Oral Mucosa
Functions of the Oral Mucosa
Secretion
The major secretion associated with the oral mucosa is saliva, produced by the salivary glands, which contributes to the maintenance of a moist surface. The major salivary glands are situated distant from the mucosa, and their secretions pass through the mucosa via long ducts; however, many minor salivary glands are associated with the oral mucosa (the salivary glands are described fully in Chapter 11). Sebaceous glands frequently are present in the oral mucosa, but their secretions are probably insignificant.
Boundaries of the Oral Mucosa
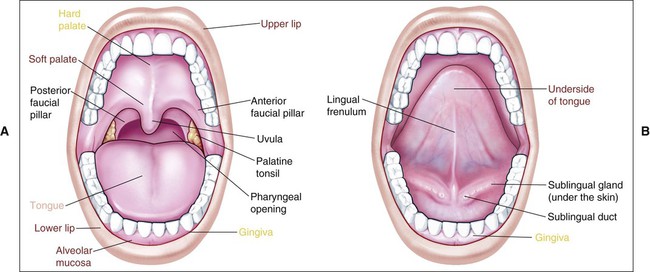
The oral cavity consists of two parts: an outer vestibule, bounded by the lips and cheeks, and the oral cavity proper, separated from the vestibule by alveolar bone and gingiva. The hard and soft palates form the superior zone of the oral cavity proper, and the floor of the mouth and base of the tongue form the inferior border. Posteriorly the oral cavity is bounded by the pillars of the fauces and the tonsils. The oral mucosa shows considerable structural variation in different regions of the oral cavity, but three main types of mucosa can be recognized, identified according to their primary function: masticatory mucosa, lining mucosa, and specialized mucosa. Figure 12-1 shows the anatomic location of each type diagrammatically, and the types are described fully later in the chapter. Quantitatively, the larger part of the oral mucosa is represented by lining mucosa, amounting to about 60% of the total area, with masticatory mucosa and specialized mucosa occupying smaller areas (25% and 15%, respectively).
Clinical Features
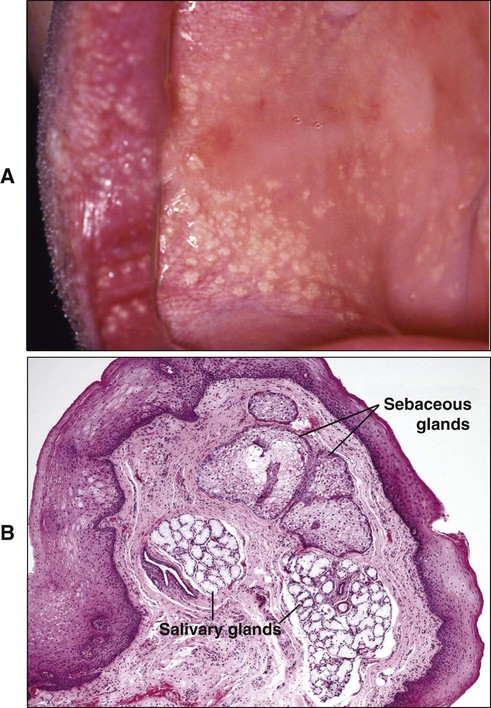
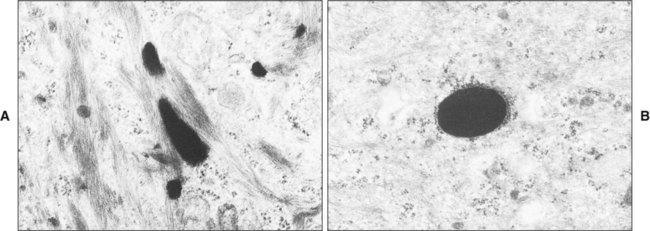
Other features that distinguish the oral mucosa from skin are its moist surface and the absence of appendages. Skin contains numerous hair follicles, sebaceous glands, and sweat glands, whereas the oral mucosa essentially only has minor salivary glands. These glands are concentrated in various regions of the oral cavity, and the openings of their ducts at the mucosal surface are sometimes evident on clinical examination (Figure 12-2, B). Sebaceous glands are present in the upper lip and buccal mucosa in about three quarters of adults and have been described occasionally in the alveolar mucosa and dorsum of the tongue (Figure 12-3). Sebaceous glands appear as pale yellow spots, also called Fordyce’s spots.
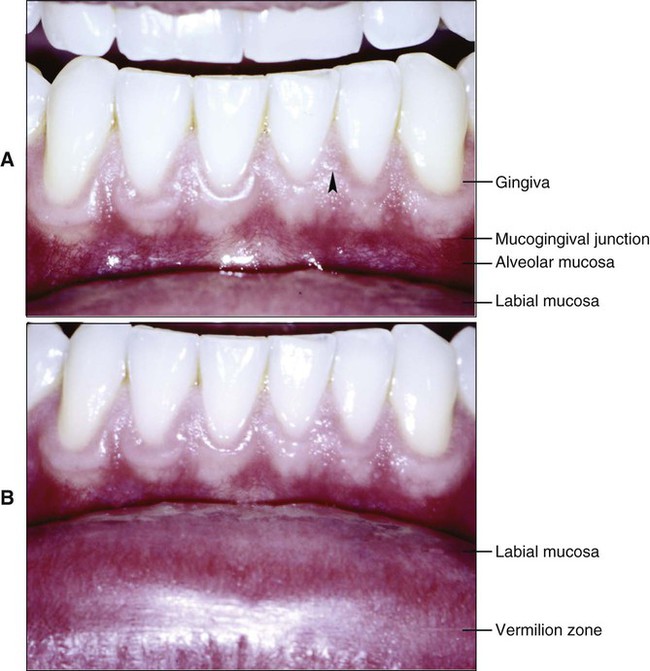
The surface of the oral mucosa tends to be smoother and have fewer folds or wrinkles than the skin, but topographic features are readily apparent on clinical examination. The most obvious are the different papillae on the dorsum of the tongue and the transverse ridges (or rugae) of the hard palate. The healthy gingiva shows a pattern of fine surface stippling, consisting of small indentations of the mucosal surface (Figure 12-2, A). In many persons a slight whitish ridge occurs along the buccal mucosa in the occlusal plane of the teeth. This line, also called the linea alba (white line), is a keratinized region and may represent the effect of abrasion from rough tooth restorations or cheek biting.
Component Tissues and Glands
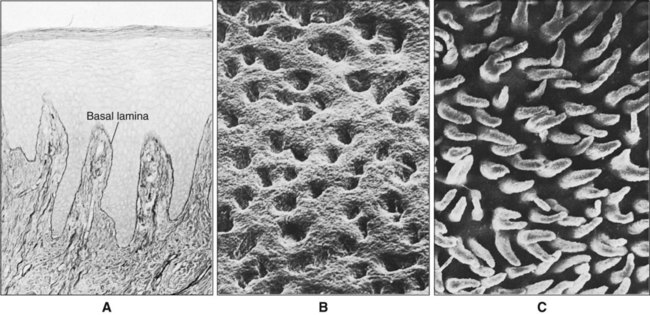
The two main tissue components of the oral mucosa are a stratified squamous epithelium, called the oral epithelium, and an underlying connective tissue layer, called the lamina propria (Figure 12-4). In the skin these two tissues are known as the epidermis and dermis. The interface between epithelium and connective tissue is usually irregular, and upward projections of connective tissue, called the connective tissue papillae, interdigitate with epithelial ridges or pegs (Figure 12-5). There is a basal lamina at the interface between epithelium and connective tissue which requires special staining to be visible by light microscopy (Figure 12-5, A).
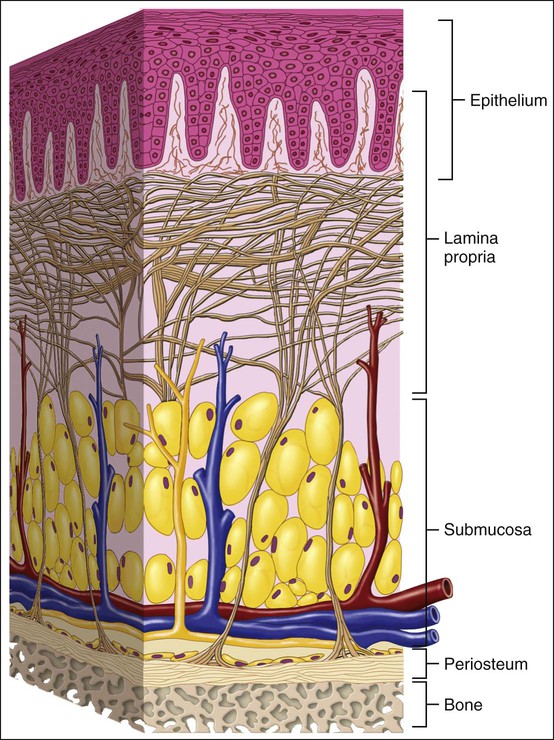
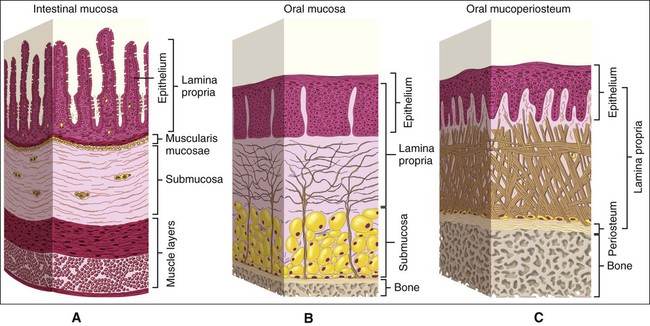
Although the junction between oral epithelium and lamina propria is obvious, that between the oral mucosa and underlying tissue, or submucosa, is less easy to recognize as compared to intestinal mucosa which clearly is separated from underlying tissues by a layer of smooth muscle and elastic fibers (Figure 12-6, A). In many regions (e.g., cheeks, lips, and parts of the hard palate) a layer of loose fatty or glandular connective tissue containing the major blood vessels and nerves that supply the mucosa separates the oral mucosa from underlying bone or muscle. The layer represents the submucosa in the oral cavity (Figure 12-6, B), and its composition determines the flexibility of the attachment of oral mucosa to the underlying structures. In regions such as the gingiva and parts of the hard palate, oral mucosa is attached directly to the periosteum of underlying bone, with no intervening submucosa (Figure 12-6, C). This arrangement is called a mucoperiosteum and provides a firm, inelastic attachment.
Oral Epithelium
As the tissue that forms the surface of the oral mucosa, the oral epithelium constitutes the primary barrier between the oral environment and deeper tissues. The oral epithelium is a stratified squamous epithelium consisting of cells tightly attached to each other and arranged in a number of distinct layers or strata. Like other epithelia, the oral epithelium maintains its structural integrity by a process of continuous renewal in which cells produced by mitotic divisions in the deepest layers mature and undergo terminal differentiation as they migrate passively toward the surface to replace those that are shed. The end phase of this pathway is indeed regarded as a unique form of programmed cell death (see Chapter 7). The cells of the epithelium thus can be considered to consist of two functional populations: a progenitor population (the function of which is to divide and provide new cells) and a maturing population (which continually differentiate or mature to form a protective surface layer).
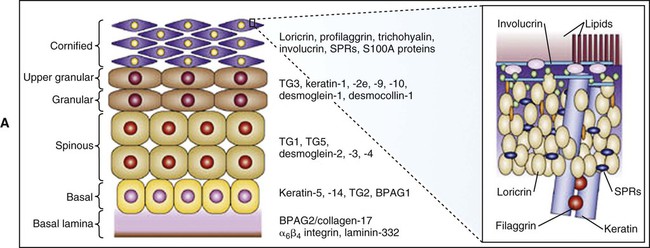
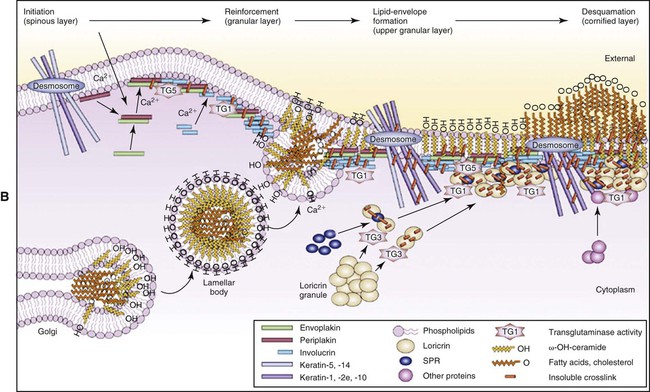
Maturing cells in stratified squamous epithelia assemble at their periphery a specialized protective layer called the cornified cell envelope, which consists essentially of keratins embedded in an insoluble amalgam of proteins surrounded by lipids (Figure 12-7). The process begins with the synthesis of an immature envelope on the cytoplasmic face of the plasma membrane. The cells produce keratohyalin granules (discussed later) which release the precursor of the intermediate filament protein filagrin. This protein aggregates keratin filaments, promoting the collapse and flattening of keratinocytes, which are thereafter referred to as corneocytes. Concurrently, a number of other proteins, including involucrin, loricrin, trichohyalin, and small proline-rich proteins are synthesized. These proteins are cross-linked by transglutaminases in relation to the aggregated keratin filaments just below the plasma membrane. This cornified cell envelope eventually replaces the plasma membrane of corneocytes, and becomes coated with lipid consisting mainly of ceramides, cholesterol, and free fatty acids and which acts as an essential water barrier. Corneocytes are tightly attached to each other by modified desmososmes which undergo proteolytic degradation to permit cells to desquamate.
Epithelial Maturation
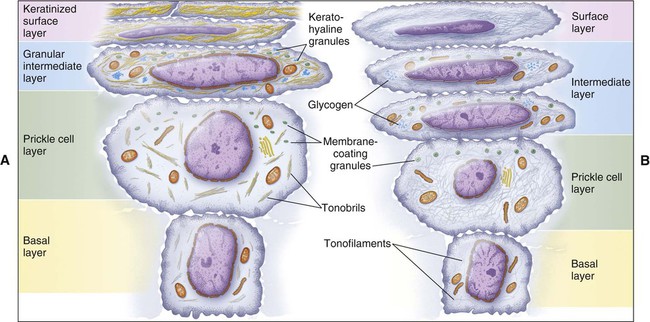
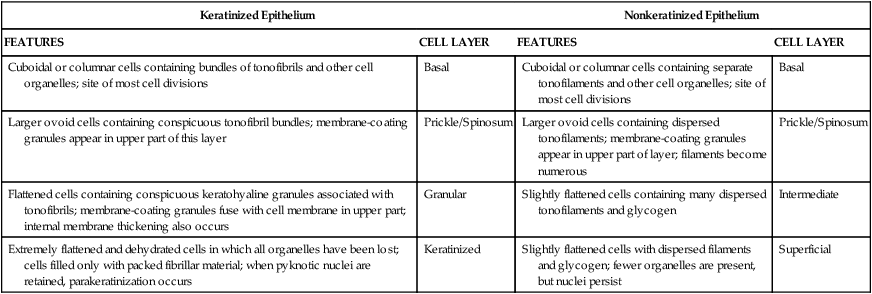
Cells arising by division in the basal or parabasal layers of the epithelium undergo a process of maturation as they are passively displaced toward the surface. In general, maturation in the oral cavity follows two main patterns: keratinization and nonkeratinization (Table 12-1).
TABLE 12-1
Major Features of Maturation in Keratinized and Nonkeratinized Epithelium
| Keratinized Epithelium | Nonkeratinized Epithelium | ||
| FEATURES | CELL LAYER | FEATURES | CELL LAYER |
| Cuboidal or columnar cells containing bundles of tonofibrils and other cell organelles; site of most cell divisions | Basal | Cuboidal or columnar cells containing separate tonofilaments and other cell organelles; site of most cell divisions | Basal |
| Larger ovoid cells containing conspicuous tonofibril bundles; membrane-coating granules appear in upper part of this layer | Prickle/Spinosum | Larger ovoid cells containing dispersed tonofilaments; membrane-coating granules appear in upper part of layer; filaments become numerous | Prickle/Spinosum |
| Flattened cells containing conspicuous keratohyaline granules associated with tonofibrils; membrane-coating granules fuse with cell membrane in upper part; internal membrane thickening also occurs | Granular | Slightly flattened cells containing many dispersed tonofilaments and glycogen | Intermediate |
| Extremely flattened and dehydrated cells in which all organelles have been lost; cells filled only with packed fibrillar material; when pyknotic nuclei are retained, parakeratinization occurs | Keratinized | Slightly flattened cells with dispersed filaments and glycogen; fewer organelles are present, but nuclei persist | Superficial |
Keratinization
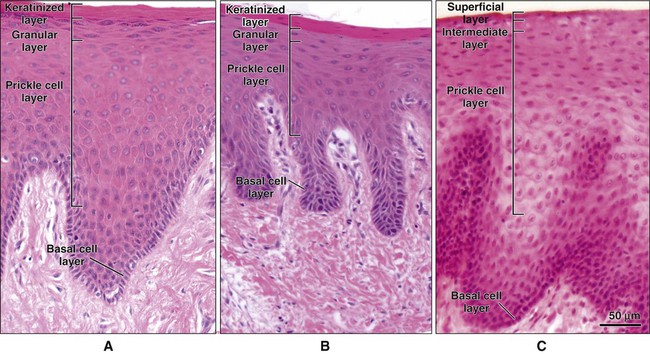
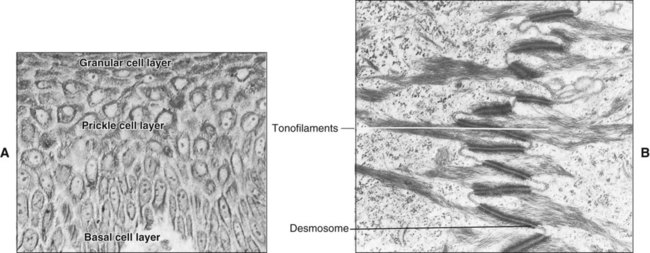
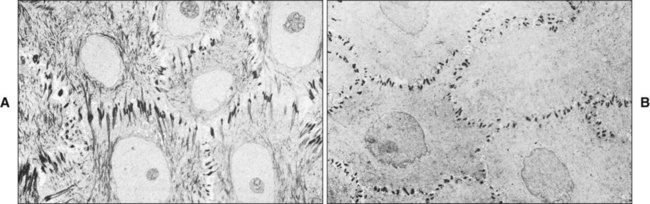
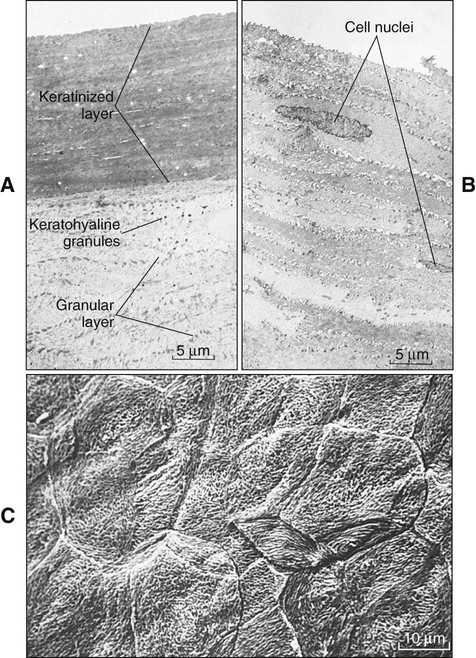
The epithelial surface of the masticatory mucosa (e.g., that of the hard palate and gingiva and in some regions of specialized mucosa on the dorsum of the tongue) is inflexible, tough, resistant to abrasion, and tightly bound to the lamina propria. It is covered by a layer of keratinized cells, and the process of maturation leading to its formation is called keratinization or cornification. In routine histologic sections a keratinized epithelium shows a number of distinct layers or strata (Figure 12-8, A). The basal layer or stratum basale is a layer of cuboidal or columnar cells adjacent to the basal lamina. Above the basal layer are several rows of larger elliptical or spherical cells known as the prickle cell layer or stratum spinosum. This term arises from the appearance of the cells in histologic preparation; they frequently shrink away from each other, remaining in contact only at points known as intercellular bridges or desmosomes (Figure 12-9). This alignment gives the cells a spiny or pricklelike profile.
The masticatory mucosa, parts of the hard palate and much of the gingiva, can show a variation of keratinization, known as parakeratinization. In parakeratinized epithelium (Figure 12-8, B), the surface layer stains for keratin, as described previously, but shrunken (or pyknotic) nuclei are retained in many or all of the squames. Keratohyalin granules may be present in the underlying granular layer, though usually fewer than in orthokeratinized areas, so that this layer is difficult to recognize in histologic preparations. Parakeratinization is a normal event in oral epithelium and does not imply disease; this is not true for epidermis, where parakeratinization may be associated with diseases such as psoriasis.
Nonkeratinization
The lining mucosa of the oral cavity, which is present on the lips, buccal mucosa, alveolar mucosa, soft palate, underside of the tongue, and floor of the mouth, has an epithelium that is usually nonkeratinized (Figure 12-8, C). In some regions, lips and buccal mucosa, the lining mucosa is thicker than keratinized epithelium and shows a different ridge pattern at the connective tissue interface. The basal and prickle cell layers of nonkeratinized oral epithelium generally resemble those described for keratinized epithelium, although the cells of nonkeratinized epithelium are slightly larger and the intercellular bridges or prickles are less conspicuous. For this reason, some prefer to avoid the term prickle cell layer for nonkeratinized epithelium. No sudden changes in the appearance of cells above the prickle cell layer occur in nonkeratinized epithelium, and the outer half of the tissue is divided rather arbitrarily into two zones: intermediate (stratum intermedium) and superficial (stratum superficiale). A granular layer is not present, and the cells of the superficial layer contain nuclei that are often plump. This layer does not stain intensely with eosin, as does the surface of keratinized or parakeratinized epithelium.
From the histologic appearance of oral epithelium it is apparent that the tissue shows a well-ordered pattern of maturation and successive layers that contain cells of increasing age (i.e., progressive stages of maturation). Furthermore, the pattern of maturation differs in different regions of the oral mucosa so that two main types can be recognized: keratinization and nonkeratinization. The next section describes the fine structure of the epithelial cell and the main events that take place at the cellular level during maturation of these two types of epithelium (Table 12-2; see also Table 12-1).
TABLE 12-2
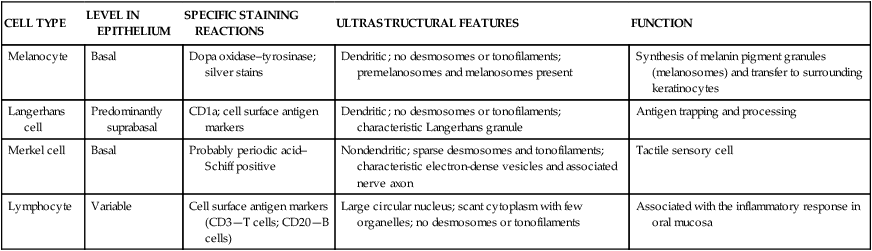
Characteristics of Nonkeratinocytes in Oral Epithelium
| CELL TYPE | LEVEL IN EPITHELIUM | SPECIFIC STAINING REACTIONS | ULTRASTRUCTURAL FEATURES | FUNCTION |
| Melanocyte | Basal | Dopa oxidase–tyrosinase; silver stains | Dendritic; no desmosomes or tonofilaments; premelanosomes and melanosomes present | Synthesis of melanin pigment granules (melanosomes) and transfer to surrounding keratinocytes |
| Langerhans cell | Predominantly suprabasal | CD1a; cell surface antigen markers | Dendritic; no desmosomes or tonofilaments; characteristic Langerhans granule | Antigen trapping and processing |
| Merkel cell | Basal | Probably periodic acid–Schiff positive | Nondendritic; sparse desmosomes and tonofilaments; characteristic electron-dense vesicles and associated nerve axon | Tactile sensory cell |
| Lymphocyte | Variable | Cell surface antigen markers (CD3—T cells; CD20—B cells) | Large circular nucleus; scant cytoplasm with few organelles; no desmosomes or tonofilaments | Associated with the inflammatory response in oral mucosa |
Ultrastructure of the Epithelial Cell
Cells of the basal layer are the least differentiated oral epithelial cells. They contain typical organelles present in the cells of other tissues as well as certain characteristic structures that identify them as epithelial cells and distinguish them from other cell types. These structures are the filamentous strands called tonofilaments and the intercellular bridges or desmosomes (see Chapter 4 and Figure 12-9, B). One name often given to an epithelial cell because of its content of keratin filaments is keratinocyte. This serves to distinguish these epithelial cells from the nonkeratinocytes that are described later.
Keratins represent a large family of proteins of differing molecular weights; those with the lowest molecular weight (40 kDa) are found in glandular and simple epithelia; those of intermediate molecular weight, in stratified epithelia; and those with the highest molecular weight (approximately 67 kDa), in keratinized stratified epithelia. A catalog of keratins has been drawn up to represent the different types. Thus all stratified oral epithelia possess keratins 5 and 14, but differences emerge between keratinized oral epithelium (which contains keratins 1, 6, 10, and 16) and nonkeratinized epithelium (which contains keratins 4, 13, and 19). An important property of any epithelium is its ability to function as a barrier, which depends to a great extent on the close contact or cohesiveness of the epithelial cells. Cohesion between cells is provided by a viscous intercellular material consisting of protein-carbohydrate complexes produced by the epithelial cells themselves. In addition, modifications of the adjacent membranes of cells occur, the most common of which is the desmosome or macula adherens (Figure 12-9, B) into which bundles of intermediate filaments (tonofilaments) insert (see Chapter 4). Adhesion between the epithelium and connective tissue is provided by hemidesmosomes, which attach the cell to the basal lamina (see Figure 12-20). Like desmosomes, hemidesmosomes also possess intracellular attachment plaques with tonofilaments inserted into them. Tonofilaments, (hemi) desmosomes, and basal lamina together represent a mechanical linkage that distributes and dissipates localized forces applied to the epithelial surface over a wide area. As discussed in Chapter 4, disease such as pemphigus can lead to the breakdown of this linkage and cause splitting of the epithelial layers.
Two other types of connection are seen between cells of the oral epithelium: gap junctions and tight junctions. As shown in Chapter 4, the gap junction is a region where membranes of adjacent cells run closely together, separated by only a small gap. Small interconnections are apparent between the membranes across these gaps. Such junctions may allow electrical or chemical communication between the cells and sometimes are called communicating junctions and are seen only occasionally in oral epithelium. Even rarer in oral epithelium is the tight, or occluding, junction, where adjacent cell membranes are so tightly apposed as to exclude intercellular space.
Cellular Events in Maturation
The major changes involved in cell maturation in keratinized and nonkeratinized oral epithelium are presented in Figure 12-10 and Table 12-1. In both types of epithelia the changes in cell size and shape are accompanied by a synthesis of more structural protein in the form of tonofilaments, the appearance of new organelles, and the production of additional intercellular material. A number of changes, however, are not common in both epithelia and serve as distinguishing features. One is in the arrangement of tonofilaments. The cells of both epithelia increase in size as they migrate from the basal to the prickle cell layer, but this increase is greater in nonkeratinized epithelium. A corresponding synthesis of tonofilaments also occurs in both epithelia, but whereas the tonofilaments in keratinized epithelium are aggregated into bundles to form tonofibrils, those in nonkeratinized epithelium remain dispersed and so appear less conspicuous (Figure 12-11). The chemical structure of keratin filaments also is known to differ between layers so that various patterns of maturation can be identified by the keratins that are present.
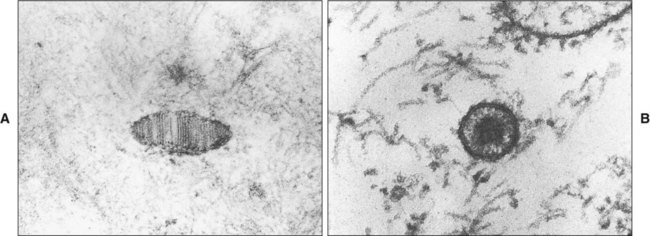
In the upper part of the prickle cell layer a new organelle appears, called the membrane-coating or lamellate granule. These granules are small, membrane-bound structures containing glycolipid. In keratinized epithelium the granules are elongated and exhibit a series of parallel lamellae. In nonkeratinized epithelium, by contrast, the granules appear to be circular with an amorphous core (Figure 12-12). As the cells move toward the surface, these granules accumulate close to the cell membrane where they release lipids that participate in establishing a permeability barrier.
Cells in the more superficial part of the granular layer develop a cornified cell envelope on the inner (intracellular) aspect of their membrane that contributes to the considerable resistance of the keratinized layer to chemical solvents (see page 283, Figure 12-7). One of the major constituents of this thickening is a protein known as involucrin. A similar, but less obvious, thickening often is seen in the surface cells of nonkeratinized epithelia. The remaining events during epithelial maturation are greatly different in keratinized and nonkeratinized epithelia and so are described separately.
Keratinized Epithelium
The most characteristic feature of the granular layer of keratinized epithelium is the keratohyalin granules, which appear as basophilic granules under the light microscope and as electron-dense structures in the electron microscope (Figure 12-13). The granules are irregular in shape and probably are synthesized by the ribosomes that can be seen surrounding them. Keratohyalin granules also are associated intimately with tonofibrils, and they are thought to facilitate the aggregation and formation of cross-links between the cytokeratin filaments of the keratinized layer. For this reason the protein making up the bulk of these granules has been named filaggrin, although they may also comprise a sulfur-rich component also called loricrin. As the cells of the granular layer reach the junction with the keratinized layer, a sudden change in their appearance occurs (Figure 12-14, A). All the organelles, including the nuclei and keratohyalin granules, disappear. The cells of the keratinized layer become packed with filaments cross-linked by disulfide bonds, which facilitates their dense packing. As part of the process, the cells modify their desmosomes into corneodesmosomes.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses