure 8-7). The cells forming tertiary dentin line its surface or become included in the dentin; the latter case is referred to as osteodentin (Figure 8-8). Tertiary dentin is subclassified as reactionary or reparative dentin, the former deposited by preexisting odontoblasts and the latter by newly differentiated odontoblast-like cells.
Pattern of Dentin formation
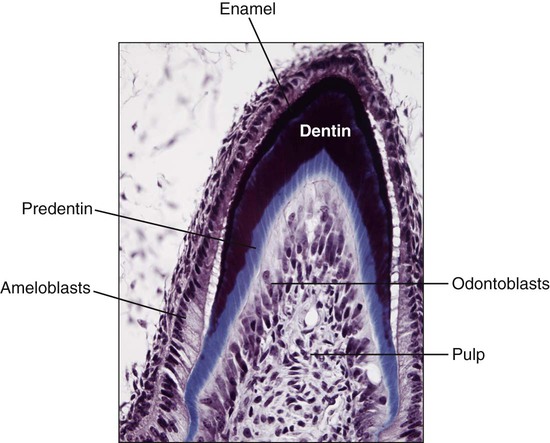
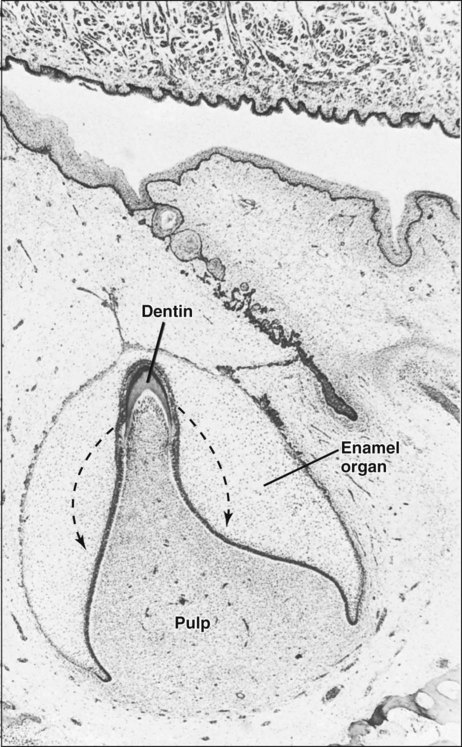
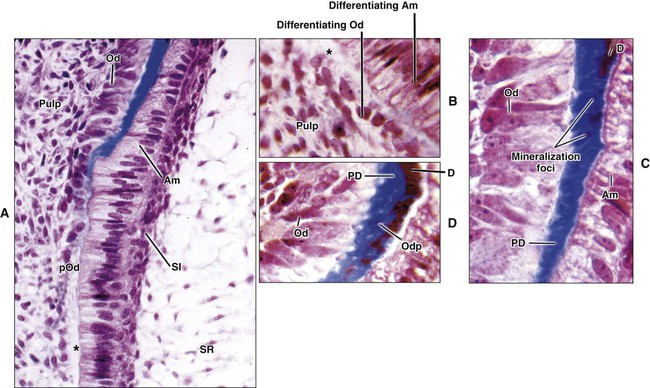
Dentin formation begins at the bell stage of tooth development in the papillary tissue adjacent to the concave tip of the folded inner enamel epithelium (Figure 8-9), the site where cuspal development begins. From that point, dentin formation spreads down the cusp slope as far as the cervical loop of the enamel organ, and the dentin thickens until all the coronal dentin is formed. In multicusped teeth, dentin formation begins independently at the sites of each future cusp tip and again spreads down the flanks of the cusp slopes until fusion with adjacent formative centers occurs. Dentin thus formed constitutes the dentin of the crown of the tooth, or coronal dentin.
Root dentin forms at a slightly later stage of development and requires the proliferation of epithelial cells (Hertwig’s epithelial root sheath) from the cervical loop of the enamel organ around the growing pulp to initiate the differentiation of root odontoblasts. The onset of root formation precedes the onset of tooth eruption, and by the time the tooth reaches its functional position, about two thirds of the root dentin will have been formed. Completion of root dentin formation does not occur in the deciduous tooth until about 18 months after it erupts and in the permanent tooth until 2 to 3 years after it erupts. During this period the tooth is said to have an open apex (Figure 8-2).
Dentinogenesis
Odontoblast Differentiation
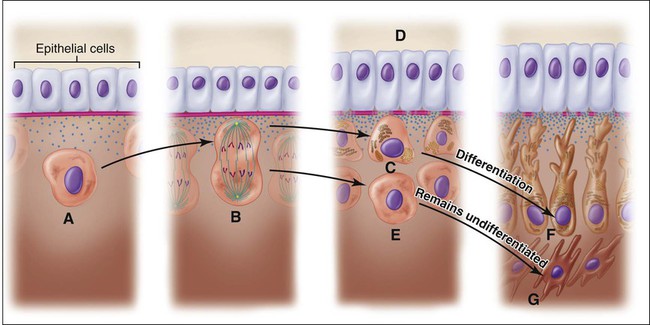
The differentiation of odontoblasts from the dental papilla in normal development is brought about by the expression of signaling molecules and growth factors in the cells of the inner enamel epithelium (see Chapter 5). Figures 8-10 and 8-11 illustrate the differentiation sequence. The dental papilla cells are small and undifferentiated, and they exhibit a central nucleus and few organelles. At this time they are separated from the inner enamel epithelium by an acellular zone that contains some fine collagen fibrils. Almost immediately after cells of the inner enamel epithelium reverse polarity, changes also occur in the adjacent dental papilla. The ectomesenchymal cells adjoining the acellular zone rapidly enlarge and elongate to become preodontoblasts first and then odontoblasts as their cytoplasm increases in volume to contain increasing amounts of protein-synthesizing organelles. The acellular zone between the dental papilla and the inner enamel epithelium gradually is eliminated as the odontoblasts differentiate and increase in size and occupy this zone. These newly differentiated cells are characterized by being highly polarized, with their nuclei positioned away from the inner enamel epithelium.
Formation of Mantle Dentin
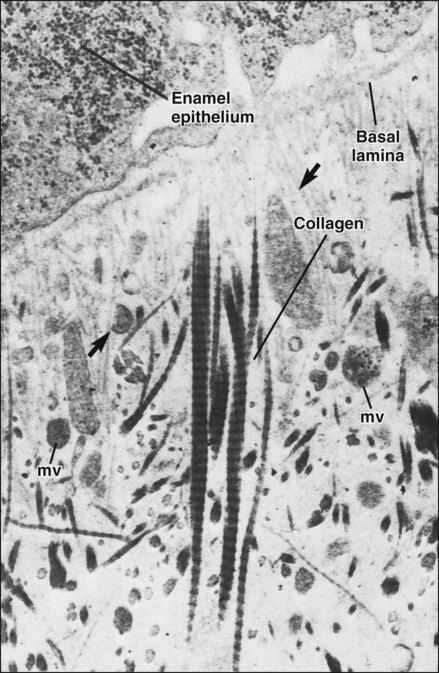
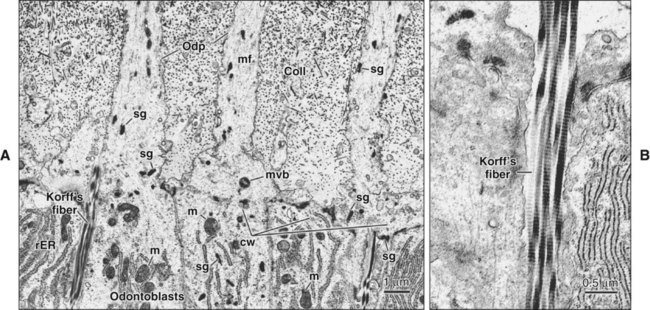
After the differentiation of odontoblasts, the next step in the production of dentin is formation of its organic matrix. The first sign of dentin formation is the appearance of distinct, large-diameter collagen fibrils (0.1 to 0.2 μm in diameter) called von Korff’s fibers (Figures 8-12 to 8-15). These fibers consist of collagen type III associated, at least initially, with fibronectin. These fibers originate deep among the odontoblasts, extend toward the inner enamel epithelium, and fan out in the structureless ground substance immediately below the epithelium. As the odontoblasts continue to increase in size, they also produce smaller collagen type I fibrils that orient themselves parallel to the future dentinoenamel junction (see Figure 8-15). In this way, a layer of mantle predentin appears.
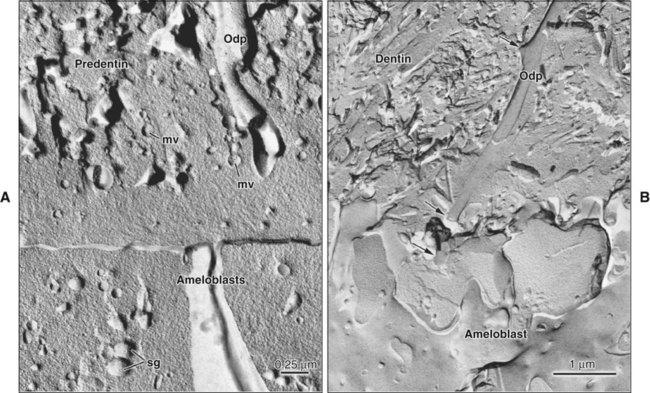
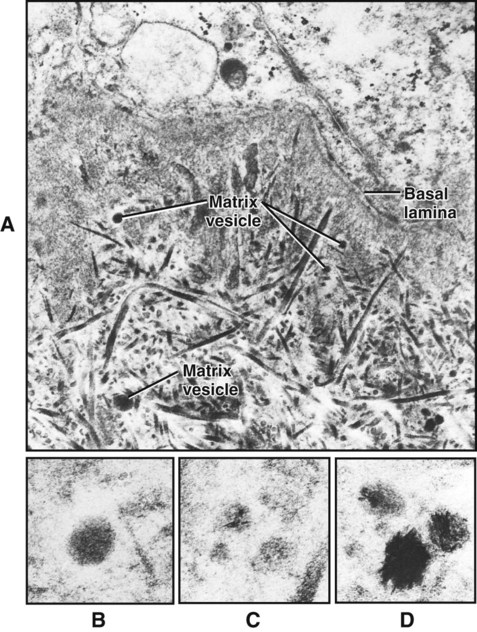
Coincident with this deposition of collagen, the plasma membrane of odontoblasts adjacent to the differentiating ameloblasts extends stubby processes into the forming extracellular matrix (Figure 8-16). On occasion one of these processes may penetrate the basal lamina and interpose itself between the cells of the inner enamel epithelium to form what later becomes an enamel spindle (see Chapter 7). As the odontoblast forms these processes, it also buds off a number of small, membrane-bound vesicles known as matrix vesicles, which come to lie superficially near the basal lamina (Figure 8-17; see also Figures 8-12 and 8-16, A). The odontoblast then develops a cell process, the odontoblast process or Tomes’ fiber, which is left behind in the forming dentin matrix as the odontoblast moves away toward the pulp (Figure 8-15). The mineral phase first appears within the matrix vesicles as single crystals believed to be seeded by phospholipids present in the vesicle membrane (see Figure 8-17). These crystals grow rapidly and rupture from the confines of the vesicle to spread as a cluster of crystallites that fuse with adjacent clusters to form a continuous layer of mineralized matrix. The deposition of mineral lags behind the formation of the organic matrix so that a layer of organic matrix, called predentin, always is found between the odontoblasts and the mineralization front. Following mineral seeding, noncollagenous matrix proteins produced by odontoblasts come into play to regulate mineral deposition. In this way coronal mantle dentin is formed in a layer approximately 15 to 20 μm thick onto which then is added the primary (circumpulpal) dentin.
Vascular Supply
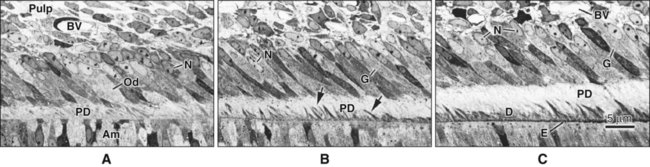
Chapter 1 stated the requirement for good blood supply during the formative phase of hard tissue formation. During dentinogenesis, interesting changes have been observed in the rat molar in the distribution and nature of the capillaries associated with the odontoblasts. When mantle dentin formation begins, capillaries are found beneath the newly differentiated odontoblasts. As circumpulpal dentinogenesis is initiated, some of these capillaries migrate between the odontoblasts (Figure 8-18), and at the same time their endothelium fenestrates to permit increased exchange. With the completion of dentinogenesis, they retreat from the odontoblast layer, and their endothelial lining once again becomes continuous.
Control of Mineralization
The problem of how mineral ions reach mineralization sites was reviewed in Chapter 1. In the case of dentinogenesis, some dispute exists because the junctions holding the odontoblasts together in a palisade arrangement are incomplete and thus leaky. Conceptually, simple percolation of tissue fluid supersaturated with calcium and phosphate ions could take place. However, calcium channels of the L type have been demonstrated in the basal plasma membrane of the odontoblast; significantly, when these are blocked, mineralization of the dentin is affected. The presence of alkaline phosphatase activity and calcium adenosinetriphosphatase activity at the distal end of the cell also is consistent with a cellular implication in the transport and release of mineral ions into the forming dentin layer.
Pattern of Mineralization
Histologically, two patterns of dentin mineralization can be observed—globular and linear calcification (Figures 8-19 and 8-20)—that seem to depend on the rate of dentin formation. Globular (or calcospheric) calcification involves the deposition of crystals in several discrete areas of matrix by heterogeneous capture in collagen. With continued crystal growth, globular masses are formed that continue to enlarge and eventually fuse to form a single calcified mass. This pattern of mineralization is best seen in the mantle dentin region, where matrix vesicles give rise to mineralization foci that grow and coalesce. In circumpulpal dentin the mineralization front can progress in a globular or linear pattern. The size of the globules seems to depend on the rate of dentin deposition, with the largest globules occurring where dentin deposition is fastest. When the rate of formation progresses slowly, the mineralization front appears more uniform and the process is said to be linear.
Formation of Root Dentin
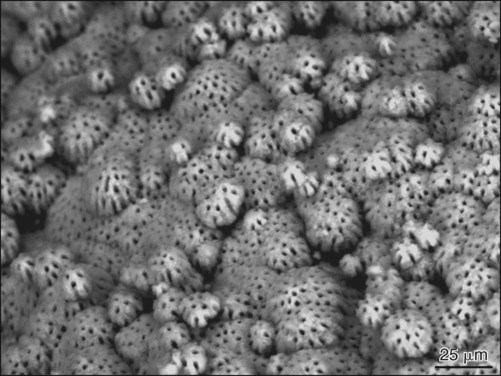
The epithelial cells of Hertwig’s root sheath initiate the differentiation of odontoblasts that form root dentin (Figure 8-21 and see Chapter 9). Root dentin forms similarly to coronal dentin, but some differences have been reported. The outermost layer of root dentin, the equivalent of mantle dentin in the crown, shows differences in collagen fiber orientation and organization, in part because the collagen fibers from cementum blend with those of dentin (see Chapter 9). Some reports also indicate that the phosphoprotein content of root dentin differs, that it forms at a slower speed, and that its degree of mineralization differs from that of coronal dentin. These possible differences, however, need to be ascertained and simply may reflect the anatomic context of root dentin rather than fundamental differences.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses