Chapter 2
Form and Function of the Oral Mucosa
The oral cavity is lined by a stratified squamous epithelium which, together with its underlying lamina propria of fibrovascular connective tissue, has a structure and function that varies from site to site in the oral cavity. It is hardly surprising, therefore, that the incidence of oral mucous membrane disease is influenced by both anatomical structure and physiological function.
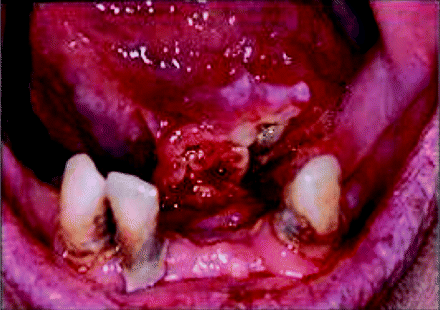
In Northern European patients, intraoral squamous cell carcinoma predominates in the ventrolateral tongue, floor of mouth and mandibular alveolus regions. These areas have been postulated to form a ‘gutter zone’ into which soluble carcinogens may pool and exert their neoplastic influence. Figure 2.1 illustrates an invasive squamous cell carcinoma arising from the floor of mouth tissue. In contrast, squamous cancers are rare on the tongue dorsum and hard palate.
Figure 2.1 Squamous cell carcinoma arising from floor of mouth tissue.
There are probably underlying structural and functional distinctions that contribute to the increased risk of oral carcinogenesis at certain oral sites. It is thus important to have a clear understanding of both the applied anatomy of the oral cavity and the cellular biology of its lining oral mucous membrane.
Applied Anatomy of the Oral Cavity
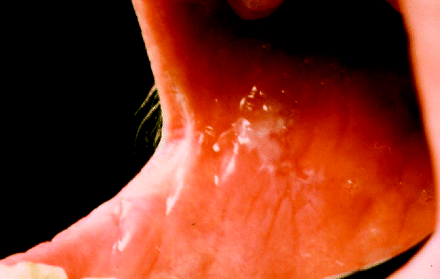
The border between the facial skin and the vermilion of the lip, the mucocutaneous junction, defines the entry to the oral cavity. The vermillion of the lip is continuous with the labial mucosa, which becomes the buccal mucosa beyond the commissure or corners of the mouth and extends backwards until it attaches to the alveolar ridges and pterygomandibular raphe. Whilst potentially malignant lesions may present not infrequently at labial commissure regions, as shown in Figure 2.2, invasive cancers affecting the buccal mucosa tend in general to arise more posteriorly.
Figure 2.2 Labial commissure leukoplakia extending to the buccal mucosa.
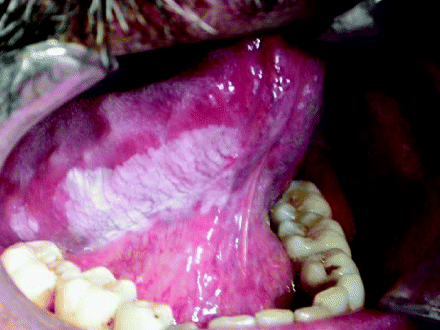
The posterior limits of the oral cavity are defined by the palatoglossal folds, the junction of the hard and soft palate and the line of circumvallated papillae that separates the anterior two-thirds of the tongue (oral or mobile part of the tongue) from the posterior third of the tongue (base of tongue). The anterior two-thirds of the tongue extends from the circumvallated papillae forward to its undersurface, where it then attaches to the floor of the mouth. The mobile tongue comprises a tip, lateral borders and both dorsal and ventral surfaces. Disease incidence varies considerably between tongue sites, with the dorsum and tongue tip rarely affected by squamous cell carcinoma, whilst the ventrolateral tongue is a particularly common site for both malignant and premalignant disease (Figure 2.3).
Figure 2.3 Leukoplakia presenting on the ventrolateral tongue. Note the extension of the lesion posterolaterally along the tongue surface.
The superior part of the oral cavity is formed anteriorly by the hard palate, which separates the oral cavity from the nose, and more posteriorly by the mobile soft palate, which extends backwards into the oropharynx.
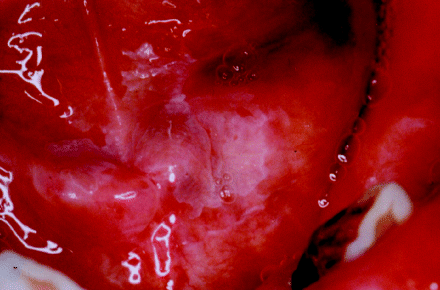
The inferior oral cavity is defined by the floor of the mouth, which is in continuity with the ventral surface of the oral tongue. Whilst the tongue occupies most of the floor, the sublingual region describes the tissues underneath the tongue tip and sides, and contains the submandibular salivary gland ducts which open anteriorly. The sublingual region is divided into left and right by the midline lingual fraenum. The vast majority of floor of mouth cancers and potentially malignant lesions arise anteriorly, as illustrated in Figures 2.1 and 2.4, respectively.
Figure 2.4 Anterior floor of the mouth showing the opening of the submandibular ducts. In this patient there is feint, homogeneous leukoplakia overlying the ducts and spreading towards the lingual mucosa.
More posteriorly sited within the floor of the mouth are the sublingual salivary gland and lingual nerve, which lie in the immediate submucosal tissues. Whilst fewer malignant lesions arise in the posterior floor of the mouth, these structures are often compromised either by local disease extension or during surgical lesion excision.
The alveolar process that bears the teeth is invested by alveolar mucosa that makes the transition to the gingiva, which forms a cuff around the necks of the teeth. Although the upper and lower alveolar ridges are anatomically distinct, both the structure and function of this mucosa and the incidence of disease are identical at both sites. Figure 2.5 shows an extensive erythroleukoplakic lesion arising from the posterior mandibular alveolus which has already transformed into an invasive carcinoma.
Figure 2.5 Erythroleukoplakia arising in the mandibular alveolar mucosa. This lesion has progressed to become an invasive squamous cell carcinoma.
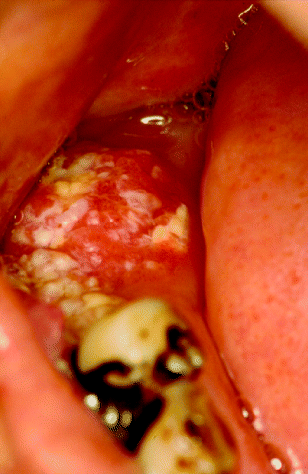
Immediately posterior to the oral cavity is the oropharynx, which comprises the palatine tonsils, the base of the tongue and the soft palate. The soft palate, the anterior and posterior faucial pillars (which surround the palatine tonsils) and the retromolar trigone (a triangular mucosal region covering the ascending ramus of mandible) together form a high-risk site for oral cancer development (Figure 2.6) [1–3].
Figure 2.6 Extensive, destructive squamous cell carcinoma arising in the soft palate and extending into the oropharynx.
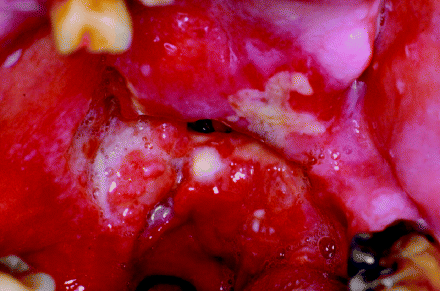
The oral mucosa lines the oral cavity and is covered by a thin film of saliva. It comprises a surface squamous epithelium with an underlying lamina propria. When the mucosa lies directly on bone it is termed mucoperiosteum, but elsewhere there is a submucosa which is composed of parenchymal fibro-adipose tissue and contains lobules of minor salivary glands and neurovascular bundles. Below the submucosa are the deep anatomical structures of the oro-facial complex, including the muscles of mastication, the major salivary glands and the maxillae.
The oral cavity is a complex environment that is adapted for the mastication and ingestion of food, taste sensation, speech and extrinsic antigen surveillance. The oral mucosa forms a physical barrier that protects the submucosal structures from the mechanical, thermal and chemical challenges associated with the consumption of food. It is also an important sensory organ that detects touch, proprioception, temperature, pain and taste sensations. The oral mucosa, along with saliva and the normal oral flora, provides protection against infection from pathogenic microorganisms. It is also an important portal for antigen sampling, priming the immune system for an appropriate and rapid response.
Microanatomy of the Oral Mucosa
The squamous epithelium is composed of keratinocytes that are organised into a multilayered, stratified structure. The epithelium has a basal cell layer, prickle cell layer, granular cell layer and keratinised or cornified layer. The basal keratinocytes are attached to the basement membrane; the epithelial–mesenchymal interface, which is continuous with the lamina propria. The basal cells are attached to the basement membrane by specialised structures called hemi-desmosomes. Desmosomes are the strong intercellular contacts between keratinocytes that preserve the integrity of the entire epithelium.
The basal cells proliferate and produce new cells that maintain the thickness of the epithelium as cells are lost by desquamation at the surface. The basal cells include stem cells and transit-amplifying cells that are organised into units. The stem cell divides to produce another stem cell and a transit-amplifying cell. The transit-amplifying cell then undergoes several rounds of mitosis, producing the large numbers of daughter cells required to populate the epithelium. The stem cell divides infrequently, but the transit-amplifying cells have a shorter cell cycle and divide more frequently. Cell cycle kinetics maintain the oral epithelium in equilibrium and enable epithelial regeneration following injury. The turnover time for oral epithelium is longer than that of gastrointestinal epithelium (4–14 days), but shorter than that for the epidermis (52–75 days). The turnover time for buccal epithelium is around 25 days and for gingiva 50 days [3].
Following rounds of cell division, the transit-amplifying cells commit to terminal differentiation. The cells loose the capacity to divide and start to accumulate cytoplasmic intermediate filaments called cytokeratins (CKs). There are numerous CK molecules that are classified into two groups: basic high molecular weight and acidic low molecular weight CKs. The CKs form pairs and have distinct spatial expression within the epithelial layers; CK5 and CK14 are expressed in the basal cells, CK4 and CK13 are expressed in non-keratinised squamous epithelium, and CK1 and CK10 are expressed by keratinised epithelium [4].
These differentiating cells form the prickle cell layer, which refers to the appearance of the ‘spiky’ profile of the keratinocytes in formalin-fixed, paraffin-embedded tissue sections stained with haematoxylin and eosin (H&E). The spikes or spines represent the desmosomal intercellular attachments. In addition to cytokeratins, several other structural proteins accumulate, for example loricrin, filaggrin and involucrin [4].
In non-keratinised squamous epithelium the surface layers are composed of polygonal keratinocytes. By contrast, in parakeratinised epithelium the keratinocytes become flattened and the nucleus becomes pyknotic (shrunken) and hyperchromatic (darker). Terminally differentiated squamous cells are eventually lost by desquamation at the surface. Orthokeratinisation is characterised by a granular cell layer between the prickle cell layer and the surface squames. The granular cell layer is replete with coarse keratohyaline bodies, which produce the typical granular appearance in H&E-stained tissue sections. The surface squamous cells, just a few cells thick, do not contain any discernable nuclear material and are termed anucleate squames.
Whilst most of the epithelium is composed of keratinocytes, there are also melanocytes (around one per ten basal keratinocytes), scattered Langerhans’ cells and neurosensory cells, Merkel cells and taste bud structures. Melanocytes produce melanin in varying amounts, accounting for racial pigmentation within the oral cavity, whilst Langerhans’ cells are antigen-presenting cells and form part of the mucosal immune defence system [5].
The lamina propria is the connective tissue or mesenchyme that lies immediately below the epithelium. It is divided into the superficial papillary layer and the deeper reticular layer. The papillary lamina propria interdigitates with the rete processes of the epithelium. The lamina propria is composed of fibrous tissue that is ramified by a rich neurovascular supply. The fibrous tissue contains fibroblasts, myofibroblasts and fibrocytes that elaborate collagen and elastin fibres along with other glycoproteins and proteoglycans that form the ground substance of the lamina propria [5].
Collagen is the most abundant constituent of the lamina propria, forming around 60% of the overall structure, and imparts mechanical strength. Together with the other proteins, a structure is produced which is well adapted for the compression and stretching that occurs during mastication, swallowing and speech. The lamina propria also contains transiting immune Langerhans’ cells, macrophages, mast cells and B and T lymphocytes which, together with the epithelial component, completes the mucosal immune defence system.
Interactions between the epithelium and the lamina propria are essential for homeostatic maintenance of the mucosa. Such interactions are essential for epithelial cell proliferation and differentiation, and are also involved in extracellular matrix remodelling. This mucosal maintenance programme is mediated by families of cell adhesion molecules, for example the cadherins (epithelia–epithelial cell adhesion molecules) and the integrins (cell–extracellular matrix adhesion molecules), along with the influence of multiple growth factors (e.g. epidermal growth factor, platelet-derived growth factor, transforming growth factor, keratinocyte growth factor) and cytokines (interleukins) [4].
Deregulation of the complex interactions between epithelium and underlying connective tissue inevitably has a part to play in the development of oral precancer and the pathogenesis of oral squamous cell carcinoma [3].
Regional Variation of the Oral Mucosa
The clinical appearance of the oral mucosa is dependent on the thickness of the epithelium, the amount of surface keratinisation, melanin pigmentation and the vascularity of the lamina propria. There are three prototypic mucosal patterns: lining mucosa, masticatory mucosa and the specialised mucosa that covers the dorsum of the tongue. These mucosal differences are summarised in Table 2.1 [3–5].
Table 2.1 Regional variation of the oral mucosa.

Lining Mucosa
The vermillion border of the lip is covered by a thin, stratified s/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


