11 Cements in Orthodontics

Photograph of the in vitro failure site for a bracket bonded with glass-ionomer cement, showing cohesive fracture through the adhesive where a layer of the material remains on the enamel
Setting Reaction and Structure
Working and Setting Times; Manipulative Variables; Film Thickness
Setting Reaction and Structure
Working and Setting Times; Manipulative Variables; Film Thickness
Setting Reaction and Structure
Working and Setting Times; Manipulative Variables; Film Thickness
Introduction
Dental cements are used for retention (luting) of preformed restorations and orthodontic bands, thermal and chemical insulators under restorative materials to provide pulpal protection, temporary or permanent restorations, and root canal sealers. This multiplicity of applications necessitates a wide variety of characteristics and a complex profile of properties. The cements may be classified by composition into four categories: (a) phosphate, which includes the zinc phosphate and silicophosphate cements; (b) phenolate, which includes the zinc oxide-eugenol and calcium hydroxide cements; (c) polycarboxylate, which includes the zinc polycarboxylate and glass-ionomer cements; and (d) resin cements. Some aspects of the glass-ionomer and resin cements have been discussed in Chapters 9 and 10.
The orthodontic application of cements is limited to luting of appliances. For acceptable performance, a luting material should possess a variety of properties: adequate working and setting times; high tensile, compressive and shear strengths; resistance to dissolution; clinically acceptable bond strength and low Adhesive Remnant Index (ARI) score (Chapter 5) following debonding; and anticariogenic potential.
Zinc Phosphate Cements
Zinc phosphate cements are widely used for cementation of orthodontic bands. These cements are generally available as hand-mixed powder-liquid systems, although some encapsulated products are marketed.
Composition
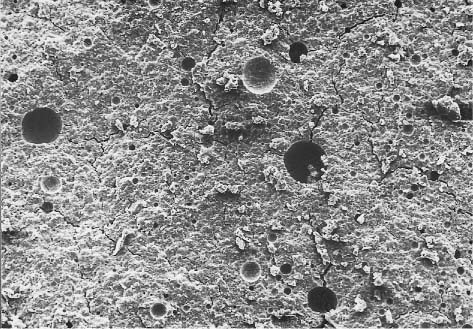
The principal active constituent of the cement powder is zinc oxide (Fig. 11.1). A small quantity of magnesium oxide may also be identified in some currently available products. Various cement powder formulations may include small amounts of additives such as silica or alumina, while other brands contain approximately 10% fluoride in the form of stannous fluoride. The latter products are generally recommended for cementation of orthodontic bands because of their anticariogenic effect from the release of fluoride (Chapter 6). The addition of magnesium oxide to these cements results in improvement of mechanical properties as well as color stability. The additions of silica and alumina also increase the mechanical properties, while providing a variety of shades for these products.
The components of the powder are sintered at 1000°–1350 °C for several hours and then ground into fine particles. This procedure reduces the reactivity of the powder and moderates the setting reaction, yielding adequate working and setting times for the cement. The magnesium oxide facilitates sintering of the zinc oxide powder; smaller powder particles produce a more rapidly setting cement.
The liquid is essentially an aqueous solution of phosphoric acid in concentrations varying from 45% to 64%, buffered by small quantities (2–3%) of aluminum phosphate and, in some products, zinc phosphate (1–9%). The metallic salts form an amorphous phosphate, resulting in stabilization of the pH of the liquid and reduction of the reaction rate; this allows the mixing of a higher quantity of powder with the liquid. The water content influences the setting rate of the cement, since it regulates the ionization of the liquid.
Setting Reaction and Structure
When the cement powder and the aqueous liquid are brought together, the acid attacks and dissolves the outer layer of the zinc oxide particles, releasing zinc ions into the liquid. At this stage, a rapid, vigorous exothermic reaction occurs, leading to the formation of hydrated zinc phosphate, as follows:
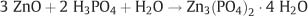
The course of the reaction is shown by the pH changes of the cement. Two minutes after mixing the pH is 1.6; after one hour the pH increases to about 4.0, and after 24 hours it ranges between 6.0 and 7.0. Although the role of aluminum in the setting reaction has not been clearly defined, it has been suggested that aluminum may produce complexes with phosphoric acid, forming a glassy zinc aluminophosphate gel on the surface of the unreacted zinc oxide particles. The incorporation of buffers into the liquid, as well as the method employed by the manufacturer for production of the zinc oxide powder, may moderate the reaction rate. The structure of the set cement consists of residual zinc oxide particles (termed “core” in some dental materials textbooks) bound together with a matrix (reaction products) of an amorphous, relatively insoluble gel of zinc, magnesium, and aluminum phosphates. The fully set cement exhibits porosity as a result of entrapment of air during mixing of the powder and liquid (Fig. 11.2).
Fig. 11.1 Secondary electron image of the powder particles of a zinc phosphate cement. Original magnification × 1000
Fig. 11.2 Secondary electron image of porosity in a set (48 hour) zinc phosphate cement. Original magnification × 200
Working and Setting Times; Manipulative Variables; Film Thickness
The powder-to-liquid ratio and the consistency of the mixed paste for the zinc phosphate cement largely depend upon the particular dental procedure and the properties desired for the cement. The powder-to-liquid ratio for the cement strongly affects the working and setting times. A thin consistency (low viscosity) is essential when the cement is used as a luting agent, to ensure adequate flow during cementation of orthodontic bands. The relatively fluid mixture is obtained by decreasing the powder-to-liquid ratio.
Several manipulative variables may affect the consistency, the working time and the setting behavior of the cement. An extended working time and a “sharp” set (very rapid increase in viscosity) are of fundamental importance in banding. A reasonable working time for zinc phosphate cements ranges between 3 and 6 minutes, and the setting time should be between 5 and 9 minutes, as specified in ANSI/ADA specification no. 96 (ISO 9917) for dental water-based cements. For optimum results, the powder should be incorporated into the liquid in small portions and at a relatively slow rate to achieve the desired consistency. With this method the exothermic setting reaction is retarded, with a reduction in heat generation. The viscosity of the mixture remains low, and the working time is optimized. In contrast, rapid mixing of the cement powder and liquid causes substantial heat evolution, with considerable decreases in the working and setting time.
Mixing of the powder and liquid over a large area of the glass slab also results in a lower temperature increase from the setting reaction and provides extended time for manipulation. The use of a cooled and dried mixing slab retards the reaction rate and prolongs the working time without influencing the setting time. In general, an extended working time allows the incorporation of greater amounts of powder into the liquid to produce a stronger and less soluble cement. However, care must be taken not to cool the slab below the dew point, since condensation from the air can cause contamination by water. Excess water may affect both the setting characteristics and the physical and mechanical properties of the cement. The addition of small quantities of powder to the liquid one minute prior to initiating mixing can prolong the setting process.
In hand-mixed products where the cement liquid is kept in a bottle, the cap should be removed only long enough to dispense the liquid portion for the mixture. Otherwise, the liquid may lose or gain water from the air, altering the setting behavior and the properties of the cement. The liquid should not be dispensed and allowed to remain on the slab for a substantial time prior to mixing, as some of the water contained in the liquid will evaporate. Prolonged spatulation should be avoided since this procedure may disturb the formation of the matrix of setting reaction products.
Since adhesion has not been documented to develop between zinc phosphate cements and orthodontic bands, retention of the bands is attained by mechanical interlocking. Thus, the film thickness of the cement placed between the band and the tooth is of paramount importance. Thin films result in substantially better cementation, as internal flaws within the cement are minimized, and there is better adaptation at the cement-tooth interface. In general, a film thickness of approximately 20 μm may be achieved with zinc phosphate cements, which is considered satisfactory since ANSI/ADA specification no. 96 suggests that an acceptable film thickness is of the order of 25 μm. The film thickness is a function of powder particle size, powder/liquid ratio, and viscosity of the cement mixture. Band placement should take place when the cement mixture flows, as the increased viscosity occurring with time may result in greater film thickness, leading to poorly retained and inadequately adapted bands.
Properties
Strength
Although an initial rapid rise in cement strength occurs in 4 to 7 minutes after mixing, reaching 50% of the final strength, the strength continues to increase at a slower rate for approximately 24 hours. Zinc phosphate cements, when properly manipulated in the luting consistency, exhibit compressive strengths ranging between 80 and 140 MPa.
The greater range of strength values reported in the literature may arise from the compositional variation for the different products. According to requirements in ANSI/ADA specification no. 96, the compressive strength for zinc phosphate cement should exceed 70 MPa. A linear relationship has been shown between the compressive strength and the powder/liquid ratio.
The (uniaxial) tensile strength of zinc phosphate cements is much lower than the compressive strength; their diametral tensile strength is typically less than about 5 MPa. The differences in tensile and compressive strength reflect the completely brittle nature of these cements. Values for modulus of elasticity in compression range for luting cements from about 9 to 13 GPa, and these cements can be considered as relatively stiff materials. The lower compressive and tensile strengths of luting cements, compared to the corresponding values for cements intended to be placed beneath restorations to serve as thermal and chemical barriers, reflect mainly the lower powder-to-liquid ratio used for the former application. Alterations in water content will influence the compressive and tensile strength of the cements.
Solubility
During the first 24 hours after mixing, zinc phosphate cements exhibit significant solubility in water, ranging from 0.04% to 3.3% by weight, presumably due to the continuing setting reaction of the cement. The dissolution rate decreases considerably after this time, when the material has fully set. A high powder-to-liquid ratio also strongly influences the solubility rate of the cements. The ions eluted within the initial setting period are magnesium and small quantities of zinc, and some release of zinc and phosphate ions may occur after completion of setting. Fluoride-containing cements exhibit a dissolution rate of 0.7–1.0% by weight, which occurs over a longer period of time than the dissolution of non-fluoride-containing cements, presumably owing to the interference of the fluoride-releasing mechanism. The solubility of zinc phosphate cements in dilute organic acids, such as lactic or acetic acid, is appreciably higher (20 to 30 times) than that in water. However, the weight of cement dissolved in water and in organic acids, as measured in vitro, does not correspond to the dis-integration rate of the cement when placed in the oral cavity.
The in vivo solubility of the cement is a highly important clinical property, since dissolution of the luting cement may result in plaque retention and subsequent development of primary caries. Additionally, continuous loss of the cement may cause loosening of the orthodontic bands.
Zinc Polycarboxylate Cements
Zinc polycarboxylate (polyacrylate) cement was introduced by Smith in 1968 and at that time represented the new generation of dental cements. This cement was the first dental material developed with adhesive potential to enamel and dentin. The polycarboxylate cements appear to combine the desirable properties of the zinc phosphate and zinc oxide-eugenol cements. The clinical indications for the polycarboxylate cements are similar to those for zinc phosphate cements.
Composition
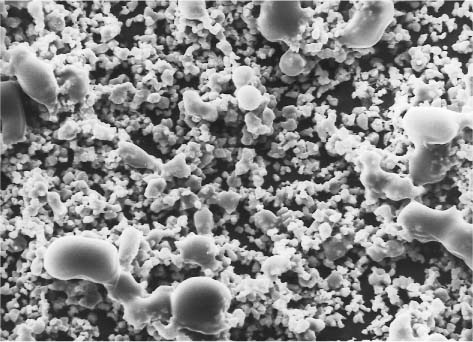
The zinc polycarboxylate cements are available as powder/liquid formulations. The powder is based upon components similar to those for the zinc phosphate cement powders (Fig. 11.3), and is mainly composed of zinc oxide with up to 10% magnesium oxide or tin oxide. Silica, alumina, or bismuth salts, and small quantities (4–5%) of stannous fluoride may be incorporated in some brands. The presence of the fluoride in these cements also increases the strength while controlling the setting time. Pigments may also be incorporated to provide a variety of shades.
The manufacturing procedure for the powder involves firing a blend of zinc and magnesium oxides between 900 8C and 1000 8C for 8 to 12 hours, grinding the sintered mass to the appropriate particle size, and reheating for another 8 to 12 hours.
Fig. 11.3 Secondary electron image of the powder particles of a zinc polycarboxylate cement. Original magnification × 1000
The liquid is an aqueous solution of a homo-polymer of acrylic acid or a copolymer of acrylic with other unsaturated carboxylic acids such as itaconic and maleic acids. The acid concentration in the liquid is approximately 40% by weight, and the molecular weight of the poly-acids varies from 22,000 to 50,000. While this high molecular weight may increase the strength of the cement, undesirable effects such as a short shelf-life and difficulties in manipulation occur because of the relatively high viscosity of the liquid. In some formulations the polyacid is a freeze-dried powder, which is then mixed with the zinc oxide powder and with tap water or distilled water or a solution of NaH2PO4.
Setting Reaction and Structure
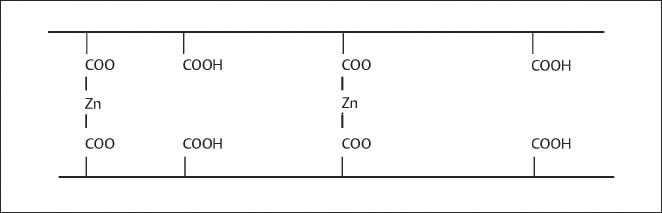
Setting of the zinc polycarboxylate cements occurs by an acid-base reaction between the zinc oxide powder and the polycarboxylic acid to form polycarboxylate salts. Upon mixing of the powder and liquid, an initial rapid stage is associated with attack of the powder particles by the acid, which releases zinc and magnesium ions. At the same time, ionization of the polycarboxylic acid takes place. This initial stage is followed by interaction between the carboxyl groups of adjacent polyacid chains and the metal ions to form cross-linked polycarboxylate salts, which act as the cement matrix (Fig. 11.4). The magnesium polycarboxylate salts have less hydrolytic stability than the corresponding zinc salts. Thus, a magnesium oxide concentration of up to 10% may yield cements with potential for higher solubility in the oral cavity.
Fig. 11.4 Schematic representation of the chemical structure of polycarboxylate salts formed between poly-acrylic chains and zinc ions
Fig. 11.5 Secondary electron image of porosity in a set (48 hours) zinc polycarboxylate cement. Original magnification × 200
In water-setting polycarboxylate cements, initiation of the acid-base reaction occurs after hydration of the freeze-dried polyacid powder particles. The set cement consists of an amorphous salt matrix in which un-reacted and partially reacted zinc and magnesium oxide particles are dispersed. An empirical formula for these salts is: Zn0·98H0·004 (CH2 · CH · COO)2·0(H2O)1·61. Both tightly bound water and loosely bound water are present in the set cement. The loosely bound water is considered to act as a plasticizer, causing weakening of the cement.
Working and Setting Times; Manipulative Variables; Film Thickness
In general, the zinc polycarboxylate cements have much shorter setting times than the zinc phosphate cements, which may be considered a potential clinical problem. Mixing of the polycarboxylate cements should be completed rapidly within 30–40 seconds. The working time varies from 2 to 5 minutes at room temperature (23 °C), and the setting time ranges from 6 to 9 minutes at 37 °C, which is an acceptable range for a luting cement. The setting reaction in water-based formulations proceeds more slowly than in the polyacrylic acid-based products, and thus the setting time is longer. The cement liquid is quite viscous, which can cause difficulty during the mixing procedure and can result in a highly porous material (Fig. 11.5). The powder should be rapidly incorporated into the liquid in large quantities to optimize the working time and setting time. Factors that influence the setting rate include the powder-to-liquid ratio, the powder composition, and the type, concentration, and molecular weight of the polycarboxylic acids.
The working time of the polycarboxylate cements can be extended, as with other cements, by lowering the temperature of the mixing slab and storing the powder in a refrigerator. However, refrigeration of the cement liquid should be avoided, because gelation may occur from the formation of hydrogen bonds. Prolonged storage at room temperature or the absence of moisture may also lead to gelation and thickening of the liquid. The use of a very cold slab is not recommended since it may cause difficulties in the mixing procedure by increasing the viscosity of the liquid.
The cement mixture should be used while it still has a glossy surface appearance. Often, a lower than optimum powder-to-liquid ratio is used in an attempt to produce a thin polycarboxylate paste comparable in viscosity to that of the more familiar zinc phosphate cement mixture. This procedure should be avoided since it will yield inferior properties for the set cement.
Polycarboxylate cements lack the relatively efficient mixing characteristics of the zinc phosphate cements because of their highly viscous liquids. Although the polycarboxylate cement mixture has a thicker consistency than that for zinc phosphate cement, it flows readily when loaded, yielding an appropriate film thickness of approximately 20 μm. This is because the polycarboxylate cements exhibit pseudoplastic behavior, undergoing shear thinning when pressure is applied during seating of orthodontic bands. A plot of shear stress as a function of strain rate has a decreased slope at high shear rates for a pseudoplastic material, whereas the linear slope remains constant for a material exhibiting newtonian viscosity, such as a zinc phosphate cement mixture. For water-mixed polycarboxylate cements, the initial viscosity occurring immediately after mixing is lower than that of the formulations in which the cement liquid contains polyacrylic acid.
Properties
Strength
Zinc polycarboxylate cements set relatively rapidly, attaining approximately 80% of their final strength within an hour. At luting consistency, the fully set cements at 24 hours after mixing have compressive strengths ranging from approximately 48 to 80 MPa, diametral tensile strengths of approximately 6 MPa and uniaxial tensile strengths ranging from 8 to 12 MPa. The modulus of elasticity of the zinc polycarboxylate cements is between approximately 3 and 6 GPa, which is about half of that of the zinc phosphate cements. These cements are substantially inferior to the zinc phosphate cements in compressive strength, but have slightly higher diametral tensile strength. The strength is improved by using a higher powder-to-liquid ratio when mixing the cement, and by the presence of/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses