The incidence of oral fungal infections has increased in recent years as a result of factors such as increased number of solid organ transplantations and the widespread use of immunosuppressive drug therapies. This article reviews the diagnosis and treatment of oral fungal infections. At one time oral fungal infections were a relatively uncommon event, but with advances in health care and polypharmacy in an increasingly aging population, these infections are becoming a more routine clinical finding. The dental practitioner therefore needs to be familiar with the diagnosis and management of oral fungal infections.
Key points
- •
Candidiasis is the most common oral fungal infection diagnosed in humans. Candidiasis may result from immune system dysfunction or as a result of local or systemic medical treatment. Because oral Candida is generally a localized infection, topical treatment methods are the first line of therapy, especially for the pseudomembranous and erythematous variants.
- •
Patients with a dental prosthesis should also be advised to disinfect the prosthesis routinely during their Candida treatment period, as the prosthesis may serve as a source of fungal reinfection.
- •
Patients should be advised that oral hygiene aids, such as toothbrushes and denture brushes, may also be contaminated and should be discarded or disinfected. A disinfection solution of sodium hypochlorite (1 part household bleach mixed with 10 parts water) may be used for non–metal-containing dental prosthesis and appliances, and oral hygiene aids.
- •
Occasionally the clinician will encounter a more resistant form of oral candidiasis, such as the hyperplastic variant or a variant that does not respond to topical therapy. Appropriate systemic therapy should be used for the treatment of these infections. In addition, a biopsy should be undertaken in individuals with the hyperplastic variant of Candida , as there is some degree of risk for malignant transformation.
- •
Deep fungal infections should be managed in association with appropriate medical specialists to rule out other systemic involvement.
- •
The dental health care provider plays an important part in the diagnosis and management of fungal disease, so clinicians should be aware of the presenting signs and symptoms of oral fungal disease.
Introduction
Fungal diseases have long affected plants, animals, and human hosts. As early as the fourth century, Hippocrates, the Father of Medicine, first described fungal disease affecting humans. Since then the approach to the diagnosis and management of fungal diseases has become more systematic. To aid in classification, management, and treatment, human fungal infections are now routinely grouped into superficial, subcutaneous, or systemic categories, based chiefly on the site of clinical involvement. This article primarily addresses superficial oral fungal infections.
Introduction
Fungal diseases have long affected plants, animals, and human hosts. As early as the fourth century, Hippocrates, the Father of Medicine, first described fungal disease affecting humans. Since then the approach to the diagnosis and management of fungal diseases has become more systematic. To aid in classification, management, and treatment, human fungal infections are now routinely grouped into superficial, subcutaneous, or systemic categories, based chiefly on the site of clinical involvement. This article primarily addresses superficial oral fungal infections.
Superficial oral fungal diseases
The most commonly occurring oral fungal infection is caused by Candida species, with Candida albicans most often encountered. Candida is an obligate organism found in the digestive and vaginal tracts of human hosts. In fact, up to 60% of all healthy ambulatory, non-immunocompromised individuals may harbor this organism in the oropharyngeal region.
Candida is a dimorphic fungus that can exist in both a yeast phase (blastospore, blastoconidial) and a hyphal (mycelial) phase. Candida reproduces by multilateral budding, and may develop either as a mycelial form, composed of long branching septae or filaments, or as spherical or ovoid yeast cells. Dimorphism is relevant to pathogenicity of the yeast and to the clinical problems of diagnosis and treatment of fungi.
Presence of sufficient quantity of Candida can indicate a pathogenic state and can occur beyond the oropharyngeal region. For instance, in United States hospitals during the period from 1995 to 2002 the frequency of candidemia ( Candida in the blood) rose from 8% to 12% with increasing frequency of C albicans species.
Candida species are assumed to cause disease by direct tissue invasion, either by inducing a hypersensitive state or by producing potent Candida toxins. These toxins help establish oral Candida (oral candidiasis) infection.
Alterations of the host environment may promote Candida growth and eventual establishment of oral candidiasis. These host-environment changes include decreased salivary flow, use of an acrylic dental prosthesis, use of broad-spectrum antibiotics, nutritional deficiencies, high carbohydrate diets, and immunosuppressive diseases and medications.
Oral candidiasis (also referred to as oropharyngeal candidiasis) is one of the most common conditions affecting the oral mucosa. C albicans usually accounts for 70% to 80% of oral isolates; however, non– C albicans species such as Candida glabrata and Candida tropicalis account for approximately 5% to 8%, respectively, of oral isolates. The tongue, followed by the palate and buccal mucosa, are the more densely populated areas of yeast in the oral cavity. The quantification of salivary Candida colony-forming units (CFU) varies with diurnal and daily fluctuation. Poor oral hygiene is associated with increased salivary Candida CFUs. Healthy individuals with yeast colonization have on average 300 to 500 CFUs of Candida per milliliter of saliva. Dental caries in children is also associated with increased carriage of C albicans , with a resulting decrease in carriage after caries treatment.
The symptoms associated with oral Candida infection can range from none to a painful burning sensation that may interfere with the ability to swallow (dysphagia) and ability to take in nutrition. Oral Candida infections have traditionally been stratified into 4 clinical subgroups: erythematous, pseudomembranous, hyperplastic, and angular cheilitis. More recently a revised classification system that stratifies Candida lesions into 2 subgroups, group I (localized) and group II (secondary), has been proposed ( Box 1 ).
-
Group I
-
Candida infections are confined to oral and perioral areas only
-
Group I acute variants
-
Pseudomembranous
-
Erythematous
-
-
Group I chronic variants
-
Hyperplastic
-
Erythematous
-
Pseudomembranous
-
-
Candida -associated lesions
-
Denture stomatitis
-
Central papillary atrophy
-
Angular cheilitis
-
-
Keratinized primary lesions superinfected with Candida
-
Leukoplakia
-
Lupus erythematosus
-
Lichen planus
-
-
-
Group II
-
Candida infections that are generalized and oral infections are secondary infections. These lesions are typically associated with rare inherited immune defects.
Group I lesions are localized to the oral cavity and do not involve any other cutaneous or mucosal sites. Group I lesions consist of 3 different clinical presentations: pseudomembranous candidiasis, erythematous candidiasis, and hyperplastic candidiasis. Pseudomembranous and erythematous candidiasis may also be subgrouped into acute or chronic presentations. There are also group I Candida -associated lesions, such as angular cheilitis, central papillary atrophy, denture-associated stomatitis, and keratinized lesions of other disorders that are commonly superinfected with Candida .
Group II lesions are also known as secondary lesions, and may have oral and extraoral sites of infection. Group II lesions are typically associated with rare inherited immune defects, and are not reviewed in this article.
Classification of Group I Candidal Lesions (Localized to Oral Cavity)
Pseudomembranous candidiasis
Pseudomembranous candidiasis, commonly referred to as thrush, was first described by Langenbeck in 1839 as a fungus in buccal aphthae in a patient with typhus. Pseudomembranous candidiasis can appear as white or yellow plaques on any mucosal surface, and can be classified as acute or chronic. The plaques represent an overgrowth of the yeast on the mucosa leading to epithelial-cell desquamation and a concomitant accumulation of bacteria, keratin, and necrotic tissue. These lesions can be either confluent or discrete. The plaques are readily removable, leaving a raw underlying surface. Pseudomembranous Candida is most often found in those with immune-system dysregulations such as infants, the elderly, individuals with immune diseases, or those taking immunosuppressive medications ( Figs. 1–3 ).
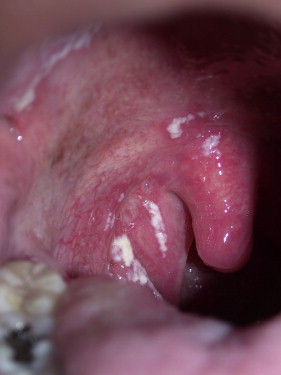
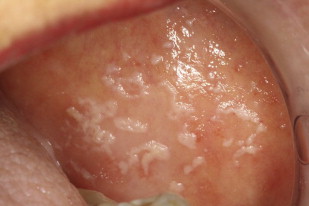
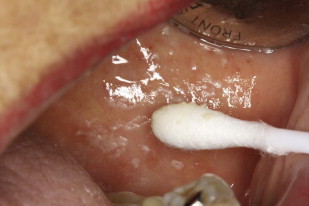
Erythematous candidiasis
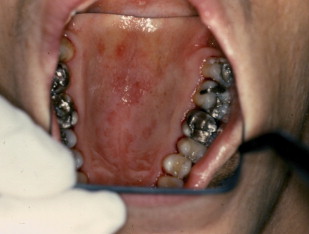
Erythematous candidiasis is sometimes referred to as atrophic candidiasis. The term atrophic originates from the Greek term for ill-fed, atrophos . Typically this variant of candidiasis appears as erythematous patches on any mucosal surface, and can be classified as acute or chronic. More often the affected surfaces are the palate and the dorsal tongue, where it can cause depapillation.
Erythematous candidiasis is most often associated with the use of broad-spectrum antibiotics or corticosteroids ( Fig. 4 ).
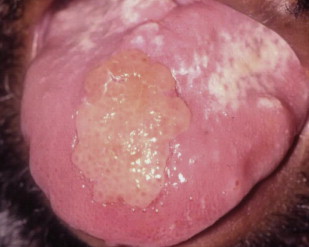
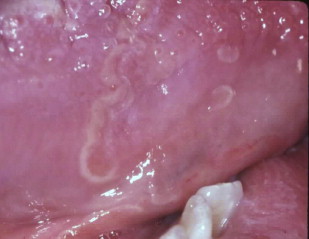
Chronic hyperplastic candidiasis
Chronic hyperplastic candidiasis (candidal leukoplakia) is the third clinical candidal variant. Typically described as a white plaque presenting on the commissural region of the buccal mucosa, it cannot be readily wiped off. Other sites affected, in decreasing order, include buccal commissures, palate, and tongue.
According to several investigators, chronic hyperplastic Candida infection is associated with a diagnosis of dysplasia. Samaranayake and Wallace MacFarlane suggest that up to 15% of all chronic hyperplastic candidal infections may progress to dysplasia. The role of Candida in epithelial dysplasia is unclear, and may in fact be related to a host-environmental change as a result of the dysplasia (see the article on oral cancer elsewhere in this issue). Nevertheless, the possibility that Candida may be associated with epithelial dysplasia necessitates close follow-up of patients with chronic recalcitrant hyperplastic Candida . Biopsy of suspected chronic hyperplastic candidiasis is recommended to establish the diagnosis differential from other leukoplakias and to determine if dysplasia is present ( Figs. 5 and 6 ).
Group I Candida -Associated Lesions
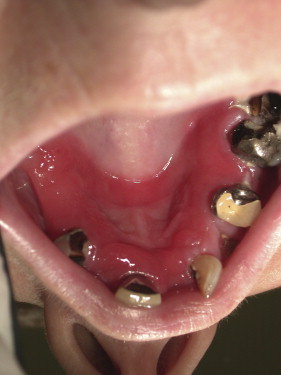
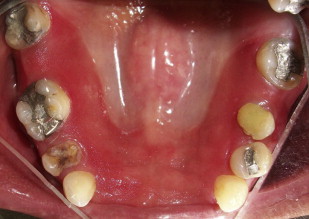
Denture-associated stomatitis
Denture-associated stomatitis, or denture sore mouth, is a chronic inflammatory condition of the denture-bearing mucosa caused in part by the erythematous variant of Candida . The porosity of the acrylic allows for fungal and bacterial contamination in the denture. Moreover, the relatively acidic and anaerobic microclimate underneath the denture provides an ideal environment for yeast growth, and the denture shields the mucosa from the saliva and salivary functional properties of local immunity. The denture or any acrylic-containing dental appliance can serve as a continuous source of inoculation for bacteria and fungi. Therefore, disinfection of dentures and dental appliances plays a key role in eradicating this form of candidal infections.
Disinfection of non–metal-containing acrylic dentures and dental appliances may be achieved with a 10-minute soak in a 0.5% solution of sodium hypochlorite (1 part household bleach mixed with 10 parts water). In patients with denture-associated stomatitis, microwave disinfection of non–metal-containing acrylic dentures was compared with treatment with nystatin mouth rinses. Microwave disinfection, at once per week for 2 treatments, was as effective as a 14-day course of topical nystatin antifungal therapy for treating denture-associated stomatitis. Disinfection of metal-containing dentures or dental appliances is somewhat more difficult to assess. There are few randomized clinical trials on denture disinfection. Investigators have proposed the use of chlorhexidine solutions, hydrogen peroxide, and antifungal medication as a means of disinfecting metal-containing dental appliances.
A Cochrane Database review published in 2009 concludes that there is a lack of evidence about the comparative effectiveness of the different denture-cleaning methods and that future research should focus on comparisons between mechanical and chemical methods ( Figs. 7 and 8 ).
Central papillary atrophy
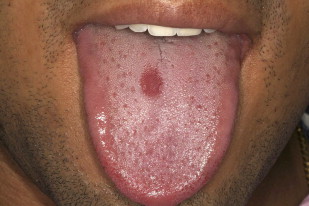
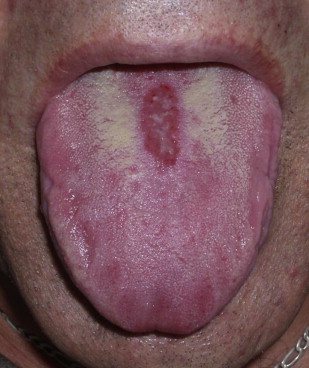
Central papillary atrophy (median rhomboid glossitis) is relatively uncommon, with prevalence of less than 1%. Men are affected 3 times more often than women. The condition clinically presents as a rhomboid-shaped hypertrophic or atrophic plaque in the mid-dorsal tongue. Central papillary atrophy has been associated with chronic Candida infection. Moreover, there is little evidence that central papillary atrophy lesions resolve completely with antifungal treatment, and currently the importance of Candida species in the etiology of central papillary atrophy is unknown ( Figs. 9 and 10 ).
Angular cheilitis
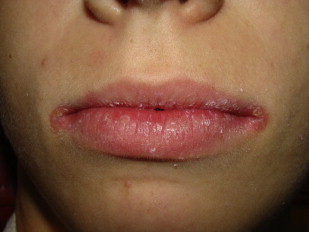
Angular cheilitis is a mixed bacterial-fungal infection commonly noted at either or both corners of the mouth. Other agents associated with angular cheilitis include staphylococci and streptococci. The typical presentation is erythematous fissuring at the commissures, often accompanied by a pseudomembranous covering. Angular cheilitis may also affect the anterior nostril region. Contributing factors include facial wrinkling along nasolabial folds and reduced occlusal vertical dimension, both of which promote a moist environment conducive to growth of etiologic agents.
Another less cited contributing factor to angular cheilitis is nutritional deficiencies. Thiamine, riboflavin, iron, and folic acid deficiencies have all been implicated in cases of angular cheilitis ( Fig. 11 ).
Host Factors Associated with Candidiasis
Oral candidiasis is associated with a decrease in the host defense mechanism, which may have many causes and can be local or systemic in nature. One local cause of reduced host defense mechanisms is medications. For example, inhaled corticosteroids are now routinely used in the management of bronchial asthma and chronic obstructive pulmonary disease to suppress airway inflammation. Inhaled corticosteroids have reduced or eliminated the need for systemic corticosteroids in the treatment of these diseases. Inhaled corticosteroids are associated with an increased risk for oral candidiasis, especially the erythematous variant. Alsaeedi and colleagues reviewed 9 randomized clinical trials with a total of 3976 patients with chronic obstructive pulmonary disease who were treated with inhaled corticosteroids. A risk ratio of 2.1 for oral Candida infections was reported for those patients treated with inhaled corticosteroids. It is postulated that steroids suppress localized cellular immunity and phagocytosis, thereby promoting the establishment of Candida .
Likewise, impaired salivary flow may also predispose an individual to candidiasis through a reduced presence of localized cellular immunity. With decreased salivary flow there are decreased salivary antimicrobial proteins secreted, such as lactoferrin, sialoperoxidase, and lysozyme, all of which have antifungal effects. Moreover, adequate salivary flow serves to dilute and remove potential pathogenic organisms from the oral mucosa. Thus, decreased salivary flow may be associated with oral candidiasis. Xerostomia, a subjective complaint of oral dryness, is a reported side effect of more than 500 medications. Xerostomia does not always correlate with a decrease in salivary-gland output. Many medications known to decrease salivary output have anticholinergic properties. Classes of medications know to decrease salivary output includes antihistamines, tricyclic antidepressants, certain antihypertensives, hypnotics, and sedatives.
Similarly, systemic medications can also affect the local environment in the oral cavity. Broad-spectrum antibiotics increase susceptibility to Candida infection by altering local oral flora that naturally inhibits candidal growth. Other systemic medications associated with onset of oral Candida infections include antineoplastic agents, immunosuppressive agents (such as azathioprine and glucocorticosteroids) and anticholinergic agents.
Other factors have also been implicated in oral candidiasis, including smoking, diabetes mellitus, endocrinopathies, immunosuppressive conditions, malignancies, and nutritional disorders.
Diagnosis of Oral Candida Infections
Superficial Candida infections usually have a characteristic clinical appearance, and diagnosis is often based on clinical findings. Subjective complaints of localized oral burning are sometimes empirically treated for candidiasis by physicians and dentists.
Objective assays in the diagnosis of oral candidiasis include exfoliative cytology, imprint culture, swab culture, salivary assays, and mucosal biopsy.
Exfoliative cytology is useful in differentiating between yeast and hyphal forms of Candida , but is less sensitive than culture methods. A representative sample from the infected site is transferred to a microscopic slide and is treated with potassium hydroxide (KOH), Gram stain, or periodic acid-Schiff (PAS) stain. Candidal hyphae will appear as clear tubes with KOH staining as the KOH clears organic material from the background, making the fungi more readily apparent. With Gram staining, hyphae and yeast will appear dark blue, whereas PAS-stained specimens will appear red to purple.
Imprint culture involves using a sterile foam pad treated with growth media. The pad is placed on the infected site and then transferred to an agar medium for culture. Similarly, contents from an oral swab of the suspected site are transferred to growth media and cultured for candidal growth. Salivary rinse techniques involve the patient rinsing with a sterile phosphate-buffered saline rinse. The expectorant is concentrated by centrifugation, and the sample is inoculated on an agar growth plate.
Mucosal biopsy can also be used to ascertain Candida infection. A sample of tissue obtained by biopsy should be placed in 10% formalin and ideally should be submitted to an oral maxillofacial pathologist. The clinician should communicate with the pathologist the patient’s medical and drug history and the suspected fungal diagnosis, so that special staining techniques such as PAS can be ordered.
General characteristics of fungi seen on direct microscopic examination of specimens include budding yeast cells (eg, blastoconidia), pseudohyphae, and hyphal structures. Budding yeast cells and pseudohyphae are characteristic of most Candida species and occur in a few other genera, such as Trichosporum and Geotrichum . Budding yeast cells without pseudohyphae are characteristic of C glabrata , Cryptococcus species, and Histoplasma capsulatum .
Exfoliative cytology, swab cultures, and biopsy can be used to qualitatively determine the presence of oral Candida species, but these techniques are less sensitive than culture methods. Imprint culture and salivary rinse techniques allow quantification of Candida , and can help differentiate between normal oral candidal levels and increased levels associated with oral candidal infections. Regardless of the technique chosen, a microbiology and/or oral maxillofacial pathology laboratory service is required for a definitive diagnosis. Secondary to increased costs associated with these assays and difficulty in assessing required laboratory services, many clinicians routinely treat oral candidal infections empirically.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


