3
Oral pain and movement disorders in aging
A CLINICAL SCENARIO
Clinical scenario
Mrs. G. is an edentulous 77-year-old woman being treated for chronic hypertension who experienced a major depressive illness recently. She had no prior psychiatric history and has never taken psychotropic drugs. She was referred to a psychiatrist, and during her first psychiatric assessment, she complained of moderately intense oral pain and displayed repetitive spontaneous chewing movements. The psychiatrist diagnosed a major depression and tardive dyskinesia, and prescribed quetiapine (Seroquel®, Astra Zeneca Group of Companies, London, UK), which is a second-generation antipsychotic drug. Shortly after this, Mrs. G’s physician referred her for a dental assessment.
INTRODUCTION
Orofacial pain is a common problem that occurs in at least 1 in 10 adults but in more than half of the elderly population (Madland and Feinmann, 2001). It is classified according to source or cause of the pain (Table 3.1). The complex category encompasses three distinct entities: (1) primary headache disorders; (2) neuralgias; and (3) persistent idiopathic orofacial pain, which includes burning mouth syndrome, arthromyalgia, atypical odontalgia, and atypical facial pain (Woda et al., 2005).
Table 3.1 Source and distribution of chronic orofacial pain in older people
(adapted from Woda et al., 2005)
.
| Source | Distribution |
| Primary headache disorders | |
| Migraine | 3%–5% lifetime prevalence for chronic daily headache |
| Cluster headache | |
| Tension-type headache | |
| Hemicrania continua | |
| Others | |
| Neuralgias | |
| Essential trigeminal neuralgia (tic douloureux) | Prevalence: 1.6% >age 84 (women: 6.3%; men: 2.7%) |
| Symptomatic trigeminal neuralgia | Rare |
| Postherpetic neuralgia | 20% 3 months after rash |
| Glossopharyngeal neuralgia | Rare |
| Central neuralgia | 11% after stroke |
| Persistent idiopathic orofacial pain | |
| Burning mouth syndrome (stomatopyrosis) | 1.7% (1%–15%) |
| Arthromyalgia (temporomandibular disorder) | 7.7% |
| Atypical odontalgia (phantom tooth pain) | 3%–6% postendodontic treatment |
| Atypical facial pain | 3% |
Chronic orofacial pain in people over 60 years presents particular diagnostic and management challenges (Riley et al., 1998). It can originate peripherally from an intraoral, extraoral, musculoskeletal, or neural source, or in the central nervous system (CNS). Either way, the pain is confounded by age-related changes exacerbated by dental disorders, tooth loss, denture use, malnutrition, cancer, and by various drugs. Trigeminal and postherpetic neuralgias, along with other atypical orofacial pains, occur primarily in the older age group. Indeed, age-related changes in the CNS, and neurodegenerative conditions affecting monoamine transmission and cognitive processes, such as Parkinson’s and Alzheimer’s diseases, have a significant influence on how pain is processed, reported, and assessed (Cole et al., 2008). Moreover, oral dyskinesias in people who wear dentures can be particularly painful. Chronic pain upsets quality of life by disturbing cognitive, psychological, and functional status of elders as they grow frail (Nitschke and Müller, 2004), although we should not underestimate the adaptive and coping skills of people in old age (Vickers and Boocock, 2005; Brondani et al., 2007).
This chapter presents the relevant disease processes by which chronic orofacial pain and disorders of oral movement occur along with related management approaches suitable for frail elders.
PAIN ASSESSMENT IN ELDERS
We know quite well how elderly people report or complain about pain in general (Hadjistavropoulos et al., 2007), but we know much less about responses to chronic orofacial pain. Self-reporting is the most reliable source of information about an older patient’s pain, even in the presence of mild to moderate dementia. However, several factors contribute to underdetection of pain and delayed pain management. Elders commonly have difficulty communicating their pain and distress, probably because of denial, stoicism, or neurological impairment; however, compared with younger adults, they are usually less angered or distressed by chronic pain (Vickers and Boocock, 2005). Single pain assessment scales are unreliable for patients with moderate to severe dementia, particularly when complicated by delirium or depression (Hadjistavropoulos et al., 2008). Consequently, reports from caregivers along with signs of grimacing, guarding, or reluctance to eat are more reliable indicators of pain (Ettinger, 2000; Pautex et al., 2006). Patients who cannot communicate or who are agitated and unresponsive to neuroleptic drugs can use a map or drawing of the face to help locate and record the source of the pain. Similarly, a short trial of acetaminophen or narcotics helps to clarify the extent of the pain. These situations, along with fear of analgesic-induced side effects and dependence, can delay diagnosis and adequate pain relief, which can in turn lead to delirium or cognitive deterioration in frail elders (Morrison et al., 2003). Consequently, all complaints of pain should be taken seriously, and changes in mood, eating, social behavior, or sleep should prompt a search for a source of pain.
CHANGES IN PROCESSING PAIN
Overall, older people have a similar or slightly higher pain threshold but lower pain tolerance compared with younger people (Gagliese, 2007). Loss of natural teeth does not seem to alter thresholds of orofacial pain (Blanchet et al., 2008), although tooth loss and old age decrease the density of myelinated fibers in the trigeminal nerve (Nonaka et al., 2001; Ishikawa et al., 2005). Damage to the neural pathways that detect noxious stimuli can result in orofacial neuropathic pain or “neuralgia.”
Neuralgias are caused by various events ranging from peripheral nerve damage to increased excitability of neurons in the CNS (Woda and Salter, 2008). Central transmission of noxious stimuli may be facilitated by advancing age due to reductions in neurotransmitters, such as serotonin and norepinephrine (Iwata et al., 2004; Karp et al., 2008). Dopamine is also implicated in descending inhibition pathways, but how it contributes to pain in the dopamine-deficient Parkinson’s nervous system is unclear (Mylius et al., 2009).
OROFACIAL PAIN
Orofacial pain, which occurs in about one in five elders in the United States, arises primarily from diseases affecting the teeth, periodontium, jaw joints, and oral mucosa (Riley et al., 1998). Jaw pain and facial pain seem to occur more frequently in women than in men. Painful mucosal ulcers and stomatitis are particularly common in elderly denture-wearers (Jainkittivong et al., 2002). Chronic periodontitis is not usually painful (Brunsvold et al., 1999), whereas inflammatory vesiculoerosive diseases (e.g., lichen planus; erythema multiforme, mucous membrane pemphigoid), oral mucositis from chemotherapy and head-neck radiation therapy, and some malignant and premalignant lesions are usually very painful (Silverman, 2007).
Neuralgic pain
Central and peripheral neuropathic pain is persistent and caused by a primary lesion or dysfunction of the nervous system. About 1% of the general population suffer from it, but it is more prevalent in elderly people with arteriosclerosis, cerebral vascular disease, diabetes, cancer, chronic inflammatory conditions, shingles, or immune deficiencies. The peripheral type is associated typically with trigeminal neuralgia, postherpetic neuralgia, and painful diabetic neuropathy (Schmader, 2002; Hadjistavropoulos et al., 2007).
Trigeminal neuralgia
This neuralgia, also called tic douloureux, is a very distressing pain that strikes suddenly, intensely, recurrently, and uncontrollably (De Simone et al., 2008). It occurs with a lifetime prevalence of up to 1.6% in the general population, but more so among women than men (3:2), and the 1-year prevalence when aged 85–94 years reaches about 6% in women and 3% in men (Schwaiger et al., 2009). People older than 50 years are more susceptible to trigeminal neuralgia probably because cerebellar vascular loops pulsate at this age against the trigeminal nerve, especially as the root of the nerve enters the pons in the brainstem where there can be a loss of central myelin (Rappaport et al., 1997). Consequently, excitation of the small-diameter afferent sensory nerve fibers of the face can cause paroxysmal pain. However, the exact cause and pathogenesis of the neuralgia are unclear. The painful jabs are always acute, intense, and excruciating, often described as electrical discharges. Typically, they are brief, but can occur in clusters lasting 1–2 min. They recur several times a day, followed by a refractory period. Most often the pain is unilateral, limited to the infraorbital portion of the maxillary branch of the trigeminal nerve, and begins in the upper lip, gums, or teeth, on the side of the nose, or occasionally around the mental nerve. Accurate diagnosis is based on very definite painful attacks in response to tactile stimulation of specific trigger zones, while neurological responses are normal between attacks. Facial pain manifesting in other ways is unlikely to be trigeminal neuralgia.
Management of trigeminal neuralgia
Anticonvulsants, such as carbamazepine or oxcarbazepine, are the first approach to managing trigeminal neuralgia. It might be necessary to supplement the anticonvulsant with a muscle relaxant, such as a gamma-aminobutyric acid (GABA)-enhancing drug (e.g., baclofen, clonazepam), or to switch to a different anticonvulsant (e.g., lamotrigine, pregabalin, or levetiracetam). However, when pharmacotherapy is unsuccessful, relief for the neuralgia might be obtained with percutaneous controlled radiofrequencies to produce a lesion in the Gasserian ganglion, surgical microvascular decompression of the nerve rootlets, or radiosurgery to relieve the pain (Sekula et al., 2008). Botulinum toxin has been used intradermally to reduce the pain of trigeminal neuralgia (Zuniga et al., 2008) and other types of focal chronic neuropathic pain including postherpetic neuralgia (Ranoux et al., 2008). It can reduce pain intensity and mechanical allodynia, while sparing thermal sensibility.
However, there is no consensus on how best to manage trigeminal neuralgia in people who are frail. If anticonvulsant medications fail, obviously, the less invasive and risky percutaneous approach is preferable, although this can produce a numb and very painful area on the face (anesthesia dolorosa) in a small proportion (<5%) of patients (Rasmussen, 1965). It remains a difficult challenge.
Other trigeminal lesions
Ischemic, compressive (e.g., tumors, aneurysms), demyelinating (e.g., multiple sclerosis), traumatic, or infiltrative lesions associated with the trigeminal nerve origin can cause symptoms of trigeminal neuralgia, although without the trigger spots or the refractory relief. However, even if these atypical forms respond to anticonvulsants, further investigations are needed to locate the source of the problem because of neoplastic possibilities (e.g., meningioma). Numbness of the chin or other discomforts in the gum and teeth innervated by the inferior alveolar nerve suggests a malignancy within the mandible unless there is a more obvious dental source of the symptoms. Typical neuralgic pain associated within the distribution of other cranial nerves (e.g., glossopharyngeal neuralgia in the tonsillar fossa) can occur, but very infrequently, and it is treated much like trigeminal neuralgia.
Postherpetic neuralgia
More than two-thirds of herpes zoster cases occur in people over 60 years of age. The general incidence of herpes zoster based on data from a health maintenance organization in the United States in the early 1990s was estimated at 215 per 100,000 person-years (100,000 person-years = 10,000 persons followed for 10 years; or 20,000 persons for 5 years), representing a 64% increase compared with the preceding 30 years (Donahue et al., 1995). It is also about sixfold higher in older people, possibly because of an age-related decline in cell-mediated immunity to the varicella zoster virus. Older persons who are immunocompromised are particularly at risk.
Acute pain is often experienced prior to the appearance of the shingles rash (Figure 3.1).
Figure 3.1 Herpes zoster “shingles” rash distributed along the mandibular branch of the trigeminal nerve.
(Photograph courtesy of Dr. Michelle Williams, Vancouver, BC, Canada.).
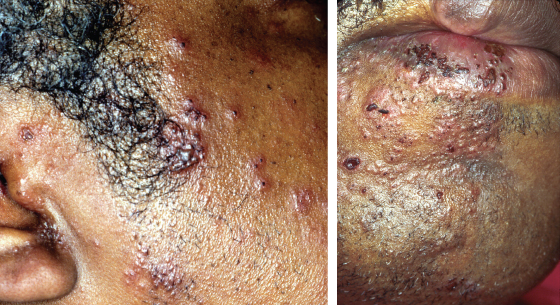
The rash resolves within 4 weeks and the pain decreases, but painful responses to normal stimuli along with an itch are common. The prevalence of pain at 1 month after the onset of the rash rises sharply after age 50 years (Choo et al., 1997; Stankus et al., 2000). Postherpetic neuralgia—pain persisting more than 3 months after the onset of the rash—occurs in about one in five people over 50 years of age, but drops to less than 1 in 10 people after a year. The ophthalmic division of the trigeminal nerve is more usually affected than the other two divisions.
Management of postherpetic neuralgia
Unfortunately, the acute antiviral treatment (famciclovir, valaciclovir) for herpes zoster does not reliably prevent the neuralgia even if taken within 72 h following onset of the rash. A vaccine reduced the incidence of neuralgia by about two-thirds in older populations (Oxman et al., 2008). However, the standard management includes anticonvulsants (e.g., gabapentin, pregabalin), tricyclic antidepressants (e.g., amitriptyline), 5% or 10% lidocaine topical cream or patch, and tramadol, an opioid analgesic (Moulin et al., 2007; Tyring, 2007). Botulinum toxin has been used intradermally with some success (see management of trigeminal neuralgia above). Anticonvulsants and topical lidocaine are the first-line treatment, while the narcotics are reserved for refractory pain.
Central neuralgia
Acquired lesions along the trigeminothalamic nociceptive pathways can cause severe facial pain resembling trigeminal neuralgia or continuous unpleasant sensations (dysesthesias). In contrast to essential trigeminal neuralgia, a sensory deficit occurs in or adjacent to the painful area, and the pain more commonly involves both sides of the body. Central neuralgia occurs in about 1 in 10 people following a stroke (Bowsher, 2001). Sensitivity to thermal pain decreases following thalamic strokes, and heat-pain thresholds are lower in patients with Parkinson’s disease (Djaldetti et al., 2004). There is no doubt that central pain is a dreadful condition and notoriously refractory to treatment even with narcotics or anticonvulsants such as pregabalin (Gray, 2007).
Persistent idiopathic orofacial pain
Acute orofacial pain is usually from a toothache but conditions identified as persistent idiopathic orofacial pain (Table 3.1) are less localized or likely to respond favorably to dental treatment. Consequently, they are often attributed incorrectly to psychological disturbances (Lipton et al., 1993).
Burning mouth syndrome
A burning sensation in the mouth lasting at least 4–6 months without mucosal lesions or other clinical findings occurs occasionally between 50 and 70 years of age. This “burning mouth syndrome” (stomatodynia, stomatopyrosis) disturbs a small (1%–15%) proportion of the population, with women more commonly (sevenfold) affected than men (Ship et al., 1995). Typically, the pain involves the tip and anterior two-thirds of the tongue, but it can involve the lips, hard palate, and edentulous alveolar ridge. Pain levels can be intense with a typical crescendo pattern peaking in the evening, reduced when eating, and absent overnight. The onset is usually spontaneous without an obvious cause, although in one-third of cases it is associated with recent dental treatment, illness, or drug treatment. Xerostomia, dysgeusia, and decreased taste can accompany the pain, but it is unclear whether these are part of the primary condition.
Other conditions have been associated with the syndrome, notably oral mucositis, nutritional (e.g., vitamin B complex, zinc) deficiencies, anemia, diabetes, hormonal changes, Sjögren’s syndrome, neuropsychiatric disorders, and antibiotic treatments (Ship et al., 1995). It has also been associated with Parkinson’s disease (Clifford et al., 1998) and dopamine deficiency (Jääskeläinen et al., 2001).
There is some evidence that the burning sensation originates in the tongue because of abnormal sensory thresholds for heat (Grushka and Sessle, 1991) and taste (Grushka and Sessle, 1991; Eliav et al., 2007), and small-fiber sensory neuropathy in biopsy specimens from the tongue of people who complain of the burning sensation (Lauria et al., 2005).
Management of burning mouth syndrome
Patients with burning mouth syndrome can be managed successfully with a low dose of a tricyclic antidepressant (e.g., amitriptyline 10–75 mg) at bedtime, or 300–600 mg of an anticonvulsant (e.g., gabapentin) three times daily. Other pharmacological treatments have been suggested but with limited evidence, such as clonazepam 1 mg tid sucked for 3 min (Gremeau-Richard et al., 2004); or alphalipoic acid supplements 200 mg three times daily (Femiano and Scully, 2002).
Atypical odontalgia
Constant and moderately intense toothache locali/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


