Chapter 6
Shaping the Root Canal System to Promote Effective Disinfection
Ove A. Peters
Department of Endodontics, University of the Pacific, San Francisco, CA, USA
Frank Paqué
Department of Preventive Dentistry, Periodontology, and Cariology, University of Zurich, Zürich, Switzerland
Introduction
It has been well established over the past 50 years that the endodontic disease, in other words, the presence of apical periodontitis, has a microbial pathogenesis (1, 2). Consequently, root canal treatment is performed to treat endodontic disease by eradicating bacteria from the root canal space.
Furthermore, it is currently accepted that disinfection and subsequent obturation of the root canal space requires mechanical enlargement of the main canals (3), and the vast majority of techniques and instruments today are based on this objective.
Root canal shaping thus serves two main purposes in canal disinfection: direct mechanistic elimination of intracanal tissue and pathogens and providing optimal space for irrigant and medicament delivery (4–6). Clinical success appears to be enhanced in the presence of better disinfection (7, 8) using antimicrobials delivered into the shaped root canal. Consequently the process of root canal preparation should be considered a main driver of clinical success in endodontics.
Using conventional anaerobic culturing, widely varying clinical preparation techniques have been associated with a reduction of intracanal bacteria to a level of 10–100 CFU/ml (9–14). However, it is highly unlikely that under clinical conditions any root canal will be rendered sterile.
This finding is not incompatible with clinical success. In fact, abundant reports of clinically successful as well as failure cases confirm the presence of bacteria and immune cells inside root canal systems in both healed cases and cases with residual lesions. (15–17). Figure 6.1 illustrates an isthmus area of a mandibular first molar after single-visit endodontic therapy, in which bacteria are present encased in dentin shavings.

Figure 6.1 Light microscopic view of a transverse section through the apical portion of the mesial root of a right mandibular first molar (a) The surgical artifact (AT) into the root dentin did not reach or damage the instrumented mesiolingual and mesiobuccal canals, which were incompletely obturated with gutta-percha cones. The isthmus (IS) connecting the canals is magnified in (b); the area indicated with the black arrowhead is further magnified in stages in (c) and (d), respectively. Note the un-instrumented isthmus with arcading profiles of Howship’s lacunae (HL) clogged with blue-stained bacterial mass (BA). Original magnifications: (a) ×316; (b) ×344; (c) ×3240; (d) ×3400.
(Nair et al. (17). Reproduced with permission of Mosby, Inc.)
The fate of such bacteria remaining in inaccessible canal areas, regardless of clinical technique (18), has been a matter of speculation: do they die from starvation (19), are they possibly killed by sealer components (20), or can they persist and cause posttreatment disease (21)?
It was demonstrated that well-filled and hence arguably well-shaped canals may still be associated with failing root canal treatments, mainly because of the fact that microorganisms remain in inaccessible areas of the root canal system (18, 21). Consequently, rather than sterilizing root canal systems, intracanal procedures are tailored to reduce microbial burden so that it becomes compatible with success by creating conditions that prohibit regrowth and persistent infection. It has been held that a threshold exists that may be reached with different canal shaping paradigms depending on numbers, virulence, and location of surviving microorganisms. Therefore, the main objective of root canal shaping is to provide an environment, in which apical disease is prevented or the body’s immune system can achieve the healing of apical disease. At the same time shaping is limited by the overall root anatomy and the deficiencies of currently available instruments.
Effect of canal preparation on intracanal bacteria
Using contemporary instruments, root canals can be prepared to desired shapes without obvious procedural errors as reviewed extensively earlier (4, 5, 22). However, it is obvious that mechanical canal preparation does not create contact with the full radicular surface and therefore all mechanical preparation is incomplete. The areas in red in Figure 6.2 demonstrate the canal surface that was not mechanically shaped; more specifically, it did not change by more than 20 µm, which was the resolution of the system. This observation is confirmed by histological and scanning electron microscopic observation that typically reveal mechanically unshaped areas (see inset in Figure 6.2).
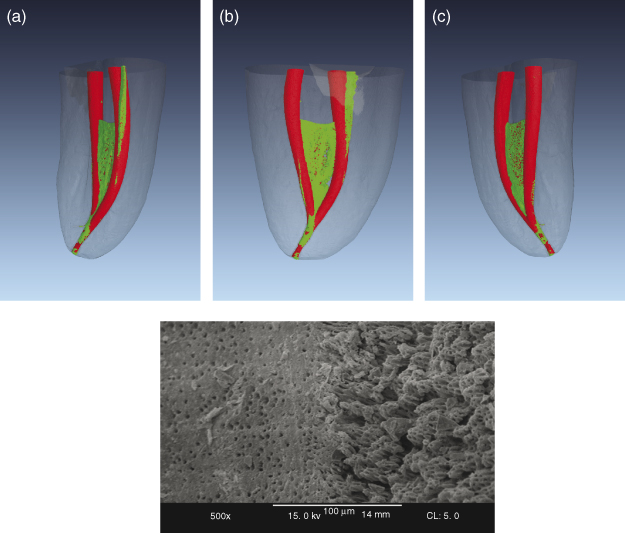
Figure 6.2 (a–c) Microcomputed tomography (MCT) images of the effect of root canal shaping of the mesial root canal system of a mandibular molar, seen from three different angles. Un-instrumented canal areas (static voxels) are depicted in green and instrumented canal areas in red. Note the isthmus area and other sections that were not altered during shaping. Final apical shaping was done with ProTaper Universal F3. Inset shows a scanning electron micrograph (×500 magnification) of areas that were in contact with an endodontic instrument (a) and a section of canal wall that shows the so-called calcospherites of the mineralization front after the removal of the pulp tissue by NaOCl.
Nevertheless, it has been demonstrated that mechanical instrumentation, alone or in combination with an inert flushing solution such as saline, is effective in significantly reducing bacterial load. Several authors have investigated the direct antimicrobial effect of canal instrumentation. First, in 1958, Ingle and Zeldow (23), using aerobic culturing methods, determined that the majority (65/89) of the clinical cases they sampled harbored bacteria prior to instrumentation. Only 13 of those became bacteria-free (=culture negative) after the first appointment. In their study turbidity of the culture medium was used as the sole indicator for bacterial presence and the authors cautioned that various numbers of microorganisms were needed to cause such turbidity.
Subsequently, Byström and Sundqvist (24) expanded this observation under conditions of anaerobic culturing in a clinical study of 15 single-rooted teeth diagnosed with apical periodontitis. They found that after the first appointment, in which canals were shaped with Hedström files to an apical size 40 to 1 mm short of the radiographic root length, intracanal bacterial numbers were reduced by 102–103 and even further in subsequent appointments. However, in 8/15 cases, complete elimination of microorganisms was not achieved even after five appointments (24).
Siqueira and colleagues (25) also used saline to irrigate root canal prepared with nickel–titanium (NiTi) hand and rotary instruments in-vitro. Utilizing anaerobic culturing, these authors found that mechanical instrumentation was effective in removing more than 90% of bacterial cells from the root canal. This effect appeared to depend on apical sizes since preparation with NiTi K-files to size 40 was significantly more effective in removing bacteria compared to GT hand files or ProFile rotaries used to shape to sizes 20 (0.12 taper) and 28 (0.06 taper), respectively (25).
Falk and Sedgley (26) introduced yet another experimental technique in the laboratory: genetically modified bacteria. They inoculated 30 permanent canines with bioluminescent Pseudomonas fluorescens and determined bacterial elimination by the reduction of luminescence, thus waiving the need for culturing. Using saline irrigation, enlarging canals to an apical size 60 was more effective than to size 36 and similarly effective than enlargement to size 77 (all 0.04 taper) (26).
In summary, these data demonstrate that mechanical canal preparation alone is effective in reducing, though not eliminating, bacterial load. Regardless of the study design—benchtop, or in situ—the presented evidence does not aid, however, in determining an apical preparation size. There are several reasons for this; first, experimental data that suggest larger preparation size does not take structural weakening or preparation errors into account. Second, the delivery of antimicrobials to remove bacteria more effectively follows different parameters than the purely mechanical removal of canal contents. To conclude, there are other factors to be considered in a clinical setting.
Working length and patency
It has been held that both mechanical removal of microorganisms and irrigation efficacy are dependent on canal shape (27, 28); however, the latter depends also on factors such as sodium hypochlorite turnover, the amount of available chlorine, contact time, and factors such as accessibility (for a more detailed review of endodontic irrigants, see Chapter 7 in this book).
Accessibility of the contaminated root canal areas is key to disinfecting efficacy. Thus, overall canal anatomy and in particular canal dimensions play an integral role in radicular shaping. Two basic concepts govern a numerical approach to root canal shaping procedures: working length (WL) and apical size, recently also described as working width (WW) (29).
Clinical research has indicated that root canal fillings that are extending beyond the root apex are associated with less success. Therefore, procedures confined to the root canal space (30) seem to be able to address most of the cases of endodontic treatment needs.
Varying concepts have been proposed regarding specific WLs (31), partially depending on preoperative diagnoses. Frequently, vital cases are clinically successful despite apparent short fills. However, with existing apical lesions and contaminated canal systems, WL definitions closer to the apex have been adopted. Classic and more recent studies suggest that a WL between 0 and 2 mm from the radiographic apex results more often in success (healing of apical lesions) than shorter or longer fills (7, 32, 33). However, apical anatomy and the relation between bacterial biofilms and clinical symptoms are complex (34) and can be addressed only partially by mechanical root canal shaping.
Figure 6.3 illustrates that the bacterial colonization in the apical canal portion ends a small distance inside the root canal, probably related to the geometry at that site and specifically the ability for the host defense to interact with microoganisms where blood supply occurs. In consequence WL for teeth with necrotic pulps and apical periodontitis should be set as close as possible the end of the confines of the root canal using radiographs and electronic devices.
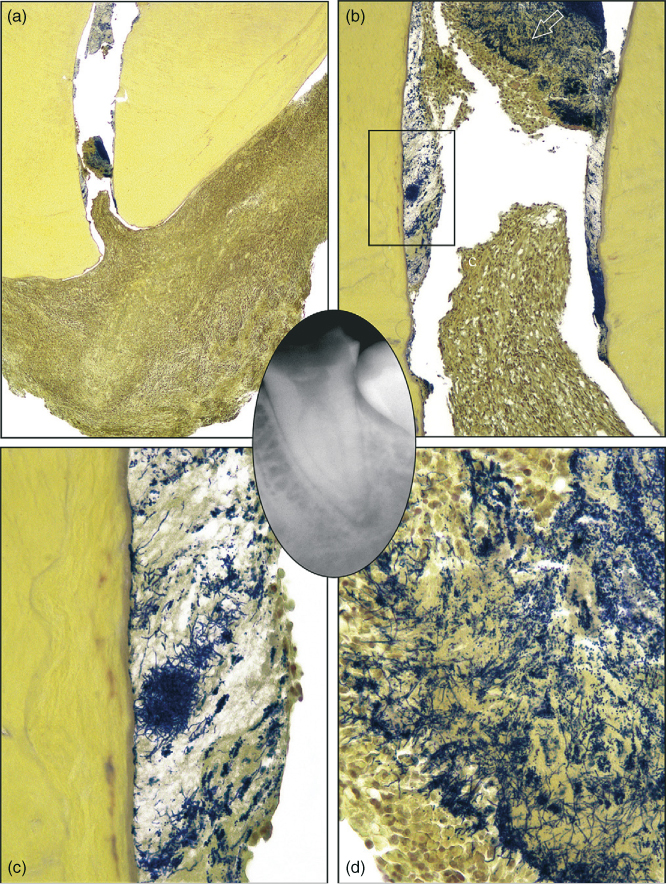
Figure 6.3 Grossly carious single-rooted mandibular second molar extracted with the apical periodontitis lesion attached. The radiographic size of the lesion was less than 5 mm (inset). The tooth was symptomatic. (a) The section passing approximately at the center of the foramen. The overview shows granulomatous tissue ingrowth at the very apical canal (Taylor’s modified Brown and Brenn, original magnification ×16). (b) Details of the apical foramen region. A biofilm is present covering the root canal walls, and a dense bacterial aggregate is evidenced more coronally. Empty spaces are shrinkage artifacts (original magnification ×100). (c) Higher magnification of the area demarcated by the rectangle in (b). Bacterial filamentous forms prevail, and the extracellular component is abundant at this level (original magnification ×400). (d) Higher magnification of the bacterial aggregate indicated by the arrow in (b). Different morphotypes are present. Note the concentration of polymorphonuclear neutrophils in contact with the biofilm surface (original magnification ×400).
(Ricucci and Siqueira (34). Reproduced with permission of Elsevier, Inc.)
Current strategies in this regard include the use of a patency file, e.g. a size #10, K-file, that is gently pushed slightly through an apical foramen without actually enlarging it (35). However, it is not firmly established if patency is beneficial in terms of canal disinfection of even counterproductive.
Indeed, one of the alleged reasons for not using apical patency is possible mechanical injury of apical tissues (36) or extrusion of infected debris through the apical foramen (37), a condition classically related with postoperative pain and with the delay in the healing process of apical pathology (36, 38, 39). On the other hand, in-vitro research has indicated that even if a patency file is contaminated, it will be disinfected by the 5.25% NaOCl present in the root canal during the shaping procedures (40).
The extrusion of debris appears to be more likely if the apical foramen is patent (41, 42). Certainly, mechanical cleaning of the foramen is unlikely, because the foramen is not enlarged and debris is not mechanically removed with a small file (43).
More recently, it was found that maintaining apical patency did not increase the incidence of postoperative pain (44) and that its benefits exceed the possible injury it might cause. This is because it is intended exclusively to prevent dentinal chips being compacted into the apical region and forming a plug that can interfere with maintaining WL (43). Of note, microcomputed tomography studies have demonstrated that dentin shavings are compacted into canal spaces not directly in contact with instruments (see below).
Other described advantages of this procedure include the sense that it improves apex locator accuracy, helps to maintain WL, reduces preparation errors such as ledges and blockages, and refines the tactile sense of the clinician during apical shaping (45). No apical transportation has been shown in the majority of root canals when using a #8 or a #10 file to maintain apical patency (46); however, using large file diameters for the purpose of apical patency may cause apical transportation (47).
Recently, in-vivo studies have suggested that maintaining apical patency may not only help to achieve the mechanical objectives of the shaping procedure of root canal treatments avoiding the accumulation of debris at the apex, but also that the use of a patency file allows better delivery of irrigants to the apical third. However, the mere presence of NaOCl in the apical third does not guarantee the proper cleaning of the root canal.
It should be stressed at this point that the use of a patency file is distinct from “apical clearing,” which is defined as a technique to remove loose debris from the apical extent, and involved sequentially rotating files two to four sizes larger than the initial apical file at WL, and then rotating the largest apical file again after a final irrigation and drying (48); this technique may be useful after hand instrumentation (49) but no such effect has been shown after rotary instrumentation.
A major question when defining WL in an attempt to enhance antimicrobial efficacy of canal preparation is where the root canal ends and how close to that point clinicians can estimate their WL. Histological studies indicated the presence of the transition of pulp to apical tissue as well as dentin and cementum in the area of the constriction (50). However, a constriction in the classic sense may be absent (51). In fact, high-resolution tomographic studies suggest more complicated apical canal configurations than previously shown.
The term working length is defined in the Glossary of Endodontic Terms as “the distance from a coronal reference point to the point, at which canal preparation and obturation should terminate” (35). The anatomic apex is the tip or the end of the root determined morphologically, whereas the radiographic apex is the tip or end of the root determined radiographically (35).
It is well established that root morphology and radiographic distortion may cause the location of the radiographic apex to vary from the anatomic apex. The apical foramen is the main apical opening of the root canal. It is often eccentrically located away from the anatomic or radiographic apex (50, 51). Kuttler’s classic investigation showed that this deviation occurred in 68–80% of teeth (50). An accessory foramen is an orifice on the surface of the root communicating with a lateral or accessory canal (35). They may be found as a single foramen or as multiple foramina. The apical constriction (minor apical diameter) is the apical portion of the root canal with the narrowest diameter.
Probably owing to its importance as a clinical entity, the apical root canal third and the foramen have been the topic of numerous systematic investigations (50, 52) beginning in the middle of the past century. Dummer and coworkers (51) reported four basic variations in the apical canal area that included about 50% of cases where a constriction was present. They also reported 6% of their cases, where the constriction was probably blocked by cementum (51). The cementodentinal junction is the region where the dentin and cementum are united; this is the point at which the cemental surface terminates or refers to the apex of a tooth (35). Of course the cementodentinal junction is a histological landmark that cannot be located clinically or radiographically.
Langeland (53) reported that the cementodentinal junction does not always coincide with the apical constriction. The location of the cementinodentinal junction also ranges from 0.5 to 3.0 mm short of the anatomic apex (50, 51).
Therefore, it is generally accepted that the apical constriction is most frequently located 0.5–1.0 mm short of the radiographic apex. It has been pointed out by Ricucci (30) that the presence of significant anatomical variation makes the direct clinical use of these average values as end points of canal preparation difficult. Further problems exist in locating apical landmarks and in interpreting their positions on radiographs.
Apical size and taper
The second factor to be considered is the apical width or the preparation size. It is an ongoing matter of debate, which apical enlargement and more specifically shape would lead to an optimal reduction of intracanal microbial load (54).
Principles of a standardized root canal preparation were based on concepts of apical canal geometry developed in the 1950s (50), which suggested apical canal diameters of 0.27–0.33 mm. However, a detailed anatomical assessment indicates that the concept for such a standardized root canal preparation (7) to an apical stop coronal to the constriction may be problematic because the “classical” singular constriction was not present in over 50% of the canals evaluated (51) and because of the possible sequelae of canal overenlargement, in particular vertical root fracture.
To complicate matters, it had been established by Shovelton (55) that intracanal bacteria colonize predentin and penetrate into dentinal tubules, to perhaps half the tubule length or in fact even deeper (56). It was therefore suggested to select a preparation size that removes this infected layer mechanically to maximize chances of success. Unfortunately, it appears that hand instruments and first generation rotaries (e.g., Quantec) were unable to completely remove predentin in-vitro (57). Moreover, this goal is likely unattainable even with modern root canal instruments such as the self-adjusting file (SAF, Figure 6.4a,b). For this file, microCT experiments suggested that even in relatively round and straight canals in maxillary anteriors under optimal conditions, about 9% canal area remains un-instrumented (58). More detailed analyses revealed that under these conditions, only about 57% of the canal surface had a dentin layer of 100 µm removed (Figure 6.4c).
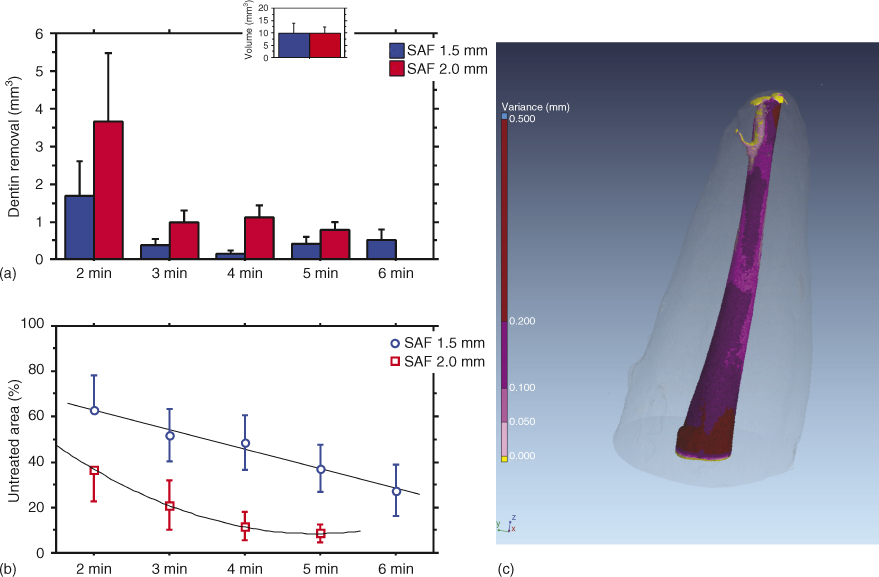
Figure 6.4 Effect of progressive preparation of root canals with the self-adjusting file (SAF) 1.5 and 2.0 mm. Initial canal volumes in both groups (n = 10 each) were statistically similar (inset). Preparation was done with 1.5 mm SAF (blue symbols) or 2.0 mm SAF (red symbols). (a) Canal volume increases with preparation over time. (b) Reduction in un-instrumented canal surface with progressive preparation. Data approximated by a linear regression line (blue) and second-order polynomial curve (red; untreated area). (c) Color-coded model for an anterior central incisor that was shaped with the 2 mm SAF for 4 min. Different amounts of surface area changes are indicated.
(a and b: Peters et al. (58). Reproduced with permission of Elsevier, Inc.)
This makes the role of irrigation solutions in canal disinfection even more relevant. Generally, irrigants are delivered using a syringe and needle system. Historically, a study using radiopaque liquids indicated that the apical penetration of the irrigants is only 1 mm beyond the needle tip (59). Furthermore, Usman and colleagues (28) demonstrated that the amount of irrigant delivered increased with the number of recapitulations. This information and regularly available needle types (27 and 30 gauge, equivalent to 0.42 and 0.31 mm, respectively) suggest that shaped canal diameter and curvature are relevant in irrigation efficacy.
It is common understanding that bacterial biofilm establishes on root canal surfaces; what is not settled is the best strategy to remove such biofilm. It seems like mechanical forces, such as in canal shaping, irrigant flow, and the selection of suitable irrigation solutions may be required (60).
Whether or not a smaller apical preparation allows sufficient antimicrobial action to take place (12, 61, 62) is a matter of continuous debate. Table 6.1 lists data in support of a variety of apical preparation sizes.
Table 6.1 Summary of evidence published in the past decade to suggest apical preparation geometry. Note that the vast majority of these studies were performed in-vitro. Also, there appears to be a very wide variation for favored apical sizes and several studies with inconclusive findings.
| Favored size | Article | Conclusion | Design |
| Small | (63) | Increased apical enlargement of curved canals did not result in a complete apical preparation, whereas it did lead to the unnecessary removal of dentin. | In-vitro |
| Small | (61) | There was no significant difference in intracanal bacterial reduction when Ni–Ti GT rotary preparation with NaOCl and EDTA irrigation was used with or without apical enlargement preparation technique. It may not be necessary therefore to remove dentin in the apical part of the root canal when a suitable coronal taper is achieved to allow satisfactory irrigation of the root canal system with antimicrobial agents. | In-vitro |
| >#20 | (64) | Apical canal geometry was affected differently by six preparation techniques; preparations with GT instruments to an apical size #20 left more canal surface untouched, which might affect the ability to disinfect root canal/> |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


