26 RESIN-BONDED FIXED DENTAL PROSTHESES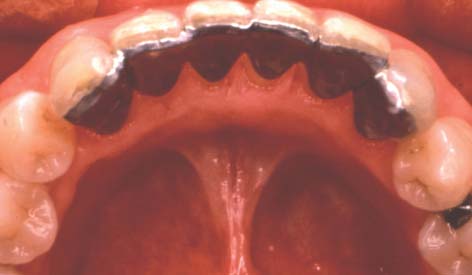
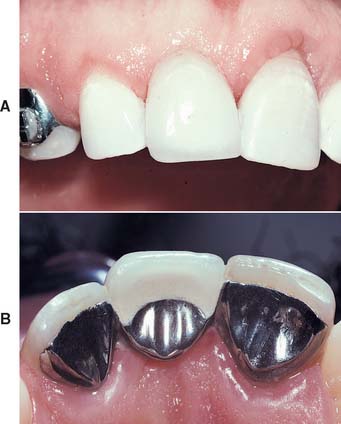
Resin-bonded fixed dental prostheses (FDPs) have had variable popularity since the technique for splinting mandibular anterior teeth with a perforated metal casting was described by Rochette in 1973.1 His work suggested an alternative to conventional metal-ceramic fixed FDPs and its substantial removal of tooth structure needed to create strong, anatomically contoured, and esthetic restorations (see Chapter 7). A prosthesis that requires minimal removal of tooth structure is appealing, particularly for abutment teeth that are intact and caries free. The primary goal of the resin-bonded partial FDP is the replacement of missing teeth and maximum conservation of tooth structure.
The advent of electrolytic etching of the metal surface to provide micromechanical retention for metal adhesion to enamel led to the technique’s broad application.2 The restoration is simple in concept and consists of one or more pontics supported by thin metal retainers bonded lingually and proximally to the enamel of the abutment teeth (Fig. 26-1). These conservative prostheses depend on bonding between etched enamel and the metal casting and require precise and defined metal engagement of the abutment. The early use of these bonded retainers tested the limits of its application. There was a limited understanding of the treatment planning required and of the degree of resistance and retention form necessary. In general use, poor design of the early retainers (with some bonded only to lingual enamel) was compounded by the difficulty of properly etching the metal. As a result, several failures occurred, and the technique was used more conservatively from 1986 to 1996. During and since that period, design parameters have been enumerated and tested clinically.3–5 Such designs, combined with new technologies for adhesive bonding of resin to most alloys, have led to a simpler, more reliable prosthetic procedure that complements the dentist’s prosthodontic armamentarium.
DEVELOPMENT OF RESIN-BONDED FIXED DENTAL PROSTHESES
Bonded Pontics
The earliest resin-bonded prostheses were extracted natural teeth or acrylic teeth used as pontics bonded to the proximal and lingual surfaces of abutment teeth with composite resin.6,7 The composite resin connectors were brittle and required supporting wire or a stainless steel mesh framework. These bonded pontics were limited to short anterior spans and had a limited lifetime, with degradation of the composite resin bond to the wire or mesh and subsequent fracture. Such restorations should be presented to patients only as short-term replacements.8–10
Cast Perforated Resin-Bonded Fixed Dental Prostheses (Mechanical Retention)
In 1973, Rochette1 introduced the concept of bonding metal to teeth by using flared perforations of the metal casting to provide mechanical retention. He used the technique principally for periodontal splinting but also included pontics in his design. Howe and Denehy11 recognized the metal framework’s improved retention (over bonded pontics) and began using FDPs with cast-perforated metal retainers bonded to abutment teeth and metal-ceramic pontics to replace missing anterior teeth. Their design recommendation, extending the framework to cover a maximum area of the lingual surface, suggested little or no tooth preparation. Patient selection limited these FDPs to mandibular teeth or situations with minimal occlusal contact. The restorations were bonded with a heavily filled composite resin as a luting medium.
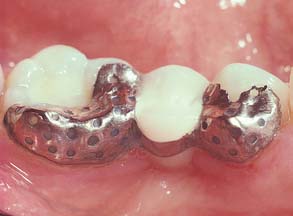
This concept was expanded to replacement of posterior teeth by Livaditis.12 Perforated retainers were used to increase resistance and retention. Cast retainers were extended interproximally into the edentulous areas and onto occlusal surfaces. The design included a defined occlusogingival path of placement by tooth modification, which involved lowering the proximal and lingual height of contour of the enamel on the abutment teeth. These restorations were placed in normal occlusion; many have survived and have been seen on recall for up to 13 years (Fig. 26-2). Despite this success, the perforation technique presents the following limitations:
Clinical results with the perforated technique were monitored for 15 years in a study at the University of Iowa.14 The results from this well-controlled study suggest that for anterior FDPs, the perforated retainers have a 50% failure at 110 months and 63% at about 130 months (Table 26-1).
Table 26-1 ESTIMATE TIME TO 50% FAILURE (DEBONDINGS) IN STUDIES WITH AT LEAST 10-YEAR MEAN
| Study | Months to 50% failure |
|---|---|
| UNIVERSITY OF IOWA | |
| Perforated design | 110 |
| Etched metal* | 250 |
| UNIVERSITY OF MARYLAND | |
| Etched metal† | 190 |
* Boyer et al, 1993: 143 anterior and 30 posterior fixed dental prostheses (FDPs).14
Etched Cast Resin-Bonded Fixed Dental Prostheses (Micromechanical Retention—“Maryland Bridge”)
A technique for the electrolytic etching of cast base metal retainers was developed at the University of Maryland by Thompson and Livaditis.2,15 Etched cast retainers have definite advantages over the cast-perforated restorations:
During the course of this work, the need for a composite resin with a low film thickness for luting the casting became apparent. This led to the first generation of resin cements, which allowed micromechanical bonding into the undercuts in the metal casting created by etching and simultaneously provided adequate strength and allowed complete seating of the cast retainers. Comspan,* the first of these cements, was moderately filled (60% by weight) with a film thickness of approximately 20 μm.16 Such cements are not chemically adhesive to the metal.
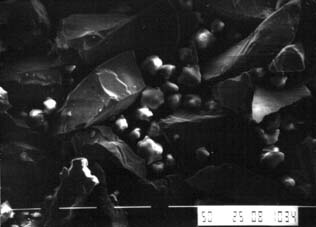
Electrolytic etching of base metal alloys proved to be critically dependent on the base metal alloy and attention to detail in the laboratory. Initial etching methods were developed for a nickel-chromium (Ni-Cr) alloy† and a nickel-chromium-molybdenum-aluminum-beryllium (Ni-Cr-Mo-Al-Be) alloy.17‡ These methods were followed by simplified techniques,18 chemical etching,19 or gel etching.20 They all yield similar results, provided that the technique is optimized for a specific alloy.21 Proper etching requires evaluation of the alloy surface with a scanning electron microscope. The degree of undercut created by this etching process can be seen in Figure 26-3. Lack of attention to detail can result in electropolishing or surface contamination.22 With time, both severely degrade bond strengths in a moist environment.
Highly variable results were reported for dental laboratories in etching the same alloy.23 Etching and bonding techniques were adopted on the basis of bond strength testing of specimens subjected to only 24 hours or 7 days of water exposure. When resin-to-metal test specimens were aged for 6 months in water and then thermally stressed by 10,000 or more thermal cycles, large reductions in bond strengths were recorded.24,25 Therefore, data from specimens that have not been aged and thermally stressed should be viewed skeptically. Even particle abrasion provides initially high resin-to-metal bonds, which can degrade to almost zero with time.26
Well-researched and tested resin systems for direct adhesion to metal surfaces have now completely supplanted metal etching as retention mechanisms.27 This is discussed subsequently.
Macroscopic Mechanical Retention Resin-Bonded Fixed Dental Prostheses (“Virginia Bridge”)
As a result of concerns about etching base metal and the desire to use alternative alloys, several methods have been developed to provide visible macroscopic mechanical undercuts on the inner surface of FDP retainers. The first was developed at the Virginia Commonwealth University School of Dentistry and is known as the Virginia Bridge.28 It involves a “lost salt crystal” technique. On the definitive cast, the abutments are coated with a model spray, and a lubricant is then applied. Within the outlines of the retainers, specially sized salt crystals* (150 to 250 μm) are sprinkled over the surface in a uniform monolayer, leaving a 0.5-mm border without crystals at the periphery of the pattern. This is followed by application of a resin pattern. After pattern investment, the salt crystals are dissolved from the surface of the pattern. Adequate bond strengths are possible with this method,29 but casting thickness must be increased to allow for the retention layer. Although no long-term results have been reported with this technique, it does allow the use of almost any metal-ceramic alloy. In general, however, macroscopic mechanical retention methods have been supplanted by direct adhesion with resin, which is possible for most casting alloys if the correct surface treatment is provided.
Chemical Bonding Resin-Bonded Fixed Dental Prostheses (Adhesion Bridges)
During the 1980s and early 1990s, when etched castings were the method of choice for retention of resin-bonded FDPs, extensive research was under way in Japan to develop adhesive systems for direct bonding of metal for this application. The first of these resin systems† is based on a formulation of a methyl methacrylate polymer powder and methyl methacrylate liquid modified with the adhesion promoter 4-methacryloxyethyl-trimellitic anhydride (4-META).30 It was developed with a unique tri-n-butylborane catalyst system that is added to the liquid before combining with the powder. On base metal alloys, Super-Bond has the highest initial bond strengths of any adhesive resin system. Unfortunately, the hydrolytic stability of these bonds over time depended on the alloy’s Cr-Ni ratio.31,32 Its advantages include its lower elastic modulus and higher fracture toughness in comparison with bisphenol-A-glycidyl dimethacrylate (bisGMA)–based resin cements,33,34 yielding better clinical results with less well-adapted castings.35 This system had shown poor clinical results with bonding high–gold content alloy retainers to abutment teeth.36 However, alloy primers have been developed that provide a more stable bond to noble alloy surfaces,37,38 which has been confirmed in a clinical study.39
Super-Bond’s introduction was followed by a bisGMA-based composite resin luting cement* that is modified with the adhesion promoter 10-methacryloxydecyl dihydrogen phosphate (MDP). MDP’s chemical structure and use are described in the literature.40
Panavia resin luting agent has shown excellent bonds to particle-abraded Ni-Cr and chromium-cobalt (Cr-Co) alloys41,42 as well as tin-plated gold and gold palladium-based alloys.21,43
Panavia has a tensile bond to etched enamel (10 to 15 MPa) comparable with that of the traditional bisGMA low-film thickness composites (e.g., Comspan and Conclude). The combination of metal electrolytic etching, followed by application of an adhesive such as Panavia, does not improve the tensile bond to the alloy, and its strength is actually slightly lower than that of the bond of Panavia to airborne particle abraded (sandblasted) base metal alloys.44 The most recent versions are Panavia 21 and Panavia F 2.0, the latter being a dual cure system (chemical and visible light) that releases fluoride. Both incorporate a self-etching primer system (ED Primer) for bonding to enamel and dentin.
Tin plating of noble alloys allows resin-to-metal bond tensile bond strengths only slightly lower than those for either the electrolytically etched or particle abraded Ni-Cr-Be alloys (18 to 30 MPa). However, tensile bond strengths are certainly greater than the bond to etched enamel.45,46 Tin plating of the metal surface also requires particle abrasion of the alloy surface just before bonding for adequate tin nucleation sites (Fig. 26-4).47,48 Tin plating can be completed in the dental laboratory, chairside, or intraorally to achieve metal bonding. One popular tin-plating system† (Fig. 26-5A) involves the use of a tin amide solution, which is applied to the metal surface with a saturated cotton pledget held on the end of a battery-powered probe (4 V). The probe is grounded elsewhere on the metal (Fig. 26-5B). Tin-plating times are usually 5 to 10 seconds, and a light gray surface is produced. Plating is followed by copious rinsing with water and drying; the adhesive resin is then applied.
Particle abrasion of the alloy surface with 50 μm alumina before bonding or tin plating not only creates a roughened, higher surface area substrate for bonding but also creates a molecular coating of alumina.49 The alumina on the surface aids in oxide bonding of the phosphate-based adhesive systems (e.g., Panavia to alloy surfaces). Studies of this bonding mechanism are also reinforced by laboratory data on bonding to alumina and zirconia surfaces.50–52
These adhesive systems have now shown nearly the same degree of long-term clinical bonding (since 1983 in Japan) as the conventional composites on etched base metal (since 1981 in the United States).15 Laboratory data support their efficacy. The favorable findings for direct adhesion to base metal have rendered alloy etching and macroscopic retention mechanisms obsolete.53 This simplifies the laboratory and clinical procedures for placement of resin-bonded FDPs.
A laboratory method for resin bonding to both base and noble metal is the Rocatec System.* In this method, the metal surface is initially particle abraded with 120-μm alumina. This is followed by abrasion with a special silicate particle–containing alumina (Fig. 26-6). This second particle abrasion step deposits a molecular coating of silica and alumina on the alloy surface. Silane is then applied to the surface, making it adhesive to composite resin. Various silane application techniques have been compared by Norling and colleagues.54 The Rocatec system has been compared with Panavia for bonding to a range of surfaces and is adequate in this regard.25,27,49 However, it requires careful laboratory technique and is generally confined to bonding composite resin veneers to alloy castings because of the concern that the silane-treated surface may become contaminated before or during the clinical bonding procedures.
Since the mid-1990s, several competing primer and cement systems for direct adhesive bonding to noble alloys have evolved (Table 26-2). All entail particle abrasion of the alloy surface, followed by application of a primer and then the resin cement. These primers have been studied in the laboratory55–59 and evaluated clinically.39,60 Their usage is justified and simplifies bonding to both gold- and palladium-based alloys.
Table 26-2 Adhesive Resin Cements and Primers for Noble Alloys
| Cement | Primer | Manufacturer |
|---|---|---|
| Bistite II DC | Metaltite | Tokyuyama Dental |
| CBMetabond | MTL-V Primer | Parkell Inc. |
| Linkmax | Metal Primer II | GC America |
| Multilink | Multilink Primer | Ivoclar Vivadent |
| Panavia 21 | Alloy Primer | Kuraray America |
| Self Adhesive Systems | ||
| Maxcem | Kerr Dental | |
| Rely X Unicem | 3M-ESPE | |
Changing the method of attachment of the resin to the metal framework does not change the design of the framework itself, because the limiting factor in the system is still the bond of resin to enamel. The evolution of Japanese designs for resin-bonded FDPs61 has paralleled that in North America and Europe. There is an almost universal agreement concerning the need for mechanical retention of the framework to limit the stress on the bond interfaces (resin-to-metal and resin-to-enamel) and in the composite resin, which can become fatigued with time.62–64
DESIGN CONCEPTS
The guidelines for optimum design of resin-retained FDPs have been empirically derived. The principle underlying these restorations has always been that it is necessary to cover as much enamel surface as possible, as long as occlusion, esthetics, or periodontal health is not compromised. To emphasize the significance of maximum enamel coverage, Crispin and associates65 reported 3-year failure rates of up to 50% when using small bonded areas and minimal retention designs.
The initial designs of etched cast retainers included an “interproximal wraparound” concept developed to resist occlusal forces and provide a broader area for bonding. Enamel preparations consisted of creating occlusal clearance, placement of occlusal/cingulum rests, and lowering the lingual and proximal height of contour, thus creating proximal extensions.
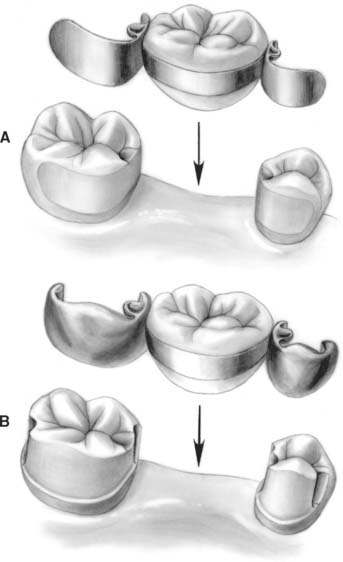
Frameworks should seat in an occlusogingival direction and should have no facial-lingual displacement (Fig. 26-7A). The contemporary design has improved retention with well-placed and precise grooves on abutment teeth (Fig. 26-7B). It is detailed in the following sections. Contemporary mouth preparations, in an effort to minimize failures, do not preserve as much tooth structure as did their predecessors; nevertheless, they are still limited to enamel and conform to conservative design principles. The new designs have been tested in laboratory studies.66,67
ADVANTAGES
When used appropriately, resin-bonded FDPs offer several advantages over conventional fixed prosthodontics (Box 26-1). Because of the unique preparation design, minimal tooth structure needs to be removed. In general, the preparation is confined to enamel only. Because of the conservative nature of the preparation, the potential for pulpal trauma is minimized. Anesthesia is not routinely used during tooth preparation (without anesthesia, it is possible to monitor the proximity of the preparation to the dentoenamel junction by the patient’s comfort level). The prosthesis can often be kept entirely supragingival; as a result, periodontal irritation is kept to a minimum. In a periodontal evaluation of restorations that averaged 10 years in service, the periodontal response was not significantly different from that for unrestored contralateral teeth.68 Only when the retainer gingival margins were less than 0.5 mm from the gingival crest was there a correlation with a detrimental gingival response. Concurrently, impression making is simplified because of the supragingival margins. Because the abutment teeth are, in addition to being nonsensitive, maintained with normal proximal contacts, fabrication of traditional interim restorations (see Chapter 15) is usually not required other than in selected patients. However, judicious placement of composite resin is important for maintaining occlusal clearances after the final impression and until the final restoration is bonded.69 (see Fig. 26-14.) Chair time is significantly reduced in comparison with that for conventional fixed prosthodontics, and the cost incurred by the patient is lower. Both may be reduced by as much as 50%.70
Box 26-1 Advantages and Disadvantages of Resin-Bonded Fixed Dental Prostheses
ADVANTAGES
Minimal removal of tooth structure
Minimal potential for pulpal trauma
Anesthesia not usually required
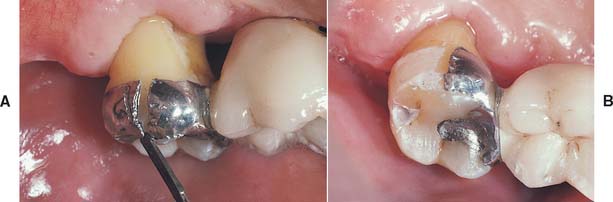
The restoration can be rebonded, in which particle abrasion and adhesive resin systems are used (as long as the debonding occurred with no sequelae involving the abutment teeth). If one retainer remains bonded, it can be gently loosened with a monobevel, single-ended instrument and a soft mallet. Any deformation of the metal framework in relation to the tooth structure can cause a crack to propagate through the luting composite resin. The monobevel chisel is positioned at an incisal or occlusal edge at an oblique angle to the long axis of the tooth along a mesial or distal line angle. The mallet should be used lightly (limited by patient response); repeated tapping causes debonding. However, with mechanically retentive designs, which include grooves and slots, the framework may require sectioning and removal of the sections (Fig. 26-8). As an alternative, ultrasonic scalers have been proposed to help remove partially debonded prostheses.69 Ultrasonic scalers with special tips are available for this specific purpose. They are applied at the incisal and gingival margins, but the procedure requires a high-power setting and can take considerable time. The debonding rate with rebonded restorations is high,71 and design modifications and a new or alternative restoration should be considered.
DISADVANTAGES
The primary disadvantage associated with resin-bonded FDPs relates to the fact that their longevity is less than that of conventional prostheses. This has been the subject of considerable investigation. Studies of first-generation etched metal FDPs at the University of Iowa (more anterior prostheses than posterior) and the University of Maryland (more posterior prostheses than anterior), with an average service time of more than 10 years, have revealed relative success. According to the results, an estimated 50% will fail after 250 months and 190 months, respectively (see Table 26-1).14,72,73 These studies also indicate that the rate of debondings does not increase with time.
In a study conducted in a private practice setting, contemporary designs with a mean service time of 6 years achieved a 93% success rate.3 This differs with the findings in a multicenter study in Europe, in which debonding rates increased with the time after placement (almost 50% at 5 years) and were related to preparation design, luting agent selection, and the area of placement within the dental arch.74 Another European study revealed retention rates of 60% at 10 years for early designs. In one study, posterior and mandibular resin-bonded FDPs demonstrated higher dislodgment rates,75 which may have resulted from occlusal forces (see Chapter 4) and increased isolation difficulty during the bonding procedure.71,76 In light of these studies, the likelihood of eventual debonding should be discussed with the patient before treatment. In comparison, a meta-analysis of clinical studies of conventional FDPs indicated a doubling of the failure rate for every 5 years of service from 0 to 15 years.77 When these results are projected from 15 years to 20 years, a 50% failure rate for conventional FDPs would take about 20 years.73
Extensive enamel modifications are required with retentive design to the proximal and lingual surfaces of the abutment teeth (see Fig. 26-7B). If the restoration is removed, composite resin bonding could restore the enamel contours, but transition to a more traditional prosthesis is likely. Enamel is limited in thickness, which requires precision in design and preparation with attention to detail.78 Enamel lingual surfaces of anterior teeth are almost always thinner than 0.9 mm.79
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses