Chapter 21
Organ and Bone Marrow Transplantation
On average, over the past 10 years there have been more than 26,000 solid organ transplant procedures in the United States annually, with nearly 95,000 persons on the Organ Procurement and Sharing Network (OPTN) waiting list for transplant organs.< ?xml:namespace prefix = "mbp" />
Key outcomes after transplantation include (1) survival of transplant recipients and (2) the function of transplanted grafts. Reported 1-year patient survival rates are highest for kidney and pancreas recipients, ranging from about 95% to 98%; the median survival rate with renal transplantation from living donors has increased from 17 years in 1988 to nearly 38 years at 2010. Corresponding survival rates at 1 year for liver, intestine, and heart transplant recipients were approximately 86% to 88%, about 83% for lung, and lowest for the small number of heart-lung recipients at around 58%.
TABLE 21-1 Patient Survival Rates After Organ Transplant Procedures
| Organ With No. of Transplants | Patient Survival Rate | |
|---|---|---|
| Deceased Donor | Living Donor | |
| Kidney: 16,067 |
||
| 1-year survival | 95.0% | 98.2% |
| 5-year survival | 81.0% | 90.6% |
| Pancreas alone: 122 |
||
| 1-year survival | 97.9% | |
| 5-year survival | 88.7% | |
| Pancreas after kidney: 214 |
||
| 1-year survival | 97.3% | |
| 5-year survival | 93.9% | |
| Kidney-pancreas: 825 |
||
| 1-year survival | 95.1% | |
| 5-year survival | 86.6% | |
| Liver: 5817 |
||
| 1-year survival | 87.1% | 87.9% |
| 5-year survival | 73.3% | 77.3% |
| Intestine: 69 |
||
| 1-year survival | 81.4% | |
| 5-year survival | 56.2% | |
| Heart: 2085 |
||
| 1-year survival | 87.6% | |
| 5-year survival | 73.9% | |
| Lung: 1473 |
||
| 1-year survival | 83.6% | |
| 5-year survival | 53.4% | |
| Heart-lung: 26 |
||
| 1-year survival | 73.8% | |
| 5-year survival | 46.5% | |
* Total of 27,281 solid organ transplants in 2008.
† Numbers based on 2008 statistics.
Data from OPTN/STTR Annual Report, 2008.
Because more organ and bone marrow transplant procedures are being performed successfully and technologic proficiency will continue to increase, so that more posttransplantation patients are surviving longer, more of these patients will be seeking dental care. Although there are no precise guidelines drawn from evidence-based clinical trials for the dental management of organ transplant patients, a number of principles have been identified that apply in the care of the dental patient before, during, and after the solid organ transplant process. This chapter reviews the epidemiology, pathophysiology, process, complications, and dental management recommendations for these patients.
Definition
Organ transplant procedures are common today and may be performed in several organ systems, depending on the mix of recipients and donors. Heart, liver, kidney, pancreas, heart-lung, bone marrow, and other transplants may be available for the appropriate recipient. The ideal combination involves the transplantation of an organ from an identical twin to the other twin (syngeneic). The next best match for organ survival is transplantation of an organ from one living relative to another (allogeneic), followed by transplantation of an organ between living nonrelated people (xenograft). Each of these combinations, however, is limited by the fact that unless at least two organs are present, the donor cannot survive. Thus, these types of matches are basically limited to kidney and bone marrow donors. Nevertheless, recent studies have shown success with transplantation of a portion of a liver or pancreas from living donors. The largest organ pool for transplantation is cadaver organs, but the match also is poorest.
Incidence and Prevalence
Dr. Joseph E. Murray, a Nobel laureate, performed the first successful human organ transplant procedure in Boston in 1954, using a kidney donated by the patient’s identical twin brother. Then Murray performed the first successful kidney allograft transplant procedure in 1959, applying total body irradiation for immunosuppression, and the first successful human kidney cadaver transplant procedure in 1962, introducing Imuran (azathioprine), an immunosuppressive drug. Before these early transplant procedures, patients with renal failure, hypertension, and azotemia were undergoing renal dialysis, which was not nearly as successful in addressing the deficits in kidney function. Since those early days, more than one-half million kidney transplant procedures have been performed in the United States.
Since 1987, all organ transplant procedures performed in the United States have been reported to the OPTN. Since that time, renal transplants have averaged over 10,000 per year nationwide.
The first human heart transplantation was performed in 1967 and at that time the 1-year survival rate was only about 20%. At present, more than 70,000 heart transplantations have been performed at the rate of approximately 3400 per year at more than 250 U.S. hospitals. The 1-year survival rate is more than 87%, with the 5-year survival rate over 70% (see
The first orthotopic liver transplantation was performed in 1963. However, the first transplant resulting in an extended survival of 13 months was not achieved until 1967. By 1980, only 300 liver transplants had been performed, with a 1-year survival rate of only 28%. At present, more than 6000 liver transplant procedures are performed each year, with about the same number on the waiting list for a liver transplant. More than 100,000 liver transplantations have been performed in the United States. Survival rates for patients receiving transplants in 1988 were 75.5% at 1 year and 68.6% at 2 years after the operation. Currently, the survival rates have increased to approximately 80% for 1 year and 70% for 5 years
The first pancreas transplant procedure, which also included a duodenum and a kidney, was performed in 1966, by Kelly and Lillehei at the University of Minnesota, in a patient with diabetic nephropathy. Now more than 1000 pancreas transplantations are performed each year. The 5-year survival rate is nearly 90%. Recently, pancreatic islet cell transplant procedures also have shown considerable success.
In 1990 only five lung transplant procedures had been performed. Since that time, more than 10,000 lungs have been transplanted at 119 centers, most within the last 5 years. The first heart-lung combination transplant procedure was performed in 1981. Since that time, more than 1000 have been performed, with a 1-year survival rate of almost 60%.
To date, only a few small bowel transplant procedures have been performed (some combined with liver transplantation) at only a few transplant centers (in Cambridge, England, and London, Ontario, and in Pittsburgh and Omaha). However, much progress is being made in this area for the treatment of end-stage intestinal failure, with a current 1-year survival rate of approximately 70%.
The modern era of bone marrow transplantation (BMT) was ushered in by the seminal experiments of Jacobsen (1950) and Lorenz (1951) and their colleagues, who demonstrated that mice could be protected from the lethal irradiation that was used to treat severe aplastic anemia or leukemia by shielding the spleen or with intravenous infusion of bone marrow. By 1956, several laboratories had demonstrated that the protective effects against otherwise lethal doses of total body irradiation were caused by the colonization of the recipient bone marrow by the infused donor cells.
In 1957, E. Donnell Thomas described the clinical technique of large quantities of marrow infused intravenously into patients with leukemia with safety and efficacy and demonstrated a transient bone marrow transplant in humans. In 1958, BMT was performed in six victims of a radiation accident.
Most early BMTs were performed in only terminally ill patients, and the grafts could not be evaluated because the patient did not live long enough. The few successful allogeneic grafts were followed by lethal immunologic reaction of graft-versus-host disease (GVHD). Many years of research, first in rodents and then in dogs and monkeys, paved the way for the ultimate success of BMT in humans. Recent advances in knowledge of histocompatability typing and in the prevention of GVHD, as well as more supportive measures for the patient, have resulted in greater success and more frequent BMTs.
The first attempts at transplantation in the 1950s and 1960s all were followed by increased activity in transplantation that resulted in very poor survival rates. A period followed during which few transplants were attempted. Increased research activity in the 1960s led to development of techniques that dealt with the major limiting factor in organ transplantation rejection of the organ by the host immune system.
With the development of effective immunosuppressive agents, improved surgical techniques (including percutaneous biopsy of solid transplanted organs to monitor rejection), and the acceptance of the concept of “brain death” as a definition for determining the death of potential donors, major advances in organ transplantation have occurred.
Transplantation of the heart, liver, and kidney is no longer considered an experimental procedure and is available as a treatment option for selected patients with end-organ disease. Transplantation of the pancreas also is considered a major treatment option for uremic diabetic patients who are receiving a kidney transplant.
The International Bone Marrow Transplant Registry reported that more than 20,000 patients had received allogeneic bone marrow transplants between 1955 and 1987. More than 50% of these were performed during the 3 years between 1985 and 1987. By 1991, that figure had doubled, and through 1999 nearly 90,000 BMTs had been performed. BMT is the treatment of choice for patients with myelogenous leukemia and for those with other blood dyscrasias (see
For many patients in whom a remission from leukemia cannot be achieved or the disease has not responded to chemotherapy, stem cell transplantation is an option. Currently, more than 15% of patients with end-stage disease will survive after stem cell transplantation.
Etiology
The most common indications for heart transplantation are cardiomyopathy and severe coronary artery disease.
Pathophysiology and Complications
All candidates for heart, liver, and bone marrow transplantation have severe end-stage organ disease and would die without transplantation. Patients with end-stage renal disease can be kept alive by hemodialysis. However, the quality of life for such patients can be greatly improved by renal transplantation. Patients with severe diabetes also can be kept alive with daily insulin injections, but their lives also may be greatly improved by pancreas transplantation.
Clinical Presentation
Signs and Symptoms
Signs and symptoms for the diseases necessitating transplantation are discussed in the chapters indicated:
• Advanced cardiac disease (
• Advanced liver disease (
• End-stage renal disease (
• Advanced diabetes mellitus (
• Red blood cell disorders and white blood cell disorders (
Laboratory Findings
Laboratory findings of particular importance in the care of dental patients who are scheduled for a transplant procedure include bleeding time, platelet count, white blood cell (WBC) count with differential, prothrombin time, hematocrit, partial thromboplastin time, blood urea nitrogen, aspartate aminotransaminase, serum creatinine, specific gravity of urine, serum bilirubin, alkaline phosphatase, and urinary levels of albumin and other proteins. Elevation of aspartate aminotransaminase, alkaline phosphatase, prothrombin time, and serum bilirubin would suggest advanced liver disease. Increased bleeding time, low platelet count, decreased WBC count, and decreased hematocrit are associated with many of the blood dyscrasias. Elevation of serum creatinine and blood urea nitrogen and increased specific gravity of urine and proteinuria are associated with advanced renal disease. In addition, a low hematocrit, prolonged partial thromboplastin time, and decreased WBC count can be found in patients with advanced renal disease. These patients may be potential bleeders, are prone to infection, and will experience buildup of toxic levels of drugs that are metabolized by the liver or kidney, depending on the organ involved (
Medical and Surgical Management
Immunosuppression
The immunosuppressive agents now used for most heart, liver, kidney, and pancreas transplantations are cyclosporine, azathioprine, prednisone, and an antilymphocyte agent (
TABLE 21-3 Current Immunosuppressive Agents for Use in Early Posttransplantation Period
| Agent | Class | Mechanism of Action |
|---|---|---|
| Prednisone | Corticosteroid | Blocks cytokine gene transcription |
| Cyclosporine | Calcineurin inhibitor | Inhibits IL-2 gene transcription |
| Reduces activation of T cells | ||
| Tacrolimus | Calcineurin inhibitor | Inhibits IL-2 gene transcription |
| Reduces activation of T cells | ||
| Azathioprine | Nucleoside inhibitor | Impairs DNA synthesis |
| Inhibits B cell proliferation | ||
| Mycophenolate mofetil | Nucleoside inhibitor | Impairs DNA synthesis |
| Inhibits B cell proliferation | ||
| Rapamycin (sirolimus) | TOR inhibitor | Inhibits tyrosine kinase |
| Everolimus (RAD) | TOR inhibitor | Inhibits tyrosine kinase |
IL, Interleukin; TOR, Transplanted organ rejection.
Data from Little, JW, Rhodus, NL: Dental treatment of the liver transplant patient. Oral Surg Oral Med Oral Pathol, 73(4):419-426, 1992.
Anti-interleukin-2 receptor antibodies (IL-2Rα), daclizumab (humanized anti-IL-2Rα) and basiliximab (chimeric anti-IL-2Rα) have proved quite successful in furthering the immunosupression. Another important new drug is sirolimus, which is a macrocyclic lactone and blocks IL-2 receptors. Organ rejection and the need for antilymphocyte globulin have been reduced by sirolimus.
Total body irradiation (1000 cGy) has been the most effective means of conditioning the bone marrow graft recipient. Cyclophosphamide usually is given in the immunosuppressive phase before (4 to 5 days) transplantation. Busulfan also has been used for conditioning the graft recipient. Cyclosporine, prednisone, and methotrexate are used after marrow transplantation to prevent or ameliorate GVHD.
Surgical Procedure
Heart Transplantation
Heart transplantation involves the surgical removal of the heart from the donor by one surgical team and the removal of the recipient’s diseased heart and the attachment of the donor’s heart to the major vessels of the recipient’s heart by a second surgical team (
Heart-Lung Transplantation
In addition to the surgical protocol for cardiac transplantation, the lungs are generally combined in patients with primary pulmonary hypertension, pulmonary fibrosis, cystic fibrosis, certain congenital heart conditions, and primary cardiac disease accompanied by secondary pulmonary hypertension. Heart-lung transplantation also involves the surgical removal of the heart and lungs from the donor by one surgical team and the removal of the recipient’s diseased heart and lungs and the attachment of the donor’s heart and lungs with anastomotic sites at the trachea, the right atrium, and the aorta by the second surgical team (
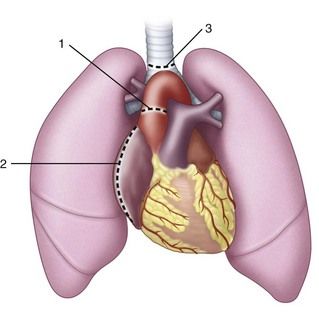
FIGURE 21-2 Anastomotic sites in a completed combination heart-lung transplant: 1, aorta; 2, right atrium; 3, trachea.
An additional complication, however, that may be seen with heart-lung combination transplantation is the implantation response. The incidence of this particular complication is highest between 4 and 21 days after transplantation. It is a reversible condition characterized by fever, tachypnea, diffuse pulmonary infiltrates on the chest radiograph, decreased partial pressure of oxygen in arterial blood (Pao2) and increased partial pressure of carbon dioxide in arterial blood (Paco2). The cause is multifactorial, involving lymphatic interruption, ischemia, denervation, and surgical trauma. The patient may need short-term mechanical ventilation. Acute rejection of the transplanted organs also is a major complication and constitutes the most common cause of death within the first year.
Liver Transplantation
Liver transplantation involves the excision of the diseased recipient’s liver with reconstruction of the vena cava, portal vein, and biliary tree (

FIGURE 21-3 Completed liver transplant procedure. Vascular anastomoses include the suprahepatic vena cava, infrahepatic vena cava, portal vein, and hepatic artery. A choledochocholedochostomy biliary reconstruction is depicted.
(Redrawn from Kaplowitz N, editor: Liver and biliary diseases, Baltimore, 1992, Williams & Wilkins.)
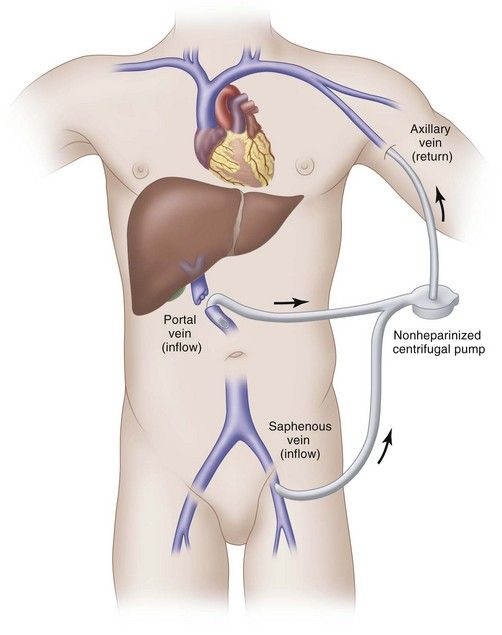
FIGURE 21-4 Venovenous bypass. The portal vein is divided, and devascularization of the liver is completed. Then the portal vein is cannulated and a second cannula is placed in the iliac vein via the saphenous vein. The blood is pumped by a centrifugal pump through a nonheparinized system and returned to the patient by way of a cannula placed in the axillary vein. Flows of 2 to 5 L/min are commonly attained. Flow must be maintained above 700 to 1000 mL/min to prevent thrombosis.
(Redrawn Kaplowitz N, editor: Liver and biliary diseases, Baltimore, 1992, Williams & Wilkins.)
Liver transplant procedures have been performed in patients with hemophilia, and their factor VIII deficiency subsequently was corrected. Obviously, then, factor VIII is produced by the liver and not endothelial cells.
Small Bowel Transplantation
Few small bowel transplantations (SBTs) have been performed. Because of its quasi-experimental nature, use of SBT currently is restricted to patients with end-stage intestinal failure in whom conventional treatments were of no benefit. Intestinal failure is characterized by the inability to maintain nutrition and intestinal fluid and electrolyte balance. Several causes of this condition are recognized, the most common being massive small bowel resection with consequent short gut syndrome, which results in rapid intestinal transit without proper absorption of nutrients.
The surgical technique for SBT is shown in
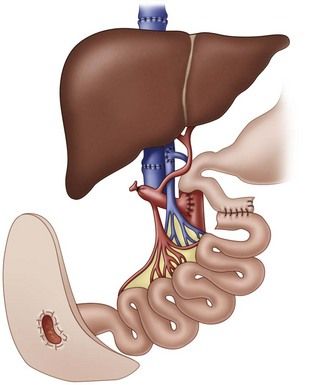
FIGURE 21-5 Small bowel transplantation. Anastomoses are at the superior end of the small intestine at the jejunum, and the terminus is exteriorized in a stoma. Here the liver also has been transplanted.
(Redrawn from Asfar S, Zhong R, Grant D: Small bowel transplantation, Surg Clin North Am 74:1197-1207, 1994.)
Kidney Transplantation
Patients who have a living related donor available for kidney transplantation usually are admitted to the hospital 2 days before transplantation. When a kidney recipient is to receive a cadaver kidney, the patient is admitted to the hospital on an urgent basis. Current preservation techniques allow kidney storage for up to 72 hours.
The indications for renal transplantation generally include chronic renal disease or end-stage renal disease (ESRD). ESRD is a progressive, bilateral deterioration of kidney nephrons that results in uremia and, ultimately, death. ESRD is associated with glomerulonephritis, nephrosclerosis, pyelonephritis, diabetic nephropathy, congenital renal disorders, drug-induced nephropathy, obstructive uropathy, and hypertension.
Accompanying hypertension, diabetes, congestive heart failure, infections, volume depletion, urinary tract obstruction, hypercalcemia, and hyperuricemia must be constantly managed. After conservative care (i.e., drug therapy, dietary restrictions, management of underlying disease), to control waste products, fluid balance, and electrolyte levels becomes inadequate, renal dialysis is the next step. As renal disease progresses, and more nephrons are destroyed, azotemia cannot be controlled, and the patient must have the blood artificially filtered. Therefore, the therapy of peritoneal dialysis or hemodialysis begins.
More than 80,000 patients are currently being maintained on hemodialysis in the United States. Hemodialysis treatment must be performed every 2 to 3 days, and each such treatment requires 4 to 5 hours. The cost in terms of time, money, patient inconvenience, and so on is enormous, and this management mode is extremely confining (see
The recipients of a kidney transplant require immunosuppressive preparation so that they will not reject the graft. As with all major transplant procedures, the major problem in renal transplantation is rejection of the graft. The type of preparation depends on the nature of the underlying renal disease. Intensive chemotherapy commonly is used. Chemotherapy with cytotoxic agents (azathioprine) or steroids (prednisone) and administration of antilymphocyte globulin (ALG) are effective. However, greater success has been attained with cyclosporine instead of prednisone or azathioprine.
With the immunosuppression, the patient is rendered susceptible to infection and poor wound healing. Sepsis is a major complication in renal transplant recipients. Adrenal function may be suppressed and, likewise, endogenous cortisol production (

Box 21-1 Signs of Overimmunosuppression in Posttransplant Patients
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses