Human immunodeficiency virus (HIV)-associated oral disease among people living with HIV infection includes oral candidiasis, oral hairy leukoplakia, Kaposi sarcoma, oral warts, herpes simplex virus ulcers, major aphthous ulcers or ulcers not otherwise specified, HIV salivary gland disease, and atypical gingival and periodontal diseases. Diagnosis of some oral lesions is based on clinical appearance and behavior, whereas others require biopsy, culture, or imaging for definitive diagnosis. Management strategies including pharmacologic and nonpharmacologic approaches are discussed in this article. Dentists also need to be cognizant of the potential oral side effects of HIV antiretroviral medications.
Key points
- •
Human immunodeficiency virus (HIV)-associated oral lesions are numerous and diverse and may relate to opportunistic infections that occur in the setting of immune suppression.
- •
Presumptive HIV oral lesion diagnosis based on clinical appearance and lesion behavior may be sufficient for some benign-appearing lesions; whereas worrisome ulcerative lesions/masses require definitive diagnosis, usually based on histopathology.
- •
Patterns of oral disease prevalence and incidence have changed with improved HIV disease management and use of highly active antiretroviral therapy (HAART).
- •
HAART-related immune reconstitution inflammatory syndrome has reactivated some oral diseases and has resulted in other medication oral side effects in some patients.
- •
Pharmaceutical and nonpharmaceutical management are important considerations for HIV-associated oral lesions.
Introduction and epidemiology
Introduction
Human immunodeficiency virus (HIV) infection affects the host by targeting the CD4 positive T-lymphocyte population. HIV viral particles bind with lymphocytes and use the lymphocyte as a host factory, where additional HIV viral particles are produced. During this repeated process of viral replication, the lymphocyte is exhausted and destroyed, resulting in fewer T-helper lymphocytes available to protect the host from a variety of viral, fungal, bacterial, and protozoal opportunistic infections and other neoplastic diseases ( Fig. 1 ).
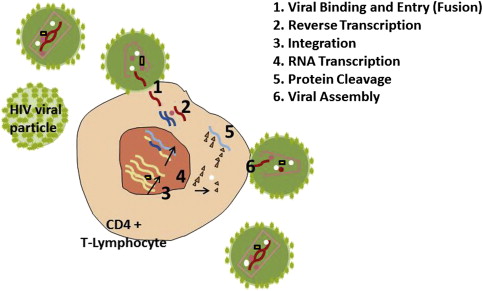
HIV disease management revolves around prevention, identification of the infected patient, and treatment of HIV with antiretroviral medications, as well as treatment of opportunistic infections and other conditions that arise. When medical management is effective at suppressing replication and reducing HIV viral loads to undetectable levels for prolonged periods, the patient is more likely to remain healthy and disease free. The standard of care is highly active antiretroviral therapy (HAART) regimens, using combinations of medications that target several stages of the HIV life cycle. The first anti-HIV medications developed in 1987, such as zidovudine, were nucleoside analogue reverse-transcriptase inhibitors and worked at stage 2, reverse transcription (see Fig. 1 ). In 1996, HIV protease inhibitors, such as indinavir, that worked at stage 6 were introduced. The newest classes of antiretroviral drugs are the fusion inhibitors and entry inhibitors (or CCR5 antagonists), working at stage 1 of viral binding and entry; and the integrase inhibitors working to block integration at stage 3.
Antiretroviral therapy is recommended for all HIV-infected individuals with the strength of evidence for the recommendation based on the pretreatment CD4 cell count. The strongest confidence in the need for therapy is among those with CD4 counts less than 500 cells/mm 3 , or those who are pregnant, or within the first 2 weeks of diagnosis of an opportunistic infection, regardless of CD4 count. Adherence to the antiretroviral medication regimens has long been a concern, with reasons for regimen switching including virologic, immunologic, or clinical failure and drug toxicity or intolerance. Several multiclass combination antiretroviral medications have been developed to reduce the pill burden for patients.
Nature of the problem
Oral lesions caused by opportunistic diseases occur in patients with HIV infection, whether or not they are on HAART. Although occurrence of some HIV-associated oral lesions has decreased after introduction of HAART, such as Kaposi sarcoma (KS), oral hairy leukoplakia (OHL), HIV-related gingival and periodontal disease, and major aphthous ulcers (MAU), others such as oropharyngeal candidiasis (OPC) have persisted. Some oral diseases, such as oral warts and HIV salivary gland disease, seem to have increased among those on HAART.
The oral mucosal epithelium lining the mouth plays an important role in protection of the host against infectious microorganisms. In HIV disease, the oral epithelium undergoes molecular alterations that place the host at increased susceptibility to comorbid oral infections. Although HAART is effective at increasing the quantity of CD4 lymphocytes, it provides incomplete immune recovery of the oral epithelial cells, leaving the host with increased susceptibility to oral disease and also exposing the patient to the toxic side effects of HAART. Chronic HIV disease is believed to silence the expression of multiple cellular proteins that in healthy individuals function to combat cellular stress and provide strong innate immune responses, further enhancing mucosal disease risk.
Definition
Orofacial manifestations of HIV in both adults and children were classified in the 1990s by collaborative expert groups using the following scheme :
- 1.
Lesions strongly associated with HIV infection
- 2.
Lesions less commonly associated with HIV infection
- 3.
Lesions seen in HIV infection (adults) or strongly associated but rare in children (children).
The lesions in each category are presented in Tables 1 and 2 . These expert consensus publications also provided presumptive and definitive diagnostic criteria and treatment considerations. Presumptive diagnostic criteria refer to the clinical features of the lesions, including signs and symptoms, whereas definitive criteria require specific laboratory tests.
| Group 1: Lesions strongly associated with HIV infection |
|
| Group 2: Lesions less commonly associated with HIV infection |
|
| Group 3: Lesions seen in HIV infection |
|
| Group 1: Lesions commonly associated with pediatric HIV infection |
|
| Group 2: lesions less commonly associated with pediatric HIV infection |
|
| Group 3: lesions strongly associated with HIV infection but rare in children |
|
Symptom criteria
In 2009, the Oral HIV/AIDS Research Alliance of the US AIDS Clinical Trials Group updated the HIV oral lesion case definitions in adults to make diagnosis easier for medical care providers and investigators in clinical trials and epidemiologic studies. In addition to clinical descriptors, patient-reported symptoms and lesion duration were added to the diagnostic matrix.
Prevalence/Incidence
HIV/AIDS
-
Prevalence (end of 2009):
- •
784,701 people in the United States were alive with a diagnosis of HIV infection (prevalence rate of 268.6 per 100,000 population).
- •
476,732 persons in the United States were alive with an AIDS diagnosis (prevalence rate of 155.3 per 100,000 population).
-
Incidence:
- •
Approximately 47,500 people in the United States newly acquire HIV each year, giving an incidence rate of 18.8 per 100,000, with the highest rates among individuals aged 25 to 34 years, men, and blacks/African Americans.
-
Mortality:
- •
All causes of death of persons with an HIV diagnosis in 2009 are estimated at 21,015 or 7.2 per 100,000 population.
With the constant rate of new HIV infections outpacing the death rate of persons with HIV disease, the numbers of individuals at risk for oral lesions related to HIV disease is increasing.
HIV oral lesions
Across the globe and throughout time, OPC has been the most common HIV oral lesion among all ages, genders, and races, with pseudomembranous candidiasis being the most readily diagnosed variant. Prevalence of various oral lesions varies greatly in the literature depending on the population characteristics, research study design, methods for assessment, geographic and clinic location, and time period of the study. General oral lesion prevalence ranges are given in Table 3 .
| HIV Oral Lesion | Prevalence in Adults (%) | Prevalence in Children (%) |
|---|---|---|
| Oral candidiasis | 0–94 | 18–72 |
| OHL | 0–43 | 0 |
| KS | 0–38 | 0 |
| Non-Hodgkin lymphoma | 0–5 | — |
| Linear gingival erythema | 0–22 | 0–48 |
| Necrotizing ulcerative gingivitis | 0–17 | — |
| Necrotizing ulcerative periodontitis | 0–23 | — |
| HSV ulcers | — | 1.7–24 |
| HIV salivary gland disease | — | 3–50 |
| Aphthous ulcers | — | 5–7 |
Although HAART therapy has resulted in some reduction of HIV oral lesions, they continue to persist in many populations. In a 2 year-follow-up retrospective study in Birmingham, Alabama among patients receiving HAART from 2000 to 2006, at least 1 HIV oral lesion was seen by medical providers in 35.8% of patients. OPC was most common, occurring in 74.9% of those with lesions, followed by oral herpes simplex virus (HSV) lesions in 10.4%, oral ulcers in 9.1%, and OHL in 3.2%. The potential for OHL to have been misdiagnosed as OPC is discussed in this report. Less commonly found were KS, undetermined stomatitis, and salivary gland swelling. Of those with HIV-related lesions, 59% had 1 lesion and 41% had at least 2 oral lesions. In this cohort, the person-visit incidence rate and person-visit recurrence rates for all episodes of HIV oral lesions were 2.15% and 0.66%, respectively. In the multicenter Women’s Interagency HIV Study cohort of HIV-infected women in the United States, the incidence rate (first events/person-visits) of erythematous or pseudomembranous OPC decreased from 7.35% to 3.43% after introduction of HAART, whereas incidence rates of OHL minimally declined from 2.54% to 2.35%, and incidence rates of warts minimally declined from 0.62% to 0.59%.
Worldwide/Regional Incidence and Mortality
Whereas some HIV oral lesions such as OPC, non-Hodgkin lymphoma (NHL), and HSV ulcerations are reported among HIV-infected populations on all continents, others are more common in certain geographic areas ( Table 4 ). Geographic distribution of HIV oral lesions is affected by HIV risk behaviors, gender, HIV management efficacy, social behaviors, race/ethnicity, socioeconomic status, and other undetermined local and regional environmental factors. Although many oral lesions contribute to morbidity, only the AIDS-defining malignancies contribute to the patient’s overall mortality.
| Oral Lesion | Geographic Location | Mortality |
|---|---|---|
| Fungal Infections | ||
| OPC | All continents | None |
| Viral Infections | ||
| OHL | North and South America, Europe and Asia | None |
| HSV ulceration | All continents | None |
| Human papillomavirus warts | North and South America and Europe | None |
| Neoplasms | ||
| KS | North America and Europe; sub-Saharan Africa | Pre-HAART 5-y survival: 30.7% Post-HAART 5-y survival: 62.5% |
| NHL | All continents | Pre-HAART 5-y survival: 16.2% Post-HAART 5-y survival: 46.0% |
| Bacterial Infections | ||
| Necrotizing ulcerative gingivitis/periodontitis | North America and Europe | None |
| Necrotizing stomatitis | Africa, Asia, and developing countries | None |
| Other Conditions | ||
| HIV salivary gland disease | Africa, Asia, and Southeast Asia; North America and Europe | None |
| MAU | North America and Europe | None |
Introduction and epidemiology
Introduction
Human immunodeficiency virus (HIV) infection affects the host by targeting the CD4 positive T-lymphocyte population. HIV viral particles bind with lymphocytes and use the lymphocyte as a host factory, where additional HIV viral particles are produced. During this repeated process of viral replication, the lymphocyte is exhausted and destroyed, resulting in fewer T-helper lymphocytes available to protect the host from a variety of viral, fungal, bacterial, and protozoal opportunistic infections and other neoplastic diseases ( Fig. 1 ).
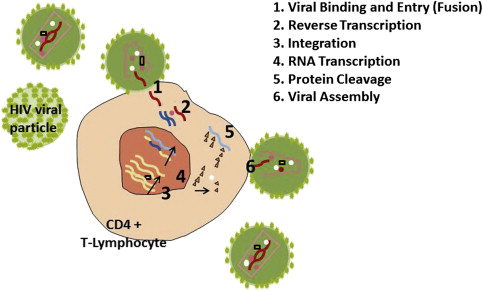
HIV disease management revolves around prevention, identification of the infected patient, and treatment of HIV with antiretroviral medications, as well as treatment of opportunistic infections and other conditions that arise. When medical management is effective at suppressing replication and reducing HIV viral loads to undetectable levels for prolonged periods, the patient is more likely to remain healthy and disease free. The standard of care is highly active antiretroviral therapy (HAART) regimens, using combinations of medications that target several stages of the HIV life cycle. The first anti-HIV medications developed in 1987, such as zidovudine, were nucleoside analogue reverse-transcriptase inhibitors and worked at stage 2, reverse transcription (see Fig. 1 ). In 1996, HIV protease inhibitors, such as indinavir, that worked at stage 6 were introduced. The newest classes of antiretroviral drugs are the fusion inhibitors and entry inhibitors (or CCR5 antagonists), working at stage 1 of viral binding and entry; and the integrase inhibitors working to block integration at stage 3.
Antiretroviral therapy is recommended for all HIV-infected individuals with the strength of evidence for the recommendation based on the pretreatment CD4 cell count. The strongest confidence in the need for therapy is among those with CD4 counts less than 500 cells/mm 3 , or those who are pregnant, or within the first 2 weeks of diagnosis of an opportunistic infection, regardless of CD4 count. Adherence to the antiretroviral medication regimens has long been a concern, with reasons for regimen switching including virologic, immunologic, or clinical failure and drug toxicity or intolerance. Several multiclass combination antiretroviral medications have been developed to reduce the pill burden for patients.
Nature of the problem
Oral lesions caused by opportunistic diseases occur in patients with HIV infection, whether or not they are on HAART. Although occurrence of some HIV-associated oral lesions has decreased after introduction of HAART, such as Kaposi sarcoma (KS), oral hairy leukoplakia (OHL), HIV-related gingival and periodontal disease, and major aphthous ulcers (MAU), others such as oropharyngeal candidiasis (OPC) have persisted. Some oral diseases, such as oral warts and HIV salivary gland disease, seem to have increased among those on HAART.
The oral mucosal epithelium lining the mouth plays an important role in protection of the host against infectious microorganisms. In HIV disease, the oral epithelium undergoes molecular alterations that place the host at increased susceptibility to comorbid oral infections. Although HAART is effective at increasing the quantity of CD4 lymphocytes, it provides incomplete immune recovery of the oral epithelial cells, leaving the host with increased susceptibility to oral disease and also exposing the patient to the toxic side effects of HAART. Chronic HIV disease is believed to silence the expression of multiple cellular proteins that in healthy individuals function to combat cellular stress and provide strong innate immune responses, further enhancing mucosal disease risk.
Definition
Orofacial manifestations of HIV in both adults and children were classified in the 1990s by collaborative expert groups using the following scheme :
- 1.
Lesions strongly associated with HIV infection
- 2.
Lesions less commonly associated with HIV infection
- 3.
Lesions seen in HIV infection (adults) or strongly associated but rare in children (children).
The lesions in each category are presented in Tables 1 and 2 . These expert consensus publications also provided presumptive and definitive diagnostic criteria and treatment considerations. Presumptive diagnostic criteria refer to the clinical features of the lesions, including signs and symptoms, whereas definitive criteria require specific laboratory tests.
| Group 1: Lesions strongly associated with HIV infection |
|
| Group 2: Lesions less commonly associated with HIV infection |
|
| Group 3: Lesions seen in HIV infection |
|
| Group 1: Lesions commonly associated with pediatric HIV infection |
|
| Group 2: lesions less commonly associated with pediatric HIV infection |
|
| Group 3: lesions strongly associated with HIV infection but rare in children |
|
Symptom criteria
In 2009, the Oral HIV/AIDS Research Alliance of the US AIDS Clinical Trials Group updated the HIV oral lesion case definitions in adults to make diagnosis easier for medical care providers and investigators in clinical trials and epidemiologic studies. In addition to clinical descriptors, patient-reported symptoms and lesion duration were added to the diagnostic matrix.
Prevalence/Incidence
HIV/AIDS
-
Prevalence (end of 2009):
- •
784,701 people in the United States were alive with a diagnosis of HIV infection (prevalence rate of 268.6 per 100,000 population).
- •
476,732 persons in the United States were alive with an AIDS diagnosis (prevalence rate of 155.3 per 100,000 population).
-
Incidence:
- •
Approximately 47,500 people in the United States newly acquire HIV each year, giving an incidence rate of 18.8 per 100,000, with the highest rates among individuals aged 25 to 34 years, men, and blacks/African Americans.
-
Mortality:
- •
All causes of death of persons with an HIV diagnosis in 2009 are estimated at 21,015 or 7.2 per 100,000 population.
With the constant rate of new HIV infections outpacing the death rate of persons with HIV disease, the numbers of individuals at risk for oral lesions related to HIV disease is increasing.
HIV oral lesions
Across the globe and throughout time, OPC has been the most common HIV oral lesion among all ages, genders, and races, with pseudomembranous candidiasis being the most readily diagnosed variant. Prevalence of various oral lesions varies greatly in the literature depending on the population characteristics, research study design, methods for assessment, geographic and clinic location, and time period of the study. General oral lesion prevalence ranges are given in Table 3 .
| HIV Oral Lesion | Prevalence in Adults (%) | Prevalence in Children (%) |
|---|---|---|
| Oral candidiasis | 0–94 | 18–72 |
| OHL | 0–43 | 0 |
| KS | 0–38 | 0 |
| Non-Hodgkin lymphoma | 0–5 | — |
| Linear gingival erythema | 0–22 | 0–48 |
| Necrotizing ulcerative gingivitis | 0–17 | — |
| Necrotizing ulcerative periodontitis | 0–23 | — |
| HSV ulcers | — | 1.7–24 |
| HIV salivary gland disease | — | 3–50 |
| Aphthous ulcers | — | 5–7 |
Although HAART therapy has resulted in some reduction of HIV oral lesions, they continue to persist in many populations. In a 2 year-follow-up retrospective study in Birmingham, Alabama among patients receiving HAART from 2000 to 2006, at least 1 HIV oral lesion was seen by medical providers in 35.8% of patients. OPC was most common, occurring in 74.9% of those with lesions, followed by oral herpes simplex virus (HSV) lesions in 10.4%, oral ulcers in 9.1%, and OHL in 3.2%. The potential for OHL to have been misdiagnosed as OPC is discussed in this report. Less commonly found were KS, undetermined stomatitis, and salivary gland swelling. Of those with HIV-related lesions, 59% had 1 lesion and 41% had at least 2 oral lesions. In this cohort, the person-visit incidence rate and person-visit recurrence rates for all episodes of HIV oral lesions were 2.15% and 0.66%, respectively. In the multicenter Women’s Interagency HIV Study cohort of HIV-infected women in the United States, the incidence rate (first events/person-visits) of erythematous or pseudomembranous OPC decreased from 7.35% to 3.43% after introduction of HAART, whereas incidence rates of OHL minimally declined from 2.54% to 2.35%, and incidence rates of warts minimally declined from 0.62% to 0.59%.
Worldwide/Regional Incidence and Mortality
Whereas some HIV oral lesions such as OPC, non-Hodgkin lymphoma (NHL), and HSV ulcerations are reported among HIV-infected populations on all continents, others are more common in certain geographic areas ( Table 4 ). Geographic distribution of HIV oral lesions is affected by HIV risk behaviors, gender, HIV management efficacy, social behaviors, race/ethnicity, socioeconomic status, and other undetermined local and regional environmental factors. Although many oral lesions contribute to morbidity, only the AIDS-defining malignancies contribute to the patient’s overall mortality.



