CHAPTER 12 Psychopharmacology
Antipsychotic and Antidepressant Drugs*
According to the National Institutes of Mental Health, 20% of the population have a diagnosable mental disorder in their lifetime.35 The Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) classifies many types of mental disorders.10 Schizophrenia, which affects 0.5% to 1% of the population, is the most severe of the psychiatric disorders, and disturbances of mood (affective disorders) are the most common. Approximately 10% to 25% of women and 5% to 12% of men have at least one major depressive episode during their lifetimes. Schizophrenia and affective disorders are episodic and progressive disorders. Although appropriate treatment tends to improve the course of a disorder, it rarely produces a cure.
Psychotherapeutic drugs have contributed much to our understanding of mental illness and have reduced hospital bed occupancy by mental patients to a tenth of what it was before their use. Pharmacotherapy has permitted individuals who would otherwise have been hospitalized long-term to be more integrated into society. The Surgeon General’s report on mental health has indicated that more people could benefit from treatment with psychotropic agents than are currently being treated.35 In daily practice, the dentist can expect to treat patients taking psychotherapeutic agents for various mental disorders. These agents may contribute to oral pathology or become a factor when rendering dental care.17 Many patients being treated for mental illness report poor oral health, but it is not all due to their treatments; poor oral health is also correlated to older age, race, unemployment or financial strain, and smoking behavior.27
Brain imaging, molecular biology, and genetic studies promise to improve the understanding of psychotic disorders. Results from such research have identified changes that may relate to the pathology of these conditions. Key findings include the observation of genetic, structural, and functional changes in schizophrenia, bipolar disorder, and depression. Inconsistencies between the symptoms and observable signs still exist that challenge our understanding and treatment of psychiatric disorders. Brain imaging studies have shed some light on this variability. Imaging has shown that the onset of schizophrenia is associated with a patchy loss of cerebral cortex that progresses to a more global and severe loss.37 The symptoms seen in a particular patient may vary depending on where the process starts and how far it continues. For children with early-onset schizophrenia, the cell loss is found in most areas of the cortex 5 years after diagnosis.52 Difficulties in categorizing these illnesses can result in inconsistent treatment outcomes.
Psychotic states such as schizophrenia are treated with antipsychotic drugs, sometimes also referred to as neuroleptics; depression is treated with antidepressant drugs, and manic illness (and bipolar disorder) is treated with lithium salts and some anticonvulsant drugs. Neuroses (e.g., anxiety), which are less severe psychiatric disorders, are treated with antianxiety agents and are discussed in Chapter 13. Selective serotonin reuptake inhibitor (SSRI) antidepressants are also effective in treating several anxiety disorders, including obsessive-compulsive disorder, panic disorder, post-traumatic stress disorder, and social anxiety disorder. These drugs may be referred to as delayed action antianxiety agents because the effect develops slowly over several weeks. Sympathomimetic stimulants are effective treatments for attention-deficit/hyperactivity disorders and narcolepsy, and they are discussed in Chapter 6. New results from imaging and gene microarray studies reveal differences and similarities between the classification of brain disorders. In an individual patient, various agents may be needed for the particular spectrum of symptoms. Because of the perceived stigma associated with psychiatric illness, the psychiatric disorder itself (which may lead to poor self-insight), or the effects of the psychotropic drugs, patients may forget or be reluctant to discuss their disorder with dentists or to provide complete information regarding their treatment.56
MAJOR PSYCHIATRIC DISORDERS
Schizophrenia
In schizophrenia, the patient’s ability to function is markedly impaired because of disturbances in thought processes. These disturbances increase the likelihood of adverse social outcomes, such as unemployment, poverty, social isolation, and suicide.4
Neither the etiology nor the pathogenesis of schizophrenia is known. Current thinking suggests that a genetic predisposition and early injury (in utero or early childhood) may set the stage for neurodevelopmental changes that ultimately manifest as behavioral difficulties later in life (often in early adulthood) and that may be triggered by a stressful lifetime event.35 For the disease to manifest itself, the effects of several cumulative factors likely combine to result in the clinical disorder.4 Evidence from brain imaging suggests that broad areas of the brain can lose cell mass in this disorder, suggesting a problem in regulation of brain growth, repair, or cell pruning, rather than dysregulation of a single neurotransmitter. Genetic survey chip studies have also identified differences in neuronal structure and function (e.g., myelination, neuroimmune and mitochondrial genes) suggesting a neurodegenerative cause rather than a specific transmitter disruption.31
Dopamine hypothesis
The role of dopamine in schizophrenia has been complicated by the identification and cloning of several dopamine receptor subtypes and the discovery that other neurochemicals, either independently or by regulating dopamine, may be involved in the disease process. Dopamine innervation is extensive and contributes to the activity in the basal ganglia, cerebral cortex, hippocampus, amygdala, thalamus, and cerebellum. Hyperactive dopaminergic neural pathways offer a simple and attractive mechanism to explain schizophrenia, but this is an incomplete explanation for the disease. It is justified to assume, however, that the parkinson-like motor disturbances, which are common side effects of the antipsychotics, result from blockade of dopamine transmission in the basal ganglia (Table 12-1). Five dopaminergic cell groups are considered important either for the therapeutic actions of antipsychotic drugs or for side effects of the antipsychotic agents. Table 12-2 summarizes these dopamine cell groups and their proposed relationship to activity.
TABLE 12-1 Therapeutic and Adverse Effects Associated with Some Receptors Bound by Common Antipsychotic and Antidepressant Drugs
| RECEPTOR OR PROCESS BLOCKED | THERAPEUTIC EFFECT OF BLOCK | ADVERSE EFFECTS RESULTING FROM BLOCKADE OF RECEPTOR OR PROTEIN |
|---|---|---|
| Histamine (H1) | Sedation, anxiolysis, antiallergy effect | CNS depression, hypotension, dry mouth, weight gain |
| Muscarinic | Reduction of extrapyramidal side effects | Dry mouth, blurred vision, sinus tachycardia, constipation, urinary retention, memory dysfunction |
| Adrenoceptor | ||
| α1-Adrenergic | Memory dysfunction, postural (orthostatic) hypotension, reflex tachycardia, epinephrine reversal, dizziness, dry mouth, weight gain, priapism | |
| α2-Adrenergic | Blockade of presynaptic autoregulation, increasing CNS 5-HT and NE | Priapism |
| Dopamine (D2) | Amelioration of the positive signs and symptoms of psychosis | Extrapyramidal movement disorders, sexual dysfunction, dry mouth, weight gain |
| 5-HT reuptake | Reversal of depression | Gastrointestinal disturbance, sexual dysfunction, activating effects, dry mouth |
| NE reuptake | Reversal of depression | Dry mouth, urinary retention, erectile dysfunction, CNS stimulation, tremor, proconvulsant |
| Dopamine reuptake | Antidepressant effect (?) | Psychomotor activation, psychosis, proconvulsant action (?), dependence |
5-HT, 5-Hydroxytryptamine; CNS, central nervous system; NE, norepinephrine.
TABLE 12-2 Dopaminergic Cell Groups and Their Relationship to Actions and Side Effects of Antipsychotic Drugs
| CELL GROUP | RELATIONSHIP/ACTION |
|---|---|
| Mesolimbic and mesocortical | Considered the major therapeutic targets for antipsychotics |
| Nigrostriatal | Essential for motor function, related to the motor side effects of antipsychotics |
| Tuberoinfundibular | Regulation of hormones, especially inhibition of prolactin secretion; thought to mediate side effects such as galactorrhea and infertility |
| Chemoreceptor trigger zone | Receptors thought to mediate the antiemetic actions of antidopaminergic drugs |
| Medullary-periventricular | May mediate the actions of antidopaminergic drugs on appetite |
Other neurotransmitters
Other neurotransmitters have been implicated in psychotic behavior and are of interest regarding the cause of psychotic symptoms. Phencyclidine-induced psychosis is proposed to be an even better model for schizophrenia than the dopamine hypothesis. The predominant action of phencyclidine is thought to be blockade of N-methyl-d-aspartate (NMDA)–type glutamate (glutamatergic) receptors. NMDA receptors are polyagonist receptors, involving glutamate, glycine, and polyamines. Stimulation of NMDA by glutamate can be therapeutically problematic. There is more recent interest, however, in glycine transporter blockade as a possible indirect alternative to the use of glutamate receptor agonists for therapy for schizophrenia.30 The hallucinogens lysergic acid diethylamide and mescaline can induce hallucinations and delusions and are thought to produce their actions through 5-hydroxytryptamine type 2 (5-HT2) receptors.2 Anticholinergics, cannabinoids, and sigma receptor agonists are other drugs that can induce psychosis-like reactions.
Affective Disorders
The affective illnesses are expressed as dysregulations of mood. There are many types of affective disorders categorized in DSM-IV, but for purposes of this discussion it is sufficient to consider only depression and mania. Most individuals have had reactive, or secondary, depression with feelings of sadness or grief associated with a personal loss.10 In normal circumstances, such reactions are related to specific causes, are not incapacitating, and are generally short-lived (1 to 2 weeks). In contrast, for a mentally ill patient, depression is a severe, disabling disorder characterized by reclusiveness and nonverbalization that may last for extended periods (2 to 5 weeks). The patient is sad most of the day; gains little pleasure from activities; and may have other signs, such as weight loss, irritability, insomnia, feelings of guilt, agitation, or difficulty in concentrating.10
A serious consequence of depression is an increased risk of suicide. Depression is also a risk factor or is comorbid with other diseases, such as sleep disorders, weight changes, sexual disorder, pain disorders, anxiety disorders (and panic attacks), drug abuse, psychoses, myocardial infarction, and coronary artery disease. This kind of depression is called primary, endogenous, unipolar, or major depressive disorder. A variant of depression is seasonal affective disorder, which is triggered by changes in seasons and can respond to treatment with intense lights.10
Manic individuals may also have alternating periods of severe depression, in which case the disorder may be referred to as bipolar (manic-depressive) illness. The first episode of psychopathology is often depression, sleep disorders, or anxiety, but psychopathology later progresses to include manic episodes.11 The incidence of bipolar disorder may be 5% of the population and 45% of community mental health patients.3 Three forms of bipolar disorder have been distinguished. Bipolar I involves cycles of mania and depression. Bipolar II involves cycles of hypomania and depression. Hypomania is a less severe form of mania that does not have psychotic features (e.g., hallucinations). Bipolar III is mania associated with the use of antidepressants. Bipolar II patients only rarely convert to bipolar I, and patients with bipolar II disorder may have the highest risk for suicide.
There is great interest in identifying the underlying brain processes that may help explain depression and bipolar disorders. Heterogeneity in the expression and causes of depression has made this process challenging, however. As shown by imaging, the brain structural changes in depression include changes in the prefrontal cortex, the cingulate49 the insula, and the temporal lobe. The hippocampus and amygdala have been implicated.36 Cerebral blood flow and tissue glucose use are poorly correlated with depression in wide regions of the cortex, in contrast to the case for bipolar or normal subjects.12 In light of these imaging results, it is unlikely that depression is due to a change in a single neurotransmitter. Paradoxically, gene survey results suggest that bipolar disorder is similar to schizophrenia because both are viewed as degenerative disorders,49 whereas major depressive disorder may involve a more specific cause—glutamate/γ-aminobutyric acid imbalance.
Historical Development of Antipsychotic and Antidepressant Drugs
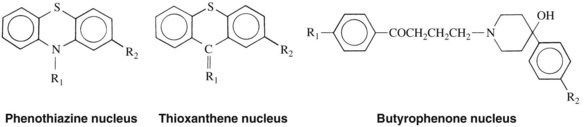
Most of the psychotherapeutic properties of the psychoactive drugs were discovered by accident. In 1950, while attempting to develop antihistaminic agents, the Rhône-Pauline Laboratories in France synthesized the phenothiazine chlorpromazine (Figure 12-1). The unusual neuroleptic property of chlorpromazine was noted, and the drug was used to treat schizophrenic patients in 1952. The discovery of chlorpromazine and other phenothiazines made possible the outpatient treatment of psychotic disorders.
A characteristic of many antipsychotic drugs is interference with multiple neurotransmitter systems. In addition to blocking dopaminergic receptors, many can block α-adrenergic and serotonergic receptors and alter functions of cells. These drugs have many side effects that can be related to these multiple receptor actions (see Table 12-1). The next phase of development focused on drugs with selective drug receptor binding. In the case of antipsychotic drugs, action at multiple receptor types may provide therapeutic advantages. The current challenge is to identify which combination of actions is most beneficial.
The tricyclic antidepressants (TCAs) were synthesized in an attempt to produce more specific antipsychotic agents (note in Figures 12-1 and 12-5 the chemical similarity of TCAs and phenothiazines). It was soon recognized, however, that imipramine, a prototypic TCA, was more beneficial in treating depression than in treating schizophrenia. TCAs were the mainstay for treating depression for many years. Their use has been limited, however, by the numerous side effects resulting from their actions on many nontherapeutic receptor sites. Efforts to develop better agents have been rewarded with a newer class of drugs, SSRIs, with fewer adverse effects than TCAs and fewer drug interactions than MAO inhibitors. These drugs have revolutionized the use of antidepressant medication. Subsequently, the selective serotonin and NE reuptake inhibitors and the selective NE reuptake inhibitors were developed.26
St. John’s wort is a botanical remedy with a long history of use for depression. It is now thought that the active principle is the compound hyperforin (instead of hypericin as had been believed).9 It has been characterized as a weak super–reuptake inhibitor with effects on NE, serotonin, dopamine, glycine, GABA, and glutamate reuptake.
Although the psychoactive properties of lithium salts were noted in 1949,6 lithium carbonate was not developed and widely recognized as an effective treatment for manic-depressive illness until 1970. More recent studies have focused on a possible neuroprotective role for lithium salts. The anticonvulsant drugs carbamazepine and valproic acid were investigated for treatment of mania (bipolar disorder) in the early 1980s, and since that time their use and that of other anticonvulsant drugs has been increasing. When used for this purpose, these drugs are referred to as mood stabilizers.
ANTIPSYCHOTIC DRUGS
The principal drugs effective in the treatment of schizophrenia are dopaminergic receptor antagonists. Five dopamine receptors (D1 through D5) have been cloned. Some of these receptors express extra long or short variants. The possibility that each of these receptors may subserve a different physiologic function illustrates the complexity of the dopaminergic system. D1 and D5 have similar actions and often increase cyclic 3′,5′-adenosine monophosphate synthesis, whereas D2, D3, and D4 are thought to decrease cyclic 3′,5′-adenosine monophosphate synthesis. Interest has now focused on the relative specificity and affinity of the antipsychotic agents for each of the dopamine receptors. The older typical antipsychotics (e.g., phenothiazines) and the atypical agents (e.g., clozapine) are dopamine antagonists. Clinical potency as an antipsychotic drug relates most closely to blocking D2 receptors (Table 12-3). The affinity of clozapine for the D1 and D4 receptors relative to D2 is proportionally greater, however, than that of the older agents. In postmortem brain samples from schizophrenic patients, there is an increase in the number of D2 receptors but not D1 receptors.
TABLE 12-3 Comparison of Relative Receptor Antagonist Affinities of Typical and Atypical Antipsychotic Drugs

Although there is little doubt that dopamine is involved in schizophrenia, other neurotransmitter systems may also play a crucial role in this disease. In addition to dopamine, neurotransmitters such as 5-HT, glutamate, NE, glycine, and GABA have been implicated in schizophrenia, suggesting that this is a very complex and multifaceted illness and that many mechanisms may be involved in the disease process.2 Preliminary gene chip microarray analysis has shown changes in signal transduction, transcription, and metabolic enzymes associated with altered regulation of certain genes in schizophrenia.31
Chemistry and Structure-Activity Relationships
Phenothiazines and thioxanthenes
The basic ring structure of the phenothiazines is illustrated in Figure 12-1. Substitutions at R1 divide the phenothiazine antipsychotics into three major groups. One group, represented by chlorpromazine, has an aliphatic chain at C1. Compounds such as chlorpromazine with three carbons in the chain linked to an amine (–CH2–CH2–CH2–N(CH3)2) have antipsychotic properties, whereas compounds with only two carbons, such as promethazine, are usually more antihistaminic or anticholinergic in nature and possess few antipsychotic effects. A second group, represented by thioridazine, has a piperidine ring at R1 attached to the carbon chain. These phenothiazines are usually less sedating than the aliphatic agents but more sedating than the next group. A third group, represented by prochlorperazine, contains a piperazine ring on the carbon chain at R1. Drugs in this group are the most potent of the three as antipsychotic agents but are also the most likely to produce extrapyramidal side effects. Minor changes in the structure of these molecules can increase or abolish antipsychotic activity. The thioxanthene antipsychotics, represented by thiothixene, are closely related to the phenothiazines and are formed when the nitrogen of the central ring is replaced by a carbon atom.
Butyrophenones
The butyrophenone antipsychotics are not chemically related to the phenothiazines, but contain a stereochemically related nucleus (see Figure 12-1). The only butyrophenone antipsychotic available in the United States is haloperidol. Droperidol, another butyrophenone, is marketed as an antipsychotic in some countries, but is occasionally used in the United States primarily to reduce nausea and vomiting associated with anesthesia and surgery. Combined with the opiate fentanyl, it is also used to achieve deep sedation (see Chapter 48).
Dihydroindolones
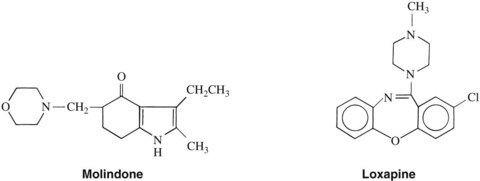
The structure of molindone is shown in Figure 12-2. This compound is not structurally related to the phenothiazines, thioxanthenes, or butyrophenones. The pharmacologic and clinical profile of molindone resembles that of the piperazine group of phenothiazines very closely. Ziprasidone, another dihydroindolone, is pharmacologically an atypical antipsychotic and is discussed subsequently.
Dibenzoxazepines
Loxapine (see Figure 12-2) is the only dibenzoxazepine available in the United States. The structure of this compound contains seven members in its central ring and resembles a TCA. Loxapine does not seem to have antidepressant activity, however. Similar to molindone, this drug has a clinical and pharmacologic profile similar to that of piperazine phenothiazines.
Dibenzodiazepines
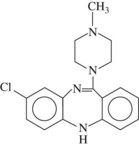
Clozapine (Figure 12-3) is the only dibenzodiazepine available in the United States. Its chemical structure closely resembles that of loxapine, but in contrast to loxapine, it is classified as an atypical antipsychotic in light of its low risk for producing extrapyramidal side effects. Clozapine is reported to improve positive and negative symptoms of schizophrenia and may reverse the progression of schizophrenic symptoms. Clozapine also has muscarinic, 5-HT2,6,7, α1-adrenergic, and D1, D2, and D4 receptor blocking properties. Use of clozapine can be accompanied by significant toxicity, especially agranulocytosis, seizures, and hypotension. Myocarditis and cardiomyopathy may rarely occur.
Thienobenzodiazepines
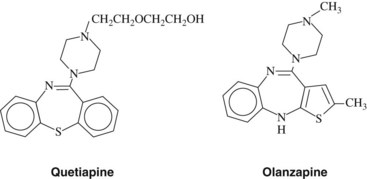
Olanzapine (Figure 12-4) is an atypical antipsychotic approved for clinical use. Its inhibitory actions at monoamine synapses are similar to the actions of clozapine except that olanzapine has a higher affinity for D2 receptors (see Table 12-3). It is associated with fewer adverse effects than clozapine, particularly agranulocytosis.
Other drugs expressing atypical antipsychotic activity
Other atypical antipsychotic drugs are available. Quetiapine (a dibenzothiazepine) (see Figure 12-4) is effective for positive and negative symptoms. Ziprasidone (a dihydroindolone) has actions similar to risperidone. Ziprasidone carries warnings for inducing long QT syndrome. Aripiprazole is a dihydrocarbostyril derivative with a unique spectrum of action. Aripiprazole has been found to act as a partial agonist at D2, D3, and 5-HT1A receptors and to act as an antagonist at 5-HT2A receptors. It is reported to produce minimal side effects commonly associated with antipsychotic drugs. This seems to be a matter of degree because typical dopamine-blocking side effects are seen in a few patients.
Pharmacologic Effects
Antipsychotic effects
There are several dopaminergic pathways in the central nervous system (CNS) (see Table 12-2) that, when antagonized by the antipsychotics, can explain their therapeutic efficacy and some of their side effects. The antipsychotic action may be ascribed to blockade of the mesolimbic/mesocortical tract, which plays an important role in behavior, arousal, salience (i.e. alerting, significance), positive reinforcement, cognitive function, communication, and psychological responses. Although blocking mesolimbic/mesocortical dopamine is thought to be central to antipsychotic efficacy, the inhibition of positive reinforcement may contribute to the high rate of discontinuation of treatment, which averages 74% over 18 months of therapy. Extrapyramidal motor dysfunction results from blockade of the nigrostriatal pathway, and endocrine disorders (amenorrhea, dysmenorrhea) result from the blockade of the hypothalamic-adenohypophyseal system. Two effects of antipsychotics may relate to blockade of dopamine receptors in the brainstem. Blockade of dopamine receptors in the medullary chemoreceptor trigger zone is thought to contribute to the antiemetic actions of antipsychotic drugs. Blockade of dopamine receptors in the medulla or brainstem may also play a role in appetite dysregulation. There are also dopamine interneurons in the olfactory bulb and retina. Olfactory changes occur in schizophrenia and Parkinson’s disease, but these have not yet been related to dopamine. In the retina, dopaminergic cells may regulate light adaptation.55
The potent anticholinergic activity of clozapine, the 5-HT2A receptor–blocking effect, and the limited occupation of D2 receptors (<60%) at therapeutic doses all may contribute to its atypical profile. Olanzapine, risperidone, and paliperidone have lower D2 receptor binding and higher 5-HT2 receptor binding and are relatively free of dyskinesias.25 Quetiapine has a therapeutic effect and a side-effect profile similar to olanzapine.
Extrapyramidal effects
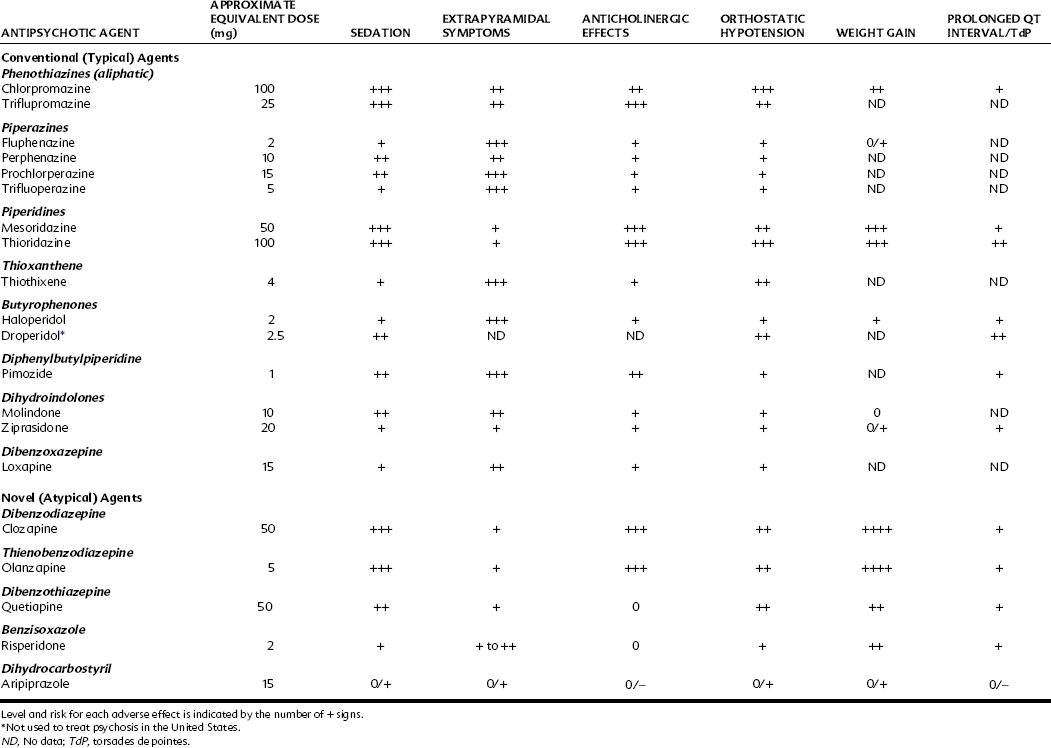
The extrapyramidal side effects produced by antipsychotic drugs include acute dystonias, a Parkinson-like syndrome, akathisia, and tardive dyskinesia. The different types of phenothiazines produce varying degrees of extrapyramidal side effects; in descending order of most to least potent are the piperazines, aliphatics, and piperidines. These compounds follow the reverse ranking order regarding their anticholinergic potency, which may explain why fluphenazine and haloperidol, weak anticholinergics, commonly produce extrapyramidal side effects, whereas thioridazine, a more potent anticholinergic drug, produces fewer motor disturbances. More recently, the role of 5-HT2 receptors in reducing extrapyramidal symptoms and dystonias has been considered. As can be seen in Table 12-3, chlorpromazine is a more potent inhibitor of 5-HT2 than it is for the muscarinic receptor. In addition, many newer antipsychotic drugs having atypical properties also block 5-HT2 receptors. Molindone and loxapine are similar to chlorpromazine in their potential for causing extrapyramidal reactions. Haloperidol may have some unique effects on motor function. It is metabolized to a potentially neurotoxic metabolite, which may adversely affect the dopaminergic cells in the substantia nigra, inducing Parkinson’s disease. Haloperidol also blocks sigma receptors. Sigma receptors in the red nucleus have been shown to participate in the generation of dystonias (oculogyric crisis and torticollis) associated with neuroleptic use. This observation may be particularly important for facial dystonias because sigma receptors are also expressed in cranial nerve nuclei.
Anti-Parkinson drugs (see Chapter 15) may be used to antagonize certain antipsychotic-induced motor disturbances, but levodopa is not helpful in this regard. Because dopamine receptors are blocked by the antipsychotic drugs, levodopa, the precursor of dopamine, is less effective in treating drug-induced parkinsonism than the anticholinergics, antihistamines, and amantadine, which act through other mechanisms.
This side effect is frequently not seen until the antipsychotic drug is either withdrawn or reduced in dosage. The only consistently effective treatment for tardive dyskinesia has been increasing the dose of the antipsychotic that caused it in the first place. Such a procedure leads to a vicious cycle and a serious therapeutic dilemma. Typical antipsychotics cause this side effect. Clonazepam may be helpful in mild cases, and botulinum toxin may allow control of overactive muscle groups.50 Atypical agents such as clozapine, risperidone, and aripiprazole seem to have a reduced liability for tardive dyskinesia. Aripirazole may be helpful for treating some cases, but long-term experience is not yet available. The low incidence of extrapyramidal side effects and the apparent absence of tardive dyskinesia are additional reasons for the interest in the atypical antipsychotic drugs.
Endocrine system
Endocrine system alterations result partly from actions on the hypothalamus. Most of the endocrine effects of antipsychotics are related to disturbances in the secretion of pituitary hormones. Particularly prominent is hyperprolactinemia elicited by blockade of dopamine receptors. Stimulation of dopamine receptors in the adenohypophysis normally inhibits prolactin release. Chlorpromazine may cause lactation and amenorrhea or delay ovulation and menstruation in women and cause gynecomastia and impotence or decreased libido in men. For atypical agents, risperidone has the highest incidence of these effects, and clozapine and aripiprazole have the lowest.48 The urinary excretion of estrogens, progestins, and 17-hydroxycorticosteroids is decreased by chlorpromazine. Diuretic and antidiuretic effects have been shown in animals and humans, although a weak diuresis seems to be the predominant effect in humans. The relative lack of effect of clozapine on dopamine receptors of the anterior pituitary accounts for its mild endocrine side effects. Weight gain and diabetogenic effects have been observed for several atypical antipsychotic agents. Olanzapine is most likely to produce weight gain.
Autonomic nervous system
The following side effects of the phenothiazines may result from their antimuscarinic properties: blurring of vision; constipation; and decreased sweating, salivation, gastric secretion, and intestinal tone. Phenothiazines also have antihistaminic, antitryptaminergic, and antiadrenergic properties that complicate further the overall pattern of their CNS and peripheral activities. Dry mouth associated with a series of neuroleptics was found to correlate significantly with their blocking potency at α1-adrenergic, H1-histaminergic, and D1-dopaminergic receptors.45 The autonomic effects of haloperidol, molindone, and loxapine are similar, although weaker, than the effects of the phenothiazines. Clozapine and olanzapine have the reported side effect of hypersalivation.47 Often this phenomenon is most prominent during sleep; the mechanism responsible for it is unknown. Although the autonomic effects of antipsychotics can be annoying, tolerance usually develops to these reactions.
Cardiovascular system
Orthostatic hypotension is a result of CNS and peripheral (α-adrenergic receptor blocking) actions of antipsychotics (see Table 12-1), whereas tachycardia and increased coronary blood flow derive from central compensatory cardiovascular reflexes. The aliphatic and piperidine phenothiazines and clozapine are the most likely, and the piperazines the least likely, to cause orthostatic hypotension (Table 12-4). In the emergency treatment of phenothiazine-induced vasomotor collapse, epinephrine is contraindicated because the α-adrenergic receptor–blocking action of the phenothiazines may cause “epinephrine reversal” and an even greater reduction in blood pressure. NE or phenylephrine, both of which lack significant β2-adrenergic receptor stimulation, is preferred in these circumstances. Chlorpromazine has a direct depressant effect on the heart and an antiarrhythmic action that may be caused in part by its local anesthetic effect. Vascular reflexes mediated by vasomotor centers of the brainstem are depressed by chlorpromazine. Haloperidol rarely causes pronounced hypotensive effects, but tachycardia is a common side effect.
Many antipsychotic and antidepressant agents have been found to cause or exacerbate the condition known as long QT syndrome (see Chapter 24). This syndrome is associated with/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses