10
Material-related Adverse Reactions in Orthodontics
Introduction
Fixed appliances
Metals
Nickel allergy – epidemiology in relation to orthodontics
Clinical aspects of hypersensitivity reactions to nickel and chromium in dental alloys
Hypersensitivity reactions to chromium in dental alloys
The possible role of oral exposure (orthodontic devices) in preventing sensitization to nickel
Summary and recommendations, including nickel-free alternatives
Bonding and banding materials
Composition, degradation/release
Hypersensitivity reactions to resin-based materials
Removable appliances
Acrylic base plates and activators
Soft retainers and trainers
Miscellaneous materials
Elastic bands and chains
Concluding remarks
References
Introduction
Unintended direct or indirect side effects of orthodontic treatment (e.g. root resorption, bone loss, white spot/caries lesions, dental hard tissue wear or cracking in relation to brackets and bands, discolorations) are well recognized and quite extensively described in the literature. Before the initiation of treatment, the orthodontic patient must be thoroughly informed and should have consented to the risks along with some degree of discomfort. Among 260 Norwegian orthodontic patients, the vast majority of patients reported that pain and intraoral ulcerations were significant aspects of orthodontic treatment (Kvam et al. 1987; Kvam et al. 1989). Eighteen percent of the adult patients reported to have ulcerations often or always, and 9% classified their intraoral ulcerations as ‘bad wounds’. Material-related adverse reactions (toxic/allergic) to orthodontic appliances can thus – unless particularly severe – be misdiagnosed or go undetected.
Fixed Appliances
Metals
Nickel Allergy – General Epidemiology
Nickel is an allergen which can cause immediate or delayed hypersensitivity reactions. Nickel has been found to be the most prevalent chemical contact allergen among the general population of the industrialized world (Dickel et al. 1998; Uter et al. 1998; Sertoli et al. 1999; Johansen et al. 2000; Veien et al. 2001; Marks et al. 2003). The exposure can be through skin, mucous membranes, diet, inhalation or implants (Hostýnek 1999). The risk of developing nickel allergic hypersensitivity (NAH) is high in some industries and occupational settings whereas for the general population the risk has been attributed to the wide use of nickel-containing alloys for e.g. jewelry, coins, tools, household utensils, dental alloys and orthopedic implants (Hostýnek 2002). The incidence of NAH is estimated to be around 22% of the female and 4.7% of the male dermatology patients in the USA and Europe (Hostýnek et al. 2002a). The increasing incidence of NAH in some countries, most markedly among women, has been attributed mainly to the wearing of nickel-containing alloys in fashion and lifestyle products. Regulations of permissible nickel levels in consumer products intended for intimate and prolonged skin contact appear to have reversed the trend, at least among the younger generations (Johansen et al. 2000; Nielsen et al. 2001; Veien et al. 2001; Jensen et al. 2002).
History of atopy is recognized as having a bearing on irritant contact dermatitis, but not on allergic contact dermatitis, as the latter does not seem to be more prevalent among people with atopy (Hostýnek et al. 2002a).
Nickel Applications in Orthodontics
In orthodontics nickel is one of the most commonly used metals, being a component of, for example, stainless steel and superelastic and shape-memory wires. Nickel is incorporated in the composition of all austenitic stainless steel alloys to stabilize the austenite phase. In comparison with the 8–12% nickel in stainless steel formulations the nickel content in nickel-titanium (Ni-Ti) and copper Ni-Ti (Cu-Ni-Ti) formulations amounts to 55–65%. The metal composition of archwires as well as brackets may vary slightly among brands (Brantley 2001; Eliades et al. 2001).
Release of Nickel from Dental Alloys
Corrosion Aspects, Laboratory Studies
Leakage of metal ions from orthodontic appliances into the saliva is preceded by disintegration of the alloy either by corrosion or mechanical abrasion. Microscopy studies of orthodontic fixed appliances have shown that after 10 months intraoral wear, corrosion is present on all intraoral metal appliances (Kratzenstein et al. 1985).
A number of studies have reported on metal ion release from dental alloys under various laboratory conditions (Toms 1988; Grimsdottir et al. 1992; Kim and Johnson 1999; Eliades and Athanasiou 2002; Schiff et al. 2006; House et al. 2008; Khuta et al. 2009; Luft et al. 2009). The clinical relevance of results obtained in vitro has, however, been questioned, and the lack of similarity with the clinical situation when using non-agitated, non-replenished artificial storage media has been emphasized (Eliades et al. 2002). Laboratory studies clearly demonstrate that physical factors such as surface structure, pH and the oxygen state at the material surface markedly influences the corrosiveness of dental alloys (Barrett et al. 1993; Geurtsen 2000; House et al. 2008; Khuta et al. 2009; Luft et al. 2009). It is thus clear that the presence of passivating oxide films on the surface decreases the corrosion rate while abrasion, polishing, low pH and high chloride content in the surrounding liquid contributes to the loss of these passivating films and subsequently increases the corrosiveness. Further, inhomogeneous soldered surfaces are more prone to corrosion, in particular connecting points between different alloys are highly susceptible to corrosion (Gjerdet and Hero 1987; Eliades 2007; House et al. 2008). In general the corrosiveness of a dental alloy depends on several physical and environmental factors and is not directly proportional to, for example, the nickel content.
Due to the methodological limitations of laboratory corrosion models in vitro results cannot be extrapolated directly to the clinical situation. From studies of retrieved orthodontic appliances it is, however, evident that corrosion takes place during oral services (Fig. 10.1) (see below). Further, discoloration of the underlying bonding material and tooth surface during orthodontic treatment has been linked to corrosion in the crevices of the bracket bases (Gwinnett 1982; Maijer and Smith 1982; Eliades et al. 2001). Some authors have raised concern that discoloration is possible not only of bonding material but also of the enamel (Maijer and Smith 1982).
Fig. 10.1 Recycled brackets. Color discrepancies indicate metallurgic changes compared with as-received brackets.

Laboratory studies estimate that the average release of metals from a simulated full-mouth orthodontic appliance is 40 µg nickel and 36 µg chromium per day, respectively, which is well below the average dietary daily intake of nickel and chromium consumed by Americans (300–500 µg nickel and 5–100 µg chromium) (Park and Shearer 1983). This is somewhat higher than results of nickel intake, in, for example, Denmark, which in a normal diet has been estimated to be in the range of 60–260 µg/day for a ‘normal’ diet and 70–480 µg/day when food rich in nickel was ingested (Veien and Andersen 1986).
In vitro studies have documented the cytotoxicity of nickel ions, which, for example, inhibit the proliferation of commonly used cell lines, however in doses which are far beyond the concentration levels estimated to be released in vivo (Hensten-Pettersen et al. 2001). On the other hand nickel ions in clinically relevant doses influence the release of inflammation mediators from human oral epithelial cells (Schmalz et al. 1997), activate monocytes and endothelial cells, and suppress or promote the expression of intercellular adhesion molecules in endothelial cells (Noda et al. 2002; Wataha et al. 2002). The clinical significance of these observations is so far not clear.
Nickel in Body Fluid Samples from Orthodontic Patients
Saliva
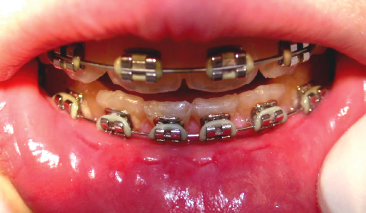
While the corrosion potential of orthodontic alloys has been studied quite intensively in vitro, less information exists on corrosion of orthodontic appliances in the oral cavity environment during orthodontic treatment. Corrosion products may be taken up directly through the oral mucosa, which in orthodontic patients often presents a compromised barrier due to mechanical irritation (Fig. 10.2) from intra-oral appliances, or through the lining of the gastrointestinal tract.
Fig. 10.2 Significant labial impression of a fixed appliance indicating intimate contact between the oral mucosa and the appliance.
The study of metal ion levels in saliva is complicated by the continuous release pattern of saliva. It is well known that factors such as temperature, quantity and quality of saliva, plaque, physical and chemical properties of food and liquids, and oral health conditions may influence the results. Further, physiological variables, such as time of the day, general health conditions, diet and salivary flow rate have significant impact. It is therefore generally recognized that large inter- and intra-individual variations are seen when analyzing saliva samples for metal content. Despite the methodological limitations, a number of papers have reported on metal ion levels in saliva samples from orthodontic patients. Studies have thus found that initial slightly elevated saliva levels of metal ions gradually decline during orthodontic treatment (Kratzenstein et al. 1988; Agaoglu et al. 2001; Gjerdet et al. 1991; Petoumenou et al. 2008) while some have found no significant differences between appliance and non-appliance wearers (Kocadereli et al. 2000). Some authors have hypothesized that a passivation layer might be formed on the metal surfaces in the oral environment, thereby decreasing the release of metal ions (e.g. Gjerdet et al. 1991).
In general reports on this issue have confirmed the large inter- and intra-individual variations in the concentrations of both nickel and chromium in saliva sample studies (Kratzenstein et al. 1988; Gjerdet et al. 1991; Kerosuo et al. 1997; Kocadereli et al. 2000) The unreliability of saliva sampling and processing have been emphasized and so far no definitive conclusions can be drawn regarding quantification of the range of metal ion release from orthodontic appliances in vivo, which, however, seems to be within that of daily intake through water and food (Eliades et al. 2003a; Kolokitha and Chatzistavrou 2008).
Blood/Serum/Urine
Concern has repeatedly been raised that metal ions from intra-oral alloys may be released to the saliva and once swallowed may accumulate in blood and organs. A few studies have attempted to elucidate whether metal ions from orthodontic appliances can be retrieved in blood or serum samples drawn from orthodontic patients. In this field too the results are conflicting. Some studies could not find measurable amounts of nickel in blood samples taken before and after orthodontic treatment with full-mouth fixed appliances (Bishara et al. 1993) while others have found a statistically significant increase in the amount of chromium after 2 years of orthodontic treatment whereas the same trend, although not statistically significant, has been observed with serum nickel levels (Agaoglu et al. 2001). The elevated chromium levels reported are far below toxic levels, and possible implications on the immune system are unclear.
One study of 18 orthodontic patients found significantly elevated urine nickel levels 2 months after placement of orthodontic appliances (Menezes et al. 2007). The biological significance of this finding is not known and more studies seem warranted to elucidate these aspects.
Influencing Clinical Factors
Wear/Friction
Toothbrushing has been shown to increase elemental release from dental alloys, particularly from base-metal alloys and when toothpaste is used (Wataha et al. 1999, 2003). Although these studies did not include orthodontic alloys, it is of interest to the orthodontic field that brushing with toothpaste caused an almost universal increase of metal ions release, particularly from nickel-based alloys (Wataha et al. 1999, 2003). The laboratory data cannot be directly extrapolated to the clinical situation. It has, however, been suggested that the use of low-abrasive toothpastes might be advisable when nickel-based alloys are used. It has been hypothesized that the high nickel release from the nickel-based alloys might at least partially be due to the loss of protective surface oxides during brushing.
Further to mechanical activation of an intraoral appliance by toothbrushing, for example, movement of archwires and friction between brackets and ligatures may lead to further corrosion-enhanced release of metal ions from the appliance (Eliades and Athanasiou 2002; House et al. 2008).
Recycling
Recycling of brackets may increase the release of metal ions substantially (Huang et al. 2001, 2004; Sfondrini et al. 2010). The higher ion release from recycled brackets has been attributed to the heating of the brackets, which is necessary to remove the bonding material. It has been hypothesized that the solder, porosity of the alloys, and grain size of alloy constituents might have caused the differences observed between brands. Differences between wing and base materials and between brands have been reported (Eliades et al. 2003b). Nickel loss initiated by corrosion and subsequent recycling may extend more than 5–10 µm below the surface (Eliades et al. 2002).
Nickel Allergy – Epidemiology in Relation to Orthodontics
There are only a few reports of the estimated prevalence of material-related side effects in orthodontics. A questionnaire survey of Norwegian orthodontists in late 1980s estimated that the incidence of material-related side effects among orthodontic patients was around 1:100 (Jacobsen and Hensten-Pettersen 1989). Reported side effects comprised both extraoral, intraoral and general effects, with headgears and fixed appliances being the most frequently implicated causes. A later survey of the same cohort of Norwegian orthodontists found that the rate of patient reactions had decreased to 1 in 300 (Jacobsen and Hensten-Pettersen 2003). Only a few verified NAH-cases were reported whereas the vast majority of reactions were categorized as unspecified reactions to fixed appliances and facebows in general. The survey did not allow any estimation of the number of possible non-diagnosed NAH-cases among the reported unspecified reactions. The reduction in the incidence of patient reactions from 1987 to 2003 was caused by a marked reduction in dermal reactions to the metallic parts of extraoral appliances. The authors suggested that this reduction was due to preventive measures such as plastic coating on headgears and avoidance of direct metal/skin contact.
A German questionnaire survey of 68 orthodontic offices estimated the prevalence of perceived allergic reactions to be 1:430 (Schuster et al. 2004). The majority of the reported reactions were extraoral (labial fissures, perioral inflammation, and eczema on the face and extremities) and most often related to the use of a headgear. The relative few intraoral reactions comprised erythema, swelling and gingivitis. The perceived allergic reactions led to discontinuation of the orthodontic treatment in only 0.03% of the cases (1:3150). In 0.07% of the cases the treatment plan and the appliances remained unchanged despite the observed reactions. In 0.13% (1:810) the orthodontic appliances were replaced with those composed of nickel-free or low-nickel materials. In a later, similar American study a prevalence of perceived nickel-related adverse reactions of <1% was reported (Volkman et al. 2007).
A Danish investigation including more than 1000 girls in active orthodontic treatment or retention aimed at elucidating whether nickel-sensitive persons were at greater risk of developing intraoral discomfort during orthodontic treatment (Staerkjaer and Menne 1990). No intraoral nickel allergic reactions were recorded. Nine patients reported extraoral discomfort. Five of these were related to headgears, one to a chin cap and one girl with a previously diagnosed nickel allergy got a rash in her face when a brass ligature was inserted for separation of teeth. She later tolerated intraoral fixed appliances without signs or symptoms of a hypersensitivity reaction. The authors found no indications that nickel-sensitive persons were at greater risk of developing discomfort in the oral cavity when wearing an intraoral orthodontic appliance.
Clinical Aspects of Hypersensitivity Reactions to Nickel and Chromium in Dental Alloys
Nickel is capable of evoking both IgE-mediated (immediate type) and cell-mediated (delayed) contact allergic reactions. Particularly in the industrial setting, volatilization of metals presents a respiratory occupational risk of type I hypersensitivity (e.g. urticaria, asthma, rhinoconjunctivitis, gastrointestinal involvement, and anaphylaxis). In contrast to dust generated in, for example, mining and construction, highly dispersible and respirable aerosols may be formed during processes such as smelting and heating (Hostýnek 2002). The delayed-type NAH reaction generally presents as dermatitis, eczema and occasionally stomatitis. Dermatitis and urticaria, the primary manifestations of NAH, are observed in the area of contact as well as at distant sites (secondary eruptions).
Quite a number of allergic reactions related to orthodontic alloys have been described in the literature and demonstrate the diversity of reactions nickel exposure may evoke in sensitized individuals.
Intraoral Reactions
Intraoral verified hypersensitivity reactions to orthodontic alloys described in case reports have ranged from almost negligible, such as a slight erythema (e.g. Hensten-Pettersen et al. 1984; Kolokitha and Chatzistavrou 2009) to large erythematous macular lesions or ulcerations (Schriver et al. 1976; Shelley 1981; Dunlap et al. 1989; Veien et al. 1994; Al-Waheidi 1995; Counts et al. 2002) and subjective symptoms ranging from mild to very painful burning sensations compromising normal oral functions (Dunlap et al. 1989; Al-Waheidi 1995; Noble et al. 2008). Further, it has been emphasized that symptoms of nickel allergy may in rare cases present as hygiene-resistant severe gingivitis (Shelley 1981; Counts et al. 2002; Pazzini et al. 2009, 2010). In sum, reports on verified intraoral adverse reactions to nickel are very rare.
Extraoral Reactions
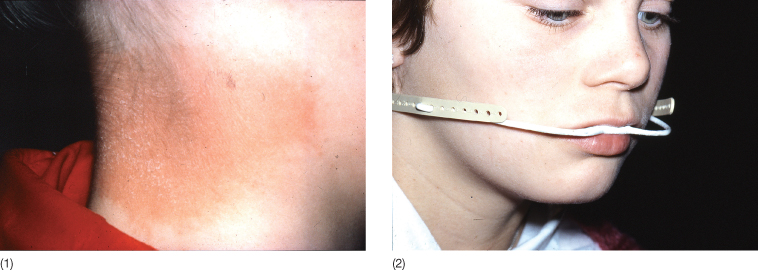
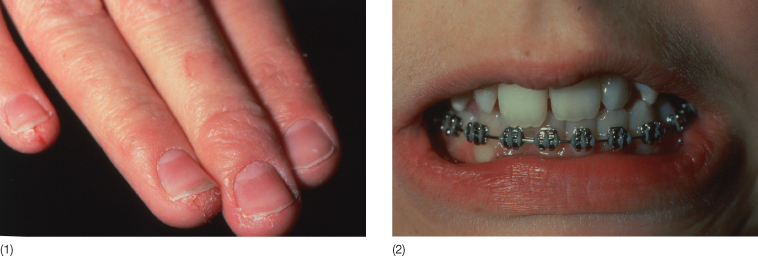
A review of published case reports on hypersensitivity reactions to intraoral nickel-containing orthodontic appliances reveals that in the vast majority of cases the lesions described were extraoral and not accompanied by any intraoral signs or symptoms of discomfort (Jacobsen and Hensten-Pettersen 2003; Kerusuo and Dahl 2007; Feilzer et al. 2008; Kolokitha and Chatzistavrou 2009). A number of reports have described skin lesions in direct contact with or adjacent to metal parts of headgears (Fig. 10.3(1,2)). Soreness, blisters and ulceration of the contacting skin areas were often seen (Greig 1983; Hensten-Pettersen et al. 1984; Burden and Eedy 1991; McComb and King 1992; Lowey 1993). Further eczematous lesions have, however, been noted in remote locations, e.g. face, fingers, arms, trunk and feet.
Fig. 10.3 (1,2) Contact dermatitis adjacent to the stainless steel part of a headgear.
(Courtesy of Professor A Hensten, University of Tromsoe, Norway.)
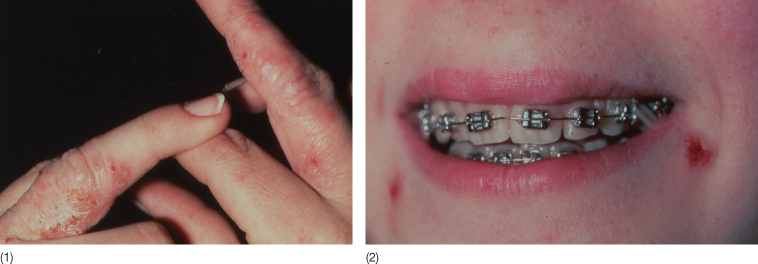
A considerable number of cases with onset or flaring of eczematous skin lesions in relation to insertion, change or activation of intraoral fixed orthodontic appliances have been reported. Very few of these were accompanied by intraoral complaints. Angular cheilitis and fissures of the lips or more severe perioral and facial eczema have frequently been described in relation to nickel-hypersensitive orthodontic patients (Hensten-Pettersen et al. 1984; Al-Waheidi 1995; de Silva and Doherty 2000; Yesudian and Memon 2003; Pigatto and Guzzi 2004; Feilzer et al. 2008; Ehrnrooth and Kerosuo 2009; Kolokitha and Chatzistavrou 2009) (Fig. 10.4). More remote eczema lesions, e.g. periorbitally, eyelids, ears, scalp, fingers, chest, back, arms and feet, have also been described (Veien et al. 1994; Bishara 1995; Ehrnrooth and Kerosuo 2009) (Fig. 10.5(1,2)). In a number of cases it has been reported how the lesions flared up, e.g. healed lesions being aggravated when the orthodontic appliance was manipulated (such as when activating archwires or inserting new wires). It is striking that a number of papers have reported that lesions aggravated when Ni-Ti wires were worn improved when stainless steel wires or titanium molybdenum (TMA) alloy wires were used as an alternative to Ni-Ti wires (Al-Waheidi 1995; Counts et al. 2002; Mancuso and Berdondini 2002; Schultz et al. 2004). Some striking remote reactions have also been reported, e.g. on the feet and trunk, in the absence of intraoral symptoms (Trombelli et al. 1992; Kerosuo and Kanerva 1997).
Fig. 10.4 Cheilitis in a nickel-sensitized 25-year-old female orthodontic patient with no intraoral symptoms. She tolerated the fixed orthodontic appliances without any skin reactions, except when Cu-Ni-Ti archwires were inserted.
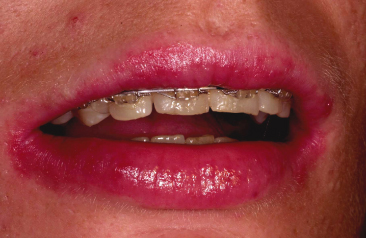
Fig. 10.5 (1) Vesicular hand dermatitis following insertion of a fixed orthodontic appliance in a 15-year-old girl with no intraoral symptoms (2). Positive skin test on chromate.
(Courtesy of Dr N Veien, Aalborg, Denmark.)
On the basis of the available case reports, it can be summarized that hypersensitivity reactions to intraoral orthodontic devices are relative rare and when they do occur, in the majority they manifest as extraoral eczematous lesions frequently in locations remote to the oral cavity. Unless particularly severe, such reactions may be misdiagnosed or may go undetected (Greppi et al. 1989).
Hypersensitivity Reactions to Chromium in Dental Alloys
Allergic contact dermatitis caused by chromate salts was first reported in 1925 and is still common. Hexavalent chromium compounds are considered the strongest sensitizers among the chromium ions. On the other hand, it is generally accepted that chromium itself does not act as a hapten, and is accordingly non-sensitizing. It is important to emphasize this difference compared with, for example, nickel. Theoretically sweat or plasma can transform metallic chromium into allergenic chromate salts. Saliva may have a similar effect on intraoral devices containing chromium (Kanerva and Aitio 1997). It is often not clear whether chromates or other metals and metal salts have caused the allergic reactions elicited by dental metals. Case reports describe patients with generalized eczematoid dermatitis following installation of dentures with metal framework (Foussereau and Laugier 1966). Skin tests were strongly positive to nickel and chromium and the dermatitis subsided after discontinuing use of the denture. In most instances in which an allergic reaction is attributed to a metallic chrome object, nickel is the actual sensitizer. A rare case of dermatitis in an orthodontic patient who was allergic to chromate, and negative on patch testing to nickel, has, however, been described (Veien et al. 1994) (Fig. 10.6(1,2)). The dermatitis appeared shortly after installation of a stainless steel orthodontic appliance and cleared when the appliance was removed. Rare cases of systemic contact dermatitis from chromate in dental cast crowns have also been reported (Guimaraens et al. 1994).
Fig. 10.6 (1, 2) Vesicular hand dermatitis which developed some months after insertion of a fixed orthodontic appliance in a 13-year-old girl with no intraoral symptoms. The oral challenge test on nickel was positive.
(Courtesy of Dr N Veien, Aalborg, Denmark.)
The Possible Role of Oral Exposure (Orthodontic Devices) in Preventing Sensitization to Nickel
There seems to be increasing evidence that long-term oral exposure to low levels of nickel substantially reduces the prevalence of hypersensitivity on later challenge (Hostýnek et al. 2002b). The history of the growing numbers of dermatology patients with NAH and studies on large cohorts of adolescents without symptoms of allergy, have led to a focus on pre-exposure to orthodontic appliances as a potential tolerance evoking factor. van der Burg et al. (1986) as the first, in a prospective study of hairdressers and nurses showed that individuals who in childhood had worn some kind of intraoral nickel-containing appliance had a markedly lower incidence of NAH than those who had not had orthodontic treatment. A number of later studies have followed cohorts of adolescents from the onset of orthodontic treatment and confirmed that early oral exposure to nickel-releasing appliances may induce a state of tolerance to nickel (e.g. Todd and Burrows 1989; van Hoogstraten et al. 1991, 1992; H Kerosuo et al. 1996; Lindsten and Kurol 1997; Mortz et al. 2002).
Summary and Recommendations, Including Nickel-Free Alternatives
According to the available literature there is a very low risk of primary sensitization through exposure to intraoral dental alloys, including orthodontic appliances (Kolokitha et al. 2008). Furthermore, patients with diagnostic tests verifying presence of NAH do not necessarily experience adverse intraoral reactions on exposure to nickel-containing dental alloys (Janson et al. 1998). On the contrary, it has been reported that even some patients with a strong positive skin test reaction to nickel may well tolerate the intraoral placement of nickel-containing alloys (Spiechowicz et al. 1984).
Although the vast majority of orthodontic patients – including those with a positive skin patch test to nickel – tolerate the wearing of conventional orthodontic appliances without material-related adverse reactions, there may be some hypersensitive Ni-allergic patients in whom appliances with high content of nickel (e.g. Cu-Ni-Ti wires) should be avoided (Kim and Johnson 1999). A number of these patients may tolerate stainless steel appliances. In a few cases it may, however, be necessary to completely avoid nickel-containing alloys. Alternative products that are nickel-free or have a very low nickel content are available (Rahilly and Price 2003; Eliades 2007). Nickel-free wires of TMA or pure titanium, or gold-plated wires, as well as plastic/resin-coated Ni-T archwires with less corrosion potential may be considered (Kim and Johnson 1999; Eliades 2007). Brackets are available in different ceramic forms, as well as polycarbonate brackets produced from acrylic polymers, titanium or even gold plated. Further, headgears without extraoral metal components are available; they have plastic studs or plastic-coated metallic studs. The successful use of cast titanium framework for rapid palatal expansion devices and activators for nickel-sensitive patients has been described (Farronato et al. 2002).
Although the information on the metal ion release from recycled brackets so far is limited, it seems justified to recommend that recycled brackets should not be used in patients with a known allergy to nickel or other metals that may be present in the bracket alloy.
Bonding and Banding Materials
Optimal material selection and application require an understanding of the chemical differences between and the physical limitations of today’s orthodontic bonding materials. Contemporary orthodontic bonding materials comprise a number of material categories, e.g. cements (zinc-phosphate; polycarboxylate, glass ionomer cements), resins, resin-modified glass ionomers and polyacid-modified composite resins. Unfortunately, the information material required by regulation (e.g. safety data sheets) is limited (Kanerva et al. 1997; Spahl et al. 1998; Michelsen et al. 2003) and recent research has documented that numerous non-declared substances of potential harmful nature may be released from, for example, resin-based dental materials (Arenholt-Bindslev et al. 2009).
Composition, Degradation/Release
Zinc Phosphate Cement
This is the reaction product of zinc oxide and a phosphoric acid solution. Some brands further contain around 10% fluoride in the form of stannous fluoride. Apart from the acidity of the phosphoric acid solution the hazardous potential of these materials is limited and when used according to the manufacturer’s instructions practically no material related adverse reactions are relevant according to the literature.
Polycarboxylate Cement
This is the reaction product of zinc oxide and a polycarboxylic acid solution. Fluoride salts may be added for anticariogenic purposes. There are no reports on materialrelated side effects when used according to the instruction manual.
Glass Ionomer Cements (GICs)
These consist of calcium fluoroaluminosilicate glass particles, which when mixed with polyacrylic acid, undergo an acid-base reaction to form polycarboxylate salts that comprise the cement matrix. No reports of side effects of conventional GICs are available in the literature.
Resin-Modified GICs and Polyacid-Modified Composite Resins (Compomers)
These materials from a biocompatibility point of view possess similar characteristics to the above. Both material categories contain a cement and a resin component, of which mainly the resin part is of interest in regard to biocompatibility (see above). Resin-modified GICs contain ion-releasing glass particles, water-soluble polyacrylic acids and light-curable monomers. Compomers were developed to combine the fluoride release of GICs with the mechanical properties of composite resins. These materials are composed of ion-releasing glass particles and a polymerizable organic matrix. In addition to conventional monomers the organic matrix of compomers contains bi-functional monomers which can react with methacrylates by radical polymerization and by acid-base reaction to bring about release of ions from glass in the presence of water.
Adhesive Resins
These consist of a mixture of monomers, mainly methacrylates (e.g. BISGMA, TEGDMA, HEMA) that vary from brand to brand. Based on the polymerization-initiation mechanism, adhesive resins can be grouped as chemically activated (two-paste), light cured or dual cured (chemically activated and light cured). Additives normally amount to 1–2% of the adhesive resins and the chemical substances added vary according to brand and the type of polymerization process they initiate and modulate. Filler particles are usually added.
Local and Systemic Toxicity of Resin-/Polymer-Based Materials (Resin-Modified GICs, Compomers and Adhesive Resins)
Common to all resin-/polymer containing dental materials is incomplete conversion during polymerization of the resin />
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


