Introduction
Our aim was to prospectively assess in-vivo color alterations of natural teeth associated with fixed orthodontic treatment.
Methods
Twenty-six consecutive patients were treated with fixed appliances bonded with a chemically cured or a light-cured resin with a split-mouth design. The spectrophotometric data of a standardized labial circular area of all teeth were recorded, before bracket bonding and after debonding and cleaning procedures. The color parameters of the Commission Internationale de l’Eclairage—L∗, a∗, and b∗ (lightness, red/green, and blue/yellow)—were measured for each adhesive and type of tooth, and the corresponding color differences (ΔE) between the interval groups were calculated. The effect of these parameters on color was assessed with 3-way mixed analysis of variance (ANOVA) and the Bonferroni comparisons test (α <0.05).
Results
Orthodontic treatment was associated with changes in color parameters. The L∗ values decreased ( P <0.001), whereas the a∗ and b∗ values increased ( P <0.001) at the end of treatment. All measured types of teeth demonstrated significant color changes (ΔE); their mean differences ranged from 2.12 to 3.61 ΔE units. Chemically cured resin was associated with greater color changes than light-cured composite.
Conclusions
The color of natural teeth is changed in various ways after fixed orthodontic treatment.
The adverse influence of orthodontic treatment with fixed appliances on the enamel has been comprehensively reviewed. However, the incidence of orthodontic bonding-induced tooth-color alterations has not been thoroughly investigated. Until recently, only a few in-vitro studies have assessed the enamel-color changes during bracket bonding and debonding. Although the results of these studies showed that some alterations in tooth color are inevitable, no matter whether these changes are visually recognizable, in-vitro tests might not be a reliable reflection of the clinical situation. Natural tooth-color determination is affected by many factors in the oral cavity, such as lighting conditions of the surrounding environment, the light scattered from adjacent perioral and gingival tissues, and the resting salivary flow rates that influence tooth hydration and, consequently, the reflective index of the underlying surface.
There are 2 common methods of analyzing in vivo the apparent tooth color: visual determination and instrumental measurement. Visual determination by comparison of teeth and shade guides is considered highly subjective but still remains the most frequently applied method in dentistry for color determination. However, several factors such as external light conditions, experience, age, fatigue of the human eye, and the inherent limitations of the contemporary shade guides can influence the consistency of visual color selection and specification. The general demand for objective color matching in dentistry coupled with rapid advances in optical electronic sensors and computer technology has made instrumental measurement devices a supplementary adjunct to visual tooth-color evaluation. Nowadays, various commercial systems, including tristimulus colorimeters, spectroradiometers, spectrophotometers, and digital color analyzers, are used in clinical and research settings for objective color specification.
The aim of this prospective clinical trial was to assess in vivo the color alterations of natural teeth associated with comprehensive orthodontic treatment, by using a reflectance spectrophotometer. The null hypothesis was that there was no statistical significant difference in the color parameters L∗, a∗, and b∗ (lightness, red/green, and blue/yellow) of the Commission Internationale de l’Eclairage (CIE) for natural teeth before and after orthodontic treatment.
Material and methods
The initial study sample consisted of 34 consecutive patients, who started orthodontic treatment at the Postgraduate Clinic, Department of Orthodontics, School of Dentistry, Aristotle University of Thessaloniki in Greece, with the following characteristics: (1) need for comprehensive orthodontic treatment by fixed appliances in both dental arches, (2) permanent dentition, (3) no severe crowding (<4 mm), (4) no plaque accumulation and gingival inflammation before bracket bonding, (5) no dental caries, prosthetic restorations, and decalcifications in the teeth under examination, and (6) no smoking habit.
During orthodontic treatment, all patients were instructed to brush regularly with white fluoride toothpaste. Daily use of chlorhexidine mouth rinses was not allowed to prevent possible extrinsic tooth staining from the various agents in these solutions. The oral hygiene status was frequently evaluated, and, when necessary, reinforcement instructions were given. The Ethical Committee of Aristotle University of Thessaloniki ensured that the procedures in this trial were conducted in accordance with the guidelines of good clinical practice. In addition, informed consent was received from all patients or their guardians.
After removal of the orthodontic appliances, the dental and periodontal statuses of each patient were examined in detail by the same clinician (A.K.). Based on the inclusion criteria of this clinical trial, 8 patients with severe gingival inflammation or decalcifications on the buccal tooth surfaces were excluded from the study. However, no patient had clinically visible enamel-color alterations indicating adverse pulp reaction. Consequently, the final study sample comprised 26 orthodontic patients (13 girls, 13 boys). Before treatment, they had a mean age of 13 years 7 months (SD, ± 2.9 years). The duration of active treatment ranged from 18 to 26 months.
All teeth were polished with nonfluoridated and oil-free pumice, rinsed, dried completely for 10 seconds, and then etched with 37% phosphoric acid gel for 30 seconds. Stainless steel brackets were bonded according to the manufacturer’s instructions with 2 resin adhesives—chemically cured (System 1+, Ormco, Glendora, Calif) and light-cured (Transbond XT, 3M Unitek, Monrovia, Calif)—by using a split-mouth design. At the end of active treatment, bracket debonding and cleaning procedures were performed by 1 clinician (A.K.) using the same debonding pliers (Ormco) and a new carbide bur (LA1172, Ormco) attached to a low-speed hand piece. The extent of the overall resin removal was determined by visual inspection of the dried enamel surfaces under the dental operating light as well as by constant examination of the surface roughness with a dental explorer.
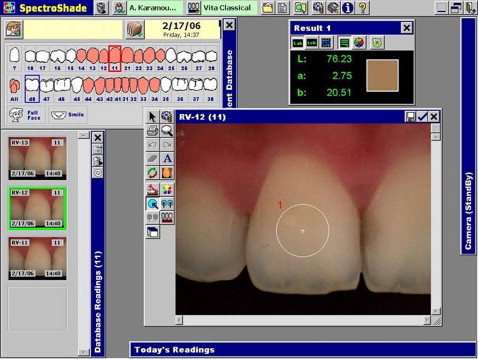
The reflectance spectrophotometer SpectroShade (LUA005, MHT Optic Research AG, Zurich, Switzerland; software version, 2.20) was used to objectively assess color alterations of natural teeth before and after orthodontic treatment. According to a previous investigation, this instrument provides precise measurements during longitudinal evaluation of tooth color in vivo. Color quantification was based on the CIE’s L∗, a∗, and b∗ color system that uses 3 parameters to define color: the L∗ coordinate is a measure of lightness similar to value in the Munsell system and ranges from 0 (black) to 100 (white); the a∗ and b∗ coordinates represent positions on red/green and yellow/blue axes, respectively. The readings of a∗ and b∗ combined provided the same information as hue and chroma in the Munsell system.
A standardized protocol of tooth preparation and in-vivo spectrophotometric color evaluation was adopted for the patients, and all measurements were performed in the same examination room with standardized lighting conditions. Thirty minutes before the measuring process, all teeth were cleansed with a plain white dentifrice with a rubber polishing cup on a low-speed hand piece and thoroughly rinsed with water to ensure complete removal of toothpaste. The proper sterile mouthpiece of the spectrophotometer’s intraoral camera was positioned at a 90° angle on the alveolar process above the respective tooth, which was centered in the yellow target box depicted on the computer screen. The spectrophotometer was calibrated before each image-capture session with white and green ceramic tiles supplied by the manufacturer. The calibration process compensated for any deviation in the quantity of illumination output from the internal light source. The spectrophotometric data of each tooth were recorded at 5 consecutive times by positioning, removing, and repositioning the intraoral camera on the saliva-wetted labial surfaces to prevent enamel-color alterations caused by drying. The resulting images were subsequently cross-referenced by using the built-in synchronized image program. Image capture verification was obtained when the total color difference (ΔE) between 2 images taken in a row did not exceed the threshold of 1 ΔE unit (default value). Images of ΔE >1 were discarded to exclude mismeasurements, and new images were captured a second time.
Maxillary and mandibular central and lateral incisors, canines, and first premolars were included in the study. Each tooth underwent spectrophotometric analysis on a standardized circular central area of the middle third of its labial surface ( Fig 1 ). The sizes of the circular area of analysis were 5 mm for the maxillary central incisors and canines, 4 mm for the maxillary lateral incisors and mandibular canines and first premolars, and 3 mm for the mandibular incisors. All images were recorded by the same operator (A.K.) using a repeated-measuring design (n = 5) at 2 time intervals: before bracket bonding and after debonding and cleaning.
The CIE color parameters (L∗, a∗, and b∗) were measured and averaged for each adhesive and type of tooth, and the resultant color differences (ΔE) between the interval groups were calculated according to the following equation:
Δ E = [ ( L ∗ i − L ∗ ii ) 2 + ( a ∗ i − a ∗ ii ) 2 + ( b ∗ i − b ∗ ii ) 2 ] 1 / 2
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


