Pulp Therapy for the Primary Dentition
Premature loss of primary teeth can lead to malocclusion and esthetic, phonetic, and functional problems; these in turn may be transient or permanent. Maintaining the integrity and health of the oral tissues is the primary objective of pulp treatment. It is important to attempt to preserve pulp vitality whenever possible; however, when this is not feasible, the pulp can be entirely eliminated without significantly compromising the function of the tooth.1,2
Histology
The pulp of a primary tooth is histologically similar to that of a permanent tooth. The odontoblasts are cells that line the periphery of the pulp space and extend their cytoplasmic processes into the dentinal tubules. These cells have several junctions, which allow intercellular communication and help to maintain the relative position of one cell to another. Below the odontoblastic layer is the cell-free zone that contains an extensive plexus of unmyelinated nerves and blood capillaries. The large blood vessels and nerves are located in the core of the pulp, and are surrounded by loose connective tissue.3 Although this description is correct during active dentinogenesis, it is now accepted that the size of the odontoblasts and the content of their cytoplasmic organelles vary throughout their life cycle and are closely related to their functional activity. The relationship between the size of the odontoblasts and their secretory activity can be demonstrated by differences in their size in the crown and in the root, and may express different dentinogenic rates in these two areas of the tooth.4
The Pulp-Dentin Complex
Dentinogenesis in Healthy State
The inner enamel epithelium and its associated basement membrane have an important role in direct odontoblastic cytodifferentiation. They present bioactive molecules, including growth factors immobilized on the basement membrane that send signals to the cells of the dental papilla, inducing the differentiation of the ectomesenchymal cells into odontoblasts.3 These cells express specific gene products that will form the highly mineralized extracellular matrix of dentin. Hydroxyapatite forms the main inorganic part of dentin, whereas the organic components consist mostly of type I collagen.5 During the postmitotic state the odontoblasts line the formative surface of the matrix and start secreting primary dentin. At the initiation of dentinogenesis, during mantle dentin formation, mineralization is achieved through the mediation of matrix vesicles. When mantle dentin formation is completed and the odontoblasts form a tightly packed layer of cells, the matrix of dentin is produced exclusively by the odontoblasts. Although the other cells of the pulp (in the subodontoblastic layer and in the pulp core) support dentinogenesis, they do not have a direct role in primary dentin secretion.6,7 As the matrix is secreted, the odontoblasts move pulpally, leaving a single cytoplasmic process embedded in a dentinal tubule in the matrix. These tubules, which increase in density near the pulp, confer the property of permeability on the dentin, a feature that has significant clinical importance.4
After secretion of the bulk of dentin during primary dentinogenesis, physiologic secondary dentin is secreted at a much slower rate throughout the life of the tooth, leading to a slow reduction in the size of the pulp chamber.8 The original postmitotic odontoblasts, responsible for primary dentinogenesis, survive for the life of the tooth, unless subjected to injury. These cells remain in a resting stage after primary dentinogenesis, and the physiologic secondary dentin formation represents a basal level of cell activity in the resting tooth stage.4
Dentinogenic Response to Injury
In pathologic conditions, such as in mild carious lesions or traumatic injuries, the secretory activity of the odontoblasts is stimulated to elaborate tertiary dentin. This will lead to focal secretion of new matrix at the pulp-dentin interface and possibly within the tubules, contributing to the histologic appearance of dentinal sclerosis at the injury site and to a decrease in dentin permeability.3,9 Thus the formation of tertiary dentin is much faster than the physiologic secondary dentin formation, so this tertiary deposition is regarded as an important defense mechanism of the pulp-dentin complex in response to either pathologic or physiologic insults (attrition).
The nature and quality of the tertiary dentin depends on its tubular structure and influences the dentin permeability of the area. Thus, in case of a mild injury, the odontoblasts responsible for the primary odontogenesis can frequently survive the challenge and are stimulated to secrete reactionary dentin beneath the injury site.10 Because the original odontoblasts are responsible for this matrix secretion, there will be tubular continuity and communication with the primary dentin matrix (Figure 22-1, A).9 Reactionary dentin might be considered as an extension of physiologic dentinogenesis. However, since it is a pathologic response to injury, it should be regarded as distinct from the primary and secondary dentinogenesis. When the injury is severe, the odontoblasts beneath the injury may die; however, if suitable conditions exist in the pulp, a new generation of odontoblast-like cells may differentiate from underlying pulp cells, secreting a reparative dentin matrix. Since this dentin is formed by a new generation of cells, there will be discontinuity in the tubular structure, with a subsequent reduction in permeability (Figure 22-1, B).11
A critical question concerns the factors responsible for triggering the stimulation of odontoblastic activity. Although there is still much to learn regarding the molecular control of cell activity in general and of odontoblastic activity in particular, one family of growth factors, the transforming growth factors (e.g., TGF-β) superfamily, has been reported to have extensive effects on the mesenchymal cells of many connective tissues.12
During tooth development, the odontoblasts secrete TGF-β, and some remains sequestrated in the dentin matrix. The sequestrated TGF-β may be released during any process leading to tissue dissolution, like formation of dental caries or the use of acid etching, for example. Thus dentin matrix should be considered not as an inert dental hard tissue but rather as a potential tissue store of a cocktail of bioactive molecules (particularly growth factors) waiting to be released, if appropriate tissue conditions prevail.3
In contrast to reactionary responses, reparative dentinogenesis represents a more complex sequence of biological processes. Migration and differentiation of pulpal progenitor cells must take place, creating a new generation of odontoblast-like cells, before matrix secretion. A series of stereotypic wound healing reactions occurs in the pulpal connective tissue, including vascular and cellular inflammatory reactions. In vitro and in vivo experiments on reparative odontogenesis demonstrate that the noninflamed pulp constitutes an appropriate environment where competent pulp cells (potential preodontoblasts) can differentiate into new odontoblast-like cells, forming reparative dentin.13,14
Factors Affecting Dentin-Pulp Complex Response to Stimuli in Primary Teeth
Although the life span of the primary teeth is shorter and the dentin is thinner when compared to permanent teeth, the dentin-pulp complex response to dental caries in human primary teeth is similar to that of permanent teeth, including a reduction in the number of the odontoblasts and an increase in the number of inflammatory cells. These are found under the very deep lesions and are less numerous at more distant regions, being almost absent in the radicular apical pulp.15
The primary dentition is frequently subjected to stimuli such as trauma or caries with associated pulp inflammation.16 The same factors affect the dentin pulp responses in both primary and permanent teeth to external stimuli.
The Deleterious Effects of Bacterial Infiltration at the Restorative Materials Margins
The involvement of bacteria in the inflammatory reaction was demonstrated by spontaneous healing of pulp exposures in germ-free animals17 and cavity surfaces that were sealed with zinc oxide–eugenol cement to prevent any bacterial contamination.18 The presence of bacteria in cavities with a remaining dentin thickness (RDT) less than 0.25 mm stimulates more severe pulp inflammatory reaction than in similar cavity preparations in the absence of bacteria.19 Thus the presence of bacteria always increases the mean grade of pulpal inflammation regardless of the RDT.20 These authors also observed that the presence of bacteria in class V cavities resulted in a significant decrease in the number of odontoblasts per unit area; this effect was more pronounced in deep cavities with RDT less than 0.5 mm than cavities with RDT greater than 0.5 mm. One can conclude that the ability to maintain an effective seal to protect the pulp from recurrent injury resulting from bacterial microleakage is a decisive factor in the clinical success of restorative products.21
However, some studies have shown pulpal inflammation in the absence of bacteria,22 clearly indicating that other factors are also responsible, even to a lesser extent, for pulp injury after restorative treatment.
The Protective Role of the Remaining Dentin Thickness
In vivo, the cavity RDT was found to be an important factor mediating pulpal inflammatory activity, particularly when the RDT was reduced below 0.25 mm.19 In class V cavities prepared in human teeth, the protective tertiary dentin area increased with decreasing RDT until 0.25 mm.19 With an RDT below 0.25 mm, a significant decrease in the number of odontoblasts was observed together with minimal reactionary dentin repair.23 The RDT significantly modifies the pulpal response: the thicker the RDT, the lower the pulpal reaction.17 The presence of an RDT over 500 µm delays the diffusion of noxious materials to the dental pulp and allows the odontoblasts to secrete a reactionary dentin, increasing the total distance between the restorative material and the pulp. Any additional decrease in the dentin thickness below 500 µm results in a significant reduction in the number of odontoblasts. The differentiation of odontoblast-like cells from progenitor pulp cells, which migrate to the injury site and secrete reparative dentin, may compensate for this reduction. This reparative dentin decreases the dentin permeability and increases the distance between the restorative material and the pulp, protecting it from noxious products. Thus the RDT appears to provide an important protective barrier against bacterial infiltration, toxins, or any noxious material applied onto the dentin.
Based on the remaining dentin thickness, three situations can be considered:
1. Initial carious lesions are present or cavity preparation is shallow (RDT greater than 500 µm): A localized reactionary dentin is secreted facing the restoration site and intratubular mineralization occurs, resulting in pulp protection by significantly decreasing dentin permeability. It has been suggested that this stimulation may be due to signaling molecules (e.g., transforming growth factors of the β family [TGF-β1], bone morphogenetic protein-2 [BMP-2]) liberated from the dentin during demineralization (see Figure 22-1, A).24,25
2. Carious lesion progression implies a deep cavity preparation (RDT less than 500 µm): These lesions may lead to partial odontoblast disintegration. Depending on the pulpal inflammatory state, progenitor/stem cells can migrate to the injury site and differentiate to yield a new generation of odontoblast-like cells. These cells are responsible for the deposition of a specific type of tertiary dentin termed reparative dentin (see Figure 22-1, B).26,27
3. During a subsequent restorative process, deep cavity preparations with RDTs between 250 and 40 µm lead to poor tertiary dentin repair activity.19 This results from impaired odontoblast dentin secretory activity due to cellular injury.28 Murray demonstrated that the mean number of intact odontoblasts found beneath this kind of cavity preparation was 36% lower than the number found beneath similar preparations with an RDT between 500 and 250 µm. This inability of odontoblasts to provide adequate pulpal repair and pulpal protection after deep cavity cutting has been supported by observations of a persistent inflammatory pulpal response and odontoblast displacement following cavity cutting.28
Clinical Pulpal Diagnosis
History and Characteristics of Pain
The history and characteristics of pain are often important in determining whether the pulp is in a treatable condition. However, children may have extensive carious lesions, often with draining paruli, with no apparent history of pain. Or if dental problems developed early (such as with nursing bottle decay), the child may have no experience of the teeth feeling any other way.2 Being aware of these limitations, the dentist should distinguish between two main types of dental pain: provoked and spontaneous. Provoked pain is stimulated by thermal, chemical, or mechanical irritants, and is reduced or eliminated when the noxious stimulus is removed. This sign frequently indicates dentin sensitivity due to a deep carious lesion or a faulty restoration. The pulp is in a transitional state in most cases and the condition is usually reversible.
Spontaneous pain is a throbbing, constant pain that may keep the patient awake at night. This type of pain usually indicates advanced pulpal damage, and the pulp is usually nontreatable. However, a final diagnosis can only be made based on clinical tests and radiographic assessment. Spontaneous, throbbing pain simulating an irreversible pulp condition can be observed when the dental papilla is inflamed from food impaction. This condition may cause bone destruction, and the pulp in these teeth may be treatable. The symptoms disappear with proper restoration of the tooth and reestablishment of an adequate contact point (Figure 22-2).
Clinical Examination
A careful extraoral and intraoral examination can be of extreme importance in detecting the presence of a pulpally involved tooth. Several signs, such as redness and swelling of the vestibulum or grossly decayed teeth with draining paruli, are definitely indicative of pulpal pathoses. In addition, attention should be paid to missing or fractured restorations or to those with carious marginal breakdown, because these may also be indicators of pulp involvement. Palpation, assessment of tooth mobility, and sensitivity to percussion are helpful diagnostic tools. Fluctuation, felt by palpating a swollen mucobuccal fold, may be the expression of an acute dentoalveolar abscess before exteriorization. Bone destruction following a chronic dentoalveolar abscess can be also detected by palpation. Comparing the mobility of a suspicious tooth with its contralateral tooth is of particular importance. If a significant difference is observed, pulpal inflammation might be suspected. Care should be taken not to misinterpret as pathologic the mobility present during the normal time of exfoliation. Sensitivity to percussion may reveal a painful tooth in which inflammation has progressed to involve the periodontal ligament (acute apical periodontitis). However, care should be taken in interpreting these tests (see Figure 22-2, A). Percussion should be done very gently with the tip of a finger and not with the end of a dental mirror to prevent exposing the child to unnecessary uncomfortable stimuli.2
Radiographic Examination
1. Deep caries with possible or definite pulp involvement.
2. Deep restorations close to a pulp horn.
3. Successful or failing pulpotomy or pulpectomy.
4. Pulpal changes, such as pulp calcifications (denticles) and pulp obliteration.
5. Pathologic root resorption, which may be internal (within the root canal) or external (affecting the root or the surrounding bone). Internal resorption indicates inflammation of a vital pulp, whereas external resorption demonstrates a nonvital pulp with extensive inflammation, including resorption of the adjacent bone.
6. Periapical and interradicular radiolucencies of bone. In primary teeth, any radiolucency associated with a nonvital tooth is usually located in the furcation area, not at the apices. This is because of the presence of accessory canals on the pulpal floor area. Thus a bitewing film is frequently a useful diagnostic aid, particularly in maxillary molars where the developing premolar obscures the furca in a periapical radiograph.
7. The dentist should be familiar with the normal factors that complicate the interpretation of radiographs in children, such as larger bone marrow spaces, superimposition of developing tooth buds, and normal resorption patterns of the teeth.2
Operative Diagnosis (Direct Pulp Inspection)
There are instances when a final diagnosis can only be reached by direct evaluation of the pulp tissue and a decision about treatment is made accordingly. The quality (color) and the amount of bleeding from a direct exposure of the pulp tissue must be assessed: profuse bleeding or purulent exudate indicates irreversible pulpitis. Based on these observations, the treatment plan may be confirmed or changed. For example, if a formocresol pulpotomy is planned, the nature of the bleeding from the amputation site should be normal (red color and hemostasis evident in less than 5 minutes with mild cotton pellet pressure). If bleeding persists, a more radical treatment should be undertaken (pulpectomy or extraction). Excessive bleeding is an indication that the inflammation has reached the radicular pulp. Conversely, if a pulp polyp is present and bleeding stops normally after coronal pulp amputation, a pulpotomy may be performed instead of a more radical procedure.2
Pulp Treatment Procedures
Conservative Treatment
Protective Base
Guidelines published by the American Academy of Pediatric Dentistry recommend placement of a protective base or liner on the pulpal and axial walls of a cavity preparation to act as a protective barrier between the restorative material and the tooth.29 Dentin is permeable and allows the movement of materials from the oral cavity to the pulp and vice versa. It was believed for several years that pulp inflammation was caused by the toxic effects from dental materials.30 Today, however, there is sufficient evidence to show that pulpal inflammation resulting from dental materials is mild and transitory, with adverse reactions occurring as the result of pulpal invasion by bacteria or their toxins.18,19,31 Continued marginal leakage with secondary recurrent caries is probably the most common cause of pulp degeneration under restorations. In deep cavities the dentin covering the pulp is thin, and the tubules are large in diameter and packed close together. This dentin is extremely permeable and should be covered with a material that seals dentin well, usually glass ionomer cement.32
The materials most recently used as cavity sealers are those that have demonstrated multisubstrate bonding ability to bond the restorative material to the tooth. These include resin cements, glass ionomers, and dentin-bonding agents. The benefits of using these materials to bond composite to tooth structure is a well-documented and accepted procedure.32 However, employing them with amalgam is more controversial. Mahler and colleagues33 observed no difference between amalgam restorations placed with and without bonding after 2 years and concluded that the use of bonding agents under traditional amalgam fillings should not be recommended. Thus, protective liners or bases should only be placed in deep cavities approaching the pulp.
Indirect Pulp Treatment
The dilemma that clinicians face lies in the assessment of how much caries to leave at the pulpal or axial floor. It is generally accepted that the carious tissue that should remain at the end of the cavity preparation is the amount that, if removed, would result in overt exposure. It is difficult to determine whether an area is an infected carious lesion or a bacteria-free demineralized zone. The best clinical marker is the quality of the dentin: soft, mushy dentin should be removed, and hard discolored dentin can be indirectly capped. The ultimate objective of this treatment is to maintain pulp vitality34 by (1) arresting the carious process, (2) promoting dentin sclerosis (reducing permeability), (3) stimulating the formation of tertiary dentin, and (4) remineralizing the carious dentin.
Clinical experience and a good understanding of the process of caries progression can allow for better control of the “partial removal caries step.” A large round bur (no. 6 or 8) can provide better results than spoon excavators.35 A chemical-mechanical approach to caries excavation known as Carisolv (Medi Team Dental, Savedalen, Sweden) has been developed. A gel made of three amino acids and a low concentration of sodium hypochlorite is rubbed into the carious dentin with specially designed hand instruments. With Carisolv, sound and carious dentins are clinically separated, and only carious dentin is removed, resulting in a more conservative preparation. When a bur is used, healthy tissue is frequently removed. The main drawback to this technique is the time needed to complete the procedure, because it is a much slower process than removing caries with a bur.36
It is current knowledge that, in the appropriate metabolic state of the dentin-pulp complex, a new generation of odontoblast-like cells might differentiate and form tubular tertiary dentin (reparative dentinogenesis).8,37 It must be emphasized that under clinical conditions, the matrix formed at the pulp-dentin interface often comprises reactionary dentin, reparative dentin, or fibrodentin formation. It is impossible to distinguish these processes in vivo, and the process might also be indistinguishable at both biochemical and molecular levels.
Presently, the materials most commonly used in IPT are calcium hydroxide, mineral trioxide aggregate (MTA), and glass ionomer. Many historical studies have examined the interaction between tooth tissues and calcium hydroxide, and more recently with MTA. The main soluble component from MTA has been shown to be calcium hydroxide. The clinical response of the tooth to both materials is based on comparable mechanisms involving the dissolution of calcium hydroxide and release of calcium and hydroxyl ions, raising the pH of the environment well above 7.0. Because dentin contains a large store of potentially bioactive molecules, it has been considered that the interaction of a high pH material, such as calcium hydroxide or MTA, may cause the release of certain of these molecules. This action is similar to that occurring during the demineralization of dentin during a caries attack, where the pH of the local environment is low.38
Studies of direct pulp capping with glass ionomer show that both patient tolerance and clinical success rates are lower with ionomer than calcium hydroxide. This finding suggests that the acidic environment created by the glass ionomer is more damaging to the pulp than the basic environment of calcium hydroxide or MTA.19,32
Interim restorative treatment, also known as stepwise caries removal, is an approach for managing caries of dentin with hand instruments without local anesthesia; it is considered a form of IPT. Massara and colleagues39 demonstrated that use of glass ionomer creates conditions that lead to remineralization and suggest that it can be recommended as a good base for indirect pulp capping.
The rationale for IPT is that few viable bacteria remain in the deeper dentin layers and that after the cavity has been sealed properly they will be inactivated. These facts argue against a two-step procedure, in which the tooth is reentered to excavate the previously carious dentin and to confirm the formation of reactionary dentin. The procedure risks creation of a pulp exposure and further insult to the pulp.40
Dentin-bonding agents have been recommended for use in direct pulp capping41 and IPT.42 However, there are some concerns regarding an IPT with these materials.38 Nakajima and coworkers43 found a significant loss of bond strength to human carious dentin when compared with sound dentin. This finding leads one to further question the integrity of the bond and subsequent ability to prevent bacterial invasion of a carious substrate.
Contrary to previous belief, IPT can also be an acceptable procedure for primary teeth with reversible pulp inflammation, provided that the diagnosis is based on a good history and proper clinical and radiographic examination, and the tooth has been sealed with a leakage-free restoration.44
The value of taking a good history complemented by a careful clinical and radiographic examination cannot be overestimated when trying to reach an accurate diagnosis. However, sometimes this cannot be achieved and the prognosis of the tooth will be affected. Figure 22-3 shows the treatment outcome of two mandibular first primary molars in the same patient. The tooth treated with an MTA pulpotomy presents internal root resorption, whereas its antimere, restored conservatively with a composite over an IPT, looks normal. These findings were probably attributable to the preoperative status of the pulp. The radicular pulp of the pulpotomized tooth was probably chronica/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


 Outline
Outline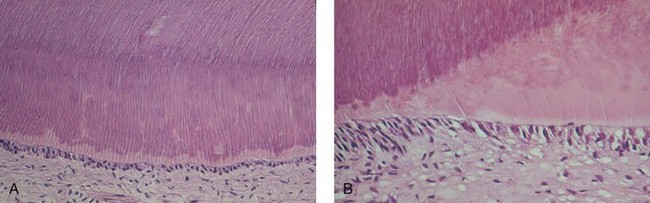
 FIGURE 22-1
FIGURE 22-1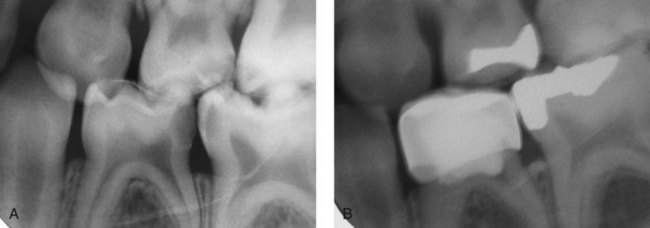
 FIGURE 22-2
FIGURE 22-2