(8.1) DIANA DUDEA, BOGDAN CULIC
(8.2) BOGDAN DIMITRIU, CONSTANTIN VÂRLAN
Chapter VIII
TOOTH DISCOLORATION
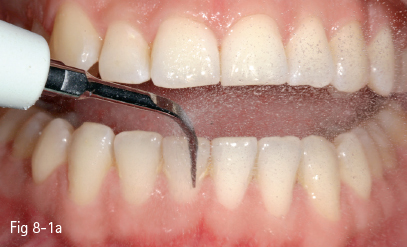
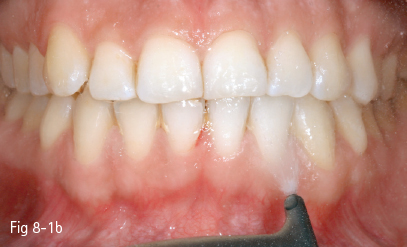
Fig 8-1 Professional prophylactic cleaning: a. Ultrasound scaling. b. Air abrasion.
Among the reasons that motivate patients to seek dental treatment is the need to improve their tooth color. Properly aligned, light-colored teeth are usually the gold standard regarding the smile’s appearance. The characteristics, as well as the importance, of dental color are undoubtedly differently perceived among individuals, both being influenced by factors such as culture, education, ethnicity, and age. However, it is unanimously recognized that the dental shape and color have a major influence on dental esthetics and, ultimately, upon the facial appearance.
Consequently, methods aimed at improving dental color are particularly important in esthetic dental treatments. The treatment methods differ, according to the etiology of color changes, and range from oral hygiene products and prophylaxis to dental bleaching and/or restorative treatments (veneers, crowns).
For the best outcome, these methods should be combined into an individual treatment plan for each patient, in accordance with the etiology and severity of the respective discoloration. Consequently, before deciding the treatment plan, a thorough clinical examination is required in order to establish the etiology, the location of discoloration to one or several teeth, and the intensity of the color changes.1,2
8.1.1 Etiology of tooth discoloration
Since the genetics of dental color are not fully understood, it is important to establish the influence of external factors on tooth color.
According to the etiological factors and the mechanism of penetration and fixation of the coloring agents into the dental structure, tooth discoloration can be classified as extrinsic, intrinsic, or internalized.3–7
8.1.1.1 Extrinsic tooth discoloration
Extrinsic tooth discoloration is caused by colorants incorporated into the mucoprotein pellicle that covers the dental surface. These stains can be removed by using oral hygiene products or prophylaxis (Figs 8-1a and 8-1b).
They are mostly produced by food colorants (natural or synthetic), but also by drugs (eg, iron-based drugs), mouth rinses (chlorhexidine, listerine, stannous fluoride, phenolic/essential oil-based), or by the activity of chromogenic bacteria. The use of tobacco (cigarette, cigar, pipe smoking, tobacco or betel chewing) is also responsible for extrinsic and internalized discoloration (Figs 8-2a and 8-2b).8–10
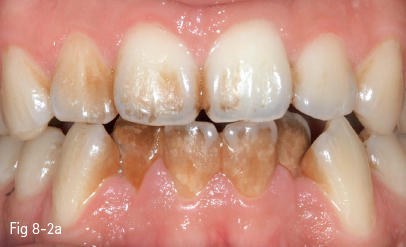
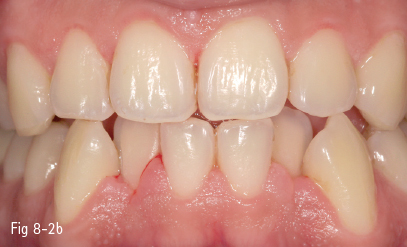
Fig 8-2 Extrinsic tooth discoloration. a. Initially b. After professional cleaning. (Images courtesy of Dr Florin Lăzărescu.)
The Nathoo classification divides extrinsic discoloration into three categories:4,5
• Nathoo type 1 (N1). The colored substances adhere to the dental surface directly or through the salivary protein pellicle; the staining is similar in color to that of the chromogenic agent (tea, coffee, other colored food and beverages).
• Nathoo type 2 (N2). These stains are caused by food colorants that change their color after fixing on the tooth surface.
• Nathoo type 3 (N3). The discoloration is caused by colorless or pre-chromogenic substances that adhere to the dental surface (eg, chlorhexidine staining); subsequently, they induce a chemical reaction responsible for pigmentation. This category includes chlorhexidine, a cationic antiseptic used as an antibacterial mouth rinse, particularly in periodontal disease; its potential to stain the teeth, mucosa, as well as restorative materials (acrylates, composites) after several weeks of use is well documented.3–9
8.1.1.2 Internalized discoloration
Internalized discoloration is caused by colorants from the oral environment that penetrate the enamel and dentin layers and bind chemically to hydroxyapatite and collagen in these structures. The adsorption of the staining molecules is due to the relative permeability of the enamel and, particularly, of the dentin; it is enhanced by structural defects occurring either during tooth formation or in a posteruptive stage.3,6,11
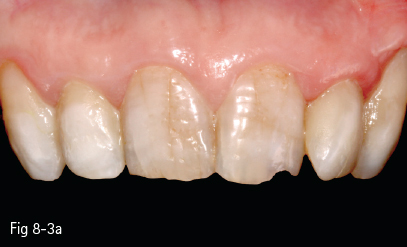
Therefore, in these patients, even if surface staining is removed through cleaning, color improvement can only be achieved by bleaching methods that remove pigments through a chemical mechanism (Figs 8-3a and 8-3b).
Both extrinsic and internalized discoloration depend not only on diet, oral hygiene habits, and dental structure, but also on other factors, including the composition of the mucoprotein pellicle on the enamel surface, responsible for the staining adsorption.12 Reduced salivary flow (in Sjögren syndrome, cervicofacial irradiation, or anticholinergic drugs) enhances the staining deposition. However, the wide range in dental color variation increases the difficulty of establishing the etiology and predicting the treatment outcome.3,7,11
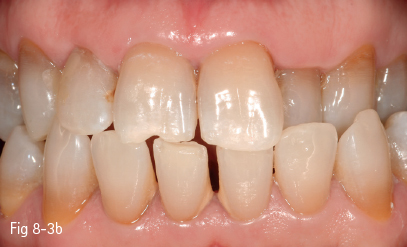
Fig 8-3 a, b Internalized discoloration. (Images courtesy of Dr Florin Lăzărescu.)
8.1.1.3 Intrinsic tooth discoloration
In these situations, the chromophore is attached to the mineralized dental structures, inducing an alteration of the light transmission. The colorant penetrates from the inside, as a result of pulp pathology (hemorrhage or necrosis, endodontic treatments), inducing local discoloration. Other causes of local discoloration are dental decays or restorative materials that can either stain the dental hard structures or cause shadows to be visible through the translucent enamel (see section 8.2). The discoloration may also have a systemic origin, which frequently induces generalized staining of the dentition.
Consequently, depending on the etiology, color changes may affect a single tooth, a group of teeth, or the full deciduous or permanent dentition.
The intrinsic discoloration is caused either by factors that act during the formation and mineralization of the tooth or by posteruptive events.
8.1.1.4 Pre-eruptive action factors
The factors with a pre-eruptive action may have different origins and pathogenic mechanisms, which explains the variation in the whitening response to chemical treatments.
Thus, certain discolorations are the result of inclusion in the dental structures of substances from abnormal metabolic chains: hyperbilirubinemia in hemolytic anemia or hemolytic jaundice; thalassemia; congenital erythropoietic porphyria; and alkaptonuria. These forms are rare and usually associated with generalized discoloration.
Developmental defects of the enamel and dentin, affecting either their organic or inorganic component, may induce white, yellow or brown discoloration that is variable in depth and more or less progressive in time. In these cases, staining is caused by the increased fixation of food colorants to the porous enamel or dentin.
These spots can be removed by microabrasion or chemical treatment, but the results are unpredictable.
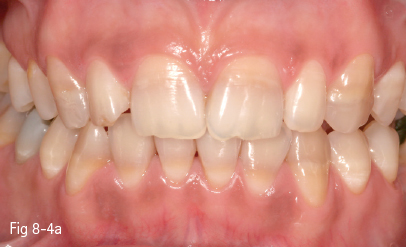
Chemicals that attach most often to the developing dental tissues are drug molecules (antibiotics – tetracycline in different formulas, or iron-based medication) (Figs 8-4a and 8-4b).
Tetracycline discoloration is due to its administration during tooth formation, but also after the tooth has erupted. The pathogenic mechanism is explained by chelation of the tetracycline molecule by the calcium ions in the hydroxyapatite of the enamel and dentin. Staining depends on the patient’s age, the duration of the administration, and the tetracycline derivative (tetracycline achromycin – yellow staining; tetracycline minocycline – black staining).11
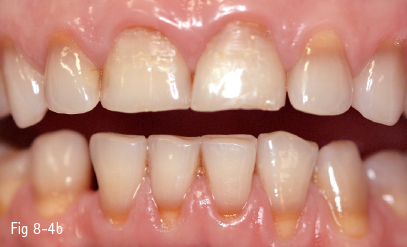
Fig 8-4 a, b Discoloration induced by tetracycline.
Tetracycline discoloration includes mild or moderate stains, uniformly distributed on the dental surface, or more severe cases characterized by horizontal bands parallel to the incisal edge. A characteristic of tetracycline discoloration is the fluorescence, which becomes visible in UV light. Stains are more intense in anterior teeth and become darker in time due to photooxidation under daily light exposure.
Tetracycline administration should be avoided up to the age of seven. Also, due to the risk of transplacental passage, this class of antibiotics is restricted during pregnancy. Tetracycline discolorations need longer exposure to bleaching products, have an unpredictable evolution, and have frequent postbleaching relapses.
Minocycline, which is administered posteruptively in prolonged cures for the treatment of acne, periodontal disease, or rheumatoid arthritis, has been associated with tooth color pathology.1,5,11–13
Dental fluorosis is caused by the alteration of enamel formation due to an increased fluoride intake. It is characterized by white opaque or brown stains (due to the adsorption of chromophore agents from the oral environment), and, in more severe cases, by lack of substance.11
Color changes due to aging
Color changes due to aging are a complex, multifactorial transformation. Among the causes are the penetration of staining agents from the oral environment into the dental tissues, the thinning and the increasing translucency of the enamel layer, as well as the deposition of secondary and reparative dentin, which has an orange-brown shade.
8.1.2 Clinical examination
A proper etiological diagnosis and a complex treatment plan require a detailed assessment of every patient, based on a thorough clinical and photographic investigation.
The medical and dental history should include:
• General disorders associated with tooth discoloration.
• History of medication or mouth rinse responsible for dental discoloration.
• History of dental discoloration: onset and progression (did it show at the time of tooth eruption, or subsequently).
• Possibility of correlating color changes with certain events (trauma, dental treatments).
• Information regarding diet and smoking.
• Oral hygiene habits, whitening products (rinse solutions, whitening pastes, gels).
• History of in-office or dentist-supervised whitening treatments.
Information on the dental color self-perception and the patient’s own desire to improve the dental shade (what “whiter teeth” means in his/her opinion) should not be omitted.
It has been suggested that it is not only important to increase the teeth lightness by a certain number of shade tabs, but also to gain a dental color that closely matches the color of the sclera – the white of the eyes.14 Esthetic questionnaires used for recording this information play an essential role in dentist-patient communication; furthermore, they may be documents with legal value.
Data regarding dental color are gathered during the patient’s clinical examination. In addition to the protocol presented in Chapter II, the following questions regarding tooth discoloration should also be answered:
• What tooth, group of teeth, or dental arch is/are affected by discoloration?
• What is the extension of the color change on the dental surface?
• What is the dental shade (expressed in the Vita Classical or 3D Master coding system)? The shade color can be either visually or instrumentally recorded.
• Is it a superficial, external staining or an inherent discoloration of the tooth structure?
• Is it a uniform discoloration, or does it appear as colored or opaque bands, spots, or lines on the dental surface?
• Are there any restorations (composite fillings, veneers, crowns), and what is their chromatic appearance in comparison with the natural dentition? Would the patient want them replaced after bleaching if they do not match the final dental color?
In addition to the clinical data, dental photography is of paramount importance. In order to have a realistic and relevant image of the baseline, the tooth should be captured next to the samples of different shade guides, either classical (Vitapan Classical) or value-based (Vitapan 3D Master; Linearguide 3D Master).
When bleaching treatments are envisaged, the Vita Bleachguide 3D Master shade guide is recommended, which is especially designed for monitoring the efficacy of tooth whitening.15 It includes high-value shade tabs. In addition, the distribution of the samples in the color space is uniform, and, on account of this, the results of whitening may be better quantified (Fig 8-5).
Single-tooth and panoramic radiographs should be obtained in order to prevent overlooking the pathology responsible for dental discoloration. In some cases, color or translucency alteration is the only sign of teeth with pulp necrosis or calcific metamorphoses. Vitality tests are also indicated in these cases (see section 8.2).11,14
8.1.3 Treatment of tooth discoloration
The medical approach to dental discoloration involves an integrative, complex treatment plan that addresses all the pathological conditions recorded during the clinical examination. Moreover, the improvement of the dental shade is often obtained through the combination of several treatment methods.
When treatment planning is considered, it is absolutely necessary to know the etiology and the clinical form of discoloration (extrinsic, internalized, or intrinsic), the intensity and the shade of staining, its location to one or more teeth, as well as other conditions that may affect the teeth: dental decays, fissures or fractures, and endodontic and restorative treatments (including their quality).
Depending on the result of the examination, the treatment plan may include:
• Prophylactic cleaning, and recommendations regarding at-home use of additional whitening products.
• Microabrasion and macroabrasion.
• Dental bleaching, with oxidative substances, in various protocols:
–At-home, patient-managed bleaching with over-the-counter products.
–Dentist-supervised bleaching with low-and medium-concentration peroxide-based products.
–In-office sessions based on high-concentration substances (for vital and/or non-vital teeth).
–Whitening methods specific for non-vital teeth.
–Partial or full coverage with composite resins or ceramic veneers, metal-ceramic or ceramic crowns.
8.1.3.1 Prophylactic cleaning and whitening products recommended for oral hygiene
Professional cleaning is aimed at removing bacterial plaque and calculus debris through scaling, followed by professional tooth brushing.7,16
Professional tooth brushing is a prophylactic method aimed at removing bacterial plaque and extrinsic staining. It is carried out with rotating instruments at low speed, using rubber cones for proximal spaces and rubber cups for vestibular and oral surfaces. It is combined with prophylactic fluoride and abrasive pastes,7,11,17 and with an air-flow projection of sodium bicarbonate particles onto the dental surface.
Products aimed at lightening the teeth may be recommended during cleaning sessions. However, whitening toothpastes and mouth rinses are frequently used by patients without a specific indication or medical control.
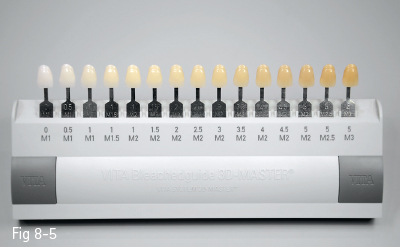
Fig 8-5 Vita Bleachguide 3D Master shade guide.
There is a myriad of oral hygiene products used as adjuvants for tooth whitening, and whitening toothpastes can have different action mechanisms.11,12,14,16
Toothpastes with an abrasive mechanism are aimed at removing extrinsic pigmentation and inhibiting its subsequent deposition. Currently, the majority of toothpastes exhibit this mechanism, their abrasive effect depending on the size and concentration of abrasive particles.17 Active abrasive agents include carbonates, calcium phosphates and pyrophosphates, alumina, and sodium bicarbonate. The last one is considered an abrasive agent with small-size particles, which can address details of the dental surface successfully.11
An exaggerated abrasive effect can affect the thickness of the enamel layer and, in addition, induce a yellowish shade, due to the underlying dentin.
Toothpastes with a chemical mechanism include those based on peroxides (hydrogen, calcium peroxides), sodium citrate, or sodium phosphates.17 Due to the short contact time of the pastes with the tooth surface, the whitening effects are limited.11,18
Toothpastes with an enzymatic mechanism, which contain the enzyme papain, are aimed at reducing the formation of bacterial plaque on the tooth surface.
Cosmetic toothpastes based on an optical effect form a pellicle, which infiltrates the tooth surface details, inducing the perception of whiter teeth.11,17 The optical effect is based on substances that change tooth color perception from a yellow to a bluish shade (bluecovarine), or on titanium dioxide.11,17–19
In addition to toothpastes, mouth rinses of various compositions contribute to improving tooth color. Some peroxide-based products may be purchased directly by the patient and used during a patient-managed dental bleaching protocol. These products will be presented in section 8.1.3.3.
8.1.3.2 Microabrasion
Microabrasion is recommended in the case of limited areas of discoloration that affect the superficial enamel structure (eg, dental fluorosis) through the combination of acids and abrasive pastes.
Macroabrasion consists of removing the superficial, discolored enamel layer over a limited area using fine diamond or carbide burs. Another option for removing the stains is air abrasion, with fine abrasive particles.
Microabrasion and macroabrasion should be regarded as intermediary methods between bleaching techniques and more invasive methods like veneers or crowns.
In the case of over-reduction, the labial surface should be restored with dental composites. Fluoridation of the abraded surface is also recommended.11,14
Dental bleaching is recommended for the treatment of internalized and intrinsic tooth discoloration and is basically aimed at altering the optical parameters of dental structures by eliminating staining molecules.
It is believed that chromophores become attached to hard dental tissues by two mechanisms:
• Interaction with the organic component in the enamel and dentin (eg, polyphenols from tea bind through five hydroxyl groups to the organic substance), forming stable compounds.3,13,20
• Calcium ion chelation (eg, hydroquinone in tetracycline).1,12,13
The removal of organic chromophores from dental structures is based on oxidative mechanisms. The bleaching products that are currently in use are hydrogen peroxide, carbamide peroxide, and sodium perborate.
Hydrogen peroxide diffuses through enamel and dentin. Under alkaline conditions, it acts as a strong oxidizing agent through the formation of free radicals, reactive oxygen molecules, and hydrogen peroxide anions.11,21,22
These compounds may cleave the double bonds that bind pigment molecules to the dental structure, or oxidize their chemical moieties, forming soluble molecules or less heavily pigmented constituents. These are believed to reflect less light, thus creating a whitening effect.1,3,12,13,16,21
However, new insights into the chemical affinity of hydrogen peroxide during its diffusion through enamel and dentin indicate its oxidizing effect on the organic compounds of dentin,23 as well as enamel.21 According to Eimar et al, in addition to the effect of hydrogen peroxide on the chromophores, which are, as a result, modified into more translucent molecules reflecting less light, hydrogen peroxide also oxidizes the transparent organic matrix into a more opaque, whiter material. This might explain the variability of the bleaching results, which may be due to the differences of the enamel concentration in organic material among individuals, as well as the lower bleaching efficacy in the elderly, as a consequence of a lower protein content in the enamel.21
Hydrogen peroxide can be used directly in different concentrations, or is generated locally by the decomposition of an organic derivate, carbamide peroxide or urea peroxide (CH6N2O3). During the bleaching treatment, carbamide peroxide is decomposed into urea (CH4N2O) and hydrogen peroxide (H2O2). As described previously, hydrogen peroxide produces reactive oxygen species able to remove or oxidize a wide variety of organic or inorganic structures.11,21
Carbamide peroxide is contained in gels with different concentrations:
• 10% (equivalent to 3.35% hydrogen peroxide).
• 15% (equivalent to 5.4% hydrogen peroxide).
• 20% (equivalent to 7% hydrogen peroxide).
• 35% (equivalent to 10% hydrogen peroxide).
Another oxidative precursor is sodium perborate, used for non-vital tooth whitening, in an endodontic application (see section 8.2).
Indications for bleaching treatment
• Vital or non-vital dischromic teeth, as the treatment of choice or in combination with subsequent prosthetic treatments.
• Internalized and intrinsic discolorations, particularly in yellow, orange, and brown shades.
• Patients in a good general health status, compliant, with adequate oral hygiene.
Contraindications for bleaching treatment
• Pregnant or lactating women.
• Disease of the oral mucosa, a burning sensation that can indicate allergic reactions.
• Severe discoloration requiring other treatment methods (veneers, crowns). In these situations, bleaching may be associated with prosthetic treatments.
• Composite or ceramic restorations that are not to be replaced. The patient should be informed that the restorations may not match the bleached teeth after treatment, since the dental materials do not respond in the same way to bleaching as the dental structures do.13,14,16,24
There are also situations that should be regarded as relative contraindications due to dischromic conditions with a poor prognosis to whitening procedures:
• Gray or bluish shades are more difficult to remove by oxidizing treatment.
• Teeth with a highly translucent aspect; bleaching may cause increased translucency that will be perceived as darker areas on the dental surface.
• Saturated color in the gingival area or in the root portion is less responsive to bleaching due to differences in the dentinal structure in this zone.14
In addition, a cautious attitude is recommended in the case of reported dental sensitivity due to gingival recession, abrasion, cervical lesions (with dentinal exposure), or in children with a larger dental pulp, in order to reduce the risk of pulpal inflammation.
At-home bleaching is based on over-the-counter, peroxide-based products that can be used by patients with or without medical recommendation and control. The commercial forms and protocols of application differ, and usually depend on the desired whitening results.
Adhesive strips containing 6% to 14% hydrogen peroxide have a configuration adapted for the maxillary and mandibular anterior teeth. The strips are recommended to be placed on the tooth surface for 30 minutes, once or twice a day.11,14,24
Peroxide-based gels, in combination with prefabricated trays, may cause lesions of the gingival fibromucosa due to the lack of intimate contact of the tray with the oral tissues, and the reflux of the whitening material. However, the most important inconvenience of the over-the-counter methods is the lack of medical supervision.
In the case of the over-the-counter methods, the treatment is not administered following a clearly formulated diagnosis, which can mask symptoms of dental conditions that should be treated by other methods (dental decays, pulp necrosis, periapical lesions). In addition, the results of treatment are not professionally monitored, and the overdose of inadequate products or protocols can induce harmful side effects, such as enamel erosion and dentin sensitivity.1,11,14,24
Dentist-supervised bleaching
Dentist-supervised bleaching is done with low- and medium-concentration peroxide gels and custom-fitted trays.
Bleaching is initiated in the dental office, following a comprehensive examination, and continued as a patient-managed treatment.
This method involves the use of 10%, 15%, and 20% carbamide peroxide gels, or the more recently introduced 7.5% to 9.5% hydrogen peroxide gels, as well as custom-fitted trays that are fabricated on models.
The following stages are involved:
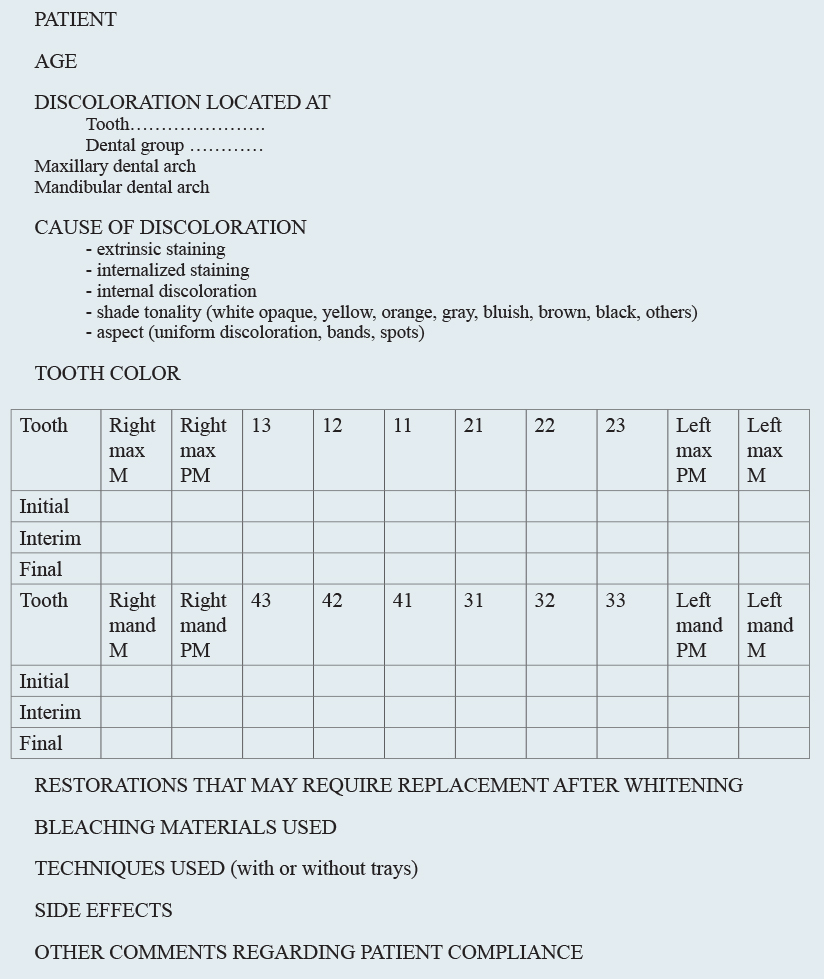
• Examination of the patient, according to the previously presented plan. The data gathered during the initial examination, the dental-color changes, and the bleaching materials and protocols used (including their side effects) could be synthetized onto a chart for each patient (Fig 8-6).
• The initial color can be expressed by using as a reference the color coding of the shade guides, or the L*a*b* values recorded with instruments. Initial photos, with the shade guides in position, are also recommended.
• Professional cleaning aims to remove plaque, calculus, and external pigmentations. If there are signs of sensitivity, the first bleaching session is postponed for several days.14
• Accurate impressions of the dental arches, with alginate or silicones (Figs 8-7 and 8-8).
8.1.3.4 Fabrication of models and trays using the thermoformation technique
This technique involves the intimate fitting of a vacuum-formed tray material – usually a plastic polyethylene foil – on the surface of the previously prepared model.
Depending on the indication, the trays may or may not include vestibular reservoirs for the whitening gel; in addition, they may have scalloped or non-scalloped gingival margins. The vestibular reservoir increases the amount of material in contact with the dental surface. Scalloped margins, parallel to the gingival margins at the cervical level, or 1 mm incisally/occlusally to it, have the advantage of reducing the contact of the peroxides with the gingiva. Scalloping is recommended in the case of fragile gingival tissues, or when highly concentrated peroxides or less-fluid bleaching gels are to be used.14 This marginal configuration reduces the gel reflux capacity, as well as the risk of gingival lesions.
Fig 8-6 Chart for dental bleaching.
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses