Chapter 12
Ozonization and Electrochemical Root Canal Disinfection
Roberta Pileggi
Department of Endodontics, University of Florida, Gainesville, FL, USA
The importance of the use of irrigants in root canal therapy is well established. Several solutions and methods have been advocated to better eradicate bacteria and tissue remnants from the root canal system. This chapter focuses on two methods that have gained popularity for the past few years: ozonofication and electrochemical activation (ECA).
Ozone therapy
Ozone was introduced to medicine, food, and chemical industries several years ago following its antibactericidal, antiviral, and antifungal properties. It is recommended in the treatment of vascular, orthopedic, and infection diseases. Structurally, ozone is a molecule composed of three oxygen atoms, commonly delivered as gas or dissolved in water, where it forms oxidate radicals (1–5). It is 1.6-fold denser and 10-fold more soluble in water (49.0 ml in 100 ml water at 0 °C) than oxygen (4).
The antimicrobial property of ozone is due to oxidation, which destroys the cell wall and the cytoplasmic membrane of microorganisms affecting osmotic stability. In a recent study where the effect of ozone on the membrane permeability and ultrastructure of Pseudomonas aeruginosa was examined, it was demonstrated that the cell inactivation was because of an increased membrane permeability and cytoplasm agglutination rather than cell lysis (6). Even with controversies related to ozone application following its toxicity, it is well established that depending on its dosage, it exhibits therapeutic properties. In medicine, it is mainly delivered as a mixture with oxygen (5). In an animal study, ozone therapy proved to be antimicrobial in infected sternal and mediastinal tissues of experimental methicillin-resistant Staphylococcus aureus animals (7).
In dentistry, ozone is applied mostly as an oxygen/ozone gas or as ozonated water. The antimicrobial effect of ozone in dentistry is controversial because of variability in the concentration of ozone and methodology utilized (8, 9).
In dentistry
In relation to caries, when Streptococcus mutans and Streptococcus sobrinus were examined following the use of an ozone-generating device for 10 s (Heolozone, USA), a reduction of both microorganisms was observed (10). In a well-controlled clinical trial, when ozone was applied on primary root carious lesions, it resulted in the arrest of lesions up to 1.5 years following initial treatment (11).
In a prospective clinical study, ozone improved noncavitated initial fissure caries in patients of high risk over a period of 3 months (12).
Polydorou et al. (13) examined the potential of ozone as an antimicrobial agent on two bonding agents to decrease the incidence of recurrent decay. They concluded that the use of ozone for 80 s in an in-vitro infected cavity model significantly reduced the presence of S. mutans. Muller et al. (14) evaluated the antimicrobial efficacy of ozone gas with photodynamic therapy in-vitro. In this study, 0.2% and 2% chlorhexidine and 0.5% and 5% sodium hypochlorite solutions were used on multispecies oral biofilm-coated discs with Actinomyces naeslundii, Veillonella dispar, Fusobacterium nucleatum, S. sobrinus, Streptococcus orali, and Candida albicans. Notably, only 5% sodium hypochlorite solution was effective in eliminating all the bacteria.
As ozone has been indicated as an antimicrobial agent, the effect of gaseous ozone application on enamel and dentin bond strength has been of interest, and several studies have demonstrated no negative influence on the bonding of the enamel and dentin bond strength (15–17). Azarpazhooh et al. (18) examined the effect of ozone treatment on tooth hypersensitivity and concluded that even though a reduction of sensitivity in the ozone group was present, this percentage of reduction was not different from the placebo. This is in agreement with the study by Elgalaid (19), who also showed that ozone was not significantly more successful than the placebo group.
In endodontics
In endodontics, most studies were focused primarily on the antimicrobial activity of ozone. These studies resulted in controversial findings depending on the microorganisms evaluated, type and concentration of ozone, and time of contact (Table 12.1).
Table 12.1 Summary of Studies Focused Primarily on the Action of Ozone in Endodontics
| Study | Type of ozone gas/water | Concentration | Time | Microorganisms | Effectiveness |
| Nagayoshi et al. (20) | Ozonated water | 4 mg/ml | 10 min | Enterococcus faecalis and Streptococcus mutans on dentinal tubules | With sonication, comparable results with 2.5% NaOCl. Less cytotoxicity |
| Virtej et al. (21) | HealOzone® | Ozone gas | Not specified | Aerobic and anaerobic bacteria | As effective as 3% NaOCl and MTAD |
| Hems et al. (22) | Ozonated gas sparged through a broth and ozonated water and ozone gas on biofilm | Ozonated gas | Not specified | Enterococcus faecalis planktonic and biofilm | Effective on planktonic but not on biofilm |
| Estrela et al. (23) | Ozonated water and gaseous ozone | Not specified | 20 min | Enterococcus faecalis | Not effective |
| Cardoso et al. (24) | Ozonated water | 24 mg/l | 20 min | Candida albicans, Enterococcus faecalis, and endotoxins | Effective immediately following application; however, not effective at later sampling Ineffective against endotoxin |
| Case et al. (25) | Ozone gas and passive ultrasound | Not specified | Not specified | Enterococcus faecalis |
Summary of studies focused primarily on the action of ozone in endodontics (by author).
Nagayoshi et al. (20) examined the antimicrobial effects of ozonated water in the dentinal tubules against Enterococcus faecalis and S. mutans. Their results suggested that ozonated water when used with sonication had approximately the same reduction of bacteria as 2.25% sodium hypochlorite and also noted that ozonated water is less cytotoxic than 2.25% sodium hypochlorite on L-929 mouse fibroblasts. Their results suggest that ozonated water not only killed the microorganisms invading the dentinal tubules but was also less cytotoxic.
Virtej et al. (21) demonstrated ozone (HealOzone®, Kavo, Biberach, Germany) to be as efficient as 3% sodium hypochlorite and Biopure MTAD (Dentsply Tulsa Dental, Tulsa, OK) on the elimination of bacteria. Lynch (26) recommend the use of high concentrations of ozone as the final step in the instrumentation of the canal system.
When the antimicrobial effect against E. faecalis of ozonated water, gaseous ozone, 2.5% sodium hypochlorite, or 2% chlorhexidine were evaluated in maxillary anterior extracted teeth, none of the irrigants completely eliminated the bacteria following 20 min of contact; however, the concentrations of ozone used were not specified (23). This result is in agreement with the finding of Case et al. (25), who demonstrated that ozone failed to eliminate more E. faecalis biofilm compared to sodium hypochlorite, even with a concentration as low as 1%.
Hems et al. (22) demonstrated that when a monobiofilm specie of E. faecalis was evaluated, ozonated water was not effective unless the sample was agitated. However, when planktonic E. faecalis cells were evaluated, ozone demonstrated antimicrobial activity following 240 s of contact.
When C. albicans was evaluated, in addition to E. faecalis, ozonated water applied for 20 min reduced the counts for both microorganisms. However, when a second sampling was taken from the root canal system, microbial counts had increased, demonstrating a lack of residual effect of ozonated water. In the same study, ozonated water was unable to neutralize Escherichia coli endotoxin in root canals (24).
Electrochemical activation
ECA is produced by applying an electric current (electrolysis) to a diluted salt solution—typically a sodium chloride solution. Direct electric current is impressed on an anode (positive charge) and a cathode (negative charge) separated by a permeable membrane. As salt solution (electrolyte) flows across the anode and cathode, the bonds of a portion of the solution constituents, H2O and NaCl, are rearranged. The solution leaving the anode chamber is known as the anolyte, and may contain oxygen gas (O2), chlorine gas (Cl2), hypochlorite ion (OCl−), hypochlorous acid (HOCl), hydrochloric acid (HCl), and unreacted NaCl. The solution leaving the cathode chamber is known as the catholyte, and may contain NaOH, H2, and unreacted NaCl (27). ECA has been used for many years and was popularized mainly in Russia and Japan (28, 29). ECA is also used in other countries in Europe and South America. The main applications of ECA are in medicine, food industry, and waste water treatment, and more recently in dentistry because of its disinfectant, antiseptic, and antimicrobial properties. ECA produces a hypochlorous acid solution that is biocompatible, antimicrobial, and possesses a detergent effect. The antimicrobial properties are characteristic of the high oxidation potential of the anolyte solution produced, while the strong detergent effect is due to the high reduction potential of the catholyte solution (28–31).
A simple schematic of the process is shown in Figure 12.1.
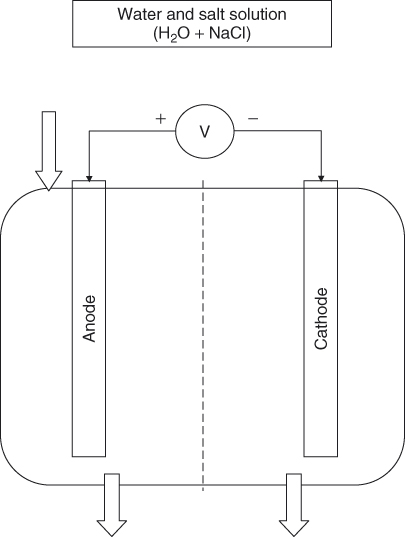
Figure 12.1 Simple schematic showing electrochemical process in an electrolyzed water generator.
In medicine, because of proven antimicrobial properties, ECA has been used in several countries during surgical procedures to disinfect endoscopy units, to prevent potential postoperative infection, and has also been used for the treatment of infectious skin, peritonitis, and to improve wound healing (32–35). The actual composition of the anolyte and catholyte will vary according to many factors, including the electrode material, electrode surface area, rate of electrolyte flow, amplitude of the electric current, and salt composition. The anolyte and catholyte have strong but opposite characteristics:
Anolyte
- Low pH, usually ranging from 2 to 8
- High oxidizing p/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


