Influence of Systemic Conditions
Many systemic diseases, disorders, and conditions have been implicated as risk indicators or risk factors in periodontal disease. Clinical and basic science research over the past several decades has led to an improved understanding of and appreciation for the complexity and pathogenesis of periodontal diseases.187 Although there is clear evidence for a bacterial etiology and there are specific bacteria (periodontal pathogens) associated with destructive periodontal disease, the presence of these pathogens does not invariably cause disease. Their absence, on the other hand, appears to be consistent with periodontal health. The role of bacteria in disease etiology and pathogenesis is discussed in Chapters 8 and 9.
Recent evidence also suggests that periodontal infections can adversely affect systemic health with manifestations such as coronary heart disease, stroke, diabetes, preterm labor, low-birth-weight delivery, and respiratory disease.167 The role of periodontal infections on these systemic health conditions is discussed in Chapter 12.
Endocrine Disorders and Hormonal Changes
Diabetes Mellitus
Diabetes mellitus is an extremely important disease from a periodontal standpoint. It is a complex metabolic disorder characterized by chronic hyperglycemia. Diminished insulin production, impaired insulin action, or a combination of both result in the inability of glucose to be transported from the bloodstream into the tissues, which in turn results in high blood glucose levels and the excretion of sugar in the urine. Lipid and protein metabolism are altered in diabetes as well. Uncontrolled diabetes (chronic hyperglycemia) is associated with several long-term complications, including microvascular diseases (retinopathy, nephropathy, or neuropathy), macrovascular diseases (cardiovascular and cerebrovascular conditions), an increased susceptibility to infections, and poor wound healing. An estimated 25.8 million individuals (both children and adults)—8.3% of the US population—have diabetes.42 Approximately 7 million of these individuals are unaware that they have the disease.
Oral Manifestations.
Numerous oral changes have been described in patients with diabetes, including cheilosis, mucosal drying and cracking, burning mouth and tongue, diminished salivary flow, and alterations in the flora of the oral cavity, with greater predominance of Candida albicans, hemolytic streptococci, and staphylococci.2,22,101,162 An increased rate of dental caries has also been observed in patients with poorly controlled diabetes.76,86 It is important to note that these changes are not always present, that they are not specific, and that they are not pathognomonic for diabetes.165 Furthermore, these changes are less likely to be observed in patients with well-controlled diabetes. Individuals with controlled diabetes have a normal tissue response, a normally developed dentition, a normal defense against infections, and no increase in the incidence of caries.239
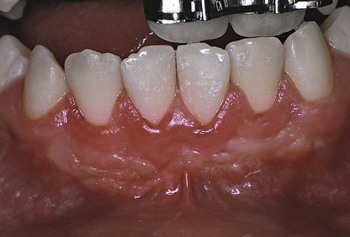
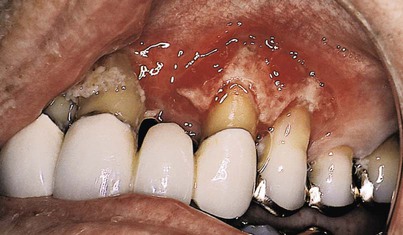
The influence of diabetes on the periodontium has been thoroughly investigated. Although it is difficult to make definitive conclusions about the specific effects of diabetes on the periodontium, a variety of changes have been described, including a tendency toward an enlarged gingiva, sessile or pedunculated gingival polyps, polypoid gingival proliferations, abscess formation, periodontitis, and loosened teeth114 (Figure 11-1). Perhaps the most striking changes in patients with uncontrolled diabetes are the reductions in the defense mechanisms and the increased susceptibility to infections, which lead to destructive periodontal disease. In fact, periodontal disease is considered to be the sixth complication of diabetes.148 Periodontitis in patients with type 1 diabetes appears to start after the age of 12 years, and it has a fivefold increased prevalence in teenagers.49 The prevalence of periodontitis has been reported as 9.8% in 13- to 18-year-old patients, and it increases to 39% in those who are 19 years old and older.
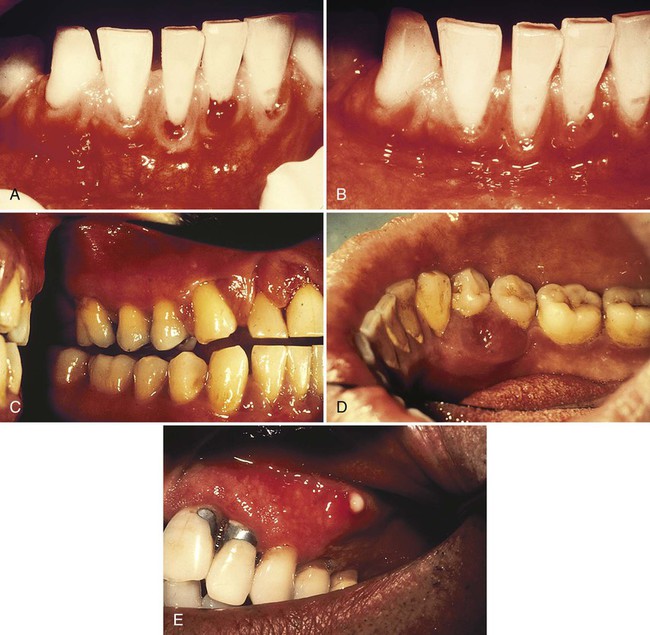
The extensive literature on this subject and the overall impression of clinicians indicate that periodontal disease in patients with diabetes follows no consistent or distinct pattern. Severe gingival inflammation, deep periodontal pockets, rapid bone loss, and frequent periodontal abscesses often occur in patients with poorly controlled diabetes and poor oral hygiene3 (Figures 11-2 and 11-3). Children with type 1 diabetes tend to have more destruction around the first molars and incisors, but this destruction becomes more generalized at older ages.48 In patients with juvenile diabetes, extensive periodontal destruction often occurs as a consequence of having more severe disease at a younger age.
Other investigators have reported that the rate of periodontal destruction appears to be similar for those with diabetes and those without diabetes up to the age of 30 years.92,233 After 30, patients with diabetes have a greater degree of periodontal destruction, possibly related to more disease destruction over time. Patients who have had overt diabetes for more than 10 years have a greater loss of periodontal support than those with a diabetic history of less than 10 years.92 This destruction may also be related to the diminished tissue integrity that continues to deteriorate over time (see later sections of this chapter for a description of altered collagen metabolism).
Although some studies have not found a correlation between the diabetic state and the periodontal condition, the majority of well-controlled studies show a higher prevalence and severity of periodontal disease in individuals with diabetes as compared with nondiabetic persons with similar local factors.14,22,39,52,111,177,179,180,236 Findings include a greater loss of attachment, increased bleeding on probing, and increased tooth mobility. A study of risk indicators for a group of 1426 patients between the ages of 25 and 74 years revealed that individuals with diabetes are twice as likely to exhibit attachment loss as nondiabetic individuals.108 The lack of consistency across studies is most likely related to the different degrees of diabetic involvement, variations in the level of disease control and the diversity of indices, and patient sampling from one study to another.
Recent studies have suggested that uncontrolled or poorly controlled diabetes is associated with an increased susceptibility to and severity of infections, including periodontitis.16,211 Adults who are 45 years old or older with poorly controlled diabetes (i.e., with a glycated hemoglobin level of >9%) were 2.9 times more likely to have severe periodontitis than those without diabetes. The likelihood was even greater (4.6 times) among smokers with poorly controlled diabetes.42 As with other systemic conditions associated with periodontitis, diabetes mellitus does not cause gingivitis or periodontitis, but evidence indicates that it alters the response of the periodontal tissues to local factors, thereby hastening bone loss and delaying postsurgical healing. Frequent periodontal abscesses appear to be an important feature of periodontal disease in patients with diabetes.
Approximately 40% of adult Pima Indians in Arizona have type 2 diabetes. A comparison of individuals with or without diabetes in this Native American tribe has shown a clear increase in the prevalence of destructive periodontitis as well as a 15% increase in edentulousness among patients with diabetes.222 The risk of developing destructive periodontitis increases threefold in these individuals.73
Bacterial Pathogens.
The glucose content of gingival fluid and blood is higher in individuals with diabetes than in those without diabetes with similar plaque and gingival index scores.79 The increased glucose in the gingival fluid and blood of patients with diabetes could change the environment of the microflora, thereby inducing qualitative changes in bacteria that may contribute to the severity of periodontal disease observed in those with poorly controlled diabetes.
Patients with type 1 diabetes mellitus and periodontitis have been reported to have a subgingival flora that is composed mainly of Capnocytophaga, anaerobic vibrios, and Actinomyces species. Porphyromonas gingivalis, Prevotella intermedia, and Aggregatibacter actinomycetemcomitans, which are common in periodontal lesions of individuals without diabetes, are present in low numbers in those with the disease.110,163 However, other studies have found scarce Capnocytophaga and abundant A. actinomycetemcomitans and black-pigmented Bacteroides as well as P. intermedia, P. melaninogenica, and Campylobacter rectus.162,214 Black-pigmented species—especially P. gingivalis, P. intermedia, and C. rectus—are prominent in severe periodontal lesions of Pima Indians with type 2 diabetes.89,267 Although these results may suggest an altered flora in the periodontal pockets of patients with diabetes, the exact role of these microorganisms has not been determined. To date, there is insufficient evidence to support the role of a specific altered microflora that is responsible for periodontal disease destruction in patients with diabetes.
Polymorphonuclear Leukocyte Function.
The increased susceptibility of patients with diabetes to infection has been hypothesized as being caused by polymorphonuclear leukocyte (PMN) deficiencies that result in impaired chemotaxis, defective phagocytosis, or impaired adherence.166,236 In patients with poorly controlled diabetes, the function of PMNs, monocytes, and macrophages is impaired.118 As a result, the primary defense mounted by PMNs against periodontal pathogens is diminished, and bacterial proliferation is more likely. No alteration of immunoglobulin A (IgA), G (IgG), or M (IgM) has been found in patients with diabetes.204
Altered Collagen Metabolism.
Chronic hyperglycemia impairs collagen structure and function, which may directly impact the integrity of the periodontium. Decreased collagen synthesis, osteoporosis, and a reduction in alveolar bone height have been demonstrated in diabetic animals.93,217 Chronic hyperglycemia adversely affects the synthesis, maturation, and maintenance of collagen and extracellular matrix. In the hyperglycemic state, numerous proteins and matrix molecules undergo a nonenzymatic glycosylation, thereby resulting in accumulated glycation end-products (AGEs). The formation of AGEs occurs at normal glucose levels as well; however, in hyperglycemic environments, AGE formation is excessive. Many types of molecules are affected, including proteins, lipids, and carbohydrates. Collagen is cross-linked by AGE formation, which makes the collagen less soluble and less likely to be normally repaired or replaced. Cellular migration through cross-linked collagen is impeded, and, perhaps more importantly, tissue integrity is impaired as a result of damaged collagen that remains in the tissues for longer periods (i.e., collagen is not renewed at a normal rate).108 As a result, collagen in the tissues of patients with poorly controlled diabetes is older and more susceptible to pathogenic breakdown (i.e., less resistant to destruction by periodontal infections).
AGEs and receptors for AGEs (RAGEs) play a central role in the classic complications of diabetes,33 and they may play a significant role in the progression of periodontal disease as well. Poor glycemic control, with the associated increase in AGEs, renders the periodontal tissues more susceptible to destruction.216 The cumulative effects of altered cellular response to local factors, impaired tissue integrity, and altered collagen metabolism undoubtedly play a significant role in the susceptibility of patients with diabetes to infections and destructive periodontal disease.
Female Sex Hormones
Gingival alterations during puberty, pregnancy, and menopause are associated with physiologic hormonal changes in the female patient. During puberty and pregnancy, these changes are characterized by nonspecific inflammatory reactions with a predominant vascular component, which leads clinically to a marked hemorrhagic tendency. Oral changes during menopause may include thinning of the oral mucosa, gingival recession, xerostomia, altered taste, and burning mouth. The changes associated with each phase of the female life cycle from puberty to menopause are briefly addressed in the online materials associated with this book. See Chapter 38 for a detailed description of these changes, including management considerations for the periodontal manifestations of hormonal changes in the female patient.
Puberty.
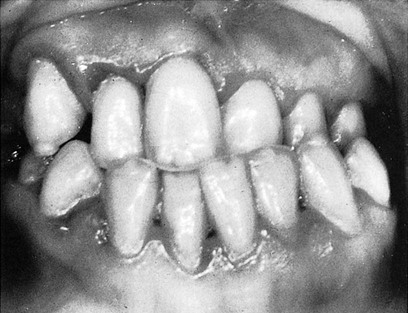
Puberty is often accompanied by an exaggerated response of the gingiva to plaque.230 Pronounced inflammation, edema, and gingival enlargement result from local factors that might ordinarily elicit a comparatively mild gingival response (Figure 11-4). As adulthood approaches, the severity of the gingival reaction diminishes, even when local factors persist. However, the complete return to normal health requires the removal of these factors. Although the prevalence and severity of gingival disease are increased during puberty, gingivitis is not a universal occurrence for all adolescents. With good oral hygiene, it can be prevented (see Chapter 18).
Menstruation.
During the menstrual period, the prevalence of gingivitis increases. Some patients may complain of bleeding gums or a bloated, tense feeling in the gums during the days preceding menstrual flow. The exudate from inflamed gingiva is increased during menstruation, which suggests that preexisting gingivitis is aggravated by menstruation; however, the crevicular fluid of normal, healthy gingiva is unaffected.116 Tooth mobility does not change significantly during the menstrual cycle.85 The salivary bacterial count is increased during menstruation and at ovulation, which occurs up to 14 days earlier.196
Pregnancy.
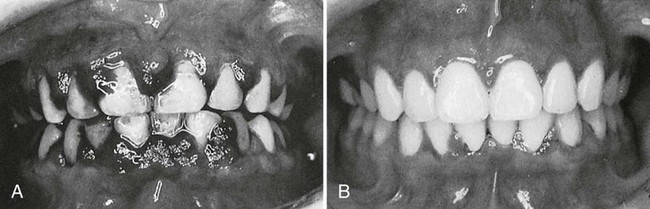
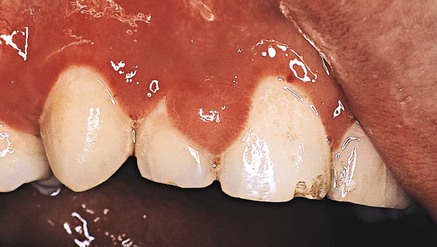
The gingival changes of pregnancy were described as early as the late 1800s, even before any knowledge about hormonal changes in pregnancy was available.25,195 As with other systemic conditions, pregnancy itself does not cause gingivitis. Gingivitis during pregnancy is caused by bacterial plaque, just as it is in nonpregnant women. The hormonal changes of pregnancy accentuate the gingival response to plaque and modify the resultant clinical picture (Figure 11-5). No notable changes occur in the gingiva during pregnancy in the absence of local factors.
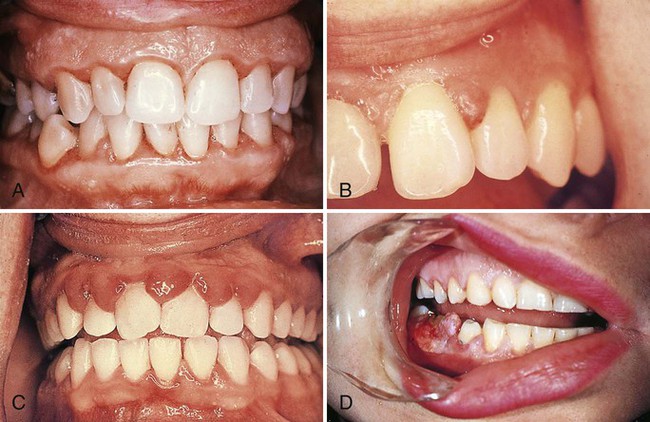
The reported incidence of gingivitis during pregnancy in well-conducted studies varies from 50% to 100%.147,154 Pregnancy affects the severity of previously inflamed areas, but it does not alter healthy gingiva. Impressions of increased incidence may be created by the aggravation of previously inflamed but unnoticed areas. Tooth mobility, pocket depth, and gingival fluid are also increased during pregnancy.117,144,198 The severity of gingivitis is increased during pregnancy beginning in the second or third month. Patients with mild chronic gingivitis that attracted no particular attention before the pregnancy become aware of the gingiva because previously inflamed areas become enlarged, edematous, and more notably discolored (Figure 11-5, A to C).
Gingivitis becomes more severe by the eighth month and decreases during the ninth month of pregnancy.147 Plaque accumulation follows a similar pattern. Some investigators report that the greatest severity is between the second and third trimesters.53 The correlation between gingivitis and the quantity of plaque is greater after parturition than during pregnancy, which suggests that pregnancy introduces other factors that aggravate the gingival response to local factors.135
Partial reduction in the severity of gingivitis occurs by 2 months postpartum, and, after 1 year, the condition of the gingiva is comparable to that of patients who have not been pregnant.53 Tooth mobility, pocket depth, and gingival fluid are also reduced after pregnancy. In a longitudinal investigation of the periodontal changes that occurred during pregnancy and for 15 months postpartum, no significant loss of attachment was observed.53
Pronounced ease of bleeding is the most striking clinical feature. The gingiva is inflamed and varies in color from bright red to bluish red.268,269 The marginal and interdental gingivae are edematous; they pit on pressure, appear smooth and shiny, are soft and pliable, and sometimes have a raspberry-like appearance. The extreme redness results from marked vascularity, and there is an increased tendency to bleed. The gingival changes are usually painless unless that are complicated by acute infection. In some cases, the inflamed gingiva forms discrete “tumorlike” masses, which are referred to as pregnancy tumors (Figure 11-5, D). Microscopically, gingival disease during pregnancy appears as nonspecific, vascularizing, and proliferative inflammation.154,269 Marked inflammatory cellular infiltration occurs, with edema and degeneration of the gingival epithelium and connective tissue. The epithelium is hyperplastic, with accentuated rete pegs, reduced surface keratinization, and various degrees of intracellular and extracellular edema and infiltration by leukocytes.244 Newly formed engorged capillaries are present in abundance.
The possibility that bacterial–hormonal interactions may change the composition of plaque and lead to gingival inflammation has not been extensively explored. Kornman and Loesche136 reported that the subgingival flora changes to a more anaerobic flora as pregnancy progresses. P. intermedia appears to be the only microorganism that increases significantly during pregnancy. This increase appears to be associated with elevations in systemic levels of estradiol and progesterone and to coincide with the peak in gingival bleeding. It has also been suggested that, during pregnancy, a depression of the maternal T-lymphocyte response may be a factor in the altered tissue response to plaque.181
The aggravation of gingivitis during pregnancy has been attributed principally to the increased levels of progesterone, which produce dilation and tortuosity of the gingival microvasculature, circulatory stasis, and increased susceptibility to mechanical irritation, all of which favor the leakage of fluid into the perivascular tissues.174,180 A marked increase in estrogen and progesterone occurs during pregnancy, with a reduction after parturition. Animal studies with radioactive estradiol have demonstrated that the gingiva is a target organ for female sex hormones.82 The severity of gingivitis varies with the hormonal levels of pregnancy.117
It has also been suggested that the accentuation of gingivitis during pregnancy occurs has two peaks: during the first trimester, when there is an overproduction of gonadotropins, and during the third trimester, when estrogen and progesterone levels are highest.147 The destruction of gingival mast cells by the increased sex hormones and the resultant release of histamine and proteolytic enzymes may also contribute to the exaggerated inflammatory response to local factors.146
Hormonal Contraceptives.
Hormonal contraceptives aggravate the gingival response to local factors in a manner similar to that seen during pregnancy; when these drugs are taken for more than 1.5 years, there is an increase in periodontal destruction.69,134,145 Although some brands of oral contraceptives produce more dramatic changes than others, no correlation has been found to exist on the basis of differences in the progesterone or estrogen content of various brands.154,193 Cumulative exposure to oral contraceptives apparently has no effect on gingival inflammation or oral debris index scores.125
Menopause.
During menopause, the usual rhythmic hormonal fluctuations of the female cycle are ended as estradiol ceases to be the major circulating estrogen.168 As a result, females can develop a gingivostomatitis. This condition occurs during menopause or during the postmenopausal period. Mild signs and symptoms sometimes appear, and these are associated with the earliest menopausal changes. Menopausal gingivostomatitis is not a common condition. The term used for its designation has led to the erroneous impression that it invariably occurs in association with menopause, whereas the opposite is true. Oral disturbances are not a common feature of menopause.260
The gingiva and the remaining oral mucosa are dry and shiny, they vary in color from abnormal paleness to redness, and they bleed easily. Fissuring occurs in the mucobuccal fold in some women, and comparable changes may occur in the vaginal mucosa.202 Microscopically, the gingiva exhibits atrophy of the germinal and prickle cell layers of the epithelium and, in some patients, areas of ulceration. The patient complains of a dry, burning sensation throughout the oral cavity that is associated with extreme sensitivity to thermal changes; abnormal taste sensations described as “salty,” “peppery,” or “sour”; and difficulty with removable partial prostheses.164
The signs and symptoms of menopausal gingivostomatitis are somewhat comparable with those of chronic desquamative gingivitis (see Chapter 19). Signs and symptoms similar to those of menopausal gingivostomatitis occasionally occur after ovariectomy or sterilization by radiation in the treatment of malignant neoplasms.
Hyperparathyroidism
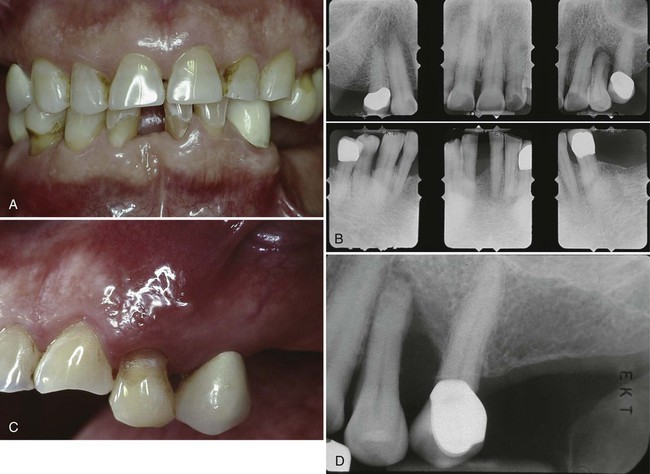
Parathyroid hypersecretion produces generalized demineralization of the skeleton, increased osteoclasis with proliferation of the connective tissue in the enlarged marrow spaces, and the formation of bone cysts and giant cell tumors.255 The disease is called osteitis fibrosa cystica or von Recklinghausen’s bone disease. Loss of the lamina dura and giant cell tumors in the jaws are late signs of hyperparathyroid bone disease, which in itself is uncommon. The complete loss of the lamina dura does not occur often, and clinicians may attach too much diagnostic significance to it. The loss of lamina dura may also occur with Paget’s disease, fibrous dysplasia, and osteomalacia.
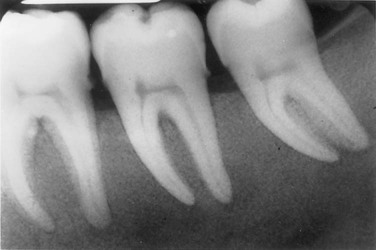
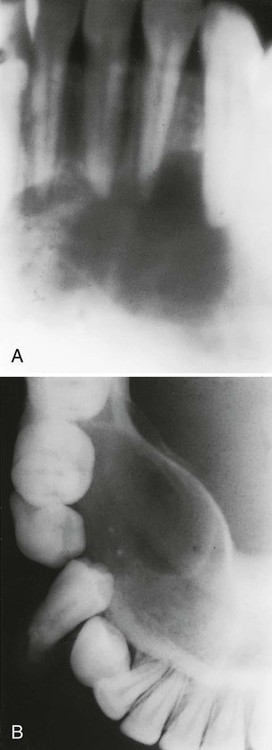
Reports have suggested that 25% to 50% of patients with hyperparathyroidism have associated oral changes.205,225,229 These changes include malocclusion and tooth mobility, radiographic evidence of alveolar osteoporosis with closely meshed trabeculae, widening of the periodontal ligament space, absence of the lamina dura (Figure 11-6), and radiolucent cystlike spaces (Figure 11-7). Bone cysts become filled with fibrous tissue with abundant hemosiderin-laden macrophages and giant cells. These cysts have been called brown tumors, but they are not tumors. More accurately, these cysts are reparative giant cell granulomas. In some cases, these lesions appear in the periapical region of teeth, and they can lead to a misdiagnosis of a lesion of endodontic origin.149 A relationship has been suggested between periodontal disease in dogs and hyperparathyroidism secondary to calcium deficiency in the diet,112 but this has not been confirmed by other studies.231
Hematologic Disorders and Immune Deficiencies
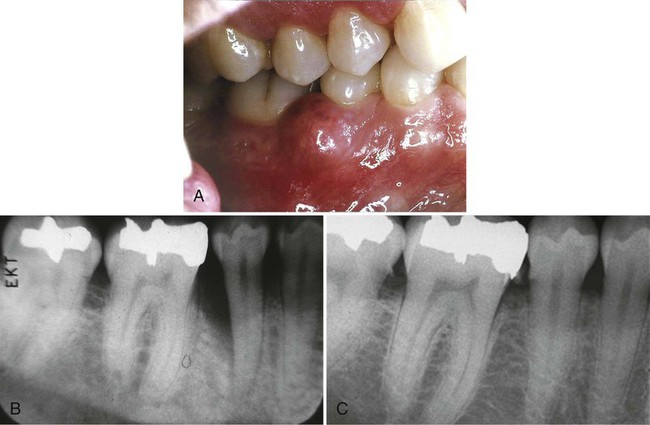
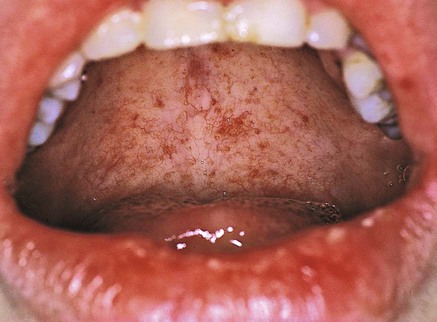
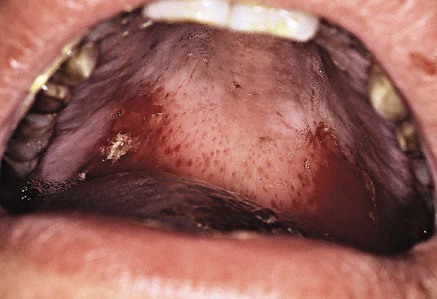
Gingival and periodontal disturbances associated with blood dyscrasias must be viewed in terms of fundamental interrelationships among the oral tissues, the blood cells, and the blood-forming organs rather than in terms of a simple association of dramatic oral changes with hematologic disease. Hemorrhagic tendencies occur when the normal hemostatic mechanisms are disturbed. Abnormal bleeding from the gingiva or other areas of the oral mucosa that is difficult to control is an important clinical sign that suggests a hematologic disorder. Petechiae (Figure 11-8) and ecchymosis (Figure 11-9) observed most often in the soft palate area are signs of an underlying bleeding disorder. It is essential to diagnose the specific etiology to appropriately address any bleeding or immunological disorder.
Deficiencies in the host immune response may lead to severely destructive periodontal lesions. These deficiencies may be primary (inherited) or secondary (acquired) and caused by either immunosuppressive drug therapy or the pathologic destruction of the lymphoid system. Leukemia, Hodgkin’s disease, lymphomas, and multiple myeloma may result in secondary immunodeficiency disorders. This section discusses common hematologic and certain immunodeficiency disorders that are not related to the human immunodeficiency virus or acquired immunodeficiency syndrome. See Chapter 26 for a detailed discussion of patients with human immunodeficiency virus infection.
Leukocyte (Neutrophil) Disorders
Disorders that affect the production or function of leukocytes may result in severe periodontal destruction. PMNs (neutrophils) in particular play a critical role in bacterial infections, because PMNs are the first line of defense (see Chapter 9). A quantitative deficiency of leukocytes (e.g., neutropenia, agranulocytosis) is typically associated with a more generalized periodontal destruction that affects all teeth.
Agranulocytosis.
Drug idiosyncrasy is the most common cause of agranulocytosis, but, in some cases, its cause cannot be explained. Agranulocytosis has been reported after the administration of drugs such as aminopyrine, barbiturates and their derivatives, benzene ring derivatives, sulfonamides, gold salts, and arsenical agents.137,153,170,197 It generally occurs as an acute disease. It may be chronic or periodic, with recurring neutropenic cycles (e.g., cyclic neutropenia).237
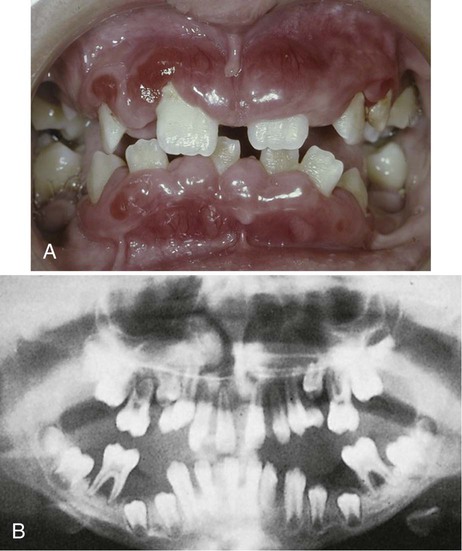
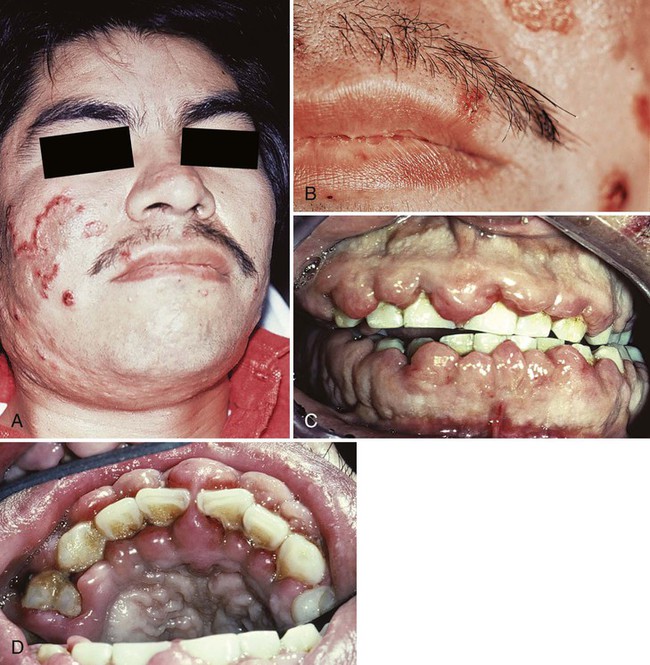
The onset of disease is accompanied by fever, malaise, general weakness, and sore throat. Ulceration in the oral cavity, the oropharynx, and the throat is characteristic. The mucosa exhibits isolated necrotic patches that are black and gray and that are sharply demarcated from the adjacent uninvolved areas.128,157 The absence of a notable inflammatory reaction caused by a lack of granulocytes is a striking feature. The gingival margin may or may not be involved. Gingival hemorrhage, necrosis, increased salivation, and fetid odor are accompanying clinical features. With cyclic neutropenia, the gingival changes recur with recurrent exacerbation of the disease.51 The occurrence of generalized aggressive periodontitis has been described in patients with cyclic neutropenia220 (Figure 11-10).
Leukemia
Leukemia is an important disease to understand and appreciate because of its seriousness and its periodontal manifestations. The leukemias are malignant neoplasias of WBC precursors that are characterized by the following: (1) the diffuse replacement of the bone marrow with proliferating leukemic cells; (2) abnormal numbers and forms of immature WBCs in the circulating blood; and (3) widespread infiltrates in the liver, spleen, lymph nodes, and other body sites.203
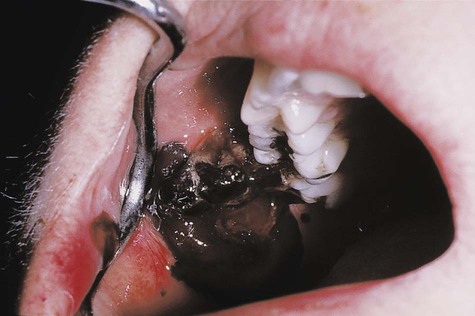
All leukemias tend to displace normal components of the bone marrow elements with leukemic cells, thereby resulting in the reduced production of normal RBCs, WBCs, and platelets, which leads to anemia, leukopenia (a reduction in the number of nonmalignant WBCs), and thrombocytopenia. Anemia results in poor tissue oxygenation, thereby making tissues more friable and susceptible to breakdown. A reduction of normal WBCs in the circulation leads to a poor cellular defense and an increased susceptibility to infections. Thrombocytopenia leads to bleeding tendency, which can occur in any tissue but which in particular affects the oral cavity, especially the gingival sulcus (Figure 11-11). Some patients may have normal blood counts while leukemic cells reside primarily in the bone marrow. This type of disease is called aleukemic leukemia.99
The Periodontium in Leukemic Patients.
Leukemic Infiltration.
Leukemic cells can infiltrate the gingiva and, less frequently, the alveolar bone. Gingival infiltration often results in leukemic gingival enlargement (see Chapter 16).
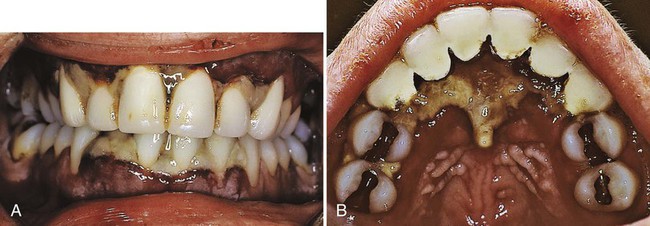
A study of 1076 adult patients with leukemia showed that 3.6% of the patients with teeth had leukemic gingival proliferative lesions, with the highest incidence seen in patients with acute monocytic leukemia (66.7%), followed by those acute myelocytic–monocytic leukemia (18.7%) and acute myelocytic leukemia (3.7%).65 It should be noted, however, that monocytic leukemia is an extremely rare form of the disease. Leukemic gingival enlargement is not found in edentulous patients or in patients with chronic leukemia, thereby suggesting that it is the accumulation of immature leukemic blast cells in the gingiva adjacent to tooth surfaces with bacterial plaque. Leukemic gingival enlargement consists of a basic infiltration of the gingival corium by leukemic cells that increases the gingival thickness and that creates gingival pockets in which bacterial plaque accumulates, thereby initiating a secondary inflammatory lesion that contributes to the enlargement of the gingiva. It may be localized to the interdental papilla area (Figure 11-12), or it may expand to include the marginal gingiva and partially cover the crowns of the teeth (Figure 11-13, C and D). Clinically, the gingiva appears bluish red and cyanotic, with a rounding and tenseness of the gingival margin. The abnormal accumulation of leukemic cells in the dermal and subcutaneous connective tissue is called leukemia cutis, and it forms elevated and flat macules and papules65,203 (Figure 11-13, A and B).
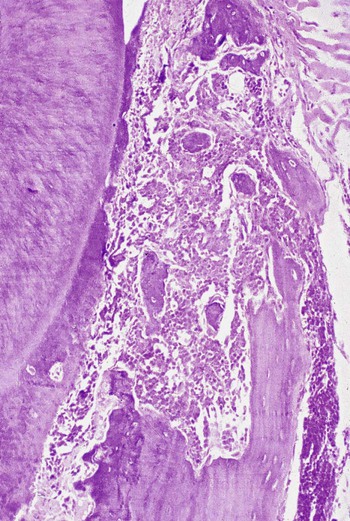
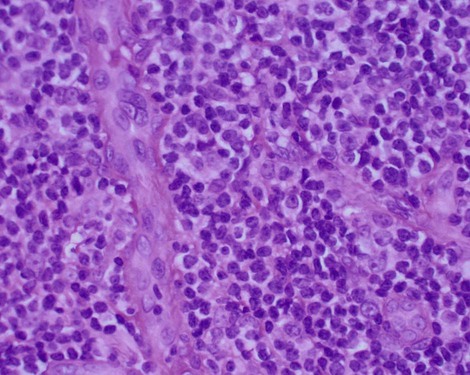
Microscopically, the gingiva exhibits a dense, diffuse infiltration of predominantly immature leukocytes in the attached and marginal gingiva. Occasionally, mitotic figures indicative of ectopic hematopoiesis may be seen. The normal connective tissue components of the gingiva are displaced by the leukemic cells (Figure 11-14). The nature of the cells depends on the type of leukemia. The cellular accumulation is denser in the entire reticular connective tissue layer. In almost all cases, the papillary layer contains comparatively few leukocytes. The blood vessels are distended and contain predominantly leukemic cells, and the RBCs are reduced in number. The epithelium presents a variety of changes, and it may be thinned or hyperplastic. Common findings include degeneration associated with intercellular and intracellular edema and leukocytic infiltration with diminished surface keratinization.
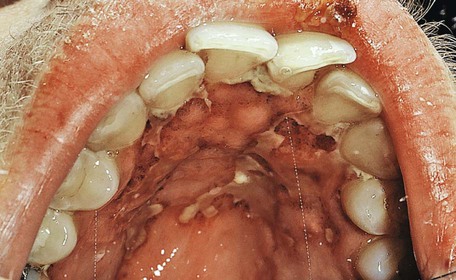
In leukemic mice, the presence of infiltrate in marrow spaces and the periodontal ligament results in osteoporosis of the alveolar bone with destruction of the supporting bone and disappearance of the periodontal fibers31,40 (Figure 11-15).
Bleeding.
Gingival hemorrhage is a common finding in leukemic patients (see Figure 11-11), even in the absence of clinically detectable gingivitis. Bleeding gingiva can be an early sign of leukemia. It is caused by the thrombocytopenia that results from the replacement of the bone marrow cells with leukemic cells and from the inhibition of normal stem cell function by leukemic cells or their products.203 This bleeding tendency can also manifest in the skin and throughout the oral mucosa, where petechiae are often found, with or without leukemic infiltrates. A more diffuse submucosal bleeding manifests as ecchymosis (see Figure 11-9). Oral bleeding has been reported as a presenting sign in 17.7% of patients with acute leukemia and in 4.4% of patients with chronic leukemia.152 Bleeding may also be a side effect of the chemotherapeutic agents used to treat leukemia.
Oral Ulceration and Infection.
Granulocytopenia (diminished WBC count) results from the displacement of normal bone marrow cells by leukemic cells, which increases the host susceptibility to opportunistic microorganisms and leads to ulcerations and infections. Discrete, punched-out ulcers that penetrate deeply into the submucosa and that are covered by a firmly attached white slough can be found on the oral mucosa.15 These lesions occur in sites of trauma (e.g., the buccal mucosa) in relation to the line of occlusion or on the palate. Patients with a history of herpesvirus infection may develop recurrent herpetic oral ulcers (often in multiple sites) and large atypical forms, especially after chemotherapy is instituted104 (Figure 11-16).
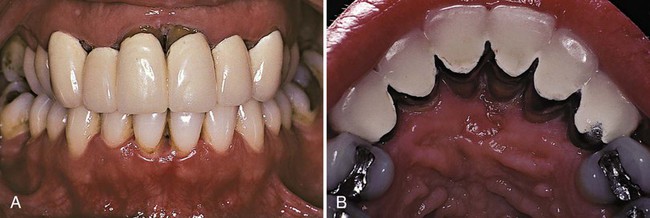
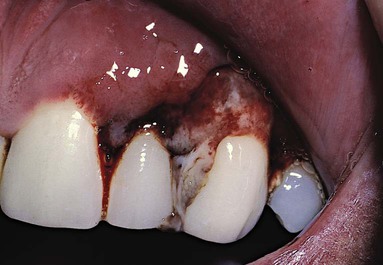
A gingival (bacterial) infection in leukemic patients can be the result of an exogenous bacterial infection or an existing bacterial infection (e.g., gingival or periodontal disease). Acute gingivitis and lesions that resemble necrotizing ulcerative gingivitis are more frequent and severe in patients with terminal cases of acute leukemia21 (Figures 11-17 and 11-18). The inflamed gingiva in patients with leukemia differs clinically from that found in nonleukemic individuals. The gingiva is a peculiar bluish red, it is spongelike and friable, and it bleeds persistently on the slightest provocation or even spontaneously in leukemic patients. This greatly altered and degenerated tissue is extremely susceptible to bacterial infection, which can be so severe as to cause acute gingival necrosis with pseudomembrane formation (Figure 11-19) or bone exposure (Figure 11-20). These are secondary oral changes that are superimposed on the oral tissues altered by the blood dyscrasia. They produce associated disturbances that may be a source of considerable difficulty to the patient, such as systemic toxic effects, loss of appetite, nausea, blood loss from persistent gingival bleeding, and constant gnawing pain. Eliminating or reducing local factors (e.g., bacterial plaque) can minimize the severe oral changes associated with leukemia. In some patients with severe acute leukemia, symptoms may only be relieved by treatment that leads to the remission of the disease.
Anemia
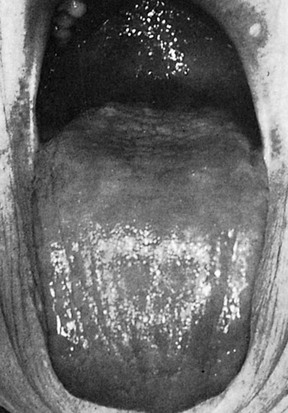
Pernicious anemia results in tongue changes in 75% of patients. The tongue appears red, smooth, and shiny as a result of atrophy of the papillae (Figure 11-21). There is also marked pallor of the gingiva (Figure 11-22). Iron deficiency anemia induces similar tongue and gingival changes. A syndrome that consists of glossitis and ulceration of the oral mucosa and oropharynx and that induces dysphagia (Plummer–Vinson syndrome) has been described in patients with iron deficiency anemia. Sickle cell anemia is a hereditary form of chronic hemolytic anemia that occurs almost exclusively in blacks. It is characterized by pallor, jaundice, weakness, rheumatoid manifestations, and leg ulcers. Oral changes include generalized osteoporosis of the jaws, with a peculiar stepladder alignment of the trabeculae of the interdental septa, along with pallor and yellowish discoloration of the oral mucosa. Periodontal infections may precipitate sickle cell crisis.197 Aplastic anemia results from a failure of the bone marrow to produce erythrocytes. The etiology is usually the effect of toxic drugs on the marrow or the displacement of RBCs by leukemic cells. Oral changes include pale discoloration of the oral mucosa and increased susceptibility to infection because of the concomitant neutropenia.
Thrombocytopenia
Thrombocytopenic purpura is characterized by a low platelet count, a prolonged clot retraction and bleeding time, and a normal or slightly prolonged clotting time. There is spontaneous bleeding into the skin or from the mucous membranes. Petechiae and hemorrhagic vesicles occur in the oral cavity, particularly in the palate, the tonsillar pillars, and the buccal mucosa. The gingivae are swollen, soft, and friable. Bleeding occurs spontaneously or with the slightest provocation, and it is difficult to control. Gingival changes represent an abnormal response to local irritation. The severity of the gingival condition is dramatically alleviated by removal of the local factors (Figure 11-23).
Antibody Deficiency Disorders
T-cell function remains normal in individuals with agammaglobulinemia. The disease, whether congenital or acquired, is characterized by recurrent bacterial infections, especially ear, sinus, and lung infections. Patients are also susceptible to periodontal infections. Aggressive periodontitis is a common finding in children who are diagnosed with agammaglobulinemia (see Figure 11-10).
Genetic Disorders
Many systemic conditions associated with or that predispose an individual to periodontal destruction include genetic disorders that result in an inadequate number or reduced function of circulating neutrophils. This underscores the importance of the neutrophil in the protection of the periodontium against infection. Severe periodontitis has been observed in individuals with primary neutrophil disorders such as neutropenia, agranulocytosis, Chédiak–Higashi syndrome, and lazy leukocyte syndrome. In addition, severe periodontitis has also been observed in individuals who exhibit secondary neutrophil impairment, which is seen in those with Down syndrome, Papillon–Lefèvre syndrome, and inflammatory bowel disease. See the online material for this chapter as well as for Chapter 6 for detailed descriptions of the periodontal manifestations of these genetic disorders and syndromes.
Chédiak–Higashi Syndrome
Chédiak–Higashi syndrome is a rare disease that affects the production of organelles found in almost every cell. It affects mostly the melanocytes, platelets, and phagocytes. It causes partial albinism, mild bleeding disorders, and recurrent bacterial infections. Neutrophils contain abnormal, giant lysosomes that can fuse with the phagosome, but their ability to release their contents is impaired. As a result, the killing of ingested microorganisms is delayed. Patients with Chédiak–Higashi syndrome are susceptible to repeated infections that can be serious and life threatening. Aggressive periodontitis has been described in these patients. Chédiak–Higashi syndrome has been described as a genetically transmitted disease in ranch-raised mink11,189 (see Chapter 6).
Lazy Leukocyte Syndrome
Lazy leukocyte syndrome is characterized by an individual’s susceptibility to severe microbial infections, neutropenia, defective chemotactic response by neutrophils, and an abnormal inflammatory response.192 Those who are diagnosed with lazy leukocyte syndrome are susceptible to aggressive periodontitis, with the destruction of bone and early tooth loss.
Leukocyte Adhesion Deficiency
Cases of periodontal disease that are attributed to LAD are rare. They begin during or immediately after the eruption of the primary teeth. Extremely acute inflammation and proliferation of the gingival tissues with the rapid destruction of bone are found. Profound defects in peripheral blood neutrophils and monocytes and an absence of neutrophils in the gingival tissues have been noted in patients with LAD.188,189 These patients also have frequent respiratory tract infections and sometimes otitis media. Both the primary and permanent teeth are affected, often resulting in early tooth loss.253
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses