Immunodeficiencies
Key Points
• Immunodeficiencies are mainly secondary to malnutrition, drugs or infections
• Immunodeficiencies predispose to infections and neoplasms
• The world epidemic of HIV continues, despite new therapies
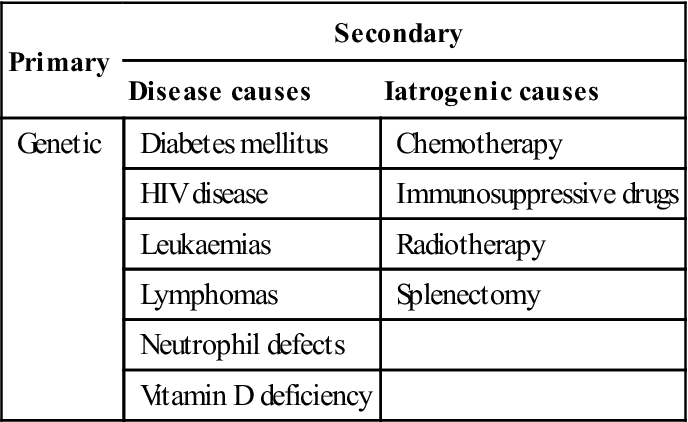
Immunodeficiencies are states that result from a defect in the immune response, and predispose to recurrent infections, especially of skin, mucosae and respiratory tract (Fig. 20.1). An immunodeficient patient is often somewhat ponderously referred to as an ‘immunocompromised host’. Human immunodeficiency virus (HIV) disease and the resultant acquired immune deficiency syndrome (AIDS) constitute a major immunodeficiency and public health issue worldwide but other important and common causes of secondary immune defects are immunosuppressive therapy and malnutrition (Table 20.1). Vitamin D deficiency is also now recognized as causing failure of immune activation. Primary (genetically determined) immune defects are mostly rare, apart from selective immunoglobulin A (IgA) deficiency.
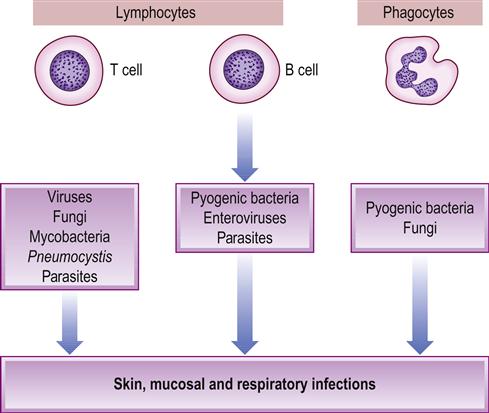
Table 20.1
< ?comst?>
| Primary | Secondary | |
| Disease causes | Iatrogenic causes | |
| Genetic | Diabetes mellitus | Chemotherapy |
| HIV disease | Immunosuppressive drugs | |
| Leukaemias | Radiotherapy | |
| Lymphomas | Splenectomy | |
| Neutrophil defects | ||
| Vitamin D deficiency | ||
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
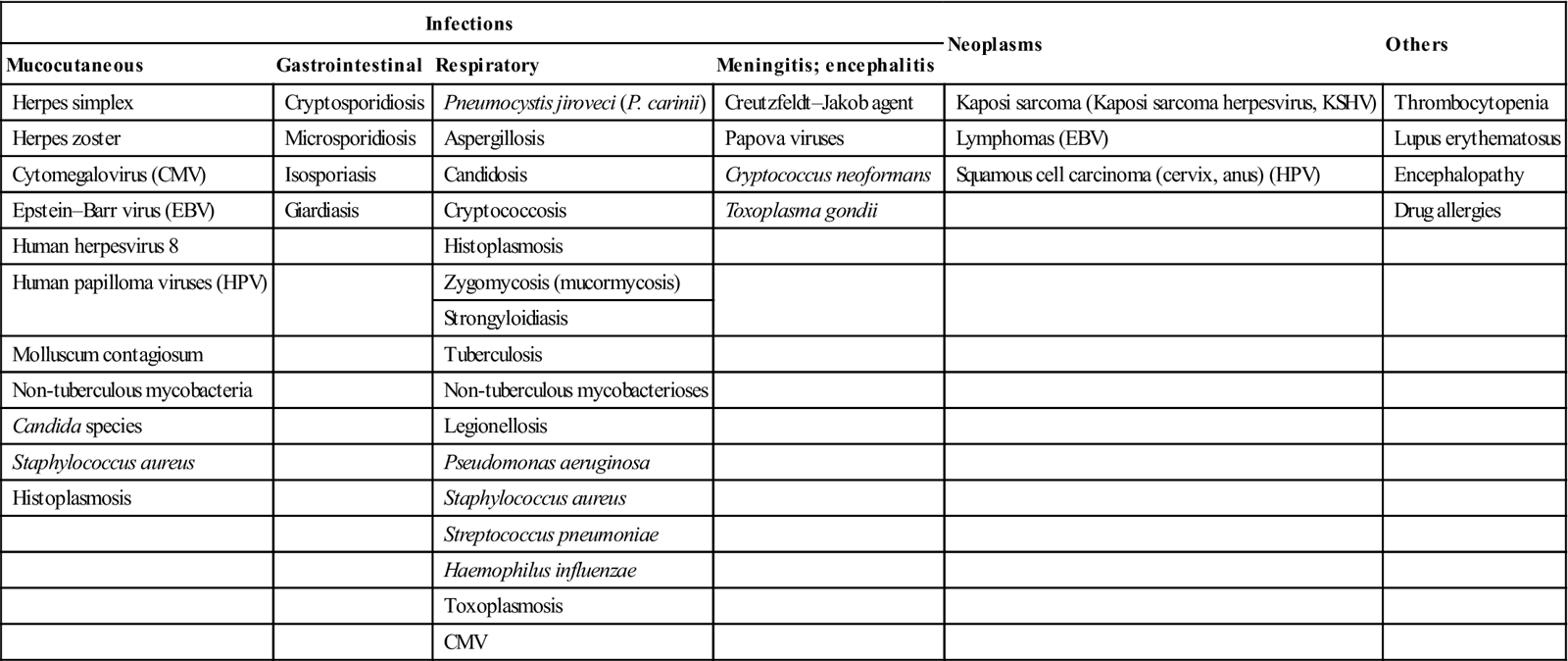
The most important consequence of all immunodeficiencies is an abnormal susceptibility to infections, frequently caused by organisms of such low pathogenicity as rarely to affect the normal individual (opportunistic infections). Candidosis, herpetic infections and Pneumocystis jiroveci (P. carinii) pneumonia (Table 20.2) are common. Immunocompromised people are also prone to various malignant neoplasms, some virally related.
Table 20.2
Complications in immunodeficient or immunosuppressed patients
< ?comst?>
| Infections | Neoplasms | Others | |||
| Mucocutaneous | Gastrointestinal | Respiratory | Meningitis; encephalitis | ||
| Herpes simplex | Cryptosporidiosis | Pneumocystis jiroveci (P. carinii) | Creutzfeldt–Jakob agent | Kaposi sarcoma (Kaposi sarcoma herpesvirus, KSHV) | Thrombocytopenia |
| Herpes zoster | Microsporidiosis | Aspergillosis | Papova viruses | Lymphomas (EBV) | Lupus erythematosus |
| Cytomegalovirus (CMV) | Isosporiasis | Candidosis | Cryptococcus neoformans | Squamous cell carcinoma (cervix, anus) (HPV) | Encephalopathy |
| Epstein–Barr virus (EBV) | Giardiasis | Cryptococcosis | Toxoplasma gondii | Drug allergies | |
| Human herpesvirus 8 | Histoplasmosis | ||||
| Human papilloma viruses (HPV) | Zygomycosis (mucormycosis) | ||||
| Strongyloidiasis | |||||
| Molluscum contagiosum | Tuberculosis | ||||
| Non-tuberculous mycobacteria | Non-tuberculous mycobacterioses | ||||
| Candida species | Legionellosis | ||||
| Staphylococcus aureus | Pseudomonas aeruginosa | ||||
| Histoplasmosis | Staphylococcus aureus | ||||
| Streptococcus pneumoniae | |||||
| Haemophilus influenzae | |||||
| Toxoplasmosis | |||||
| CMV | |||||
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Immunodeficiency diseases can affect any component of the immune system but often do not produce clinical pictures that are precisely predictable from the immune defect. Infections do vary in character as a consequence of the nature of the immune defect; the kinds of microorganisms to which the patient is exposed; and attempts at treatment (for example, broad-spectrum antibiotics used to control bacterial infections increase the risk of fungal infections such as candidosis). Thus, in T-cell disorders (such as HIV disease), cell-mediated immunity is principally affected but antibody production may also be impaired, so common infections are with fungi, viruses and certain bacteria – such as candida, herpesviruses and mycobacteria.
Secondary Immune Defects
Human Immunodeficiency Virus (HIV) Infection and the Acquired Immunodefiency Syndrome (AIDS)
HIV is the virus that can lead to AIDS. HIV (a lentivirus or retrovirus) infects CD4+helper T lymphocytes, causing AIDS after an average of 10 years in the untreated, and has spread worldwide to create a global pandemic. In 2012 there were over 2 million new infections and 34 million infections recorded worldwide; there was a three-fold rise in individuals over 50 years of age in the UK and one-third of those infected in the UK remained undiagnosed. HIV is found worldwide, and disease patterns are changing as a result of increasing travel and the emergence of new pathogens, such as multidrug-resistant tuberculosis, and the appearance of exotic infections in temperate zones. HIV weakens the immune system, putting persons with HIV or AIDS at risk for many different types of infections. In untreated AIDS, the mortality rate is virtually 100%, although antiviral drugs can significantly impede disease progress.
General Aspects
AIDS was first recognized in 1981 in young men who have sex with men (MSM) in the USA. Sporadic cases have since been identified as long ago as 1959 in Central Africa (where the virus appears to have crossed the species barrier from chimpanzee monkeys).
In June 1981, five cases of Pneumocystis carinii (now jiroveci) pneumonia were reported in five young males; all suffered from cytomegalovirus infections. Later the same year in the USA, a series of previously healthy MSM who developed Pneumocystis carinii pneumonia and mucosal candidiasis were reported, with the suggestion that infection could have been caused by a cellular inmunodeficiency. These publications present the first proven cases of what later was termed AIDS. As the first AIDS patients were MSM, it was initially believed that the illness was provoked by peripheral vasodilator drugs (‘poppers’), especially amyl nitrite, used by male homosexuals to increase sexual activity. Nevertheless, by the end of 1981, a few cases of AIDS appeared in intravenous drug users, suggesting an infectious aetiology. AIDS was also recognised in all populations, mainly in heterosexuals. After the discovery of the aetiological virus (then termed human T-lymphotropic virus: HTLV-3), retrospective analyses confirmed the presence of the causal virus, which became known as HIV, in the tissues of a Norwegian sailor who died in 1976 and an Afro-American adolescent who died in 1969, and in the plasma of a Congolese male obtained from 1959.
Ten years after HIV was defined, the use of a standardized method to quantify its plasma concentration (viral load) became adopted worldwide. The viral replication rate is stable at every stage of the disease, being estimated at about 109–10 new virions a day. Its assessment is important in the follow-up of infected patients and in the choice of therapeutic approach.
HIV transmission
HIV is transmitted via body fluids, mostly by:
■ sexual intercourse – semen, blood and possibly saliva may contain HIV
■ contaminated blood, blood products and donated organs
■ contaminated needles – intravenous drug users and needlestick injuries
■ vertical transmission (mother to child) – in utero, during childbirth and via breast milk.
HIV is spread primarily by sexual intercourse, via:
■ having unprotected sex with someone who is HIV-infected:
Not using a condom when having sex with a person who has HIV is a risk.
Unprotected anal sex is riskier than unprotected vaginal sex.
Among MSM, unprotected receptive anal sex is riskier than unprotected insertive anal sex.
Unprotected oral sex can be a risk, but is a much lower risk than anal or vaginal sex.
Less common modes of HIV transmission include:
■ being damaged or ‘stuck’ with an HIV-contaminated needle or other sharp object
■ receiving HIV-contaminated blood transfusions, blood products or organ/tissue transplants
■ receiving unsafe or unsanitary injections or other medical or dental practices
■ eating food that has been pre-chewed by an HIV-infected person
■ being bitten by an HIV-infected person
■ undergoing tattooing or body piercing using unsafe or unsanitary procedures.
HIV cannot reproduce outside the body and is not spread by:
■ air
■ water
■ insects, including mosquitoes
■ social contact, like shaking hands or sharing cups or dishes
The virus is transmitted mainly by intimate sexual contact and can infect anyone who practises risky behaviours, such as having sexual contact with an infected person or someone whose HIV status is unknown without using a condom. Sexual transmission of HIV is more likely during HIV seroconversion or in advanced AIDS, by receptive anal intercourse, or where there is genital ulceration. Saliva and breast milk may contain HIV, but transmission via the orogenital route is uncommon. Reports that HIV virions are present in a cell-free state in saliva have not been confirmed and saliva contains inhibitory factors. However, saliva transmission may result from human bites.
Heterosexual intercourse is the main mode of transmission worldwide; it is an important route in developing countries in Africa and Asia, and is increasingly important in the developed world, where it is responsible for 25–50% of cases. MSM remain a major risk group in developed countries. In the UK, the number of young men diagnosed with HIV has more than doubled in the last decade and, in 2012, almost half of new cases were infected through sex between men. With antiretroviral therapy, more people with HIV now survive and this has resulted in a rise in the number of new cases of HIV infection – especially in MSM – along with increasing use of the Internet to make contact with new partners; greater substance abuse and use of medications to treat erectile dysfunction; and a rise in unsafe sexual practices. Unfortunately, multidrug-resistant HIV (MDR-HIV) has appeared, with a rapid progression to AIDS.
HIV spread by contaminated needles and syringes is an important route in injecting drug users. Persons infected with HIV, especially those who abuse drugs intravenously or/and are sexually promiscuous, may also be co-infected with other agents, including viruses, such as HTLV-1, HTLV-2 and hepatitis viruses, and other SSIs (Ch. 32). Transmission by needlestick injury is an occasional risk for health-care professionals (HCPs; see later and Ch. 31) and others.
Transmission of HIV can also occur via infected blood or blood products, including plasma, or tissues, such as in transplantation, but screening of blood and plasma for HIV antibody and heat treatment of clotting factor concentrates have substantially reduced the risk in developed countries. All HIV antibody-positive individuals are potentially infective and must not be allowed to donate blood, organs for transplantation or semen for artificial insemination. Rarely, some seronegative but infected blood (collected during the incubation period before antibodies are detectable) can still transmit HIV. Haemophiliacs, initially a risk group for HIV, now form only a small proportion of HIV patients.
Children can contract HIV intrapartum or via breast milk; approximately one-quarter of all untreated pregnant women infected with HIV pass the infection to their babies, and the figure rises to one-third if the infant is breast-fed. Antiretroviral drugs reduce this type of transmission (Box 20.1).
2 Protease inhibitors (PIs)
3 Integrase inhibitor
Raltegravir (RAL)
4 Entry inhibitor
Fusion inhibitors
CCR5 antagonist
There is no reliable evidence for transmission of HIV by non-sexual social contact or by insect vectors such as mosquitoes or bedbugs. Studies of families of HIV-infected people have shown clearly that HIV is not spread through casual contact, such as the sharing of food utensils, towels and bedding, swimming pools, spas, telephones or lavatory seats.
Prevention of HIV/AIDS can thus be achieved by measures such as the use of condoms, the cessation of needle-sharing, screening blood and tissue before transfer to a recipient, and the use of antiretroviral drugs for children born to HIV-infected mothers.
HIV pre-exposure prophylaxis
Daily oral tenofovir disoproxil fumarate 300 mg (TDF)/emtricitabine 200 mg (FTC) is available as antiretroviral pre-exposure prophylaxis (PrEP) to reduce the risk for HIV acquisition. By 2013, four trials had compared HIV infection rates in participants randomized to receive PrEP compared with rates in participants on placebo. No serious toxicities were identified, efficacy was around two-thirds and there was no evidence of adverse effects among fetuses exposed to TDF or FTC. Daily oral TDF/FTC use has been shown to be safe in reducing the risk for sexual HIV acquisition when used consistently. The efficacy of TDF/FTC for HIV prevention is dependent on adherence to daily doses of medication; PrEP long-term safety in HIV-uninfected adults or following fetal exposure is not yet determined.
Centers for Disease Control and Prevention (CDC) guidance for clinicians considering using PrEP for adults at very high risk for HIV acquisition through heterosexual sex is as follows:
Further information is available at: http://www.cdc.gov/hiv/prep/ (accessed 30 September 2013).
HIV epidemiology
HIV-1, the most common virus causing AIDS, has spread to all continents to create a global pandemic with the most devastating consequences ever known to humankind. A further virus, HIV-2, which originated in sooty mangabee monkeys, predominates in West Africa, and has also now spread to many countries, including Central Africa, Europe, the USA and South America; at present, however, there are few HIV-2 infections in developed countries.
An estimated 20 million people have died from AIDS since the epidemic was recognized. Estimates are that over 40 million HIV cases had developed worldwide by 2008 – more than 1 in every 100 sexually active adults. The difference between numbers estimated and reported is due to underdiagnosis (especially in the developing world); under-reporting and delayed reporting; and false extrapolation of basic information derived from Africa and MSM.
Probably 75% of cases worldwide are located in Africa, about 10% in Latin America, about 7% in the USA, about 5% in Asia and slightly less in Europe. However, epidemics are progressing in Asia, especially India and China, and in Eastern Europe. In the USA, the CDC estimated that, by 2008, gay and bisexual men of all races were the group most heavily affected by HIV, accounting for 53% of all new infections, and the impact of HIV was greater among blacks than any other racial or ethnic group, with an HIV incidence rate seven times higher than that of whites and almost three times higher than that of Latinos. Currently, HIV infection is the commonest cause of death in US men aged 25–44 years, and the third commonest cause of death in women of this age.
In the UK, HIV infection is the fourth most common cause of death in men aged 25–44 years. New HIV diagnoses in the UK in 2011 showed a slight decrease on 2010 and continued a year-on-year decline from the peak in 2005. The overall decline in new diagnoses is largely due to fewer reported cases among heterosexuals, who probably acquired their infection abroad. The number of new HIV diagnoses in MSM surpassed new diagnoses in heterosexuals. Half of those diagnosed in 2011 probably acquired their infection through sex between men. Continued low numbers of HIV diagnoses were made in people who inject drugs and through other exposures. All infections acquired through receipt of blood/tissue products diagnosed since 2002 were acquired outside of the UK.
Pathogenesis
The HIV viruses are ribonucleic acid (RNA) viruses. HIV-1 and HIV-2 differ in antigens and nucleic acids but are transmitted similarly, have similar biological properties and are both capable of causing disease. HIV-2 may progress more slowly and cause less profound immunosuppression. HIV-1/HIV-2 co-infection can occasionally be acquired. However, the remaining discussion relates to HIV-1.
HIV-1 can be classified into two major groups: M, which contains ten genetically distinct subtypes (A to J), and O, which contains several very heterogeneous viruses. The B virus is found in USA, Europe and Central Africa.
HIV contains several so-called ‘core’ proteins and is surrounded by an envelope made up of ‘coat’ proteins; one of these, the viral cover glycoprotein (gp 120), interacts with a cell surface protein receptor (CD4). The external layer or cover presents 72 external projections where glycoproteins (gp41, gp120) are placed, as well as a number of proteins derived from the host cells, such as beta2-microglobulin and the alpha and beta chains of the human leukocyte antigen (HLA)-DR. Chemokine (co-)receptors are also involved, the most important being CCR5, the main monocytotropic cell receptor; chemokines (RANTES, MIP-1α MIP-2β); and the CXCR4 (fusine), which is inside the lymphotropic cells and whose natural linkage is stromal-derived factor 1 (SDF1) chemokine. There are several viral variants with different trophisms to CCR5 and CXCR4, which have the capacity for interaction with different co-receptors such as CCR2, CCR3, BONZO and BOB; they are sometimes capable of penetrating a cell using multiple co-receptors. The importance of RANTES and SDF lies in their capability to compete for the same HIV receptors, blocking the viral cycle by preventing virus penetration.
CD4 cells are mainly helper-inducer T lymphocytes, monocytes and macrophages, Langerhans cells, brain glial cells and some colonic cells. Brain tissue, in particular the cells of monocyte–macrophage lineage, can become infected with HIV. Neurological damage may also be mediated by the production of factors that affect neuronal cell function or neural transmitters by HIV-infected macrophages. Most HIV is tropic for various chemokines such as CC CKR-5 (CCR5-tropic virus or R5), involved in HIV–cell attachment; a few patients appear able to resist infection because of altered expression of this chemokine or the emergence of CXCR4-tropic virus strains (X4). Some HIV strains show dual tropism. HIV-1 variants that use the co-receptor CCR5 for entry (R5; macrophage tropic) predominate in early infection, while variants that use CXCR4 emerge during disease progression. Some late-stage variants use CXCR4 alone (X4; T-cell tropic), while others use both CXCR4 and CCR5 (R5X4; dual tropic). One coat protein, p24, is particularly antigenic and antibodies to p24 form the basis for most serological testing (the HIV test). The coat proteins are subject to considerable variability, making vaccine development difficult.
After interaction of the viral particle and receptors, the virus coat merges with the cell membrane, losing the external capsule, while the nucleocapsid penetrates the cytoplasm, releasing viral RNA. The genetic material is set inside the core, constituted by two identical RNA strands of positive polarity, nucleoproteins and several enzymes necessary to complete the viral cycle which eukaryote cells lack. HIV also contains enzymes involved in its pathogenicity – reverse transcriptase (which can transcribe DNA from RNA, i.e. in the reverse direction; hence ‘retrovirus’) and proteases. A retrotranscription is produced in the infected cell by a number of reactions in which DNA polymerase, RNAase and inverse transcriptase (from the virion) are involved. A DNA double strand develops from the RNA genome of the virus. This viral DNA is carried to the cell core, forming part of the host genome, due to the action of the viral integrase and other proteins such as Vpr, p17 and Nef, and becoming the integrated provirus. In the CD4 lymphocytes at rest, the viral genome is incompletely retrotranscribed and the integration process ends only when the cell becomes active. These proviruses lack pathogenic capability and, thus, preventing cellular activation would avoid the virus cytopathic effect.
The messenger RNA is carried to the cytosol and processed in different transcripts of different sizes, via the action of the Rev viral protein, leading to the synthesis of viral proteins by the polysomes. The absence of Rev protein would provoke the gathering of messenger RNA in the cell core.
Viral proteins synthesized and forming a single long chain have to be processed (cut) in order to become structural elements of the nucleocapsid and the viral coat, as well as to form proteins of enzymatic activity (transcriptase, ribonuclease, integrase and protease). In this process, although it is the protease action that is essential in the production of viral particles, turning the p55 pre-proteic precursors into p24, p17, p9, p6 nucleocapsid proteins and the gp160 ones into gp41 and gp120, different viral proteins such as Vif and Vpu are also involved. These are potential targets for therapy.
Finally, the viral genome, covered by the nucleocapsid, projects into the cellular membrane within a layer of proteins, such as viral beta2-microglobulin and gp41 and gp120; the virus is then released.
After primary infection with HIV, acute viraemia results in widespread dissemination of HIV. The virus is trapped in follicular dendritic cells in lymphoid tissue germinal centres and there is expansion of a subset of CD8 cells that are precursors of HIV-specific cytotoxic T cells. Proinflammatory cytokines, including interleukin-6 (IL-6), tumour necrosis factor, gamma-interferon and IL-10, are also overexpressed and may represent an unsuccessful attempt at a protective response. Dendritic cells are involved in the initiation and propagation of HIV infection in CD4 cells.
Antibodies to HIV develop and appear in the serum within 6 weeks to 6 months from infection (seroconversion) but, within this window, the patient may be seronegative (though HIV may be detected by the polymerase chain reaction; PCR). The seroconversion clinical illness is a battle between the virus and the host. Usually, the virus has been proliferating unchecked prior to this point; therefore infectivity is maximal and thereafter wanes (although never dropping to zero). There is then a variable latent period that can last years, until HIV infection manifests with HIV disease and, ultimately, with AIDS.
Clinical Features
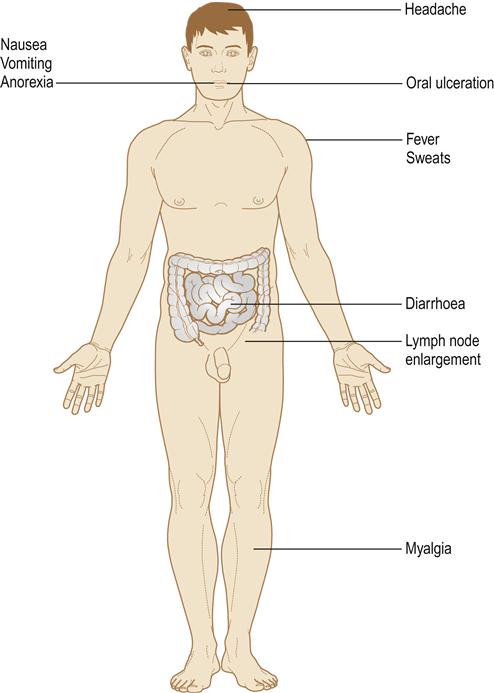
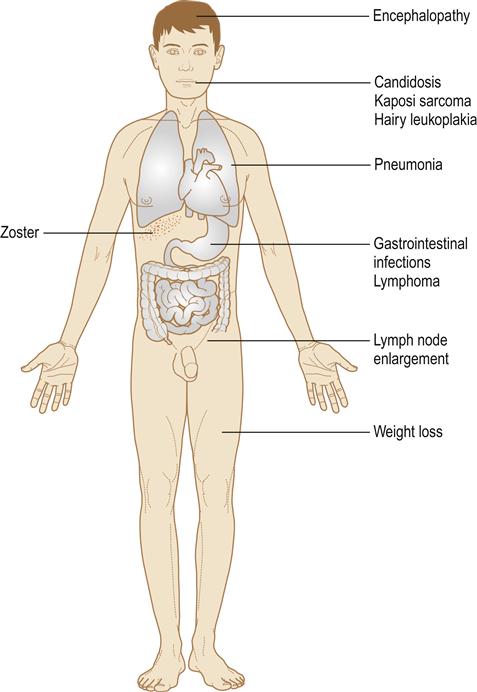
Infection with HIV may be staged according to its clinical features (Table 20.3). After primary infection with HIV, an acute viraemia results in widespread dissemination of the virus. Antibodies to HIV develop and appear in the serum within 6 weeks to 6 months of infection (seroconversion). One-third of individuals develop a clinical seroconversion illness 3–6 weeks after infection, resembling glandular fever; this is often severe and is termed the acute retroviral syndrome (ARS). Sometimes there is neurological involvement or a rash or mouth ulcers (Fig. 20.2). HIV infection may then remain asymptomatic for a median time of about 10 years. However, despite having circulating (and thus diagnostic) antibodies, people are also able to transmit the disease in the interim. Persistent generalized lymphadenopathy may become evident (Fig. 20.3). As HIV damages CD4 cells in the immune and nervous systems, the risk of development of severe immunodeficiency, and symptoms of disease, rises. HIV damage to CD4+lymphocytes leads to a lymphopenia over about 6 months to 10 years, a fall in the ratio of helper (CD4+) to suppressor (CD8+) lymphocytes, a profound defect in cell-mediated immune reactivity, and HIV disease, which manifests with infections (see Fig. 20.2) as the CD4 count falls to about 500/mm3, and the CD4+to CD8+T-lymphocyte ratio falls. HIV disease manifests in a variety of clinical syndromes, commonly including infections (Fig. 20.4), neoplasms, and neurological and autoimmune disorders (Table 20.4). The CDC definition of AIDS comprises a CD4 count of less than 200/mm3 of blood in a patient infected with HIV and a clinical condition associated with advanced HIV disease – such as pulmonary tuberculosis (TB), Pneumocystis pneumonia, recurrent episodes of pneumonia or invasive cervical carcinoma.
Table 20.3
Centers for Disease Control and Prevention (CDC) classification of HIV infection
| CDC stage | Presentation |
| 1 | Primary seroconversion illness |
| 2 | Asymptomatic disease |
| 3 | Persistent generalized lymphadenopathy (PGL) |
| 4a | AIDS-related complex (ARC) but no AIDS-defining conditions |
| 4b–d | AIDS (AIDS-defining illness) |

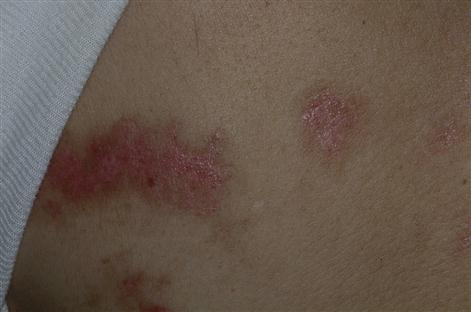
Table 20.4
Main manifestations of HIV/AIDS
| Main manifestations | Comments |
| Opportunistic infections | Mainly fungal and viral |
| Neoplasms | Mainly Kaposi sarcoma and lymphoma |
| Neurological disorders | Mainly dementia |
| Autoimmune disorders | Mainly purpura |
Patients can, however, develop a range of infections (viral, mycobacterial, fungal and parasitic), malignancies (malignant neoplasms, including melanoma, basal cell carcinoma, Kaposi sarcoma, lymphomas and squamous cell carcinomas of the skin and lip, and carcinoma of the cervix, external genitalia and perineum) and encephalopathy. Loss of weight and wasting are common in AIDS and, in Africa, is called ‘slim disease’ (Figs 20.5 and 20.6). The onset of AIDS may be influenced by the HIV virus titre, patient age, gender, drug habits, immunogenetics and other factors. Clinical disease is most likely to appear when the CD4 counts fall to low levels, where the infected person has depressed defences for other reasons (e.g. diabetes or malnutrition), and where there is high exposure to potential pathogens (e.g. TB, cryptosporidiosis or histoplasmosis).
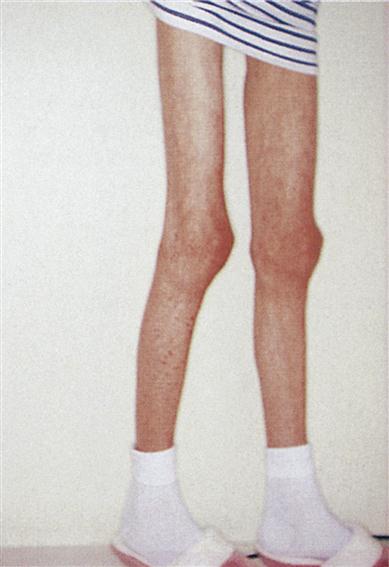
In the developed world, about 20% of HIV-infected people develop AIDS within 5 years, and 50% at 10 years; nearly all will have developed AIDS at 20 years unless treated. Some HIV-infected individuals have, or are vulnerable to, a host of co-morbid conditions, including other infections (e.g. fungal/viral infections, hepatitis, SSIs, TB), substance abuse, mental illness and homelessness.
A very small minority of people appear resistant to HIV infection, mostly because of a mutation in the CCR5 gene called CCR5-delta32, which inhibits HIV binding to CD4 cells. A very few HIV-infected persons (about 2%) inherently appear not to develop AIDS over periods as long as 15 years, mostly because they retain active cytotoxic T cells.
The 2008 CDE Case Definition for HIV infection in adults is as follows (http://www.cdc.gov/osels/ph_surveillance/nndss/casedef/aids2008.htm; accessed 30 September 2013):
AIDS-defining conditions
■ candidiasis of bronchi, trachea or lungs
■ coccidioidomycosis, disseminated or extrapulmonary
■ cryptococcosis, extrapulmonary
■ cryptosporidiosis, chronic intestinal (>1 month’s duration)
■ cytomegalovirus disease (other than liver, spleen or nodes), onset at age>1 month
■ cytomegalovirus retinitis (with loss of vision)
■ histoplasmosis, disseminated or extrapulmonary
■ isosporiasis, chronic intestinal (>1 month’s duration)
■ lymphoid interstitial pneumonia or pulmonary lymphoid hyperplasia complex
■ lymphoma, Burkitt (or equivalent term)
■ lymphoma, immunoblastic (or equivalent term)
■ Mycobacterium avium complex or Mycobacterium kansasii, disseminated or extrapulmonary
■ Mycobacterium tuberculosis of any site, pulmonary, disseminated or extrapulmonary
■ Mycobacterium, other species or unidentified species, disseminated or extrapulmonary
■ Pneumocystis jiroveci pneumonia
■ progressive multifocal leukoencephalopathy
■ Salmonella septicemia, recurrent
Opportunistic infections
Opportunistic infections are common and resistant to treatment. The pattern of infections varies geographically and with time. For example, histoplasmosis affects mainly AIDS patients from endemic areas of the USA and tuberculosis is seen mainly in AIDS in Africa. Fungal, bacterial, protozoal and viral infections that may be seen are described below.
Fungi
Pneumocystis jiroveci (P. carinii) pneumonia is a common fungal (previously thought to be a protozoon) lung infection in patients with HIV/AIDS; it is seen up to 80% and is the immediate cause of death in up to 20%. Features include cough, dyspnoea, fever and malaise. Treatment is often with co-trimoxazole or pentamidine. Clindamycin/primaquine, atovaquone and dapsone/trimethoprim are alternatives. Pneumocystis pneumonia is the most common opportunistic infection in people with HIV disease. People with CD4 cell counts under 200/mm3 have the highest risk of developing it.
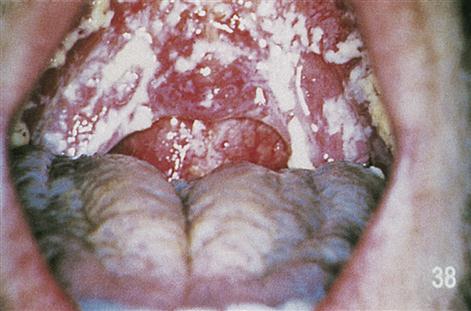
Candidosis (candidiasis) is extremely common in the mouth (Fig. 20.7) and vagina, and virtually all patients experience it. HIV infection should be suspected in a young or youngish person who develops thrush without any apparent precipitating factor.
Cryptococcus neoformans is a mycosis that is the main cause of meningitis in AIDS and is especially common in Africa. It is treated with amphotericin or/and an azole.
Histoplasmosis, usually as disseminated disease, mainly affects persons from endemic areas in the Americas. The same applies to coccidioidomycosis. Both infections are treated with amphotericin or/and an azole.
Penicillosis (infection with Penicillium marneffei) is seen in some areas of Thailand.
Bacteria
Tuberculosis is particularly common in Africa and is spreading in the West, where some 5% of patients now have lung infection with Mycobacterium tuberculosis, which is increasingly multidrug-resistant. TB can affect other sites, including the central nervous system (CNS). Since it can spread by droplets, it is becoming a major public health problem.
Atypical mycobacteria are found in about 40% of AIDS patients in the West, though double that number are actually infected. Over 95% of these are Mycobacterium avium complex (MAC) infections – M. avium and M. intracellulare.
Protozoa
Toxoplasmosis, due to the protozoon Toxoplasma gondii, affects about 15% of AIDS patients, and particularly involves the CNS. It is treated with pyrimethamine and sulfadiazine. Leishmaniasis may be seen.
Gastrointestinal protozoal infections (cryptosporidia, microsporidia, Isospora belli) often cause intractable diarrhoea.
Viruses
Herpes simplex virus can produce severe erosive lesions around the mouth, genitals, anus or other orifices, and may be seen intraorally and in the oesophagus. It can involve the skin and eyes. It is treated with aciclovir or foscarnet.
Herpes zoster can be severe, painful and incapacitating. It is treated with aciclovir or foscarnet.
Cytomegalovirus (CMV) is the most dangerous viral infection, capable of widespread lesions, especially ulceration throughout the gastrointestinal tract, and retinitis. It is treated with ganciclovir, foscarnet or cidofovir.
Epstein–Barr virus (EBV) is associated with oral hairy leukoplakia (Fig. 20.8) and with lymphomas.
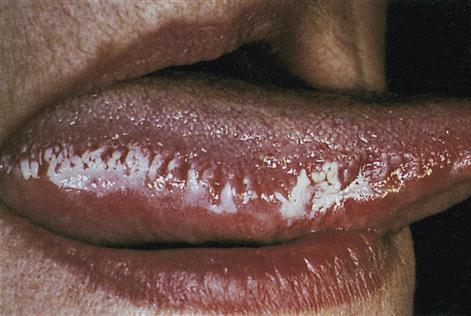
Human papillomavirus (HPV) infections produce warty lesions.
Neoplasms
Various neoplasms are seen in HIV/AIDS – often virally related.
Kaposi sarcoma (an otherwise exceedingly rare tumour of endothelial cells among elderly persons) is seen almost exclusively in sexually transmitted HIV infection in MSM, associated with human herpesvirus 8 (HHV-8; Kaposi sarcoma herpesvirus, KSHV) and accounts for about 80% of neoplasms in AIDS. It often affects the gastrointestinal tract, skin, respiratory tract, mouth or lymph nodes. It is atypical in its early age of onset, distribution (particularly in the head and neck area) and greater malignancy (Fig. 20.9).
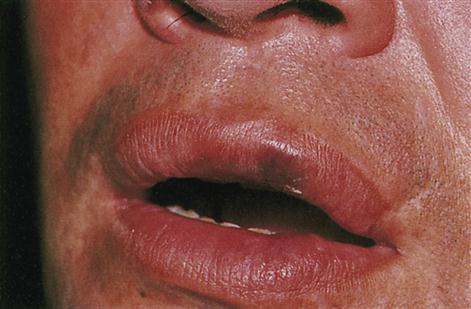
Lymphomas, mostly non-Hodgkin B cell lymphomas, are also seen in AIDS (Fig, 20.10); they frequently affect the brain and are often related to EBV. A diffuse infiltrative lymphocytosis syndrome (DILS) can cause asymptomatic bilateral parotid swelling; since patients with DILS can develop lymphomas, periodic observation is mandatory.
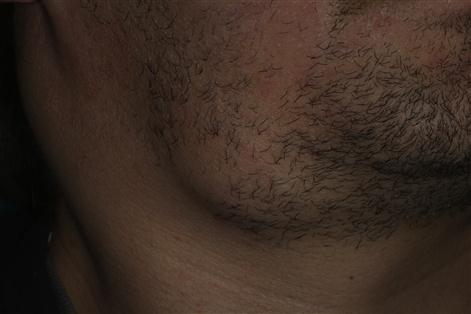
Carcinomas of the cervix and of the anus are increased in HIV disease and caused by HPVs.
Neurological disorders
HIV can cause a variety of neurological symptoms, culminating in encephalopathy (AIDS-related dementia) and death. In addition, intracranial infections (mainly parasitic, viral or fungal) are common. Up to two-thirds of AIDS patients develop a dementia complex, with personality changes, ataxia and convulsions; about 15% of AIDS patients develop CNS toxoplasmosis, while 10% develop cryptococcal meningitis. Spinal cord disease is seen in about 20% and may progress to bladder and bowel dysfunction.
Neurological manifestations can be acute, with fever, malaise, depression, fits, facial palsy and neuropathies affecting the extremities. Myelopathy or neuropathy can cause weakness of the legs, sometimes paraesthesiae and, in severe cases, ataxia and incontinence. Subacute encephalopathy affects about 30% of AIDS patients, who gradually develop a confusional state associated with fever and depression; the patient eventually may become bedridden and incontinent. Chronic meningitis may precede more typical features of AIDS and is characterized by fever, headache, meningeal signs and cranial nerve palsies (typically affecting nerves V, VII and VIII). Brain tumours, particularly lymphomas, are also common complications of HIV/AIDS. Peripheral and cranial neuropathies, and myopathy, are relatively common.
Autoimmune disorders
Autoimmune disorders, particularly thrombocytopenic purpura, are relatively common in HIV disease. Drug allergic reactions are common, mostly to co-trimoxazole, and may cause erythema multiforme.
HIV infection in children
Infection with HIV in children causes them to appear ill and stunted, and promotes staphylococcal and streptococcal infections, such as otitis media, rashes and diarrhoea, as well as fungal and viral infections.
Normal CD4/CD8 ratios may persist late in the disease and lymphopenia is uncommon. Humoral immunity is more severely affected than in adult HIV disease and may long precede changes in T-lymphocyte subsets. Bacterial infections are relatively more common because of depressed humoral immunity. Generalized lymphadenopathy and hepatosplenomegaly are often the first signs and chronic parotitis is a common presenting feature. Neurological disease develops in the great majority. Interstitial lymphoid pneumonitis is particularly characteristic. Hypergammaglobulinaemia is also severe and 13% have thrombocytopenic purpura.
B-cell lymphomas are the most common tumours but Kaposi sarcoma is rare and affects the skin particularly infrequently. Other systemic effects of HIV infection in children include renal disease and cardiomyopathy.
General Management
Diagnosis of HIV infection
Diagnosis of HIV infection is made on suspicion, especially if there is a supportive history; clinical criteria, if present; and, after appropriate consent, laboratory investigations. Lymphopenia, low CD4 counts in the blood, and a low CD4:CD8 ratio (normal value is about 2, which falls to about 0.5 in AIDS) are typical but not specific findings. HIV infection must be confirmed by testing for HIV and all results must be kept confidential. Relatives cannot give consent for an HIV test. Most HIV tests detect HIV serum antibodies, but it takes some time – 2–8 weeks – to produce enough antibodies to be detected by the antibody test. This ‘window period’ can vary from person to person and the HIV viral load may be very high at this time, as is the likelihood of transmitting HIV to sex or needle-sharing partners. Most HIV-infected people (97%) will develop detectable serum antibodies in the first 3 months, but a few will take longer, so a follow-up test more than 3 months after the last potential exposure to HIV is prudent. All those infected will have detectable serum antibodies to HIV by 6 months.
Conventional HIV serological tests sent to a laboratory give results within a week or two; rapid HIV tests can produce results in as little as 20 minutes. A positive HIV serological test result means that a person may have been infected with HIV, but all positive HIV test results, whether from conventional or rapid tests, must be verified by a second ‘confirmatory’ HIV test. Thus HIV antibody screening tests (e.g. reactive enzyme immunoassay [EIA]) should be confirmed by a positive result from a supplemental HIV antibody test (e.g. Western blot or indirect immunofluorescence assay test) or a positive result or report of a detectable quantity from any of the following HIV virological (i.e. non-antibody) tests:
■ HIV nucleic acid (DNA or RNA) detection test (e.g. PCR)
Note that a negative (i.e. undetectable or non-reactive) result from an HIV virological test (e.g. viral RNA nucleic acid test) does not rule out the diagnosis of HIV infection.
HIV testing should be offered not only as part of a sexual health screen but also to:
■ anyone with one of a number of ‘clinical indicator diseases’
■ patients with specific lifestyle or demographic characteristics
■ all adults in all health-care services if HIV prevalence is high.
No patient with signs of HIV infection should be discharged as uninfected solely on the basis of one negative HIV test result; neither should they be told that they have HIV infection without the knowledge that the laboratory has confirmed the test result using the above criteria. Treat the results of any HIV tests and a diagnosis of HIV infection or AIDS with confidentiality. Considerable care is needed regarding the social and psychological consequences of HIV infection. More information on HIV testing is available at: http://www.aidsmap.com/UK-guidelines/page/1321555/ (accessed 30 September 2013).
The mortality rate from AIDS was virtually 100% but, although there is still no effective treatment for the underlying immune defect, antiretroviral therapy (ART) employs drugs that can significantly impede the progress of HIV infection; these include nucleoside analogues, non-nucleoside reverse transcriptase inhibitors, protease inhibitors, entry/fusion inhibitors and integrase inhibitors. Treatment with a combination of these drugs is termed active ART. Early intervention with ART (Tables 20.5, 20.6, 20.7 and 20.8) is usually beneficial.
Table 20.5
Antiretroviral drugs, cautions and adverse effects: nucleoside reverse transcriptase inhibitors (NRTIs)a
< ?comst?>
| Generic name (abbreviation) | Trade name | Possible orofacial adverse effects | Other main adverse effects | Cautions with drugs used in oral health care |
| Abacavir (ABC) | Ziagen | Erythema multiforme, ulcers | Lactic acidosis, nausea, diarrhoea, life-threatening rash; human leukocyte antigen (HLA)-B*5701 screening (for hypersensitivity reaction), lipodystrophy | No significant interactions |
| Didanosine (ddI) | Videx | Erythema multiforme, dry mouth, facial lipoatrophy | Nausea, diarrhoea, lactic acidosis, mitochondrial dysfunction leading to pancreatitis, abnormal liver function, peripheral neuropathy, retinal damage | Ciprofloxacin |
| Ganciclovir | ||||
| Itraconazole | ||||
| Ketoconazole | ||||
| Emtricitabine | Emtriva | Hyperpigmentation, swelling | Lactic acidosis, pruritus, hyperpigmentation, lipodystrophy | No significant interactions |
| (FTC) | ||||
| Lamivudine (3TC) | Epivir | Dry mouth | Nausea, diarrhoea, mitochondrial dysfunction leading to pancreatitis, abnormal liver function, neuropathy, lipodystrophy, clefts | No significant interactions |
| Stavudine (d4T) | Zerit | Facial lipoatrophy | Neuropsychiatric reactions, mitochondrial dysfunction leading to pancreatitis, abnormal liver function, lactic acidosis, neuropathy | No significant interactions |
| Tenofovir disoproxil (TDF) | Viread | Facial lipoatrophy | Renal tubular damage, hypophosphataemia | No significant interactions |
| Zidovudine (azidothymidine; AZT, ZDV) | Retrovir | Erythema multiforme, hyperpigmentation, lichenoid drug reaction, facial lipoatrophy | Nausea, diarrhoea, myelotoxicity, lactic acidosis, myopathy | Fluconazole |
| Ganciclovir |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
aThere are also combinations of these; zalcitabine (dideoxycytidine; DDC) is no longer marketed.
Table 20.6
< ?comst?>
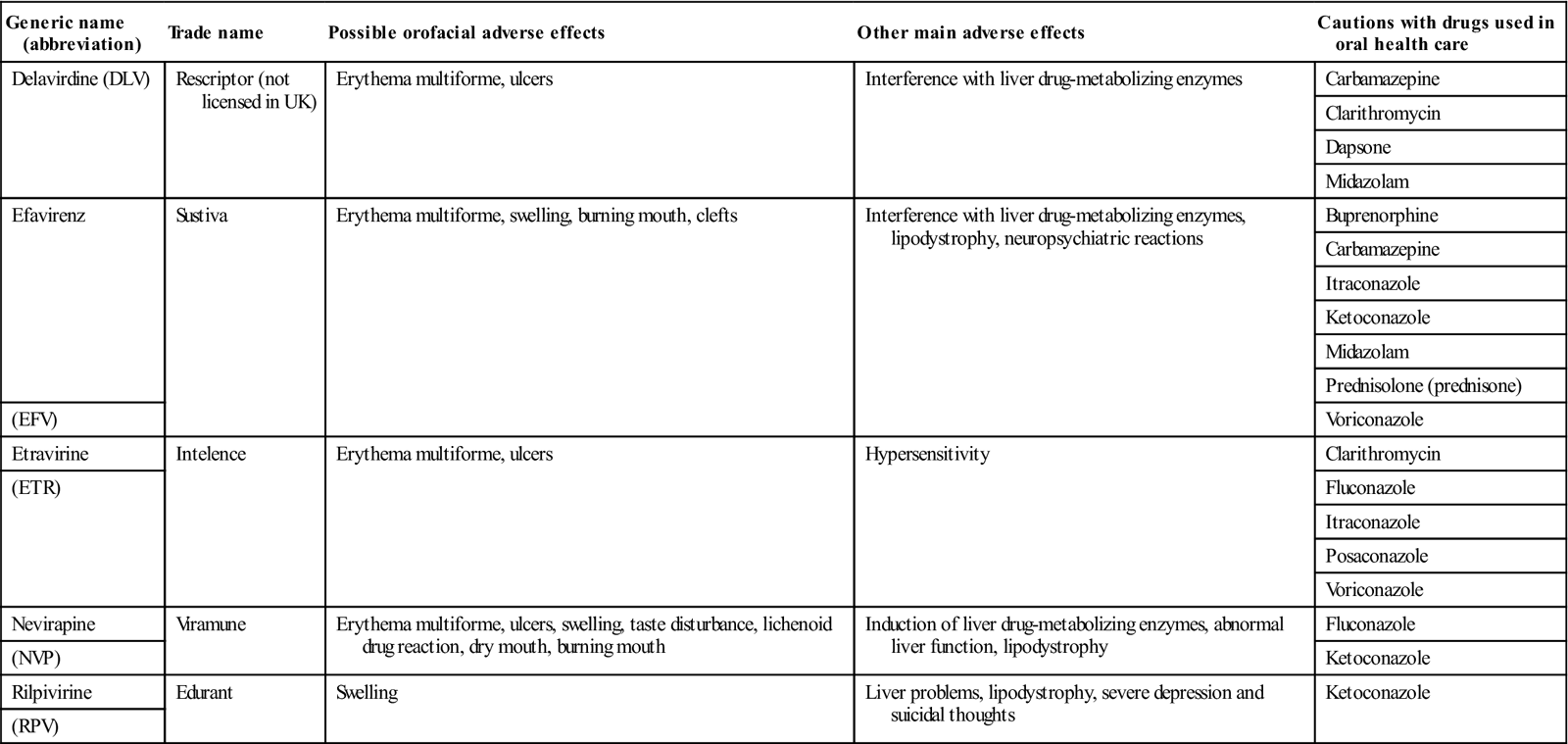
| Generic name (abbreviation) | Trade name | Possible orofacial adverse effects | Other main adverse effects | Cautions with drugs used in oral health care |
| Delavirdine (DLV) | Rescriptor (not licensed in UK) | Erythema multiforme, ulcers | Interference with liver drug-metabolizing enzymes | Carbamazepine |
| Clarithromycin | ||||
| Dapsone | ||||
| Midazolam | ||||
| Efavirenz | Sustiva | Erythema multiforme, swelling, burning mouth, clefts | Interference with liver drug-metabolizing enzymes, lipodystrophy, neuropsychiatric reactions | Buprenorphine |
| Carbamazepine | ||||
| Itraconazole | ||||
| Ketoconazole | ||||
| Midazolam | ||||
| Prednisolone (prednisone) | ||||
| (EFV) | Voriconazole | |||
| Etravirine | Intelence | Erythema multiforme, ulcers | Hypersensitivity | Clarithromycin |
| (ETR) | Fluconazole | |||
| Itraconazole | ||||
| Posaconazole | ||||
| Voriconazole | ||||
| Nevirapine | Viramune | Erythema multiforme, ulcers, swelling, taste disturbance, lichenoid drug reaction, dry mouth, burning mouth | Induction of liver drug-metabolizing enzymes, abnormal liver function, lipodystrophy | Fluconazole |
| (NVP) | Ketoconazole | |||
| Rilpivirine | Edurant | Swelling | Liver problems, lipodystrophy, severe depression and suicidal thoughts | Ketoconazole |
| (RPV) |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Table 20.7
Antiretroviral drugs, cautions and adverse effects: protease inhibitors (PIs)a
< ?comst?>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses