Chapter 8
Clinical Outcome
Accurate, predictive assessment of the behaviour of oral precancers remains elusive in clinical practice. Yet establishing and defining clinical outcomes following the treatment of patients with potentially malignant disorders is absolutely fundamental to any discussion of management protocols. It is astonishing how little agreement exists in the literature on how such data should be analysed and discussed.
Clinical outcome may be defined overall as a measurement of how a patient feels, functions or indeed survives following disease recognition and treatment. Clearly, the most significant clinical outcome and the principal reason for identification and intervention in oral precancer is the prevention of malignant transformation and halting the development of oral cancer. This specific issue, however, will be explored and analysed in Chapter 9. In this chapter we will discuss in some detail the important overall clinical outcomes that may follow on from the diagnosis and treatment of oral potentially malignant disorders.
There are a number of difficulties inherent in trying to draw conclusions applicable to individual patient prognoses. These include:
- The lack of agreed terminology in published papers.
- The wide range of different types of lesions included in studies, particularly the analysis of non-dysplastic white patches, which unfortunately seems to confound the entire oral precancer literature.
- The varied treatment regimes and uncoordinated follow-up periods involved.
Indeed, many studies in the literature are highly subjective and entirely retrospective in nature.
If one combines a range of study data obtained through the years, however, it is possible to make some very broad observations. In general, approximately 40% of dysplastic lesions probably change little or progress only slightly with time, whereas 20% may actually regress spontaneously. A further 20% will increase in size and extent, whilst the remaining 20% are at risk of progressing to malignancy. It must be emphasised that these are overall estimates based on population studies of both treated and non-treated lesions and, perhaps most significantly and very unhelpfully, do not in any way inform individual patient management.
It is surprisingly difficult to write definitively about the clinical outcome of oral precancer lesions because the available literature is so confusing in relation to terminology. There remains an enormous inconsistency between different authors and different studies. Similarly, the demonstrable lack of understanding of the natural history of oral potentially malignant disorders only confounds rather than clarifies our understanding.
What is also striking in reviewing published papers is the consensus observation that none of the perceived important and standard clinicopathological factors help to accurately predict clinical outcome [1]. Such factors include the clinical appearance of individual lesions, their anatomical site of origin, histopathological assessment, associated patient risk factor behaviour, and patient age or gender. Clinical outcome is itself, of course, influenced if not actually determined by the clinical management applied to individual cases. The fundamental problem with oral precancer remains the lack of internationally accepted treatment guidelines, a poor evidence base in the literature, and the paucity of relevant prospective, randomised clinical trials comparing different management strategies.
Many observational, anecdotal and retrospective papers have been published through the years. Unfortunately they add very little to our true understanding of the natural history and clinical outcome of oral potentially malignant disease, especially in relation to individual patient diagnosis and management. It is worth looking at two relatively recently published papers in detail because these illustrate quite well some of the inherent problems in trying to determine the true significance of clinical outcome for oral precancer patients.
Holmstrup et al. in 2006 reviewed long-term treatment outcomes for 269 premalignant lesions in 236 patients seen over a 20-year period [2]. One group of patients underwent surgical intervention and the other received no surgery. Histopathological data and risk of malignant transformation were reported for 94 surgically excised lesions and compared with 175 non-surgically treated lesions. However, as a retrospective review, the authors admit that they are unable to explain how treatment decisions were made for all of their reported cases. They stated that 12% of surgically treated lesions developed carcinoma after a mean follow-up period of 7.5 years, compared with only 4% of non-surgically treated lesions after a mean of 6.6 years follow up. It is important to appreciate, however, that the two groups were actually non-comparable. Whilst 71% of the surgically excised lesions were dysplastic and often severely so, only 12% of the non-surgically treated lesions displayed any dysplasia whatsoever and if present it was usually categorised as mild.
This is one of the commonest and most persistent difficulties found in the literature when one attempts to interpret the true relevance of clinical outcome papers. If many cases are included in analyses that have never shown dysplasia on biopsy, comparison of outcome with dysplasia patients is probably entirely meaningless. So that, despite the large number of patients included and the availability of follow-up data for up to 20 years (although the mean was actually only between 5.5 years for non-surgical and 6.8 years for surgical cases), Holmstrup’s series is a non-randomised retrospective review with effectively non-comparable groups. This renders the results of questionable or perhaps even no true biological significance.
Arduino et al. in 2009, similarly, retrospectively reviewed the outcome of 207 oral dysplastic lesions treated over a 16-year period by a variety of surgical excisions, cryotherapies or clinical observation alone [1]. They noted that over a median 4.5-year follow-up period, 39% lesions disappeared, 20% remained stable, 34% developed new dysplastic lesions and 7% developed a squamous cell carcinoma [1]. Whilst the authors of this paper found no significant differences in outcome between treated and untreated lesions, the study group was so heterogeneous and patient data collected over such a long study period that once again the results must be interpreted with real caution.
The limitations of the above non-randomised patient studies are obvious and the potential for clinician bias in treatment selection, however well meaning, severely limits scientific validity and clinical relevance. Interestingly, both the above papers share the same unsurprising conclusion that randomised controlled clinical trials are advised because of the inherent weaknesses of such retrospective reviews.
It is probably true to say that, whilst there is no strictly universally agreed hierarchy of biomedical evidence, randomised controlled trials rank much higher in terms of scientific methodology than observational studies, expert opinion and anecdotal experience. Many authors would also now add that systematic reviews and meta-analyses should rank even higher than controlled trials as they combine data from a variety of different studies and sources.
Randomised controlled trials are favoured because they probably best eliminate investigator bias and potential confounding factors by randomly assigning patients to one type of intervention or another, or indeed often to a non-intervention or placebo arm. Whilst this may be scientifically valid, such trials are not always appropriate or acceptable to patients or clinicians in the medical environment. As we have seen in Chapter 7, there have been attempts to design randomised trials for oral precancer medical treatments but these have largely proved unsuccessful, unreliable and have produced little of clinical relevance.
Many authors, as exampled above, correctly bemoan the lack of meaningful randomised trials for oral precancer management. However, there are clinical difficulties in randomising potentially malignant disease patients, especially to non-treatment study arms, when there remains such an unpredictable and biologically devastating risk of malignant transformation.
Cohort studies, on the other hand, do offer opportunities for longitudinal observational analysis of a group of people who share either a common characteristic or experience within a defined time period. There are a number of potential advantages to patient cohort studies in oral precancer research and these are summarised in Box 8.1. The longitudinal observation of a group of patients through time and the collection of data at regular intervals can actually produce data superior to those of cross-sectional and retrospective studies. Indeed, this is often the recommended methodology in observational epidemiology.
Box 8.1 Advantages of oral precancer patient cohort studies.
- Defined study population sharing common characteristics and risk factor behaviour
- Inclusion criteria facilitate recruitment of cases with proven dysplasia
- Standard diagnostic procedures
- Agreed treatment protocol and standardised intervention
- Consistent clinical decision making
- Longitudinal observation of subjects with documented clinical follow up
In the absence of multicentre, randomised, prospective clinical trials we will describe in this chapter the results of a series of patient cohort studies carried out in our dysplasia clinics in Newcastle upon Tyne in northern England. Whilst not necessarily universally applicable, these studies benefit from a defined and static patient population known to be at high risk of oral cancer, a consistent and coordinated clinical management protocol coordinated by one clinician, and a regular and consistent follow-up strategy, as outlined in Chapter 7.
Definitions of Clinical Outcome
It is important at the outset, before describing and attempting to analyse our patient cohort data, to define the parameters by which the clinical outcome and evolution of precancer disease should be assessed. As we have seen, the literature in this field is full of so many differing terminologies that meta-analysis is rendered extremely complicated, if not actually impossible. We recommend the following terms, which are summarised in Box 8.2. They are firmly based upon several observational studies we have carried out on cohorts of new patients presenting with dysplastic oral precancer lesions who have been carefully followed up subsequent to interventional laser surgery. The categories of malignant transformation and development of oral squamous cell carcinoma, however, will be discussed in Chapter 9.
Box 8.2 Definitions of clinical outcome.
| • Clinical resolution: | Patient is clinically free of oral precancer disease |
| • Persistent disease: | Oral precancer lesion persists clinically despite interventional treatment |
| • Recurrent disease: | Oral precancer lesion recurs at same site following previous excision |
| • Further disease: | New-site development of oral precancer lesion(s) |
| • Malignant transformation: | Oral squamous cell carcinoma arising at the site of a clinically recognised precursor lesion |
| • Oral cancer development: | Oral squamous cell carcinoma developing at a new site distant from precursor lesions |
Clinical Resolution (Disease-Free State)
Clinical resolution is the successful surgical removal or regression of a potentially malignant disorder resulting in clinically normal-looking oral mucosa with no further precancer lesion arising either at the site of previous disease or elsewhere in the oral cavity or oropharynx.
An example of clinical resolution following laser excision of a lateral tongue precancer lesion is shown in Figure 8.1. Figure 8.2 also illustrates an example of clinical resolution, although in this case floor of mouth scarring has created a white appearance which may be mistaken for persistent or recurrent leukoplakia. Such scarring may also give rise to confusion under VELscope® examination, where scar tissue can appear dark on tissue autofluorescence due to the increased vasculature in the immediately subjacent tissue.
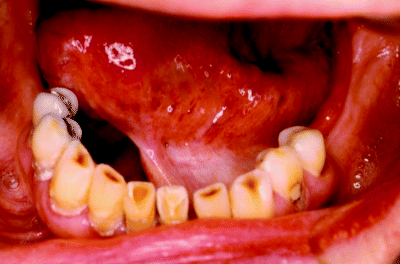
Figure 8.1 Clinical resolution following laser excision of a dysplastic leukoplakia from the lateral surface of the tongue. Two years post laser there is no evidence of further precancer disease at this site.
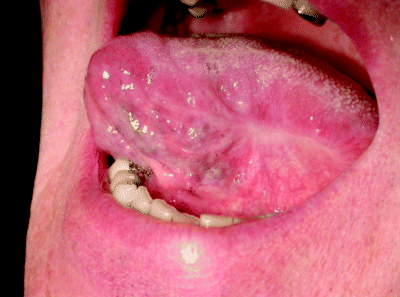
Figure 8.2 Clinical resolution following floor of mouth laser excision at 1 year postoperatively. Note the pale scar tissue which on superficial inspection may resemble leukoplakia.
Overall, in our studies we have observed that 76% of laser-treated patients may be disease-free 18 months after surgery, although this falls with increased length of follow-up periods, dropping to around 64% by 5 years. Thus, the longer patients are followed up, the more chance there is of identifying new potentially malignant disease development and this is a very important observation for clinical management strategies.
Persistent Disease
Persistent disease is the term used to describe potentially malignant lesions that persist at the same site following treatment. Thus, once postoperative healing has stabilised and despite whole lesion excision, there is evidence of a residual precancer lesion which can be seen and identified clinically.
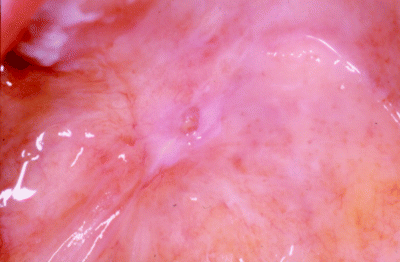
Such persistent lesions usually have an identical clinical appearance and exhibit the same histopathological features as their original presenting lesion. A clinical example of persistent leukoplakia following laser excision of a soft palate lesion is shown in Figure 8.3.
Figure 8.3 An example of persistent disease, illustrating an erosive leukoplakia at the site of a recent soft palate laser excision.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


