Surgical Reconstruction of Defects of the Jaws
Edward Ellis III.
Defects of the facial bones, especially the jaws, have a variety of causes, such as eradication of pathologic conditions, trauma, infections, and congenital deformities. The size of the defects that are commonly reconstructed in the oral-maxillofacial region varies considerably from small alveolar clefts to mandibulectomy defects. Each defect poses a unique set of problems that reconstructive surgical intervention must address. In each of these instances, restoration of normal structure is usually possible, with resultant improvement in function and appearance.
When an osseous structure is defective in size, shape, position, or amount, reconstructive surgery can replace the defective structure. The tissue most commonly used to replace lost osseous tissue is bone. Bone grafting has been attempted for centuries with varying degrees of success. Recent advancements in the understanding of bone physiology, immunologic concepts, tissue-banking procedures, and surgical principles have made possible the successful reconstruction of most maxillofacial bony defects. As such, the biology and principles of transplantation of bone are presented in this chapter.
Biologic Basis of Bone Reconstruction
A tissue that is transplanted and expected to become a part of the host to which it is transplanted is known as a graft. Several types of grafts are available to the surgeon, which are discussed later. A basic understanding of how a bone heals when grafted from one place to another in the same individual (i.e., autotransplantation) is necessary to understand the benefits of the various types of bone grafts available.
The healing of bone and bone grafts is unique among connective tissues because new bone formation arises from tissue regeneration rather than from simple tissue repair with scar formation.1 This healing, therefore, requires the element of cellular proliferation (i.e., osteoblasts) and the element of collagen synthesis. When bone is transplanted from one area of the body to another, several processes become active during the incorporation of the graft.
Two-Phase Theory of Osteogenesis
Two basic processes occur on transplanting bone from one area to another in the same individual.1–5 The first process that leads to bone regeneration arises initially from transplanted cells in the graft that proliferate and form new osteoid. The amount of bone regeneration during this phase depends on the number of transplanted bone cells that survive the grafting procedure. Obviously, when the graft is first removed from the body, the blood supply has been severed. Thus, the cells in the bone graft depend on diffusion of nutrients from the surrounding graft bed (i.e., the area where the graft is placed) for survival. A considerable amount of cell death occurs during the grafting procedure, and this first phase of bone regeneration may not lead to an impressive amount of bone regeneration when considered alone. Still, this phase is responsible for the formation of most of the new bone. The amount of bone that will form depends on the amount of viable cells that can be successfully transplanted with the graft.
The graft bed also undergoes changes that lead to a second phase of bone regeneration beginning in the second week. Intense angiogenesis and fibroblastic proliferation from the graft bed begin after grafting, and osteogenesis from host connective tissues soon begins. Fibroblasts and other mesenchymal cells differentiate into osteoblasts and begin to lay down new bone. Evidence shows that a protein (or proteins) found in the bone induces these reactions in the surrounding soft tissues of the graft bed.6,7 This second phase is also responsible for the orderly incorporation of the graft into the host bed with continued resorption, replacement, and remodeling.
Immune Response
When a tissue is transplanted from one site to another in the same individual, immunologic complications usually do not occur. The immune system is not triggered because the tissue is recognized as “self.” However, when a tissue is transplanted from one individual to another or from one species to another, the immune system may present a formidable obstacle to the success of the grafting procedure. If the graft is recognized as a foreign substance by the host, it will mount an intense response in an attempt to destroy the graft. The type of response the immune system mounts against “foreign” grafts is primarily a cell-mediated response by T lymphocytes. The response may not occur immediately, however, and in the early period the incorporation of a bone graft into the host may appear to be progressing normally. The length of this latent period depends on the similarity between the host and the recipient. The more similar they are (antigenically), the longer it may take for an immunologic reaction to appear. This type of immunologic reaction is the most common reason for rejection of hearts, kidneys, and other organs transplanted to another individual. Tissue-typing procedures, in which a donor and recipient are genetically compared for similarities before transplantation, are currently commonplace for organ transplantation but never for bone grafts.
Because of the immunologic rejection of transplants between individuals or between species, methods have been devised to improve the success of grafting procedures in these instances. Two basic approaches are used clinically: The first is the suppression of the host individual’s immune response. Immunosuppression with various medications is most commonly used in organ transplant recipients. This approach is not used routinely in oral-maxillofacial surgical bone grafting procedures because of the potential complications from immunosuppression.
Another approach that has been used extensively in oral and maxillofacial surgical procedures is the alteration of the antigenicity of the graft so that the host’s immune response will not be stimulated. Several methods of treating grafts have been used, including boiling, deproteinization, use of thimerosal (Merthiolate), freezing, freeze-drying, irradiation, and dry heating. All of these methods, potentially helpful for use in bone grafts, are obviously not helpful in organ transplants.
Types of Grafts
Several types of bone grafts are available for use in reconstructive surgery. A useful classification categorizes the bone grafts according to their origin and, thus, their potential to induce an immunologic response. Because of their origins and the preparations used to help avoid an intense immune response, the grafts have different qualities and indications for use.
Autogenous Grafts
Also known as autografts or self-grafts, autogenous grafts are composed of tissues from the same individual. Fresh autogenous bone is the most ideal bone graft material. The autogenous graft is unique among bone grafts in that it is the only type of bone graft to supply living, immunocompatible bone cells essential to phase I osteogenesis. The larger the number of living cells transplanted, the more will be the osseous tissue produced.
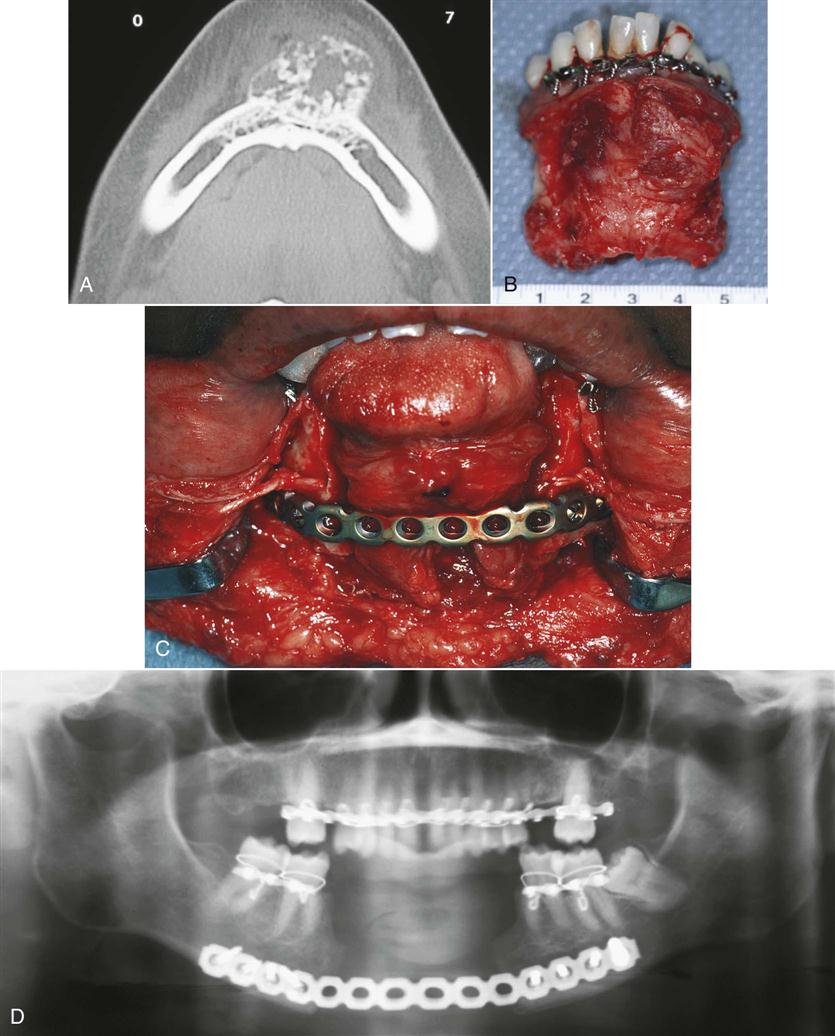
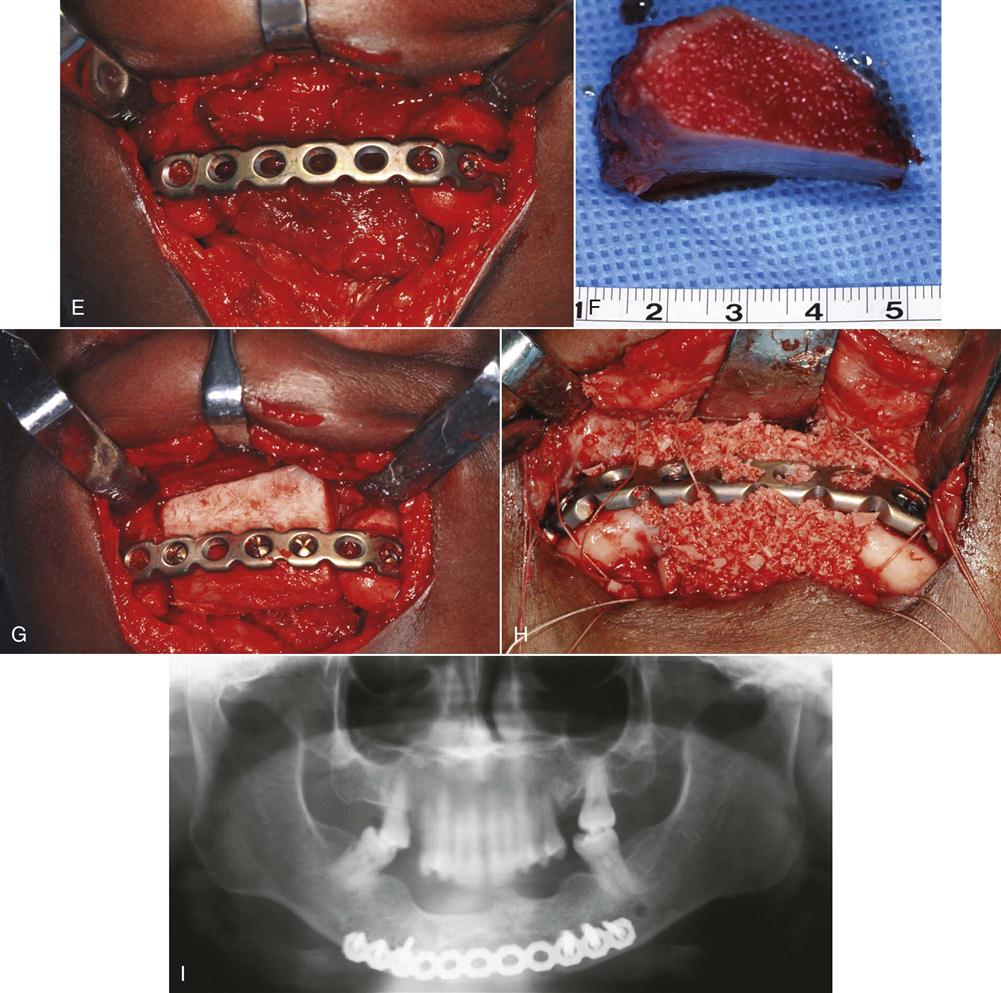
Autogenous bone is the type used most frequently in oral-maxillofacial surgery. The bone to be transplanted can be obtained from a host of sites in the body and can be taken in several forms. Block grafts are solid pieces of cortical bone and underlying cancellous bone (Figure 29-1). The iliac crest is often used as a source for this type of graft. The entire thickness of the ilium can be obtained, or the ilium can be split to obtain a thinner piece of block graft. Ribs also constitute a form of block graft. Particulate marrow and cancellous bone grafts are obtained by harvesting the medullary bone and the associated endosteum and hematopoietic marrow. Particulate marrow and cancellous bone grafts produce the greatest concentration of osteogenic cells, and because of the particulate nature, more cells survive transplantation because of the access they have to nutrients in the surrounding graft bed. The most common site for the procurement of this type of graft is the ilium. The iliac crest can be entered, and large volumes of particulate marrow and cancellous bone grafts can be obtained with large curettes. The diploic space of the cranial vault has recently been used as a site for obtaining this type of graft when small amounts of bone chips are needed (e.g., alveolar cleft grafts).
Autogenous bone may also be transplanted while maintaining the blood supply to the graft. Two methods can accomplish this: The first involves the transfer of a bone graft pedicled to a muscular (or muscular and skin) pedicle. The bone is not stripped of its soft tissue pedicle, preserving some blood supply to the bone graft. Thus, the number of surviving osteogenic cells is potentially great. An example of this type of autogenous graft is a segment of the clavicle transferred to the man/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses