Chapter 4
Ischemic Heart Disease
Coronary atherosclerotic heart disease is a major health problem in the United States and in other industrialized nations. Atherosclerosis is the thickening of the intimal layer of the arterial wall caused by the accumulation of lipid plaques. The atherosclerotic process results in a narrowed arterial lumen with diminished blood flow and oxygen supply. Atherosclerosis is the most common underlying cause of not only coronary heart disease (angina and myocardial infarction [MI]) but also cerebrovascular disease (stroke) and peripheral arterial disease (intermittent claudication).
Symptomatic coronary atherosclerotic heart disease often is referred to as ischemic heart disease. Ischemic symptoms are the result of oxygen deprivation secondary to reduced blood flow to a portion of the myocardium. Other conditions such as embolism, coronary ostial stenosis, coronary artery spasm, and congenital abnormalities also may cause ischemic heart disease.
General Description
Incidence and Prevalence
More than 70 million Americans (about 25% of the population) are estimated to have some form of cardiovascular disease, with about 13 million having coronary heart disease.< ?xml:namespace prefix = "mbp" />
Etiology
The cause of coronary atherosclerosis is not known; however, research indicates that the disease is related to a variety of risk factors. These risk factors include male gender, older age, a family history of cardiovascular disease, hyperlipidemia, hypertension, cigarette smoking, physical inactivity, obesity, insulin resistance and diabetes mellitus, mental stress, and depression. In addition to these conventional risk factors, markers of inflammation such as C-reactive protein, homocysteine, fibrinogen, and lipoprotein(a) have been found to be associated with atherosclerosis.
Before the age of 75, the risk of coronary atherosclerosis is greater for men than for women.
Elevation in serum lipid levels is a major risk factor for atherosclerosis. Increased levels of low-density lipoprotein (LDL) cholesterol pose the greatest risk for coronary atherosclerosis, whereas increased levels of high-density lipoprotein (HDL) cholesterol have been shown to reduce the risk.
Increased blood pressure appears to be one of the most significant risk factors for coronary atherosclerotic heart disease.
Cigarette smoking is the single most important modifiable risk factor for coronary heart disease (see
Patients with diabetes mellitus have a greater incidence of coronary atherosclerotic heart disease and more extensive lesions. They develop the condition at an earlier age than that typical for persons who do not have diabetes. Almost 41 million Americans have some degree of abnormal glucose tolerance (prediabetes)—a condition along with obesity that markedly increases the risk for type 2 diabetes and premature atherosclerosis.
Other risk factors for the development of atherosclerosis have emerged and include elevated levels of C-reactive protein (inflammatory marker), fibrinogen (procoagulant), plasminogen activator inhibitor (thrombolytic), and homocysteine.
Numerous studies have reported an association between periodontal disease and cardiovascular disease, raising the question of whether periodontal disease is a risk factor for cardiovascular disease.
No single risk factor is responsible for the development of coronary atherosclerosis, but many factors act synergistically. Evidence suggests that modification of those risk factors that can be controlled, such as cigarette smoking, hypertension, hyperlipidemia, and diabetes, may reduce or modify the clinical effects of the disease.
Pathophysiology and Complications
Understanding of the pathophysiology of atherosclerosis has evolved significantly over the past 2 or 3 decades. In essence, the basic model has gone from the concept of a cholesterol storage disease with a buildup of plaque on the surface of an artery, analogous to rust buildup in a pipe, to one of an inflammatory disorder of the cellular lining of the arteries, with inflammation playing a fundamental role at all stages of the disease.
Libby and Theroux
Atheroma formation is initiated by adherence of monocytes to an area of injured or altered endothelium. Usually, monocytes do not adhere to intact endothelium; however, triggers of atherosclerosis such as a high saturated fat diet, smoking, hypertension, hyperglycemia, obesity, and insulin resistance initiate the expression of adhesion molecules by the endothelial cells, thus promoting attachment. The attached monocytes then migrate into the intima of the vessel and become macrophages. Lipids derived from plasma LDLs also enter through the injured or dysfunctional endothelium, forming extracellular deposits or small pools. Macrophages then engulf lipid molecules to become foam cells, which are characteristic features of the fatty streak. Foam cells are joined by T lymphocytes, and together they produce a variety of inflammatory cytokines, which promote the migration and proliferation of smooth muscle cells and collagen to surround the foam cells, thereby forming a fibrous covering or cap. The arrival of the smooth muscle cells triggers a coalescence of the foam cells and small extracellular pools of lipid into a larger pool or lipid core. The T lymphocytes secrete cytokines that inhibit the further production of collagen, possibly leading to weakening and thinning of the fibrous cap and rendering it susceptible to rupture. With rupture or disruption of the plaque surface, tissue factor comes into contact with blood, and a thrombus is subsequently formed (
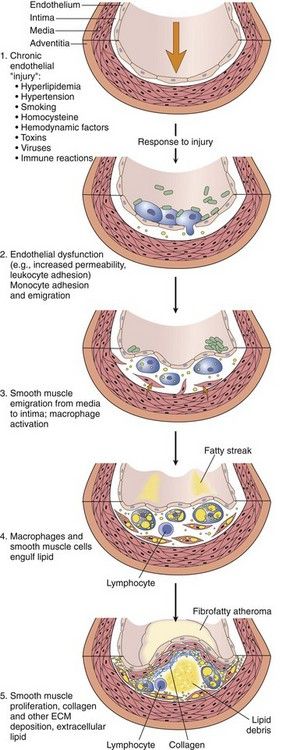
FIGURE 4-1 Evolution of arterial wall changes in the response to injury hypothesis. ECM, Extracellular matrix.
(From Schoen FJ: Blood vessels. In Kumar V, et al editors: Robbins and Cotran pathologic basis of disease, ed 8, Philadelphia, 2010, Saunders.)
Plaques may grow and proliferate outwardly, away from the lumen of the artery, or inwardly, into the lumen. With inward proliferation, the size of the lumen is progressively reduced (stenosis). Thus, blood flow may be chronically decreased, and when the demand for oxygen exceeds supply, the outcome is ischemic pain. Ischemic symptoms may be produced when occlusion reaches 75% of the cross-sectional area of the artery
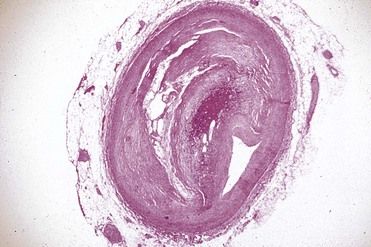
FIGURE 4-2 Photomicrograph of a cross section of a coronary artery with severe stenosis and narrowing.
(Courtesy W. O’Conner, MD, Lexington, Kentucky.)
Most acute coronary syndromes (e.g., unstable angina, myocardial infarction) are caused by physical disruption or fracture of the atheromatous plaque, most commonly of a plaque that did not cause extreme stenosis. In plaque rupture, the fibrous cap tears, allowing arterial blood to enter the lipid core, where contact with tissue factor and collagen induces platelet adhesion and aggregation and activation of the coagulation cascade. This series of events results in thrombus formation and sudden expansion of the lesion. Blood flow through the affected artery may become compromised or completely blocked.
Atherosclerosis usually is a focal disease that commonly occurs in certain areas or regions of arteries while sparing others. Those affected include the brain, heart, aorta, and peripheral arteries (
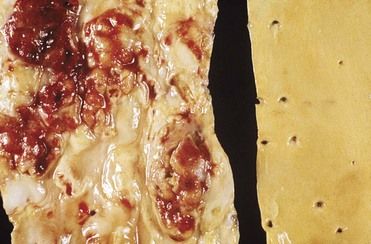
FIGURE 4-3 The segment of aorta on the left demonstrates advanced atheromatous plaques, and the specimen on the right side is unaffected.
(Courtesy W. O’Conner, MD, Lexington, Kentucky.)
The outcome of the atherosclerotic process is extremely variable. Some lesions never progress past the fatty streak phase; however, in most Western societies, the presence of frank plaques is the norm. Even so, most atheromatous plaques are not associated with clinical signs and symptoms and may never produce clinical manifestations.
Intraarterial complications of coronary atherosclerosis consist of luminal narrowing, intramural hemorrhage, thrombosis, embolism, and aneurysm. Intramural hemorrhage, which results from weakening of the intimal tissues, may lead to thrombosis. The localized blood also may serve as an irritant to precipitate a reflex reaction, resulting in spasm of the collateral vessels. Once formed, a thrombus may become encapsulated and may undergo fibrous organization and recanalization.
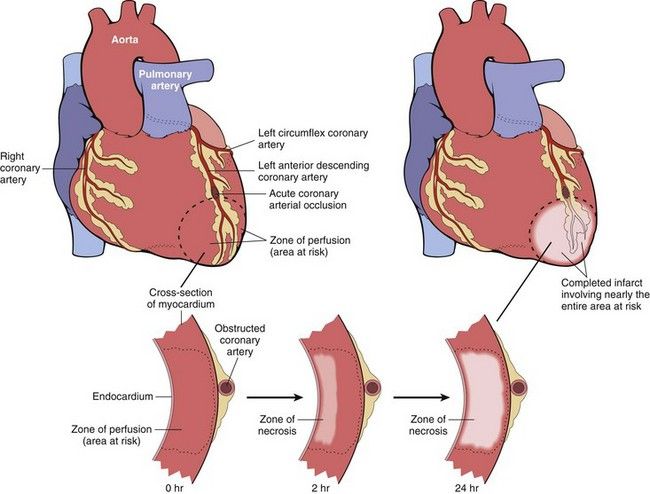
If the degree of ischemia that results from coronary atherosclerosis is significant and the oxygen deficit is prolonged, the area of myocardium supplied by that vessel may undergo necrosis. Reduced blood flow may result from thrombosis of the affected artery, a hypotensive episode, an increased demand for blood, or emotional stress. The infarct, or area of necrosis, may be subendocardial or transmural, the latter involving the entire thickness of the myocardium (
Clinical Presentation
Symptoms
Chest pain is the most important symptom of coronary atherosclerotic heart disease. The pain may be brief, as in angina pectoris resulting from temporary ischemia of the myocardium, or it may be prolonged, as in unstable angina or acute MI. Ischemic myocardial pain results from an imbalance between the oxygen supply and the oxygen demand of the muscle. Atherosclerotic narrowing of the coronary arteries is an important cause of this imbalance. The exact mechanism or agents involved in producing the cardiac pain are not known.
Angina pectoris usually is described as a sensation of aching, heavy, squeezing pressure or tightness in the midchest region. The area of discomfort often is reported to be approximately the size of a fist and may radiate into the left or right arm to the neck or lower jaw. In rare cases, it may be present in only one of these distant sites, and the patient is free of central chest pain. The pain is of brief duration, lasting 5 to 15 minutes if the provoking stimulus is stopped or for a shorter time if nitroglycerin is used. Angina is defined in terms of its pattern of symptom stability. Stable angina is pain that is predictably reproducible, unchanging, and consistent over time. Pain typically is precipitated by physical effort such as walking or climbing stairs but also may occur with eating or stress. Pain is relieved by cessation of the precipitating activity, by rest, or with the use of nitroglycerin. Unstable angina is defined as new-onset pain, pain that is increasing in frequency, pain that is increasing in intensity, pain that is precipitated by less effort than before, or pain that occurs at rest. This pain is not readily relieved by nitroglycerin. The key feature is the changing character (increasing intensity) or pattern of the pain. Patients with stable angina have a relatively good prognosis. Patients with unstable angina have a poorer prognosis and often experience an acute MI within a short time. The term acute coronary syndrome describes a continuum of myocardial ischemia that ranges from unstable angina at one end to non–ST segment MI at the other.
Patients with coronary atherosclerosis who experience prolonged pain as a result of myocardial ischemia usually have unstable angina or are having an acute MI. This pain usually is more severe and lasts longer than 15 minutes but has the same general character as that described for stable angina. Its location is the same as for the brief pain that results from temporary myocardial ischemia, and it may radiate in the same pattern. Use of vasodilators or cessation of activity does not relieve the pain caused by infarction. Neither brief nor prolonged pain resulting from myocardial ischemia is aggravated by deep breathing. Of interest, women and men report different symptoms of MI, with fewer women experiencing chest pain but more often experiencing fatigue and dyspnea.
Sudden cardiac death accounts for 300,000 deaths annually in the United States and is often, but not always, due to cardiac arrhythmia.
Palpitations of the heart (disagreeable awareness of the heartbeat) may be present in patients with coronary atherosclerotic heart disease with normal or abnormal rhythm. The complaint is not directly related to the seriousness of the underlying cardiac problem. Syncope, a transient loss of consciousness resulting from inadequate cerebral blood flow, also may occur in patients with coronary atherosclerotic heart disease.
Symptoms and signs of congestive heart failure developing as a complication of coronary atherosclerotic heart disease and its sequelae include dyspnea, orthopnea, paroxysmal nocturnal dyspnea, edema, hemoptysis, fatigue, weakness, and cyanosis. Fatigue and weakness may be manifest early in the course of heart disease, before the onset of congestive failure (see
Signs
Clinical signs of coronary atherosclerotic heart disease are few, and the patient’s clinical appearance may be entirely normal. Most clinical signs relate to other underlying cardiovascular disease or conditions such as congestive failure. Conditions such as corneal arcus and xanthoma of the skin are related to hyperlipidemia and hypercholesterolemia. Blood pressure may become elevated, and abnormalities in the rate and/or rhythm of the pulse may occur. Diminished peripheral pulses in the lower extremities may be noted, along with bruits in the carotid arteries. Panoramic radiographs of the jaws may occasionally demonstrate carotid calcifications in the areas of C3 and C4, consistent with atherosclerotic plaques in the carotid arteries (see
Laboratory Findings
Blood tests are used in the evaluation of patients with symptoms of angina pectoris to screen for abnormalities that may contribute to or worsen coronary heart disease. These tests include complete blood count to rule out anemia, thyroid function tests to exclude hyperthyroidism, renal function tests to exclude renal insufficiency, lipid screening for hypercholesterolemia, glucose screening for diabetes, homocysteine level determination, and C-reactive protein assay. Other diagnostic modalities that are specific for coronary heart disease include resting ECG, chest x-ray studies, exercise stress testing, ambulatory (Holter) electrocardiography, stress thallium-201 perfusion scintigraphy, exercise echocardiography, ambulatory ventricular function monitoring, and cardiac catheterization and angiography.
Along with physical examination and diagnostic testing, serum enzyme determinations are necessary to establish the diagnosis of acute MI and to determine the extent of infarction. Serum markers of acute MI most commonly used in clinical practice include troponin I, troponin T, and creatine kinase isoenzyme (CK-MB). These enzymes are released only when cell death (infarction) or injury to the myocyte occurs. Troponins are proteins that are derived from the breakdown of myocardial sarcomeres. For investigation of acute MI, troponin assays have largely replaced creatine kinase (CK) and CK-MB determinations because these markers are more specific in differentiating cardiac muscle damage from trauma to skeletal muscle or other organs. Very little difference has been noted between the two troponins. Plasma troponins are virtually absent in normal persons and are found only after cardiac injury. Troponins are first detectable 2 to 4 hours after the onset of an acute MI, are maximally sensitive at 8 to 12 hours, peak at 10 to 24 hours and persist for 5 to 14 days.
CK-MB is another enzymatic marker of cardiac cell injury with characteristics similar to those of the troponins; however, in addition to its presence in damaged heart, CK-MB is found after injury to skeletal muscle and other tissues. Despite this relative lack of specificity, elevated levels of CK/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses