Cardiovascular medicine
Key Points
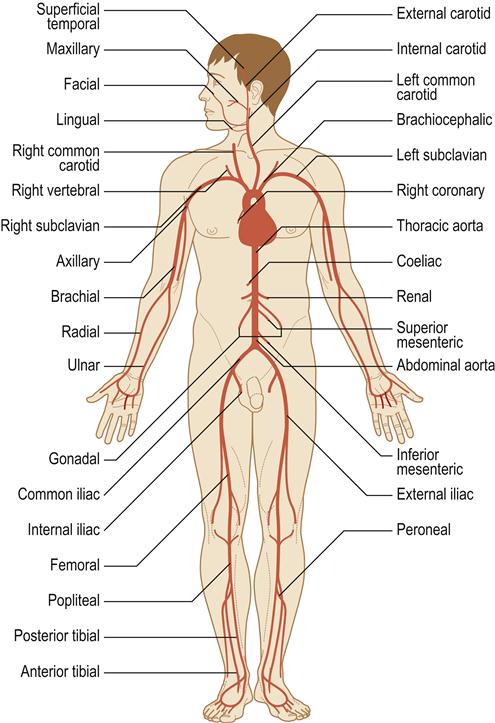
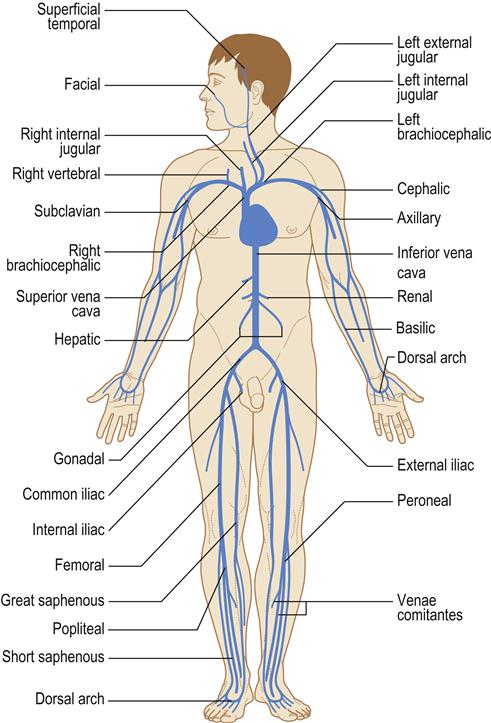
The heart is vital to life, as its essential function is pumping oxygenated blood to the brain and vital organs. Blood is oxygenated in the lungs and travels in the pulmonary vein to the heart at the left atrium, from where it flows to the left ventricle through the mitral valve. Blood leaves the left ventricle through the aortic valve to the aorta (the body’s main artery) and is distributed via arteries throughout the body. Thus the left ventricle is the most powerful chamber, as it has to move blood all around the body. Oxygen is transported on the red blood-cell (erythrocyte) pigment haemoglobin, which colours the blood red. Arteries branch to arterioles and then to capillaries to transport oxygen to the tissues (Fig. 5.1) and collect waste carbon dioxide. A vast venous network collects deoxygenated blood from all the tissues and returns it to the superior vena cava and inferior vena cava and thence to the right atrium of the heart (Fig. 5.2). This blood flows through the tricuspid valve to the right ventricle and then leaves the right ventricle through the pulmonary valve to the pulmonary artery and thence to the lungs, where it takes up oxygen. The heart thus consists of four chambers: two ventricles and two atria. It is centrally located in the chest (thorax) but, because the left ventricle is larger, the heart beat (apex beat) is found to the left side (Fig. 5.3).
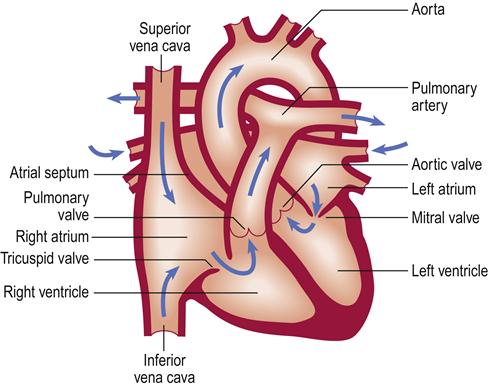
The heart rate and force of contraction are controlled by nerves (mainly vagus and sympathetic) and hormones (especially adrenaline [epinephrine], which increases both rate and force). Anxiety and exercise can increase the pulse rate and force of the heart beat.
Heart disease is a major killer; in the USA, possibly one person dies from heart disease every 30 seconds. It also causes significant morbidity and disability in many aspects of life, including effects on entitlement to drive a motor vehicle. There is a wide range of disorders that can affect the heart (Box 5.1).
Congenital Heart Disease (CHD)
General aspects
Congenital heart defects are the most common type of heart problem in children, present in about 1% of live births. Lesions may involve the heart or adjacent great vessels, in isolation or in a variety of combinations, and include a wide variety of structural defects that can lead to malfunction such as arrhythmias or flow problems. Vessel or valve stenosis or obstruction and septal defects strain the myocardium.
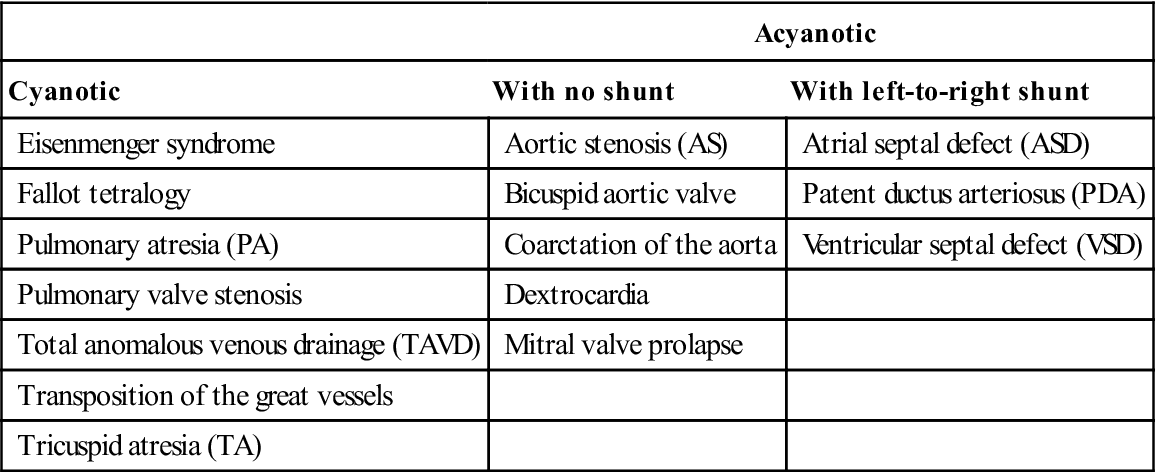
The types of CHD are summarized in Table 5.1. CHD may be: cyanotic (‘blue babies’), where there is right-to-left shunting – in general these are more severe defects; or acyanotic. Cyanosis and hypoxia can lead to neurological deficits, polycythaemia (with hypercoagulability and thromboses) and systemic infections (e.g. brain abscess). Approximately 20% of patients with CHD also have other congenital anomalies.
Table 5.1
Types of congenital heart diseasea
< ?comst?>
| Acyanotic | ||
| Cyanotic | With no shunt | With left-to-right shunt |
| Eisenmenger syndrome | Aortic stenosis (AS) | Atrial septal defect (ASD) |
| Fallot tetralogy | Bicuspid aortic valve | Patent ductus arteriosus (PDA) |
| Pulmonary atresia (PA) | Coarctation of the aorta | Ventricular septal defect (VSD) |
| Pulmonary valve stenosis | Dextrocardia | |
| Total anomalous venous drainage (TAVD) | Mitral valve prolapse | |
| Transposition of the great vessels | ||
| Tricuspid atresia (TA) | ||
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
The cause of CHD is often unidentified but some causes are genetic (e.g. Down syndrome; see Ch. 28). There are many other causes (Table 5.2; Appendices 5.1 and 5.2) and syndromes (e.g. oesophageal atresia and heart disease, and forearm/digit abnormalities and atrial septal defect [ASD]). The main acquired causes of CHD are maternal infections such as TORCH – congenital toxoplasmosis, rubella, cytomegalovirus or herpes infection – which can lead to patent ductus arteriosus (PDA), pulmonary stenosis, ventricular septal defect (VSD) and coarctation of the aorta; maternal drug use (e.g. alcohol, anticonvulsants, lithium, recreational drugs, thalidomide and warfarin); or maternal systemic disease (systemic lupus erythematosus and Sjögren syndrome [both can cause heart block], and diabetes [which is associated with an increased incidence of VSD, coarctation and transposition of the great arteries and/or a characteristic hypertrophic cardiomyopathy]); and, rarely, maternal irradiation.
Table 5.2
Main features of congenital heart disease (CHD)
| CHD | Main pathology/location | Main features |
| Aortic stenosis | Narrowing of the aortic valve | Angina, dyspnoea and syncope. Balloon valvuloplasty or surgery may be indicated |
| Atrial septal defect (ASD) | Usually near foramen ovale and termed a secundum defect. Associated in 10–20% with mitral valve prolapse | Initially acyanotic, with survival into middle age in most cases, and therefore is the most common CHD presenting in adults. In the absence of surgical correction, right ventricular failure eventually develops. Usually repaired by primary closure or by pericardial or Dacron patch |
| Bicuspid aortic valve | Valve is bicuspid instead of tricuspid. Often associated with other left-sided lesions (e.g. coarctation of the aorta or interrupted aortic arch) | Usually asymptomatic, even in athletes, but there is a high risk of infective endocarditis |
| Coarctation of the aorta | In the aorta beyond the origin of the subclavian arteries | Normal blood supply to the head, neck and upper body but restricted to the lower body. Surgery is indicated |
| Dextrocardia | Right-sided heart | Usually asymptomatic but may be part of situs inversus (all organs transposed) with bronchiectasis and sinusitis (Kartagener syndrome) |
| Ebstein anomaly | Congenital abnormality of the tricuspid valve, usually associated with ASD | Ebstein anomaly is mild in most and surgery is needed only if the tricuspid valve leaks severely enough to result in heart failure or cyanosis |
| Eisenmenger syndrome or reaction | The process in which a left-to-right shunt in the heart causes increased flow through the pulmonary vasculature (and pulmonary hypertension) which, in turn, causes increased pressures in the right heart and reversal into a right-to-left shunt | Ventricular or atrial septal defects, patent ductus arteriosus and more complex types of acyanotic heart disease can underlie this. Initially there is pulmonary hypertension, polycythaemia and a hyperviscosity syndrome with haemorrhagic and thrombotic tendencies. Later there is right ventricular hypertrophy and cyanosis. Anticoagulants and pulmonary vasodilators (e.g. bosentan) may be indicated |
| Fallot tetralogy | Ventricular septal defect, pulmonary stenosis, right ventricular hypertrophy and an aorta that overrides both ventricles. Fluconazole use in pregnancy may predispose | Cyanosis progressive from birth. Chronic hypoxia causes decreased neurological function. Episodes of acute hypoxia from infundibular spasm are life-threatening. Polycythaemia causes hypercoagulability and thrombosis. Right-to-left shunting is associated with a higher incidence of systemic infection such as a brain abscess. Surgery is indicated |
| Mitral valve prolapse (floppy mitral valve; Barlow syndrome) | The most common CHD, affecting 5–10% of the population. Seen mainly in women. Strong hereditary tendency. Seen especially in Marfan, Ehlers–Danlos and Down syndromes. May be associated with polycystic kidney disease and panic disorder | May cause no symptoms but, if there is mitral regurgitation, some patients develop pain, irregular or racing pulse, or fatigue and heart failure |
| Patent ductus arteriosus (PDA) | A persistent opening (normally closed by the third month of life) between aorta and pulmonary artery, common in prematurity and maternal rubella | Shunt is left to right, initially acyanotic, and a typical complication is right ventricular failure. Closure can be promoted in early infancy by giving intravenous indometacin, a prostaglandin inhibitor |
| Patent foramen ovale | Communication between atria | Often symptomless |
| Pulmonary atresia | No pulmonary valve exists, so blood cannot flow from the right ventricle into the pulmonary artery and to the lungs. The right ventricle is not well developed. The tricuspid valve is often poorly developed. An ASD allows blood to exit the right atrium, so the baby is cyanotic. The only source of lung blood flow is the PDA | Early treatment often includes using a drug to keep the PDA from closing, and surgery to create a shunt between the aorta and the pulmonary artery |
| Pulmonary stenosis | Narrowing of the pulmonary valve | Symptoms are breathlessness and right ventricular failure. Balloon valvuloplasty or surgery may be indicated |
| Total anomalous pulmonary venous return (TAPVR) | Blood returns from the lungs to the right rather than left atrium | Survivors also have ASD or patent foramen ovale |
| Transposition of the great vessels | Reversal of pulmonary artery and aorta origins | Cyanosis and breathlessness from birth, with early congestive cardiac failure and death in infancy unless there are associated defects providing sufficient collateral circulation (such as a patent interventricular septum or PDA) |
| Tricuspid atresia | Absence of the tricuspid valve means no blood can flow from the right atrium to the right ventricle. Thus the right ventricle is small. Survival depends on there being an ASD and usually a ventricular septal defect | A surgical shunting procedure is often needed to increase the lung blood flow and reduce cyanosis. Some children may need pulmonary artery banding to reduce blood flow to the lungs. Others may need a more functional repair (Fontan procedure) |
| Ventricular septal defect (VSD) | One of the most common CHDs. Ranges from a pinhole compatible with survival at least into middle age, to a large defect causing death in infancy if untreated. Some 90% of children with VSD have an additional cardiac defect | Initially left-to-right shunt but, with right ventricular hypertrophy, shunt may eventually reverse with late-onset cyanosis. Right ventricular failure may develop. Usually repaired by primary closure or by a pericardial or Dacron patch |
Clinical features
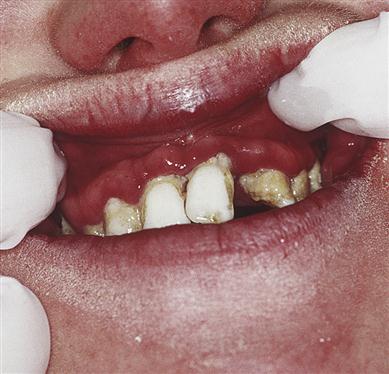
Cyanotic CHD is, in general, a much more severe defect than acyanotic (Fig. 5.4), which can lead to cardiac failure or be life-threatening or fatal without surgical intervention. In the absence of treatment, 40% of children with cyanotic CHD die within the first 5 years. Cyanosis (caused by>5 g of reduced haemoglobin per decilitre of blood) results from shunting deoxygenated blood from the right ventricle into the left side of the heart and the systemic circulation (right-to-left shunt), and leads to chronic hypoxaemia (Fig. 5.5). Patients may crouch in an effort to improve venous return but eventually polycythaemia develops – with consequent haemorrhagic or thrombotic tendencies. Finger- and toe-clubbing are associated with cyanotic CHD, but appear after about 3 months of age (Fig. 5.6). Patients with CHD are liable to a range of complications (Box 5.2).
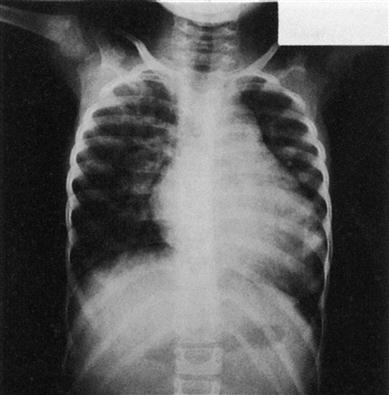
(The heart normally occupies less than half the width of the chest).
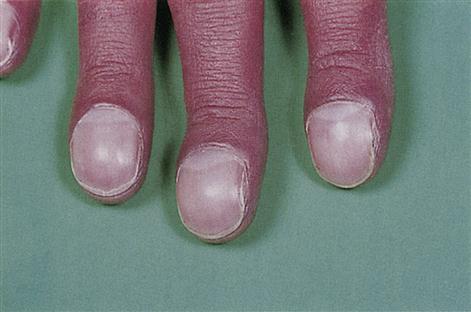
Finger-clubbing can be found in several different disorders, particularly cyanotic heart disease, chronic respiratory disease, lung cancer, Crohn disease, etc., but is sometimes benign and hereditary.
Pulmonary hypertension and, eventually, right ventricular hyper-trophy may be seen where the shunt is left-to-right and some of the output of the left ventricle is recirculated through the lungs. The shunt may eventually reverse and then cyanosis develops.
General management
An obstetric ultrasound scan is often used to screen pregnant women at around 20 weeks for signs of CHD in their unborn babies. Most defects, however, are well tolerated in utero and it is only after birth that the impact of the anatomical and subsequently haemodynamic abnormality becomes evident. In young children, chest radio-graphy may show cardiomegaly. Electrocardiography (ECG) may demonstrate an abnormal cardiac axis, ventricular hypertrophy and strain, depending on the lesion present. CHD is more commonly diagnosed through echocardiography, which has superseded intracardiac catheter studies, and confirmed by cardiac magnetic resonance imaging (MRI).
Early correction of the congenital defect, often by transvenous catheter techniques, is the treatment of choice. More complex defects may require an operation. Medical treatment may be needed for the management of pulmonary oedema, heart failure, polycythaemia, infection or emotional disturbances. Modern surgical and medical care helps children survive into adult life and patients are then often said to have adult or ‘grown-up’ CHD. Nevertheless, complications observed in adults who were previously thought to have had successful repair of CHD include arrhythmias, valve disorders and cardiac failure, and residual defects can still predispose to complications such as infective endocarditis.
Dental aspects
All dental health-care professionals (HCPs) should be certified in basic cardiopulmonary resuscitation (CPR), and the entire team should rehearse emergency protocol procedures regularly (Ch. 1).
Patients with heart disease should take their medications as usual on the day of the dental procedure, and should bring all their medications to the dental surgery for review at the time of the first appointment. The most important aspect for dentists to consider is how well the patient’s heart condition is compensated. Patients with stable heart disease receiving atraumatic treatment under local anaesthesia (LA) can receive treatment in the dental surgery. Since cardiac events are most likely to occur in the early morning, patients with cardiac disease should be treated in the late morning or early afternoon. The dental team should provide dental care with a stress-reduction protocol and with good analgesia, limiting the dosage of adrenaline (epinephrine). An aspirating syringe should be used to give an LA, since adrenaline in the anaesthetic entering a vessel may theoretically raise the BP or precipitate arrhythmias. Gingival retraction cords containing adrenaline should be avoided. Conscious sedation, preferably with nitrous oxide, can be given with the approval of the physician. General anaesthesia (GA) is a matter for expert anaesthetists in hospital.
Congestive cardiac failure may complicate management. In cyanotic CHD there may also be polycythaemia-related bleeding tendencies caused by thrombocytopenia, platelet dysfunction, coagulation defects (from liver hypoxia – causing reduced vitamin K-dependent factors) and excessive fibrinolytic activity. Thus platelets may be reduced, and the haematocrit, prothrombin time (PT [and International Normal Ratio, INR]; Ch. 8) and activated partial thromboplastin time (APTT) increased. Occasionally, there is a thrombotic tendency. A special hazard in some CHD is the development of cerebral abscess, sometimes due to oral bacteria. Leukopenia may be a factor in some right-to-left shunts. There may be susceptibility to infective endocarditis. Oral abnormalities can be associated with cyanotic CHD and may include:
Congenital Heart Disease in Other Syndromes
Down syndrome
See Chapter 28.
Noonan syndrome
Noonan syndrome, usually an autosomal dominant trait, is characterized by CHD (the commonest cardiac lesions are pulmonary stenosis, septal defects and hypertrophic cardiomyopathy), short stature, unusual facies, chest deformity, learning disability, and cryptorchidism in males. Facial features may include an elongated mid-face, hypertelorism, retrognathia, a lower nasal bridge and nasal root, a wider mouth, a more prominent upper lip and low-set ears. Abnormal vision and hearing are common. Other associations include hepatosplenomegaly and an abnormal bleeding tendency associated with low levels of clotting factors (particularly XI and XII), and with cherubism, jaw giant-cell lesions and neurofibromatosis.
Hypercalcaemia–supravalvular aortic stenosis syndrome (Williams syndrome)
Williams syndrome comprises, in its rare complete form, infantile hypercalcaemia, elfin facies, learning disability and CHD. Most cases appear to be sporadic. These children may be sociable and talkative (‘cocktail party manner’). Hypercalcaemia typically remits in infancy but leaves growth deficiency, osteosclerosis and cranio- stenosis. Cardiovascular lesions include supravalvular subaortic stenosis or other abnormalities, and may contraindicate GA. Also, masseter spasm has been reported during GA with halothane and suxamethonium, but it is not malignant hyperthermia. Dental defects may include hypodontia, microdontia and hypoplastic, bud-shaped teeth. The upper dental arch may be disproportionately wide and overlap the lower.
Acquired Heart Disease
General aspects
Most acquired heart disease is ischaemic heart disease (IHD; coronary artery disease, CAD) – the most important cardiac disease, which can lead to heart failure and is the major killer in the high-income world. Functional disorders, where there is no organic disease of the heart itself, can also cause circulatory failure, as in shock. A variety of common extracardiac diseases can aggravate heart disease, particularly if there is impaired oxygenation, as in severe anaemia.
Clinical features
Serious heart disease is not always symptomatic and patients can die suddenly from myocardial infarction (MI), despite having experienced neither chest pain nor any other symptom (“silent infarction”). Also, many patients with manifest cardiac disease can have signs and symptoms effectively controlled so that they may superficially appear well and, often, only the drug history gives a clue to the nature of any cardiac illness. Common clinical features of cardiac disease include breathlessness, chest pain, palpitations and cyanosis, while signs may be seen on the hands (clubbing, cyanosis) or deduced from measuring the BP, palpating the pulse or listening to the heart (auscultation).
General management
The heart rate, force of contraction and rhythm can be assessed from palpation of the pulse; disturbances of rhythm may signify cardiac disease.
The BP is measured by sphygmomanometry. The finding of either normal levels (pressures of 120 mmHg systolic and 80 mmHg diastolic) or grossly raised levels (>160/90 mmHg for a male of 45 years or over) is informative. However, these must be checked on further occasions, as the BP frequently increases in anxiety. In an injured patient, a falling BP is also a danger sign that must not be ignored, since it implies a serious complication such as haemorrhage or shock.
Experienced cardiologists can identify by auscultation the vast majority of innocent functional murmurs and can differentiate these from the serious murmurs of aortic stenosis, mitral regurgitation, tricuspid regurgitation, pulmonary stenosis, VSD and hypertrophic cardiomyopathy.
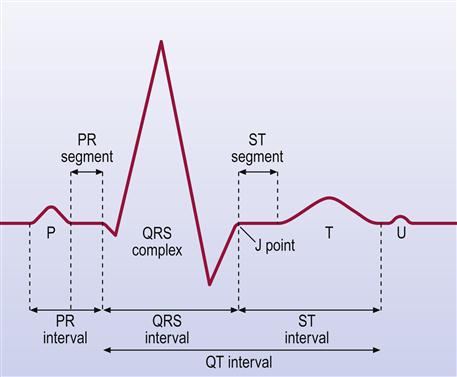
Other investigations include blood assays of cardiac enzymes (troponin, troponin T (cTnT), creatine kinase), which are raised after cardiac damage such as an MI. Chest radiography (CXR) helps detect heart enlargement (e.g. in cardiac failure or hypertension). ECG, invaluable for the diagnosis of arrhythmias and of IHD, is a graphic tracing of the variations in electrical potential caused by the excitation of the atria (Fig. 5.7). The QRS deflections are due to excitation (depolarization) of the ventricles. The T wave is due to recovery of the ventricles (repolarization). Doppler echocardiography (cardiac ultrasonography done trans-thoracically or trans-oesophageally) is a valuable non-invasive imaging tool for the heart; it is helpful in establishing a specific diagnosis and estimating the severity of various diseases by evaluating cardiac chamber size, wall thickness, wall motion, valve configuration and motion, and the proximal great vessels. This technique also provides a sensitive method for detecting pericardial and pleural fluid accumulation, and can allow identification of mass lesions within and adjacent to the heart. Cardiac MRI, scans using gadolinium, thallium-201 or technetium-99, and positron emission tomography (PET) using 18F-deoxyglucose (FDG) are also extremely useful.
Medical treatments are available to control many cardiac disorders, but surgery may be required, especially for IHD.
Dental aspects
Management implications in oral health care depend mainly on the type of intervention and the degree of cardiovascular risk (Table 5.3 and see below). Both the anxiety and the pain that may be associated with dental care can cause enhanced sympathetic activity and adrenaline (epinephrine) release, which increases the load on the heart and the risk of angina or arrhythmias. Therefore, patients with unstable angina (pain pattern changing in occurrence, frequency, intensity or duration; pain at rest) and those with MI<3 months previously should have dental treatment in a hospital environment. Patients with stable angina and those who are at least 3 months post-MI may be treated in primary care. However, appointments should be made for the late morning, with the patient’s glyceryl trinitrate (GTN) available and used preoperatively, and the use of pulse oximetry and prophylactic sedatives considered. Nitrate drugs, which include glyceryl trinitrate (GTN), isosorbide dinitrate and isosorbide mononitrate are vasodilators and act by reducing load on the heart. GTN is available for sublingual use as a tablet or a spray, or as a skin patch. Tables have a limited shelf life of about 8 weeks. Oral use works within a minute or so and lasts 30 mins but the patch takes about an hour to work and is often left on for 12 hours. The most common adverse effects of nitrates are a throbbing headache, flushing, and dizziness, and they interfere with sildenafil. Pain control is crucial to minimize endogenous adrenaline release. LA must be given with aspiration and it may be prudent to avoid adrenaline-containing LA. If a patient is taking a non-selective beta-blocker (e.g. propanolol), no more than two carpules of LA with adrenaline 1:80 000 should be administered. The combination of aspirin with other antiplatelet drugs increases the chances of significant postoperative bleeding. Furthermore, drugs such as erythromycin and clarithromycin should be avoided in long QT syndrome and in patients also taking statins (antihyperlipidaemics). In the case of intraoperative chest pain, medical assistance should be summoned and emergency management commenced, including the use of GTN aspirin and oxygen. Cardiac monitoring is desirable in many instances (Figs 5.8 and 5.9).
Table 5.3
Predictors of high cardiovascular risk
| Very high risk | Medium risk | Low risk |
| MI in previous month | Mild angina | Old age |
| Unstable or severe angina Atrioventricular block |
Myocardial infarction (MI) more than 1 month previously | ECG abnormalities only Atrial fibrillation Stroke |
| Ventricular arrhythmias | Compensated heart failure | Uncontrolled hypertension |
| Severe cardiac valvular disease | Mild heart failure | |
| Advanced heart failure |
Effective painless LA is essential. An aspirating syringe should be used since adrenaline in the anaesthetic may get into the blood and may (theoretically) raise the BP and precipitate arrhythmias. BP tends to rise during oral surgery under LA, and adrenaline theoretically can contribute, but this is usually of little practical importance. However, adrenaline-containing LA should not be given in excessive doses to patients taking beta-blockers, as this may induce hypertension and cardiovascular complications. Note: A woman on beta-blockers, who was (inexcusably) given 16 dental cartridges of LA, died from lidocaine overdosage, not from accelerated hypertension. Gingival retraction cords containing adrenaline should be avoided wherever possible. General anaesthesia for dental care in cardiac patients should be avoided unless essential.
A range of cardiovascular drugs can influence oral health care (Table 5.4), while drugs used in dentistry can interact with cardiac drugs. Aspirin may cause sodium and fluid retention, which may be contraindicated in severe hypertension or cardiac failure. Indometacin may interfere with antihypertensive agents. Macrolides and azoles may cause statins to produce increased muscle damage.
Table 5.4
Cardiovascular drugs: potential effects on oral health care
| Group | Oral adverse effects | Possible interaction or reaction |
| Alpha-blockers | Dry mouth | Orthostatic hypotension |
| Alpha-/beta-blockers | – | Enhanced pressor response to adrenaline (epinephrine) |
| Angiotensin-converting enzyme inhibitors (ACEIs) | Burning sensation Ulceration Taste changes |
Impairment by indometacin |
| Dry mouth | ||
| Angioedema | ||
| Lichenoid reactions | ||
| Angiotensin II receptor blockers (ARBs) | Facial flushing Taste disturbance Dry mouth |
Good toleration |
| Increased gag reflex | ||
| Lupoid reactions | ||
| Antihypertensives (see Table 5.8) | Predisposition to lip cancer (hydrochlorothiazide, nifedipine, triamterene, lisinopril, atenolol) | Photosensitivity |
| Antiplatelets | Purpura Bleeding |
Bleeding tendency enhanced by erythromycin |
| Beta-blockers (non-cardioselective) | Dry mouth Paraesthesia Lichenoid lesions |
Enhanced pressor response to adrenaline (epinephrine) |
| Calcium-channel blockers (CCBs) | Gingival swelling: 30% using nifedipine develop this | – |
| Angioedema | ||
| Digitalis glycosides | Dry mouth | Possible induction of arrhythmias by LA vasoconstrictor |
| Gag reflex increased | ||
| Diuretics | Dry mouth | – |
| Potassium-channel blockers | Ulceration | – |
| Statins | – | Muscle damage increased by macrolides (erythromycin, clarithromycin, telithromycin) and azoles (fluconazole, itraconazole, ketoconazole, miconazole) but rosuvastatin, pravastatin and fluvastatin do not appear to have this problem |
An association between periodontal disease and cardiovascular disease has been suggested but several evidence-based reviews question this association.
Hypertension
General aspects
Hypertension (high blood pressure) is common, often unrecognised and potentially lethal. The blood pressure is measured with a sphygmomanometer (Box 5.3), in units of millimetres of mercury (mmHg). A ‘normal’ adult blood pressure (BP) is 120/80 (systolic/diastolic) mmHg. High blood pressure (hypertension) is usually associated with a progressive rise in BP. Untreated hypertension is one of the most important preventable causes of morbidity and mortality, since it is a major risk factor for ischaemic and haemorrhagic strokes (cerebrovascular events), myocardial infarction, heart failure, chronic kidney disease, cognitive decline and premature death. The risk associated with increasing BP is continuous, with each 2 mmHg rise in systolic BP linked with a 7% rise in risk of mortality from ischaemic heart disease and a 10% increased risk of mortality from stroke. The vascular and renal damage that hypertension may cause can culminate in a treatment-resistant state. The BP is the product of cardiac output and peripheral resistance, and is dependent on the heart and vasculature, autonomic nervous system, endocrine system and kidneys. Regulatory mechanisms, though unclear, involve baroreceptors in various organs that detect changes in BP, and adjust it by altering both the heart rate and the force of contractions, as well as the peripheral resistance. Other factors are the kidney renin–angiotensin system, which responds to a fall in BP by activating a vasoconstrictor (angiotensin II), and the adrenocortical release of aldosterone in response to angiotensin II, causing fluid retention via the kidney – increasing sodium retention and potassium excretion. The most potent vasoconstrictor is endothelin-1, released by vascular endothelium in response to expansion of plasma volume, hypoxia or growth factors, and which regulates vascular tone. Nitric oxide (NO) also modulates vasodilator tone in the control of BP.
The BP will vary, depending on age, gender, ethnicity, environment, emotional state and activity. Since it rises with anxiety, measurements should be made with the patient relaxed and fully at rest. The BP is lowest at night and highest first thing in the morning. It tends to increase with age.
If the clinic BP is 140/90 mmHg or higher, ambulatory blood pressure monitoring (ABPM) is needed to confirm the diagnosis of hypertension (average value of at least 14 measurements taken during the person’s usual waking hours). Home BP monitoring (HBPM) can be used to confirm a diagnosis of hypertension, provided that:
■ BP is recorded twice daily, ideally in the morning and evening
■ BP recording continues for at least 4 days, and ideally for 7 days.
Hypertension is a persistently raised BP. There is no agreed BP cut-off point above which ‘hypertension’ exists and below which it does not. At least one-quarter of adults (and more than half of those older than 60) have high BP. Hypertension is thus common, especially with advancing age, when systolic hypertension becomes a more significant problem, as a result of the progressive stiffening and loss of compliance of larger arteries. Diastolic pressure is more commonly raised in people younger than 50 years (Table 5.5). Hypertension nevertheless is generally defined as a BP of at least 140/90 mmHg, based on at least two readings on separate occasions. Indeed, the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure defined a BP of 120/80–139/89 mmHg as pre-hypertension – a designation chosen to identify individuals at high risk of developing hypertension. The National Heart, Lung and Blood Institute (NHLBI) has even recommended that ‘pre-hypertension’ should be considered as beginning when BP is 115/75 mmHg. Hypertension may be staged as shown in Tables 5.5 and 5.6.
Table 5.5
| Stage | Designation | Level |
| Normal | Systolic: less than 120 mmHg | |
| Diastolic: less than 80 mmHg | ||
| At risk | Pre-hypertension | Systolic: 120–139 mmHg |
| Diastolic: 80–89 mmHg | ||
| Stage 1 | Hypertension | Systolic: 140/90 mmHg or higher and subsequent ambulatory monitoring (ABPM) daytime average or home monitoring (HBPM) average is 135/85 mmHg or higher |
| Stage 2 | Hypertension | Systolic: 160/100 mmHg or higher and subsequent ABPM daytime average or HBPM average is 150/95 mmHg or higher |
| Severe | Hypertension | Systolic: 180 mmHg or higher |
| Diastolic: 110 mmHg or higher |
Table 5.6
< ?comst?>
| Blood pressure (mmHg; systolic, diastolic) | ASA grade | Hypertension stage | Dental aspects |
| <140,<90 | I | – | Routine dental care |
| 140–159, 90–99 | II | 1 | Recheck BP before starting routine dental care |
| 160–179, 95–109 | III | 2 | Recheck BP and seek medical advice before routine dental care |
| Restrict use of adrenaline (epinephrine) | |||
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


