Periodontal Conditions
Bengt Sjödin and Dorte Haubek
Inflammatory reactions in the periodontal tissues are common in children and adolescents. In most cases, the inflammation is restricted to the gingival tissues. Gingivitis is characterized by the presence of gingival inflammation without detectable loss of bone or connective tissue attachment. Periodontitis in children and adolescents may, as in adults, have varying manifestations, and the present nomenclature distinguishes between aggressive (i.e., severe and rapid disease progression) and chronic (i.e., slowly developing disease, usually with low to moderate progression) forms of the disease.
As in adults, gingivitis and periodontitis in children and adolescents are primarily caused by an accumulation of microbial deposits on the teeth and in the gingival crevices, and especially in the case of gingivitis, the state of the gingiva mirrors the oral hygiene level of the individual. However, severe forms of inflammation may also signal the presence of a general disorder, especially if the gingival inflammatory reaction is out of proportion to the amount of plaque present [1].
Early treatment of periodontal diseases is usually very effective and early recognition increases the likelihood of preventing loss of tooth support or limiting the progression of the disease. Thus, good reasons exist to pay attention to inflammatory reactions in the periodontal tissues of young individuals [2]. In addition, there are other conditions, such as gingival recessions and gingival enlargements, which need the pediatric dentist’s attention.
Normal periodontal conditions
Primary dentition
The marginal edge of the primary tooth gingiva has a bulky and rounded appearance (Figure 14.1). The typical stippling of healthy gingiva develops slowly from the age of 2 or 3 years. In areas with diastema between primary teeth, the interdental tissues are comparable to saddle areas. When the molars have established proximal contacts the interproximal area is completely filled by an interdental papilla with a marginal concavity, a col, corresponding to the contact area [3].
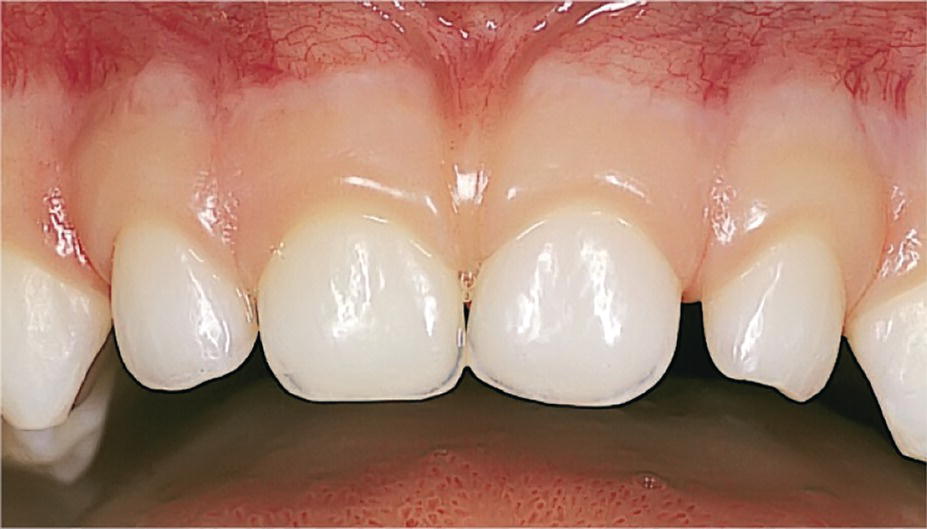
Figure 14.1 Clinically healthy primary tooth gingiva.
The connective tissue has a similar composition to that around young permanent teeth. However, compared to permanent teeth the primary teeth are associated with a thicker junctional epithelium, which may influence the permeability of the epithelial structures by, e.g., bacterial toxins. The junctional epithelium of the primary dentition is less permeable and thus more resistant to inflammation [4].
On radiographs, the alveolar bone surrounding the primary teeth has a distinct, but thin lamina dura and a comparatively wide periodontal membrane. Few trabeculae and large marrow spaces with rich vascularization are seen. The root cementum is thin and mainly cellular [4].
Permanent dentition
The exfoliation of primary teeth and the eruption of permanent teeth entail considerable morphologic and histologic changes, as described in Chapter 5.
Compared to the primary tooth gingiva, the healthy marginal gingiva around permanent teeth is thin and characterized by a pink color (Figure 14.2). After the tooth is fully erupted, the gingival margin is located on the enamel surface approximately 0.5–2 mm coronal to the cemento–enamel junction. During the period of passive eruption, i.e., the period of slow withdrawal of the marginal soft tissue, the length of the junctional epithelium is considerable in children. Although a periodontal probe is easily inserted deep along the tooth surface, there is no justification for unnecessary explorations interfering with the junctional epithelium.
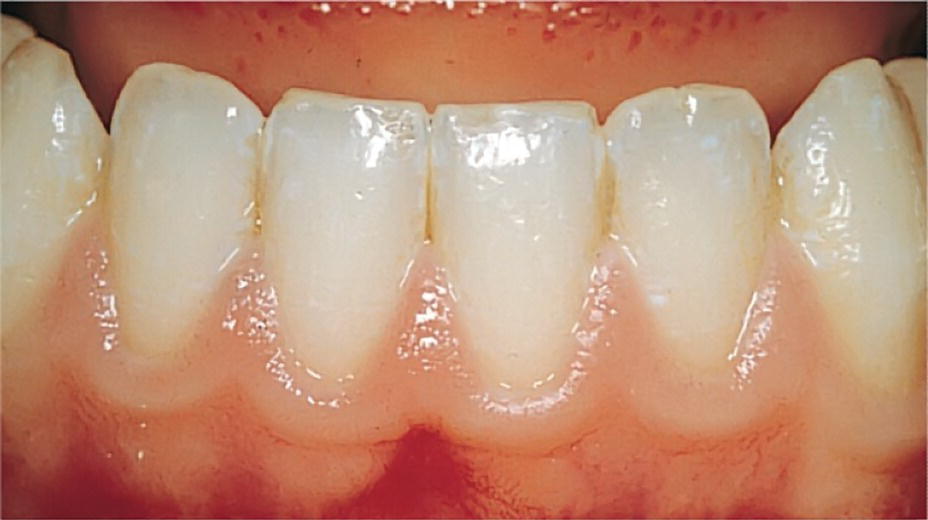
Figure 14.2 Clinically healthy permanent tooth gingiva.
Bacteria‐induced inflammatory periodontal diseases
One of the major problems in understanding the pathogenesis of periodontal diseases is the difficulty in distinguishing clearly between normal and pathologic conditions. When the gingival tissue is kept free from plaque, leukocytes will still be found migrating through the junctional epithelium towards the gingival sulcus. A few inflammatory cells may also be present in the connective tissue [4].
The balance between irritation and the individual phagocytic capacity and immunologic competence will be decisive for the severity of the disease. If plaque accumulation is minimal and the defense mechanisms are reacting normally, there will be no clinical symptoms. More pronounced plaque accumulations or defects in the defense reactions result in clinical symptoms [4].
Gingivitis
Clinical picture
The inflammatory reaction includes a vascular response and an accumulation of inflammatory cells. When the vascular response has reached a certain level, clinically noticeable signs of inflammation will occur. The marginal gingiva becomes reddish with a swollen appearance and papillae protruding from the interproximal spaces (Figure 14.3). The volume is increased, and the surface is shiny. Crevicular exudation is clinically visible, especially when light pressure is applied to the free gingiva. There is also an increased tendency towards gingival bleeding on probing.
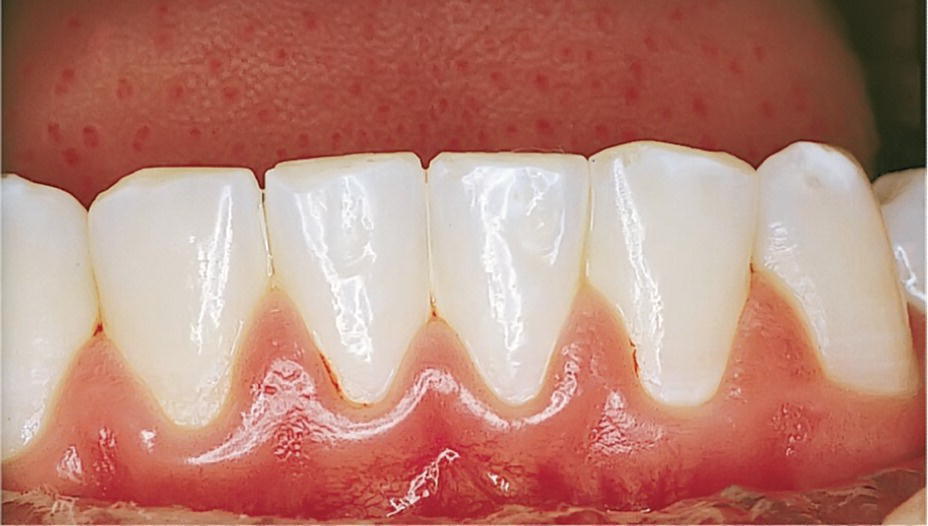
Figure 14.3 Chronic gingivitis.
The vascular and cellular reactions in the marginal gingiva should primarily be regarded as a natural defense against microorganisms. Since the causative factor is plaque accumulation, an efficient oral hygiene regimen usually will eliminate the clinical symptoms rapidly [3]. However, a new period of poor oral hygiene will result in a recurrence. Subclinical reactions and episodes of clinical gingivitis may alternate over long periods.
The diagnosis of gingivitis is based on the clinical symptoms visible to the eye, such as redness, swelling and bleeding tendency. Today, the tendency is to simplify the diagnostic criteria using gingival bleeding tendency as a measure of the gingival inflammatory condition. The gingival bleeding index (GBI) is based on one simple criterion: whether or not the marginal gingiva bleeds on gentle probing. The index is calculated as the percentage of bleeding gingival units of the total number of units examined [4].
In healthy children, gingival affection usually remains superficial, and when a child shows severe generalized longstanding gingivitis, the dentist should perform a detailed clinical examination based on an updated extensive anamnesis and may consider having the child’s general health investigated.
Age‐related differences
Epidemiologic as well as experimental studies have revealed an age‐dependent difference in the tendency to develop gingivitis during childhood. Thus, preschool children tend to be less susceptible to gingivitis than adolescents and adults [2,3]. The reasons for this difference are not fully understood, but it has been shown that, e.g., spirochetes and black‐pigmented Bacteroides, although frequently found in adults, are not regularly present in the microbial plaque of children with normal gingiva [5]. In addition, the microbial plaque of children with gingivitis has lower proportions of Fusobacterium, Eubacterium, and Lactobacillus species [5]. The increased cell proliferation and turnover of collagen, compared with adults, may also be significant. The cellular infiltrate of established gingival lesions in children is dominated by T‐lymphocytes and the adult lesions by B‐lymphocytes indicating age‐related differences in the immunologic response. The thicker junctional epithelium in the gingiva of the primary dentition, a factor that could influence the permeability of the epithelial structures, may also be important for the age‐related differences in the development of gingivitis.
Etiology
Unanimous agreement exits that gingivitis is caused by microbial plaque [5]. A large number of bacteria exist as a part of the normal ecology of the mouth. Many of these have virulence potential, but gingivitis is characterized as non‐specific in terms of predominance of any particular pathogen. Most studies of the role of microorganisms in the pathogenesis of gingivitis have concluded that the quantity of bacteria and bacterial products that accumulates locally is of prime importance in the pathogenesis of gingival inflammation. Nevertheless, gingivitis must be regarded as a multifactoral disease and a number of intrinsic as well as extrinsic factors influence the severity of its manifestation [6,7].
Factors influencing plaque formation
Calculus is formed by mineralization of microbial plaque. Supragingival calculus is predominantly found adjacent to the ducts of the major salivary glands (Figure 14.4). Subgingival calculus is particularly accumulating in areas with deepened periodontal pockets (Figures 14.5 and 14.6). The surface of the calcified deposits is rough and thus enhances bacterial colonization. Calculus is therefore deleterious to periodontal health [4].
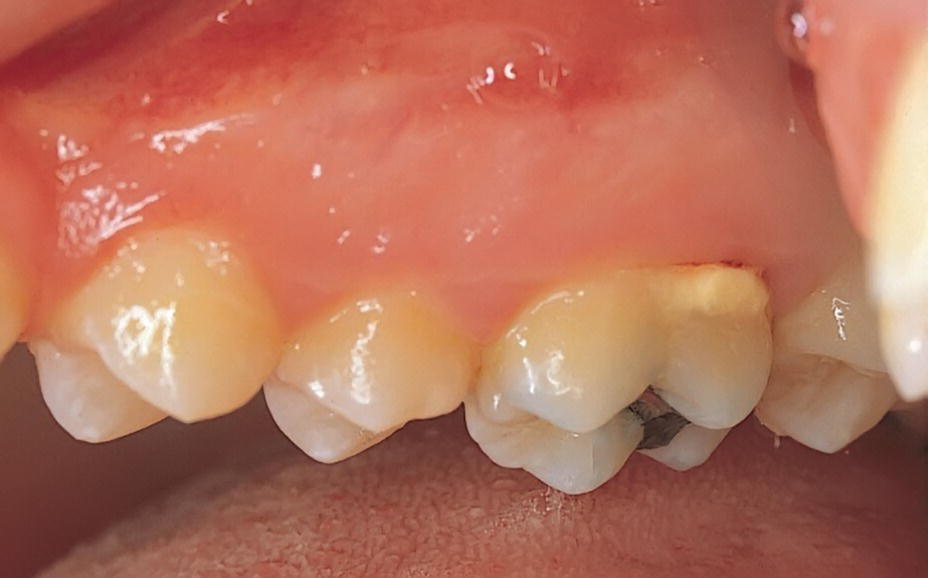
Figure 14.4 Supragingival calculus.
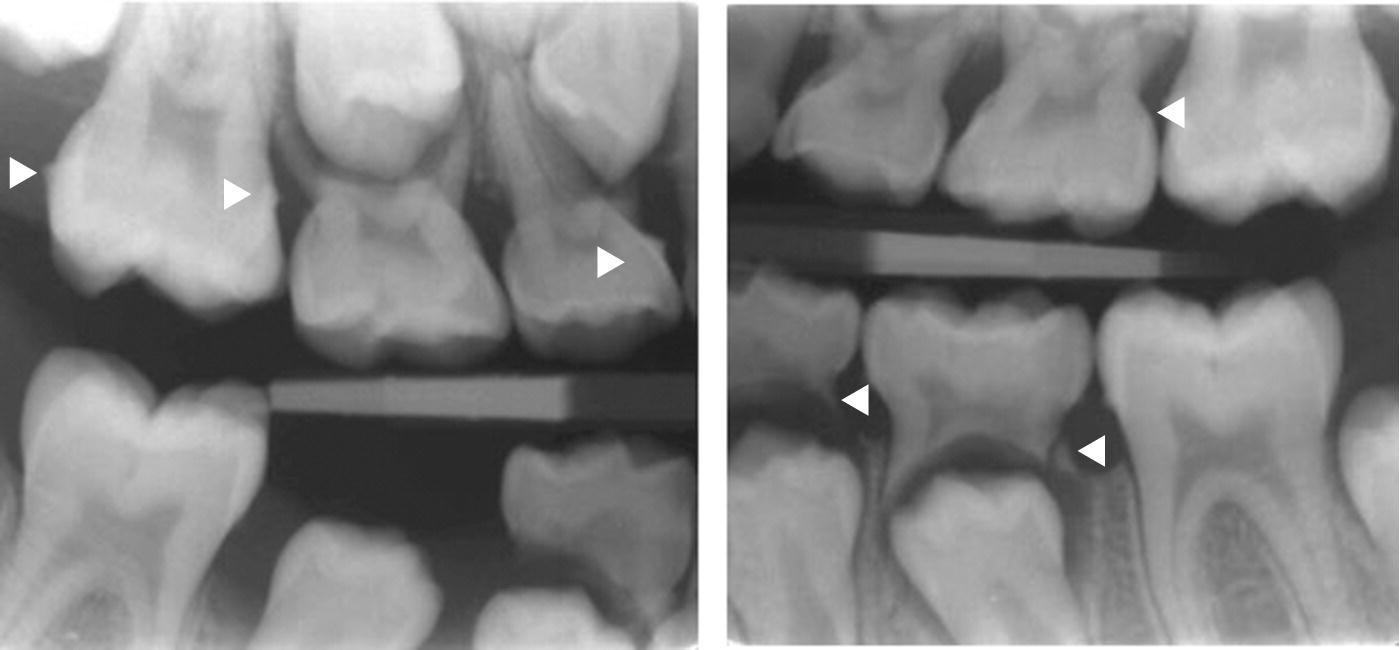
Figure 14.5 Bitewing radiographs showing proximal calculus on primary and permanent teeth. Arrow heads indicate subgingival calculus.
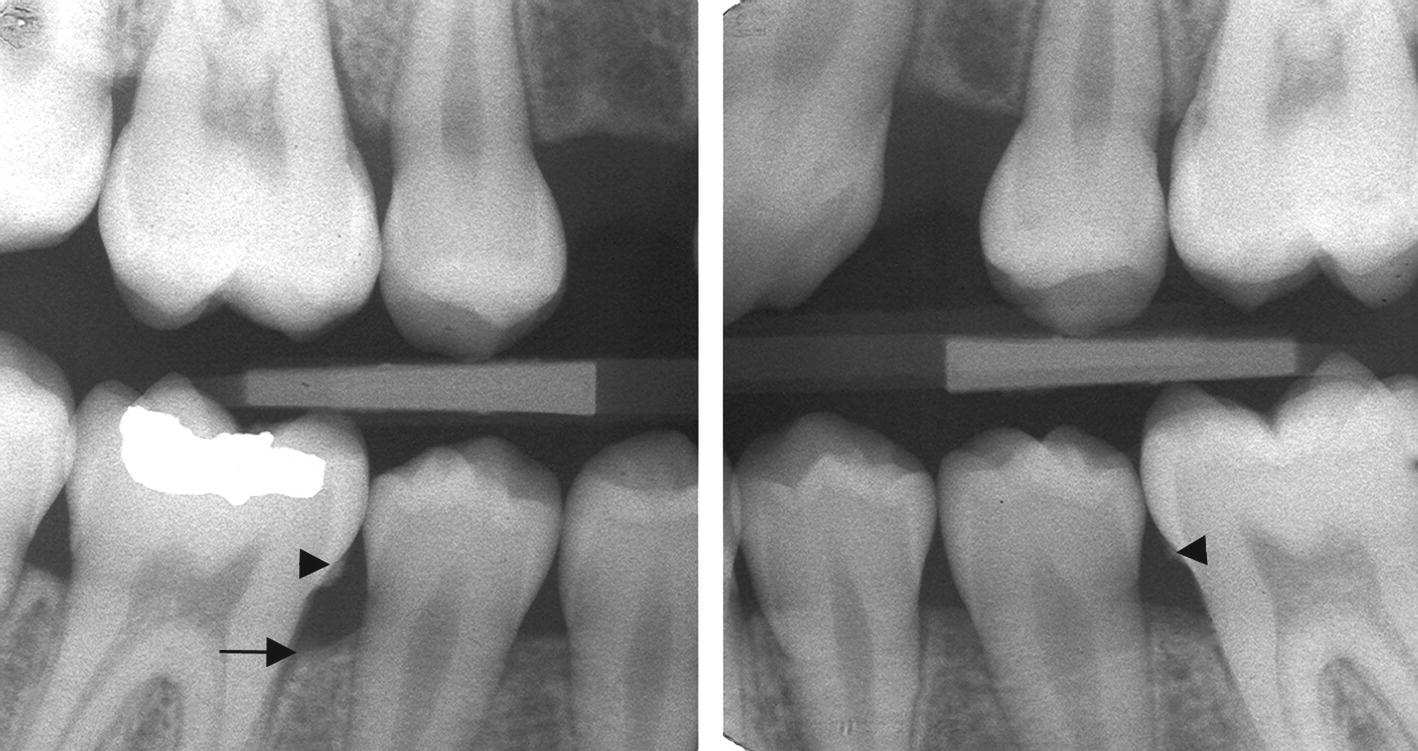
Figure 14.6 Radiographs of a 14‐year‐old boy showing proximal subgingival calculus and minor bone loss at mandibular permanent first molars. Arrow indicates bone loss and arrow heads indicate subgingival calculus.
In children, occasionally an extrinsic stain, a firmly attached black deposit on teeth, can be seen on primary and sometimes on permanent teeth [8]. It is usually a thin, darkly pigmented line located at the cervical part of tooth enamel. The etiology is unclear, but there seems to be an association with the composition of the microflora. Apart from an esthetic problem, no impairment of dental health has been reported (Figure 14.7).
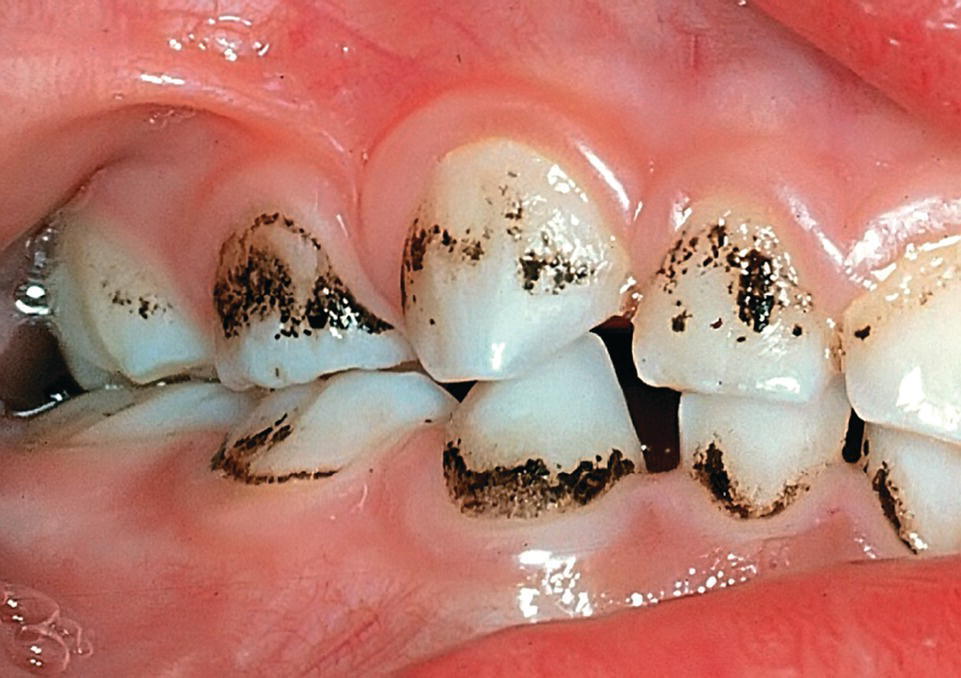
Figure 14.7 Black stains can be observed in children.
Disturbances of the enamel mineralization or deviation of the tooth morphology may cause an irregular and/or rough surface, accumulating plaque. For example, the early stages of eruption of hypomineralized teeth may be accompanied by pronounced gingivitis, which disappears later if the cervical part of the tooth has unaffected enamel (see Chapter 20).
Manifest carious lesions increase plaque accumulation and gradually impair oral hygiene. Cervical carious lesions are almost without exception accompanied by local, chronic gingivitis.
Restorations with defective margins, rough surfaces or faulty contacts will all cause chronic gingivitis due to an increased plaque accumulation. The dentist who inserts the first proximal or cervical restoration has a great responsibility for the patient’s future periodontal health.
Malocclusions do not play a dominant role in the etiology of periodontal disease, but crowding of teeth and dental anomalies may render oral hygiene measures difficult (Figure 14.8).
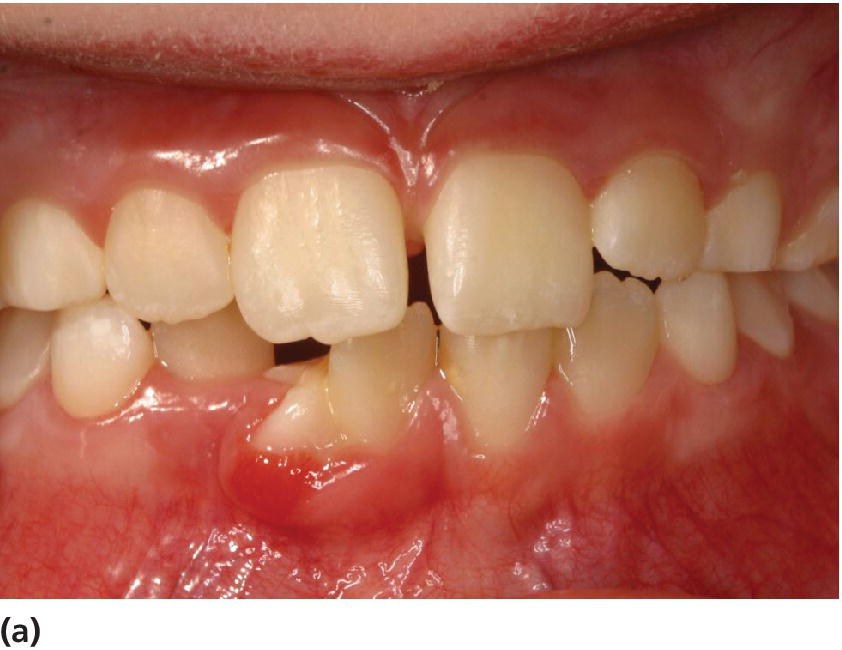
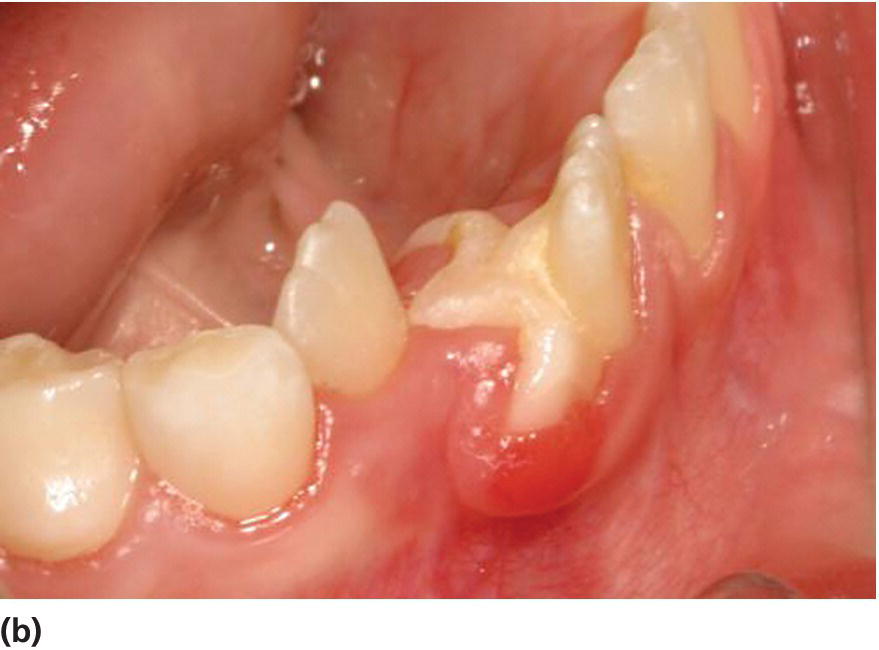
Figure 14.8 Gingivitis in relation to dens geminatus (tooth 41) in the incisal region of the lower jaw. (a) Frontal view of tooth 41, (b) Distofacial view of tooth 41.
Fixed orthodontic appliances may impair oral hygiene procedures; bands and brackets accumulate plaque (Figure 14.9) and removable plates can cause stomatitis (see Chapter 15). Any possible harm to the supporting tissues caused by the appliances must be adequately treated and controlled.
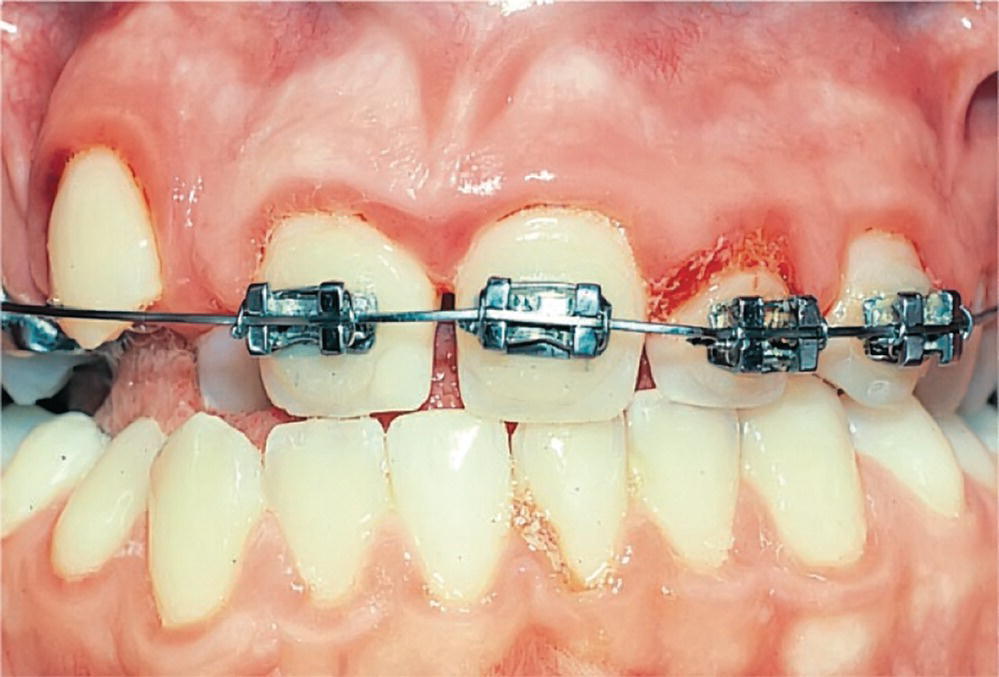
Figure 14.9 Poor oral hygiene and gingivitis in a patient undergoing orthodontic treatment.
Factors modifying the defense system
- Mouth breathing. Clinical observations and epidemiologic studies indicate an association between mouth breathing or deficient lip closure and chronic gingivitis (Figure 14.10). Mouth breathing may cause frequent drying out of the gingiva in anterior areas. It has been suggested that this may result in vasoconstriction and decreased host resistance [9].
- Hormonal changes. It is an established fact that hormonal changes contribute to the increased susceptibility to gingival affections during pregnancy. For example, “puberty gingivitis” has been described manifesting as pronounced edema in the marginal gingiva (Figure 14.11). Epidemiologic studies have shown that the incidence of gingivitis reaches a peak 2 or 3 years earlier in girls than in boys, approximately coinciding with puberty [7]. Findings of a correlation between the degree of gingivitis and parameters describing pubertal maturation further strengthen the theory of an influence of sex hormones on gingival status during puberty [2,7].
- Eruption gingivitis. This term is used to describe more intense forms of gingival inflammatory reactions around erupting permanent teeth. In areas of shedding primary teeth and erupting permanent teeth, a great risk of plaque accumulation exists. Tooth cleaning may be difficult or even unpleasant to perform leading to an inflammatory reaction. Furthermore, a gingival response is sometimes seen that is out of proportion to the degree of bacterial irritation, indicating that other factors modify the inflammatory response. It has been shown that during the phase of eruption, the epithelium displays degenerative changes at the site of fusion between dental and oral epithelia. This indicates a weak point in the epithelial barrier, and an enhanced permeability of the newly formed junctional epithelium may make the area especially vulnerable to bacterial accumulation. Another factor of importance is that once gingival inflammation has been established, the long dental epithelium of the erupting tooth may separate from the enamel creating a niche for pathogenic bacteria and a risk of deeper tissue involvement. Such an establishment of a subgingival plaque may explain why a gingival inflammatory reaction at an erupting tooth is often more difficult to cure than at a fully erupted tooth [3].
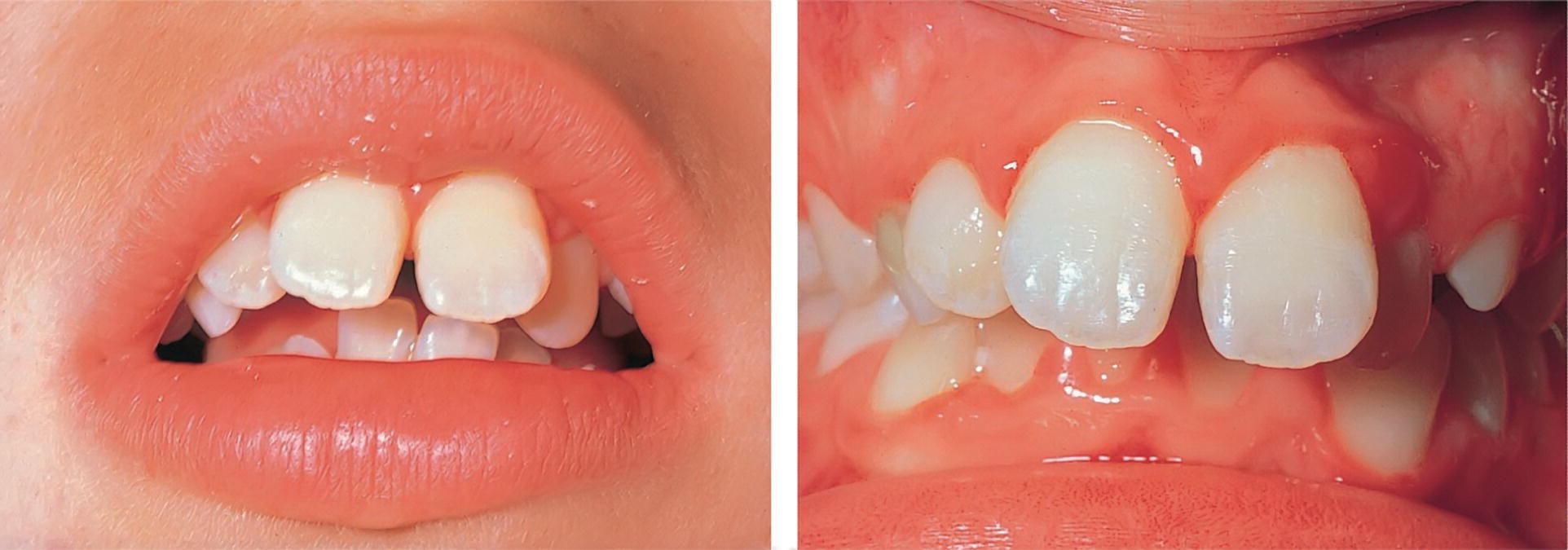
Figure 14.10 Chronic gingivitis associated with mouth breathing.
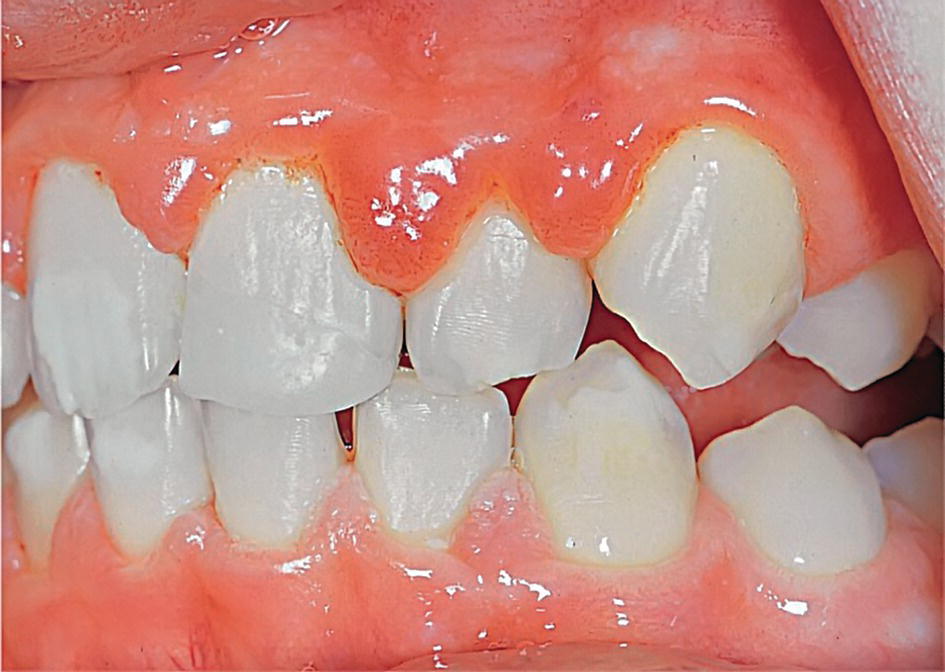
Figure 14.11 Edematous gingival inflammatory reaction during puberty.
Systemic diseases and syndromes
- Diabetes mellitus. Children with diabetes are more susceptible to periodontal diseases than healthy children. The tendency to develop chronic forms of gingivitis is most pronounced in children with poorly controlled diabetes. Consequently, children with diabetes should be given instruction and motivated early to maintain efficient plaque control [10].
- Leukemia. The most common form during childhood, acute lymphoblastic leukemia, is often accompanied by severe oral symptoms at the time of hospitalization and during the period of cytotoxic treatment. The low resistance of the tissues to infection is explained by drug interference with the replication of epithelial cells, in addition to a low number of circulating leukocytes. Therefore, plaque control is essential both before commencing cytotoxic treatment and during medical treatment [4].
- Agranulocytosis. This malignant type of neutropenia is rare in children, but as in cyclic neutropenia and chronic neutropenia, oral ulceration and periodontal manifestations are common. In chronic cases, the gingiva will become hyperplastic with granulomatous changes [4].
- Heart conditions. The severity of oral manifestations is directly proportional to the general cyanosis. The gingiva has a bluish‐red hue. As the lowered tissue respiration impairs the defense against microorganisms, children with peripheral cyanosis exhibit high gingivitis prevalence. Indications for antibiotic prophylaxis are given in Chapter 23.
Treatment
Gingivitis, involving marginal and papillary tissues, is reversible with plaque control and heals without any permanent damage of the gingiva. However, parents must bear the responsibility for plaque control in their preschool children. A simplified Bass brushing technique using a soft toothbrush is adequate in cases of the most prevalent cases of marginal gingivitis.
In case of more severe forms of gingivitis, professional tooth cleaning is usually necessary in order to secure the removal of subgingival plaque and calculus. This often needs to be performed under local anesthesia. The treatment may be supported by chemical plaque control during the initial phase. Treatment of gingivitis should also include education on the etiology of the disease [11].
Periodontitis
Clinical picture
Inflammatory periodontal disease includes both gingivitis and periodontitis. Not all patients with gingivitis will develop periodontitis. The latter term implies an ongoing inflammatory process involving deeper parts of the periodontium with loss of tooth support [4].
Periodontitis differs from gingivitis in the histologic appearance of the inflammation. In periodontitis, larger proportions of plasma cells and B‐lymphocytes are found compared to what is found in gingivitis.
Periodontitis is accompanied by few if any subjective symptoms, and the patient therefore has to rely on early diagnosis by professionals. The diagnosis is based primarily on the recording of probing pocket depth, attachment loss and/or loss of marginal bone assessed on radiographs. These methods, however, do not discriminate between current disease, previous episodes of disease and loss of periodontal support for other reasons, and therefore have to be supplemented with an evaluation of the inflammatory status. Signs indicative of ongoing disease are excessive bleeding on probing and suppuration. However, owing to the specific morphology of the gingiva around teeth under eruption or around newly erupted permanent teeth, insertion of a probe into the crevice should be avoided. In general, there is no point in carrying out systematic measurements of pocket depth or probing attachment level until the age of 12–14 years.
Classification and epidemiology
Classification
The first reports on periodontitis in children and adolescents described medically compromised individuals. Since the late 1970s, cases of otherwise healthy children and adolescents with periodontitis have been reported. Beyond the early onset, the seemingly rapid progression, a characteristic microflora, and frequently occurring localized periodontal lesions, it has been suggested that periodontitis in children and adolescents represents unique forms of disease entities differing from periodontal disease in adults. However, this consideration is still a matter of debate.
The terms prepubertal periodontitis, juvenile periodontitis, early onset periodontitis, early periodontitis, and incidental attachment loss have been used and are still used in the literature. According to the results of recent research, there has gradually been a change in the perspective, and today a common understanding is that the diagnosis, based mainly on the age of onset, cannot distinguish periodontal disease entities. In the classification system used today, the expressions “aggressive periodontitis” and “chronic periodontitis” replace the previously used terminology (Box 14.1). Both forms of disease have been subgrouped according to severity and distribution within the dentition as a localized or a generalized form [1]. Although the American Academy of Periodontology (1999) recommended that age should not be a criterion for diagnosis, this parameter is still to some extent used in the sub‐classification of aggressive periodontitis. The specific features of the localized form included circum‐pubertal onset, and the patients with the generalized form were described as usually being older, but under 30 years of age. However, the age of onset for the individual patient is obviously hard to determine since the diagnosis, using current methods of examination, is performed when the supportive tissue loss is present, possibly a long time after the actual onset of the disease. Except for age, other features are used when diagnosing periodontitis. For example, the number of teeth involved is used in the sub‐classification. The localized form mainly affects permanent first molars and incisors while the generalized form involves more or most permanent teeth. There are, however, reports of cases where the localized form progresses into a generalized form as there are cases of chronic or incipient periodontitis which progress to aggressive periodontitis [12,13].
A number of studies suggest a substantial number of cases of aggressive periodontitis to have onset in childhood and thereby affecting primary molars [14,15] (Figure 14.12). It seems like the patients included in these studies had alveolar bone loss already around primary molars, but early exfoliation of the involved primary teeth was not reported.
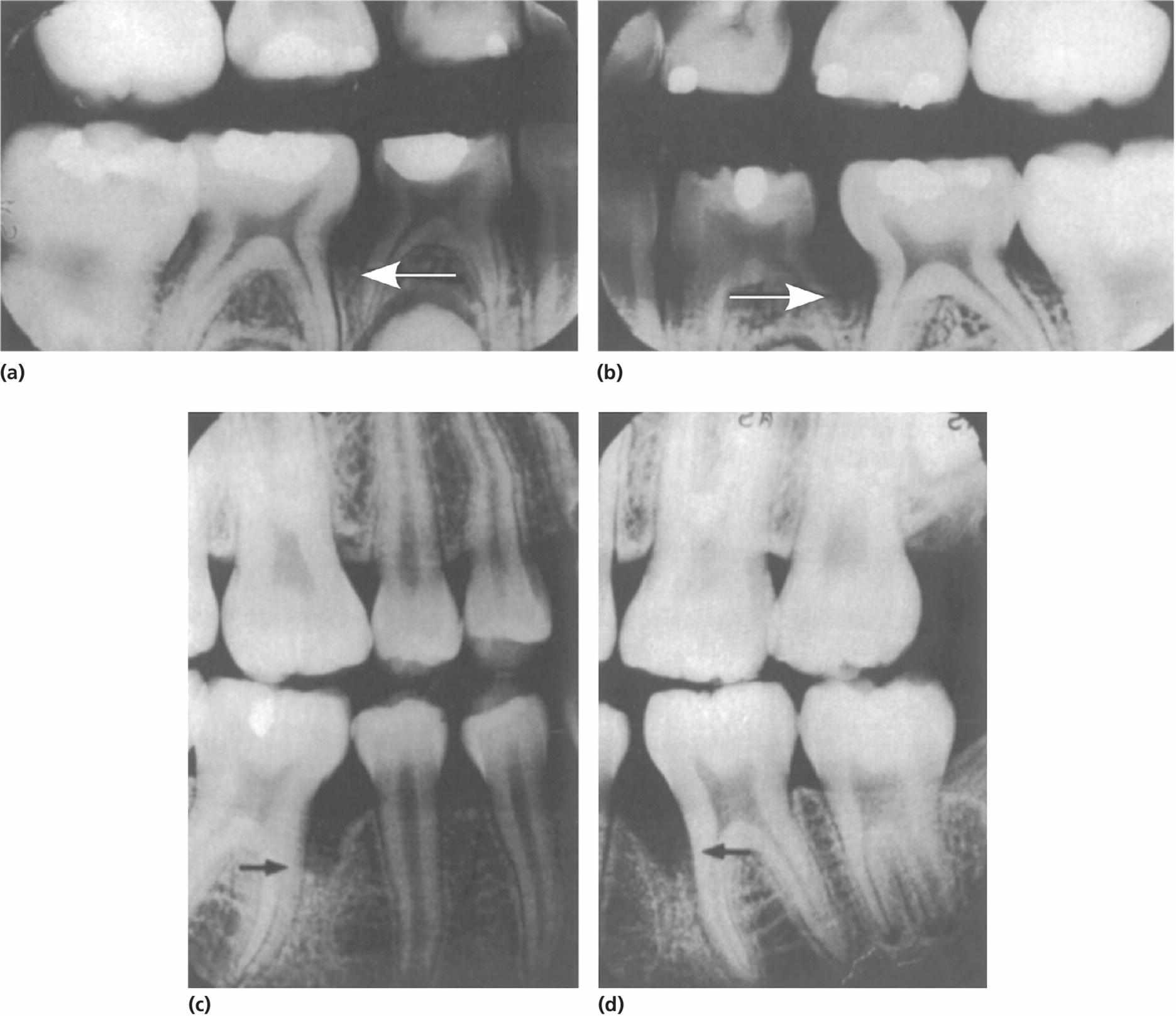
Figure 14.12 A 14‐year‐old boy with localized aggressive periodontitis (c, d). Previously obtained and filed radiographs from the age of 8 years show loss of bone support (a, b) (arrows)
The most common forms of periodontal disease in children and adolescents are single or a few sites with loss of attachment or generalized, but minor loss of attachment. The lesions are often localized at the first permanent molars, i.e., the same teeth that usually are affected in patients with localized aggressive periodontitis. Single lesions or lesions of minor extent, earlier often referred to as early periodontitis or incidental periodontitis, are today classified as chronic periodontitis. The patients usually display more plaque and calculus compared with individuals with aggressive periodontitis and also have higher gingivitis scores (Box 14.1).
Epidemiology
Whereas epidemiologic studies in young children are few, a large number of studies including teenagers have been performed. An extensive variation of the prevalence has been reported [16]. The variation may, of course, represent true differences between populations, but there is no doubt that disparate findings most likely are also due to the use of different modes of examination procedures, different criteria and methods of population sampling.
While early epidemiologic studies used measurements of clinical attachment loss (CAL) as diagnostic criteria, recent studies have used radiographic criteria. Usually, the distance between the cemento–enamel junction and the marginal bone level is measured. According to methodological studies, distances >2 mm should be regarded as a deviation from normality both in the primary and in the permanent dentition. In developed countries, the reported prevalence of radiographic bone loss is 2–13% both in children and in adolescents [17,18].
Most epidemiological studies do not distinguish between individuals with minor loss of periodontal attachment (chronic periodontitis) and individuals with severe loss (aggressive periodontitis). Most cases found in epidemiologic studies are probably individuals with minor lesions. However, longitudinal studies of adolescents have shown that individuals with minor or few lesions more frequently develop attachment at additional sites or more severe attachment loss at the original site (aggressive periodontitis) compared to adolescents with no signs of attachment loss [12,19–21].
Chronic periodontitis
Chronic periodontitis is most prevalent in adults, but may also occur in young individuals. This form of periodontitis is characterized by minor loss of periodontal support and a slow progression rate (Figure 14.6). The patients often show considerable plaque accumulation, and subgingival calculus is a frequent finding [17].
In young children, the loss of periodontal support usually manifests as single lesions at primary molars. The deepening of periodontal pockets around affected primary teeth is limited. Scandinavian studies report that 2–4% of 7‐ to 9‐year‐old children display solitary sites of radiographic bone loss in the primary dentition [22]. Most of these sites could be characterized as incidental attachment loss, associated with various types of local trauma or with factors related to the development of the dentition. This type of defect may also represent an earlier inflammatory process, which has healed. However, more importantly, it may represent an initial stage of progressive periodontal disease [14,23]. Patients with clinical or radiographic bone loss should be regarded as being at risk of developing early periodontitis.
In developed countries, most epidemiologic studies on loss of periodontal support in adolescents report frequencies of less than 5%. As in younger children, most affected individuals show solitary sites [24]. However, the number of sites per individual as well as the amount of attachment loss seems to increase with age. Usually, the first permanent molars are affected. Subgingival calculus is a frequent finding in these patients (Figure 14.6) [18].
Aggressive periodontitis
The prevalence estimates of aggressive periodontitis differ between studies, but the use of various criteria may account for some of the differences. Therefore, one should be cautious when comparing the reported prevalence figures. Most studies indicate a prevalence of less than 0.5% in adolescent populations, although some populations show a considerably higher prevalence [18]. A majority of the reported cases show localized lesions, i.e., localized aggressive periodontitis. The patients in general display moderate signs of gingival inflammation, except in diseased areas. In common, patients often display less plaque and subgingival calculus compared to individuals with chronic periodontitis. The diagnosis of generalized aggressive periodontitis is used when the patients exhibit widespread bone loss, but the level of attachment loss and number of teeth involved used as criteria in the epidemiologic studies differ [18]. In both types of disease, the periodontal pockets often, but not always, harbor subgingival calculus.
In young children, the generalized form of periodontitis is often associated with systemic diseases, although otherwise healthy cases are reported (Figure 14.13). The periodontal destruction often starts early after eruption and is usually characterized by severe gingival inflammation and may lead to premature loss of teeth. Patients should be referred to a pediatrician for medical examination [25].
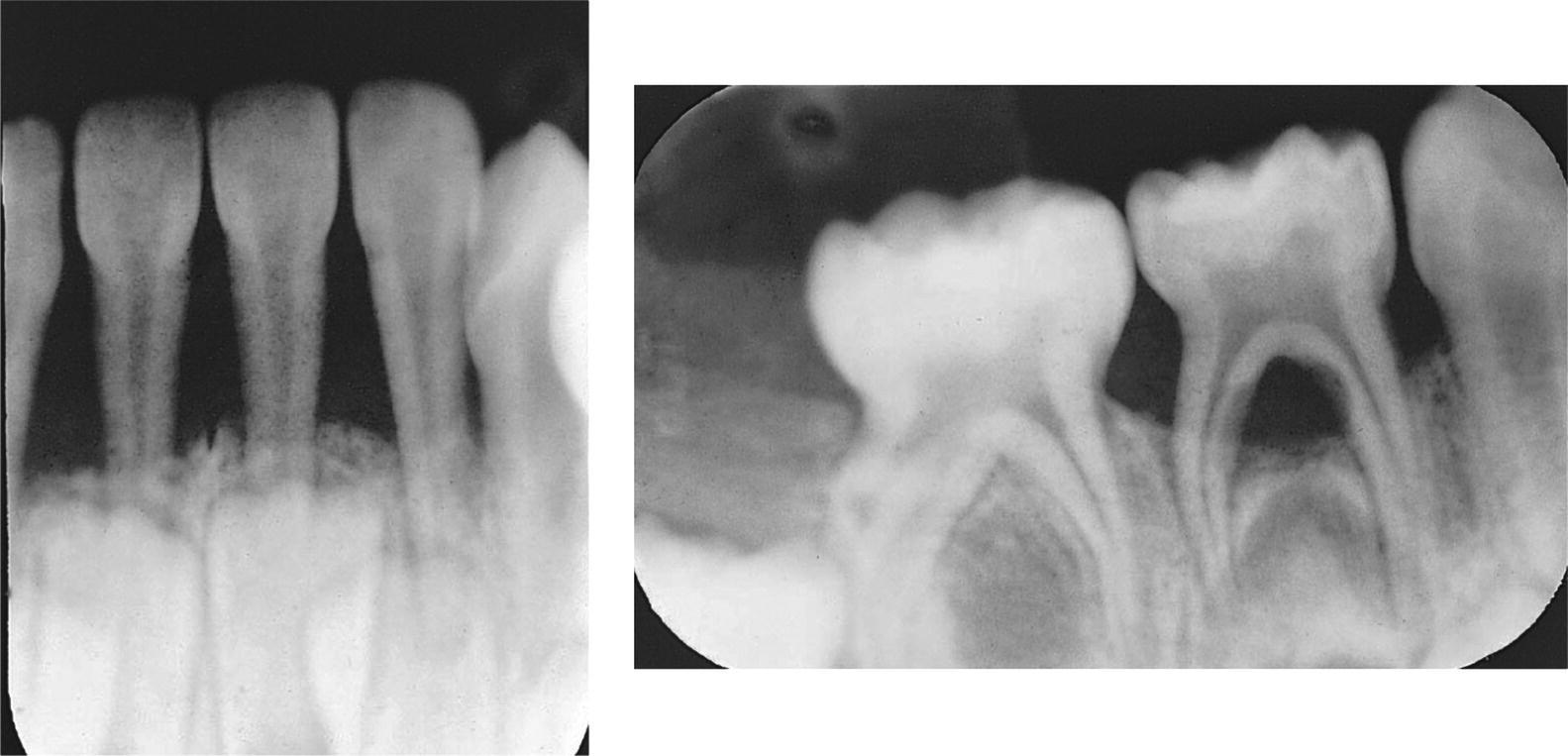
Figure 14.13 A 3‐year‐old boy with a generalized form of aggressive periodontitis. The primary teeth in all quadrants are involved.
Localized aggressive periodontitis in adolescents is a rapidly progressing disease with onset in the early permanent dentition. It is defined as mostly having a circum‐pubertal onset and usually involves permanent first molars and incisors (Figure 14.14). The inflammatory signs are often restricted to the parts of the periodontium showing attachment loss. Patients often, but not always, show smaller amount of plaque and calculus compared to patients with chronic periodontitis. Radiographs often disclose a vertical or arch‐shaped pattern of bone loss at the molars, but horizontal bone loss at incisors. The affected sites will generally bleed on probing.
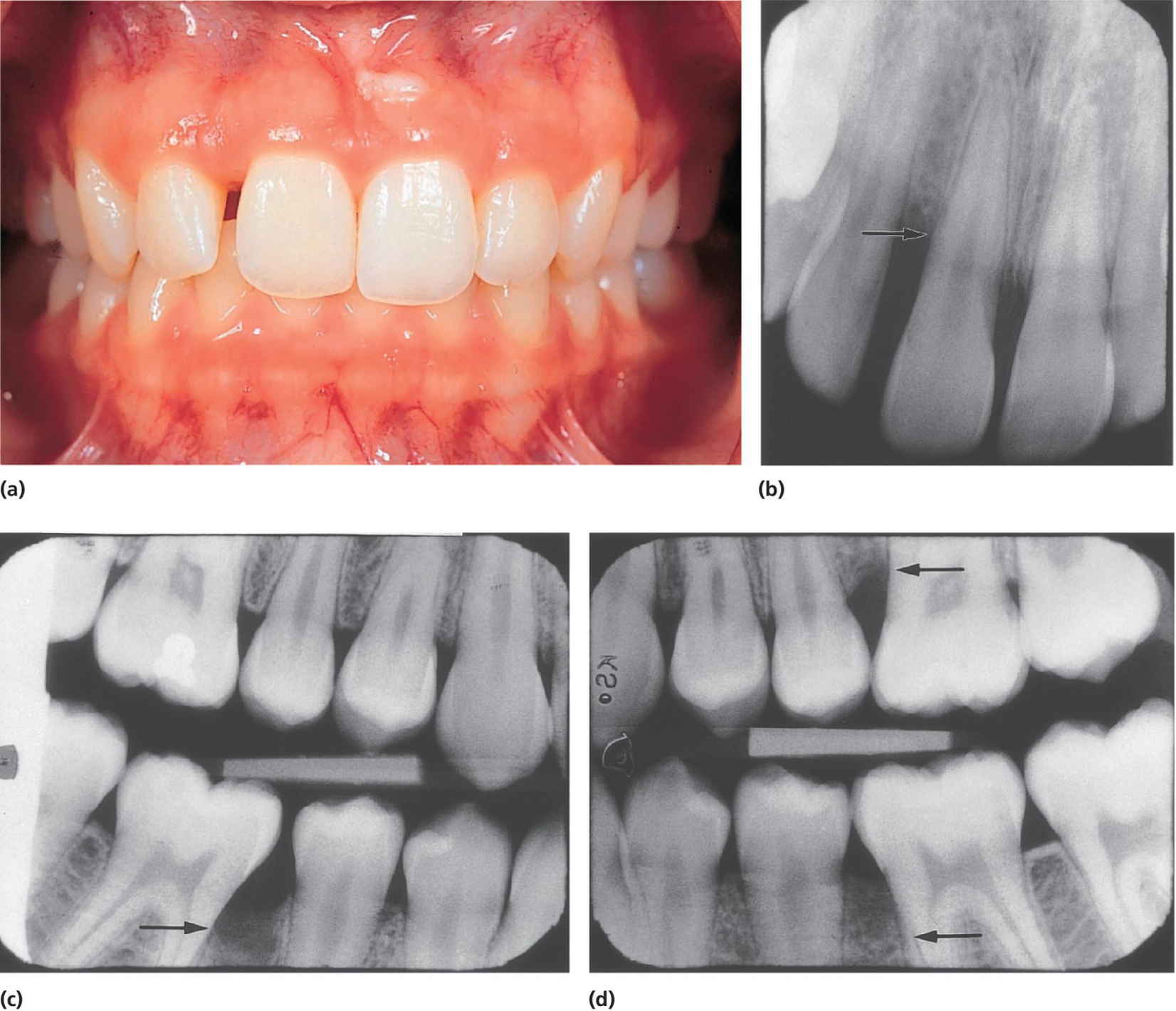
Figure 14.14 (a) A 13‐year‐old girl with a localized form of aggressive periodontitis, clinically identified with a diastema between permanent maxillary incisors. (b, c and d) The radiographs show bone destruction in the same area as well as in the permanent molar regions (arrows).
The diagnosis of generalized aggressive periodontitis is used when the patients exhibit widespread bone loss, including at least three teeth that are not first molars and incisors. This form of disease is usually accompanied by plaque, calculus, and severe inflammation.
Etiology and risk factors
General factors
Periodontitis is an infectious disease of the tooth‐supporting tissues. If the gingival tissues are exposed to microbial plaque over a long period of time, deeper parts of the periodontium may be involved and slowly destroyed by the action of the inflammatory process. If left untreated, the teeth may lose their ligamentous support. Most children and adolescents show varying degrees of gingivitis. Why some individuals do and others do not develop destructive periodontitis is not fully understood.
Although bacterial exposure is a prerequisite for the occurrence of gingivitis and periodontitis, its presence alone explains only a limited proportion of the variance in the disease expression. The wide variation of the expression of the destructive forms, e.g., progression rate, number of teeth involved and clinical appearance, implies that it can be considered a multifactorial disease.
Most studies on possible etiological factors in young individuals have focused on aggressive periodontitis cases. Research has mainly addressed various infections, variation in host response, the impact of genetic factors and person‐related factors as, e.g., ethnicity.
Microbiology
A large number of different microorganisms are found in the oral cavity [26]. An increasing body of evidence suggests that only a subgroup of these microbes may be responsible for the tissue destruction. The mere occurrence of the so‐called periodontal pathogens does not explain all aspects of disease development. The initiation of periodontitis may be a consequence of a change of the micro‐environment when the proportions of virulent microorganisms increase [27,28]. However, particular attention has been paid to the role of Aggregatibacter actinomycetemcomitans in the pathogenesis of aggressive periodontitis. This bacterium is a short facultative anerobic rod, which possesses several virulence factors. Of specific interest is the capacity to produce a leukotoxin with the ability to harm and kill human leukocytes [29,30]. Other virulence factors include cytolethal‐distending toxin which reduces the content of collagen in the tissues. It seems like A. actinomycetemcomitans also has the capacity to invade the periodontal tissues. A majority of young individuals with aggressive periodontitis harbor this species, but the mere presence of this microorganism does not necessarily indicate ongoing disease since many healthy individuals also carry this microorganism. There seems to be a variation of virulence between different clones. One clone of A. actinomycetemcomitans (JP2) is often found in adolescents of North or West African descent [30]. There is strong evidence that this highly leukotoxic clone is causally involved in the etiology and pathogenesis of localized aggressive periodontitis [30,31].
The composition of the subgingival microbiota in patients with aggressive periodontitis and chronic periodontitis in young individuals differ significantly. A. actinomycetemcomitans and other periodontal pathogens, such as Porphyromonas gingivalis, Prevotella intermedia and a number of other microorganisms, are found in dental plaque collected from young individuals with aggressive periodontitis or chronic periodontitis.
In most periodontitis cases, it seems reasonable to assume that the disease is a result of a polyinfection by different microorganisms and the interaction of this infection with the host response [32].
Host‐defense factors
The host‐defense system comprises a great number of cells and molecules whose role is to protect the body against infection. The function of polymorphonuclear leukocytes (PMNs) is an important part of the innate host‐defense system against bacterial infection. Since patients with defect function of the PMNs, for instance individuals with leucocyte adhesion deficiency (LAD), show severe periodontitis a lot of attention has been paid to the function of the PMNs. The PMNs have the ability to migrate to extravascular sites, adhere, function as phagocytes and kill microorganisms. Early findings suggested decreased chemotaxis of the PMNs in young individuals with aggressive periodontitis, but later studies have not been able to verify these results [32]. Although patients with systemic diseases, such as LAD, Papillion Lefèvre and other diseases, show simultaneous PMN cell deficiencies and severe periodontitis, this does not seem to apply to the “ordinary” young patients with aggressive periodontitis. An extensive amount of literature on the innate immune system has been published in recent years and a number of different factors are suggested to be of importance for the development of aggressive periodontitis.
The role of the specific immune responses in the pathogenesis of aggressive periodontitis has been studied over the past decades. In both chronic and aggressive periodontitis the cytokine profiles are similar, but an increased number of T‐cells and a decreased number of macrophages in the lesions of aggressive periodontitis have been found [32]. Even if there are findings showing a significant elevation in serum immunoglobulin G levels to particularly A. actinomycetemcomitans as well as to other periodontal pathogens, the relationship between the antibody level and periodontal disease is very complex, as is the relationship between the disease and the humoral immune response. The progression from gingivitis to periodontal disease is accompanied by a switch in the cell population of the lesion from T‐cells to B‐cells. There has been a debate on a similar shift from humoral to cellular immunity, possibly explaining a progression of the disease in addition to the involvement of the susceptibility of the patients. In parallel, it has been suggested that a deterioration of the periodontal status from a mild to a severe expression might be explained by a weak antibody response in patients developing generalized aggressive periodontitis [32].
Genetic factors
In some families, there is a marked aggregation of severe periodontitis. This finding, together with results from epidemiologic studies, indicates that hereditary factors may be important for the development of aggressive periodontitis. Segregation studies of affected families have revealed different modes of inheritance. Both autosomal dominant and recessive as well as X‐linked transmission have been suggested. The limited number of families in most studies may explain the inconsistent findings. However, the familial clustering of aggressive periodontitis could be an expression of enhanced susceptibility to microbial infections, in part determined by the host’s genotype. A wide variation (8–63%) of near relatives have been reported to have severe periodontitis [33,34]. It is obvious that the different results can be attributed not only to variations in the populations included, but also to diagnostic criteria and the number of relatives examined.
Even if there have been conflicting suggestions as to the modes of inheritance, it seems established that inheritance is an important factor to consider. For the clinician, this emphasizes the importance of clinical examination of children, siblings, and parents of patients diagnosed with aggressive periodontitis.
Ethnicity
Comprehensive epidemiologic surveys in the United States have demonstrated that black or Hispanic adolescents are 5–15 times more likely to develop aggressive periodontitis or chronic periodontitis compared to Caucasian adolescents. In addition, Scandinavian studies have revealed an enhanced risk of developing periodontitis in immigrant children of Asian origin compared to children of Scandinavia descent [35,36].
Modifying factors
In areas of defective restorations or manifest carious lesions, localized inflammatory reactions may progress to involve deeper parts of the periodontium. In a dentition under development, eruption disorders, such as ectopic eruption of first permanent molars and infraocclusion of primary molars, may favor plaque accumulation leading to chronic gingivitis and possibly the development of destructive periodontal disease.
Early reports often state that adolescents with localized aggressive periodontitis generally demonstrate little dental plaque and subgingival calculus. In contrast, more recent investigations have reported levels of plaque and calculus similar to cases with chronic periodontitis. Proximal subgingival calculus is often seen in areas of marginal bone loss in the primary dentition and the young permanent dentition, and is always connected with signs of chronic inflammation (Figure 14.5). Calculus is found more often in individuals with bone loss than in individuals without any sign of loss of periodontal support [13]. The exact role of calculus in the initiation and progression of the periodontal lesion is however unclear, and the finding of an association between subgingival calculus and periodontal disease does not automatically imply that calculus is of primary etiologic importance for the development of the disease. However, the rough calcified deposits facilitate bacterial colonization and should be removed.
Smoking is considered to be among the most significant risk factors for periodontitis in adults and should be regarded as a factor of importance for the initiation of the disease in adolescents as well [4]. The reasons for the enhanced risk of developing periodontitis in smokers are not fully known, but smoking‐related substances are known to act as vasoconstrictors, possibly resulting in tissue ischemia. These substances may also exert a negative effect on fibroblasts and inflammatory cells thereby affecting wound‐healing capacity.
Systemic diseases and syndromes
- Down syndrome. Periodontal disease is a common finding in children with Down syndrome. Marginal bone loss is more severe in the anterior segments, and especially in the mandible. The reasons for the high susceptibility to periodontal disease in these children are probably an impaired phagocytic function of neutrophils and monocytes in combination with poor oral hygiene [37].
- Type 1 diabetes. Most studies of adolescents with diabetes demonstrate a tendency to higher susceptibility to loss of periodontal support compared to healthy controls (Figures 14.15 and 14.16). Patients with poor metabolic control are at risk of developing periodontal disease [10].
- Hypophosphatasia. This hereditary metabolic syndrome results in low serum alkaline phosphatase activity, ricket‐like skeletal changes, and loss of alveolar bone, usually limited to the area of anterior primary teeth (Figure 14.17). The result is a precocious exfoliation of these teeth. Microscopically, teeth from affected areas exhibit aplasia and hypoplasia of root cementum, large pulp chambers, and interglobular dentin formation. Findings indicate that permanent teeth can also be affected, which puts children with hypophosphatasia at risk of developing periodontal complications during adolescence and adult life [25].
- Histiocytosis‐X (reticuloendotheliosis). This condition may cause alveolar bone destruction in connection with lesions in the jaws. Eosinophilic granuloma (histiocytosis in bone) is more frequent in the mandible than in the maxilla. Hand–Schüller–Christian disease (chronic disseminated histiocytosis) may lead to gross bone destruction extending around the roots and causing exfoliation. The treatment of the disease (corticosteroids, irradiation, and cytostatics) may produce secondary negative effects in the periodontium [25].
- Papillon–Lefèvre syndrome. This is a rare genetic disease affecting the hands and feet (keratosis palmaris et plantaris) and leading to fulminant types of periodontitis with rapid bone destruction. The oral symptoms start immediately after eruption of the primary teeth and cease after the premature loss of the first dentition only to start again after eruption of the permanent teeth [38].
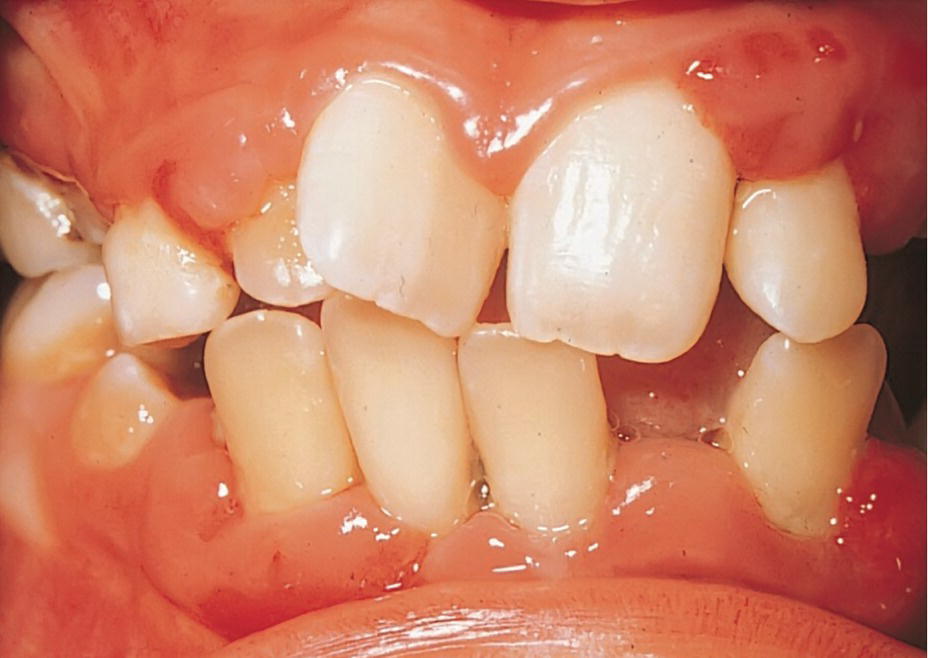
Figure 14.15 Severe periodontal involvement in a young child suffering from diabetes mellitus.
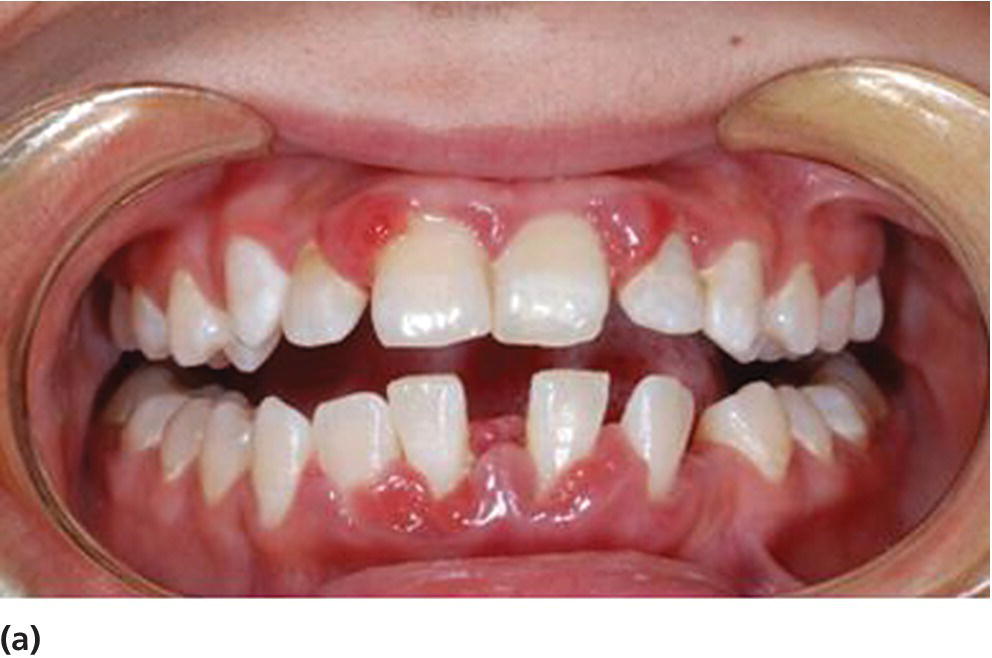
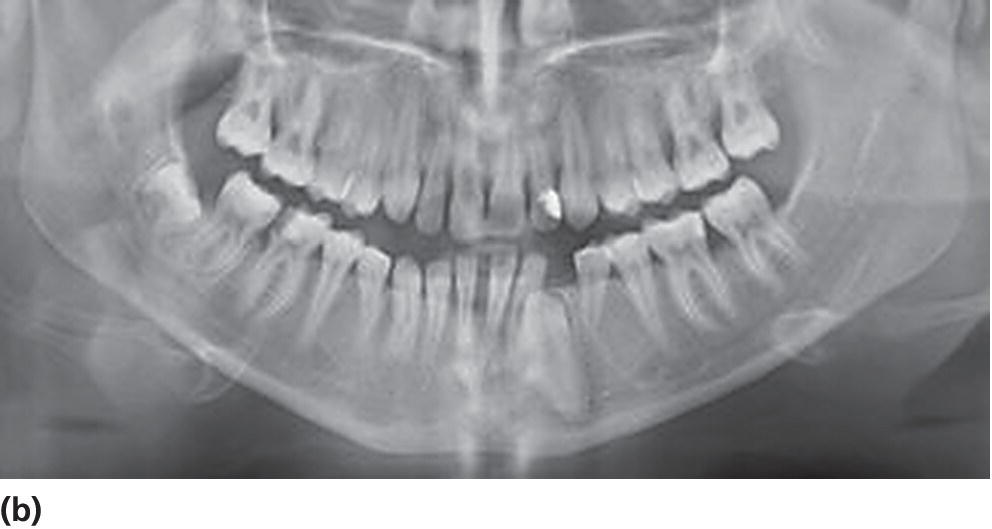
Figure 14.16 Aggressive periodontitis in a 19‐year‐old patient with diabetes mellitus with poor metabolic control. (a) Clinical picture (b) Panoramic radiograph.
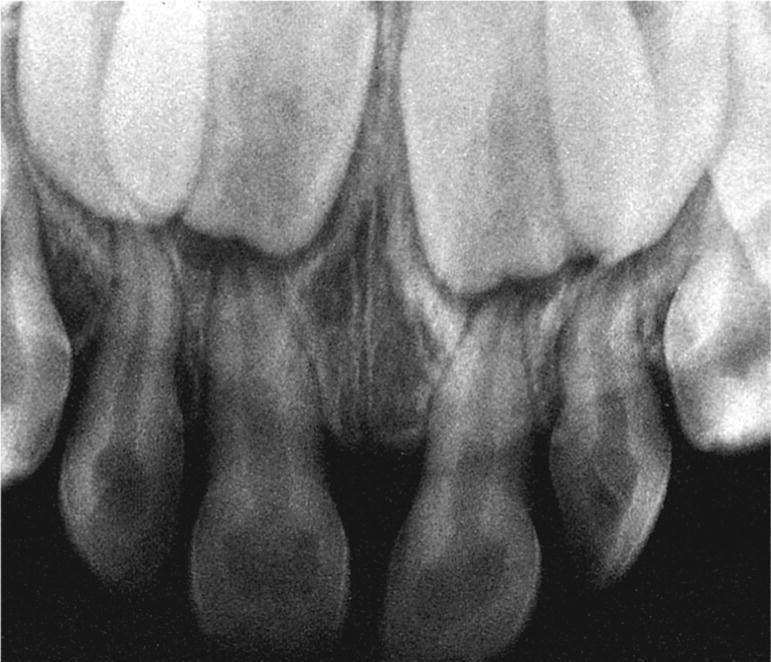
Figure 14.17 Alveolar bone loss in a child with hypophosphatasia.
Screening and treatment
Screening
Evidence is available that a proportion of young individuals with early clinical or radiographic signs of attachment loss are at risk for disease progression [15]. Data suggest also that young patients enrolled in organized dental health care have better prerequisites to stabilize or improve their periodontal conditions [20]. Although the prevalence of attachment loss is low in many populations, inclusion of periodontal assessment should be considered in the regular oral examination of children and adolescents (Box 14.2).

VIDEdental - Online dental courses


