47 Immunopathogenesis of Periradicular Disease
Following root canal invasion, endodontic pathogens and their pathogen-associated molecular patterns (PAMPs) instigate nonspecific (innate) and antigen-specific (acquired) host immune responses within pulpal and subsequently periradicular tissues. Central to the initiation of appropriate innate host responses is the cellular recognition of PAMPs by endodontic pathogen -binding members of the toll-like receptor family (TLRs), which bind PAMPs. This response induces an influx of “innate” immune cells into the periradicular region, which occurs within 3 days after pulp exposure (Kawashima et al., 1996).
Of clinical significance, this inflammatory infiltrate and concomitant bone destruction occurs within the periradicular tissue prior to pulp tissue necrosis (Suzuki et al., 1999). Although host-defensive reactions are activated to prevent further infection, perpetuation of the inflammatory response eventually elicits periradicular disease (PRD).
Epidemiologic studies reveal that PRD is remarkably prevalent, occurring in 33%–80% of adult populations (Tsuneishi et al., 2005); 35%–80% of individuals may have more than one affected tooth (De Moor et al., 2000) and as many as 14 affected teeth (Kabak and Abbott, 2005). PRD is associated with a large proportion of root-filled teeth, from 25% to 64.5% (Loftus et al., 2005), and these teeth are five to 12 times more likely to have radiographic evidence of PRD than teeth without root fillings (Petersson et al., 1986; Buckley and Spångberg, 1995).
Furthermore, teeth with inadequate root fillings are significantly correlated with PRD lesions (Chugal et al., 2003). Importantly, there is clear evidence that the presence of a periradicular lesion prior to the start of treatment is the single most significant factor influencing successful outcome of endodontic therapy (Negishi et al., 2005).
Patient susceptibility to PRD is determined by complex interactions between endodontic microorganisms, host factors, and environmental components. It is now accepted that the host’s immune response is an important constituent in the outcome of endodontic treatment, this having been previously largely ignored (Marending et al., 2005).
To be able to clinically attenuate immune responses and successfully eradicate disease, it is essential to have an appreciation of the complex pathologic processes that contribute toward PRD.
The innate cellular immune system is responsible for rapidly initiating cellular responses essential for early microbicidal host defenses. The inflammatory cellular infiltrate consists of neutrophils, macrophages, mast cells, lymphocytes, natural killer cells, plasma cells, and eosinophils.
Among the bacterial-derived moieties, lipopolysaccharide (LPS) from Gram-negative bacteria plays a key role in pulpal disease (Khabbaz et al., 2001) and subsequently PRD (Nelson-Filho et al., 2002). LPS induces periradicular bone destruction (Mattison et al., 1987) and its concentration within the root canals correlates with the presence of periradicular lesions (Horiba et al., 1991).
Nevertheless, cell wall components from Gram-positive bacteria and other virulence factors, including fimbriae and lipo protein, likely contribute toward PRD development (Wang and Stashenko, 1993).
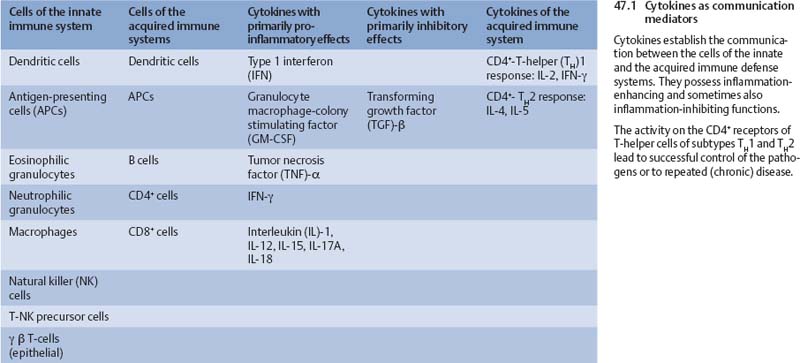
Adverse effects of endodontic pathogens may not be because of direct actions on bone-derived cells. More importantly, bacteria-derived moieties indirectly stimulate bone resorption by inducing a myriad of inflammatory mediators involved in the recruitment and maturation of osteoclasts, including cytokine expression.
Cytokines are a diverse group of small, secreted proteins acting />
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses